 Found 44 hits with Last Name = 'spyrakis' and Initial = 'f'
Found 44 hits with Last Name = 'spyrakis' and Initial = 'f' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Thymidylate synthase
(Homo sapiens (Human)) | BDBM50590309

(CHEMBL5196526)Show SMILES CC(C)C[C@H](NC(=O)CC[C@H](NC(=O)c1ccc(NCc2cnc3nc(N)[nH]c(=O)c3n2)cc1)C(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02107
BindingDB Entry DOI: 10.7270/Q2VH5SS8 |
More data for this
Ligand-Target Pair | |
Serine racemase
(Homo sapiens (Human)) | BDBM14673
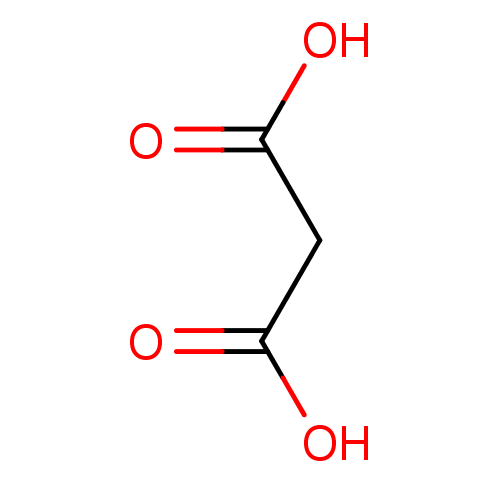
(Fragment 3 | Malonic Acid | propanedioic acid)Show InChI InChI=1S/C3H4O4/c4-2(5)1-3(6)7/h1H2,(H,4,5)(H,6,7) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 5.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant serine racemase expressed in Escherichia coli Rosetta 2 (DE3) cells using L-serine as substrate by horseradish peroxi... |
Bioorg Med Chem Lett 25: 4297-303 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.081
BindingDB Entry DOI: 10.7270/Q2CV4KHX |
More data for this
Ligand-Target Pair | |
Serine racemase
(Homo sapiens (Human)) | BDBM14673

(Fragment 3 | Malonic Acid | propanedioic acid)Show InChI InChI=1S/C3H4O4/c4-2(5)1-3(6)7/h1H2,(H,4,5)(H,6,7) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 6.60E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Inhibition of hexa-His-tagged purified human recombinant serine racemase expressed in Escherichia coli BL21 Codonplus (DE3)-RIL cells assessed as red... |
Bioorg Med Chem Lett 25: 4297-303 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.081
BindingDB Entry DOI: 10.7270/Q2CV4KHX |
More data for this
Ligand-Target Pair | |
Thymidylate synthase
(Homo sapiens (Human)) | BDBM50590310
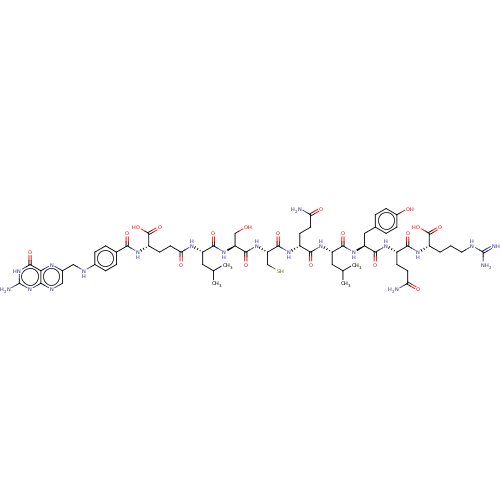
(CHEMBL5195943)Show SMILES CC(C)C[C@H](NC(=O)CC[C@H](NC(=O)c1ccc(NCc2cnc3nc(N)[nH]c(=O)c3n2)cc1)C(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)N[C@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 7.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02107
BindingDB Entry DOI: 10.7270/Q2VH5SS8 |
More data for this
Ligand-Target Pair | |
Serine racemase
(Homo sapiens (Human)) | BDBM50119563
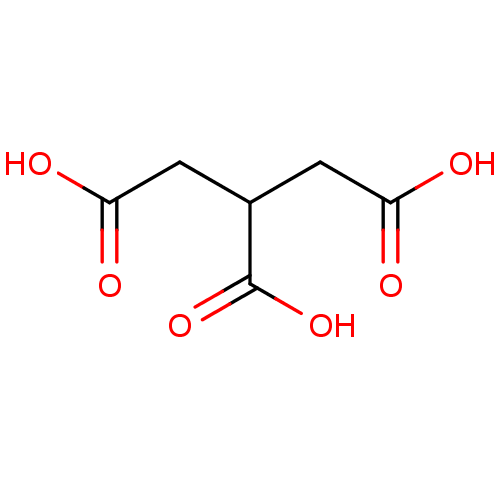
(CHEBI:45969 | TRICARBALLYLIC ACID)Show InChI InChI=1S/C6H8O6/c7-4(8)1-3(6(11)12)2-5(9)10/h3H,1-2H2,(H,7,8)(H,9,10)(H,11,12) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 1.30E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Inhibition of hexa-His-tagged purified human recombinant serine racemase expressed in Escherichia coli BL21 Codonplus (DE3)-RIL cells assessed as red... |
Bioorg Med Chem Lett 25: 4297-303 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.081
BindingDB Entry DOI: 10.7270/Q2CV4KHX |
More data for this
Ligand-Target Pair | |
Serine racemase
(Homo sapiens (Human)) | BDBM50119562
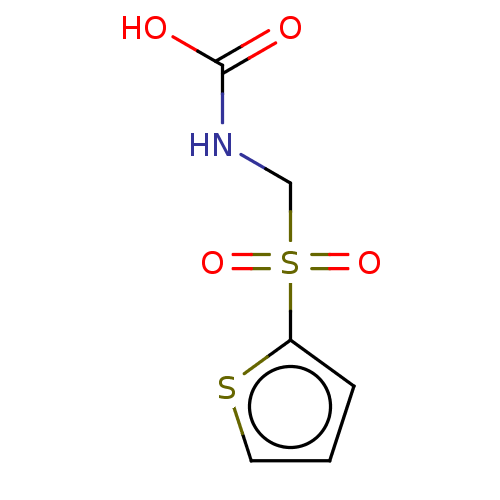
(CHEMBL3617724)Show InChI InChI=1S/C6H7NO4S2/c8-6(9)7-4-13(10,11)5-2-1-3-12-5/h1-3,7H,4H2,(H,8,9) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.50E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Inhibition of hexa-His-tagged purified human recombinant serine racemase expressed in Escherichia coli BL21 Codonplus (DE3)-RIL cells assessed as red... |
Bioorg Med Chem Lett 25: 4297-303 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.081
BindingDB Entry DOI: 10.7270/Q2CV4KHX |
More data for this
Ligand-Target Pair | |
Serine racemase
(Homo sapiens (Human)) | BDBM50119565
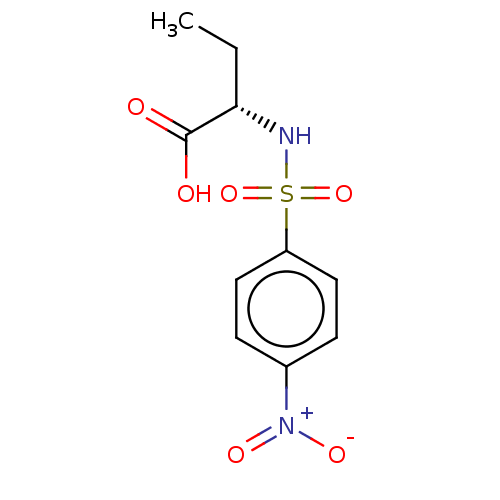
(CHEMBL3617722)Show SMILES CC[C@H](NS(=O)(=O)c1ccc(cc1)[N+]([O-])=O)C(O)=O |r| Show InChI InChI=1S/C10H12N2O6S/c1-2-9(10(13)14)11-19(17,18)8-5-3-7(4-6-8)12(15)16/h3-6,9,11H,2H2,1H3,(H,13,14)/t9-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.50E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Inhibition of hexa-His-tagged purified human recombinant serine racemase expressed in Escherichia coli BL21 Codonplus (DE3)-RIL cells assessed as red... |
Bioorg Med Chem Lett 25: 4297-303 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.081
BindingDB Entry DOI: 10.7270/Q2CV4KHX |
More data for this
Ligand-Target Pair | |
Serine racemase
(Homo sapiens (Human)) | BDBM50119564
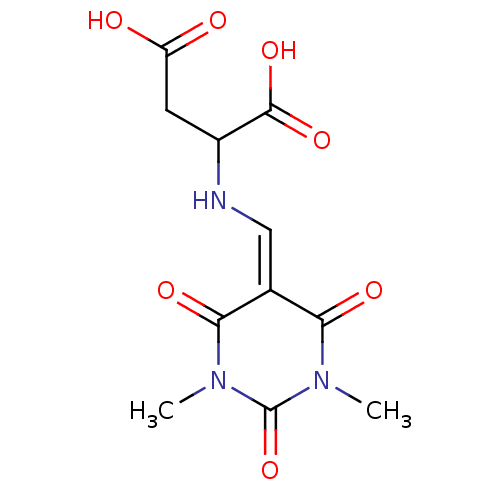
(CHEMBL3617723)Show SMILES [#6]-[#7]-1-[#6](=O)-[#7](-[#6])-[#6](=O)\[#6](=[#6]\[#7]-[#6](-[#6]-[#6](-[#8])=O)-[#6](-[#8])=O)-[#6]-1=O Show InChI InChI=1S/C11H13N3O7/c1-13-8(17)5(9(18)14(2)11(13)21)4-12-6(10(19)20)3-7(15)16/h4,6,12H,3H2,1-2H3,(H,15,16)(H,19,20) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 1.70E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Inhibition of hexa-His-tagged purified human recombinant serine racemase expressed in Escherichia coli BL21 Codonplus (DE3)-RIL cells assessed as red... |
Bioorg Med Chem Lett 25: 4297-303 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.081
BindingDB Entry DOI: 10.7270/Q2CV4KHX |
More data for this
Ligand-Target Pair | |
Serine racemase
(Homo sapiens (Human)) | BDBM50119560
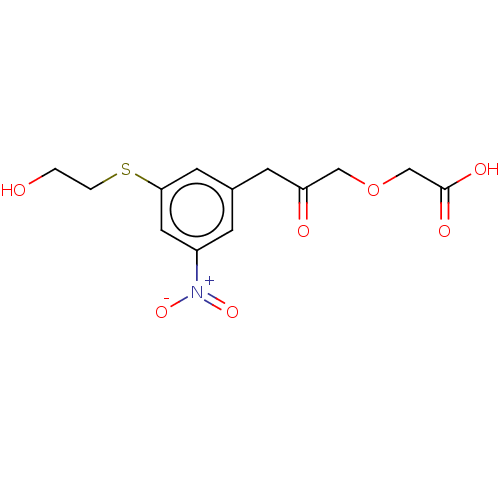
(CHEMBL3617727)Show SMILES OCCSc1cc(CC(=O)COCC(O)=O)cc(c1)[N+]([O-])=O Show InChI InChI=1S/C13H15NO7S/c15-1-2-22-12-5-9(3-10(6-12)14(19)20)4-11(16)7-21-8-13(17)18/h3,5-6,15H,1-2,4,7-8H2,(H,17,18) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.50E+6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Inhibition of hexa-His-tagged purified human recombinant serine racemase expressed in Escherichia coli BL21 Codonplus (DE3)-RIL cells assessed as red... |
Bioorg Med Chem Lett 25: 4297-303 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.081
BindingDB Entry DOI: 10.7270/Q2CV4KHX |
More data for this
Ligand-Target Pair | |
Serine racemase
(Homo sapiens (Human)) | BDBM50119561
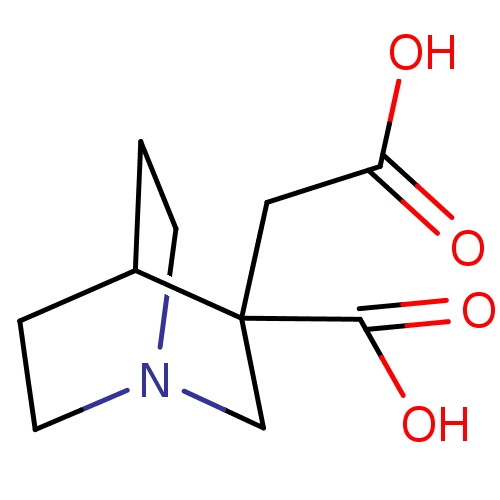
(CHEMBL3617725)Show SMILES OC(=O)CC1(CN2CCC1CC2)C(O)=O |THB:3:4:8.7:10.11,12:4:8.7:10.11,(0,2.93,;-1.3,2.21,;-2.57,2.97,;-1.32,.35,;.3,-.59,;2.12,-3.48,;-1.06,-4.26,;.27,-3.48,;.27,-1.94,;-1.06,-1.19,;-2.37,-1.94,;-2.37,-3.48,;1.9,.37,;3.19,-.37,;1.89,1.85,)| Show InChI InChI=1S/C10H15NO4/c12-8(13)5-10(9(14)15)6-11-3-1-7(10)2-4-11/h7H,1-6H2,(H,12,13)(H,14,15) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >1.00E+7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Inhibition of hexa-His-tagged purified human recombinant serine racemase expressed in Escherichia coli BL21 Codonplus (DE3)-RIL cells assessed as red... |
Bioorg Med Chem Lett 25: 4297-303 (2015)
Article DOI: 10.1016/j.bmcl.2015.07.081
BindingDB Entry DOI: 10.7270/Q2CV4KHX |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50593724
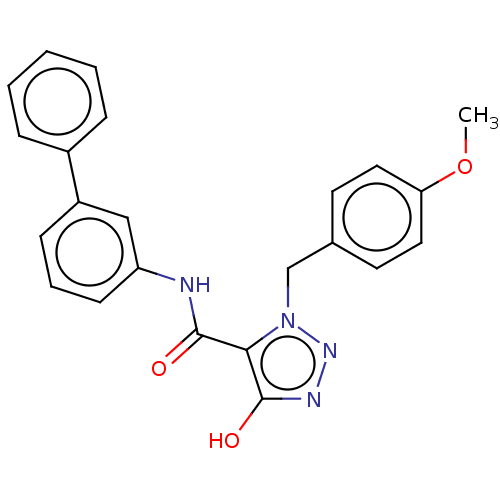
(CHEMBL5194388)Show SMILES COc1ccc(Cn2nnc(O)c2C(=O)Nc2cccc(c2)-c2ccccc2)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 69 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50593730
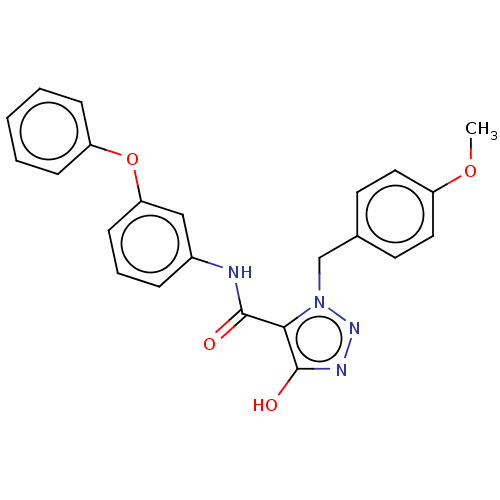
(CHEMBL5172658)Show SMILES COc1ccc(Cn2nnc(O)c2C(=O)Nc2cccc(Oc3ccccc3)c2)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50509767
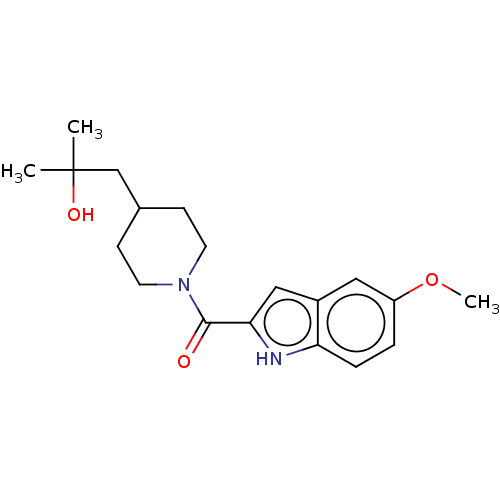
(CHEMBL4513510)Show SMILES COc1ccc2[nH]c(cc2c1)C(=O)N1CCC(CC(C)(C)O)CC1 Show InChI InChI=1S/C19H26N2O3/c1-19(2,23)12-13-6-8-21(9-7-13)18(22)17-11-14-10-15(24-3)4-5-16(14)20-17/h4-5,10-11,13,20,23H,6-9,12H2,1-3H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Casein kinase I isoform delta
(Homo sapiens (Human)) | BDBM50509981

(CHEMBL3990606)Show SMILES CN(C)Cc1n[nH]c(n1)-c1ccc2[nH]nc(-c3cccc(c3)C(=O)Nc3ccc(F)cc3)c2c1 Show InChI InChI=1S/C25H22FN7O/c1-33(2)14-22-28-24(32-30-22)16-6-11-21-20(13-16)23(31-29-21)15-4-3-5-17(12-15)25(34)27-19-9-7-18(26)8-10-19/h3-13H,14H2,1-2H3,(H,27,34)(H,29,31)(H,28,30,32) | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 134 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CSNK1D (unknown origin) using CSNK1D/Ser/Thr 11 peptide incubated for 1 hr by FRET based Z'-LYTE assay |
ACS Med Chem Lett 10: 487-492 (2019)
Article DOI: 10.1021/acsmedchemlett.8b00523
BindingDB Entry DOI: 10.7270/Q289195J |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50593732

(CHEMBL5174744)Show SMILES COc1ccc(Cc2onc(O)c2C(=O)Nc2cccc(c2)-c2ccccc2)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50593726
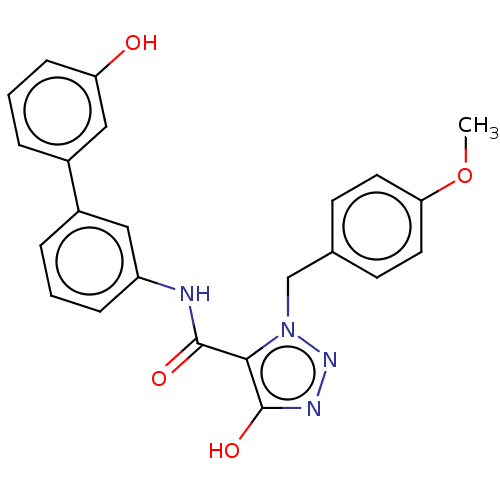
(CHEMBL5204156)Show SMILES COc1ccc(Cn2nnc(O)c2C(=O)Nc2cccc(c2)-c2cccc(O)c2)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50593727
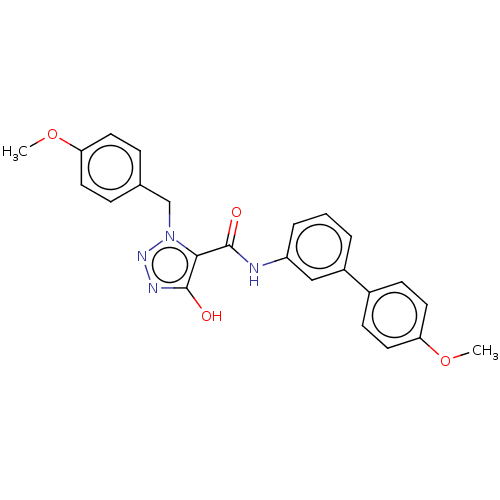
(CHEMBL5170149)Show SMILES COc1ccc(Cn2nnc(O)c2C(=O)Nc2cccc(c2)-c2ccc(OC)cc2)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 220 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50593728
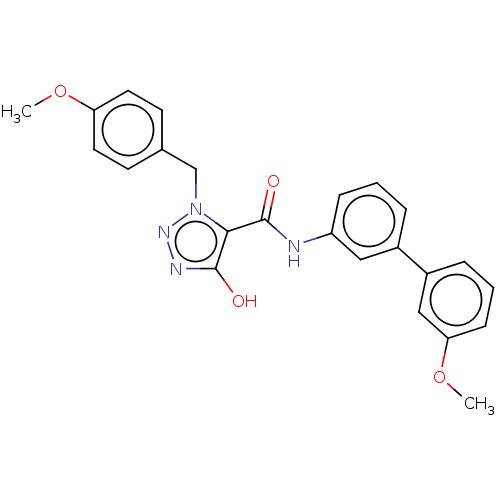
(CHEMBL5170401)Show SMILES COc1ccc(Cn2nnc(O)c2C(=O)Nc2cccc(c2)-c2cccc(OC)c2)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 260 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50593729
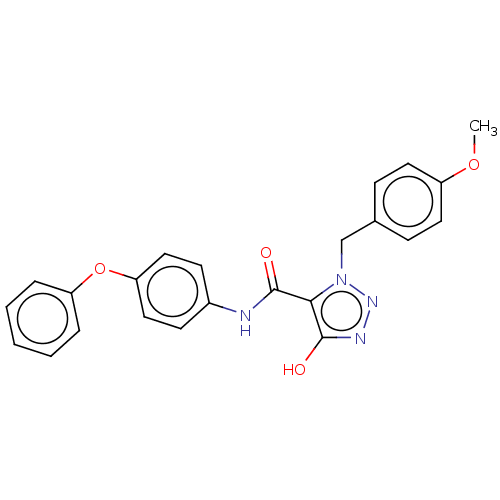
(CHEMBL5175151)Show SMILES COc1ccc(Cn2nnc(O)c2C(=O)Nc2ccc(Oc3ccccc3)cc2)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50593725
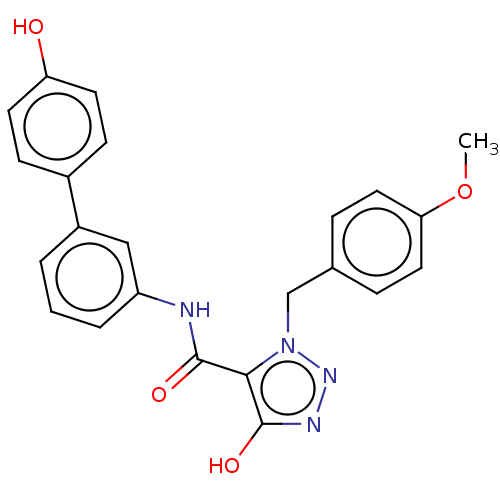
(CHEMBL5206583)Show SMILES COc1ccc(Cn2nnc(O)c2C(=O)Nc2cccc(c2)-c2ccc(O)cc2)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 310 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50277998
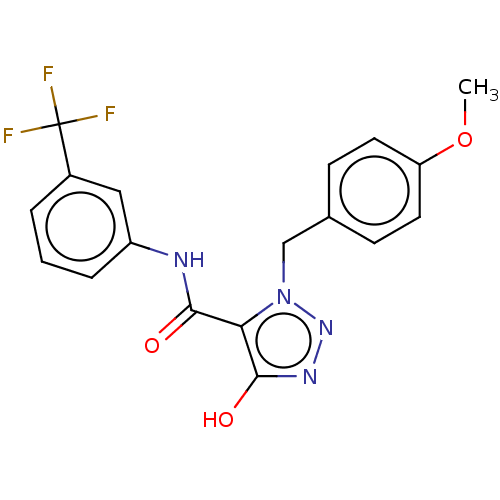
(CHEMBL4174786)Show SMILES COc1ccc(Cn2nnc(O)c2C(=O)Nc2cccc(c2)C(F)(F)F)cc1 Show InChI InChI=1S/C18H15F3N4O3/c1-28-14-7-5-11(6-8-14)10-25-15(17(27)23-24-25)16(26)22-13-4-2-3-12(9-13)18(19,20)21/h2-9,27H,10H2,1H3,(H,22,26) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 310 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Casein kinase I isoform delta
(Homo sapiens (Human)) | BDBM50509984
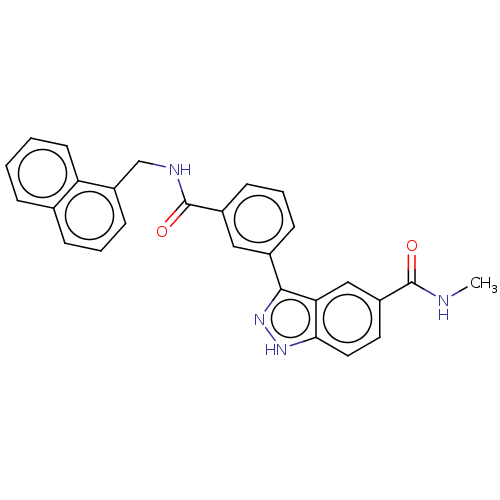
(CHEMBL4476186)Show SMILES CNC(=O)c1ccc2[nH]nc(-c3cccc(c3)C(=O)NCc3cccc4ccccc34)c2c1 Show InChI InChI=1S/C27H22N4O2/c1-28-26(32)20-12-13-24-23(15-20)25(31-30-24)18-8-5-9-19(14-18)27(33)29-16-21-10-4-7-17-6-2-3-11-22(17)21/h2-15H,16H2,1H3,(H,28,32)(H,29,33)(H,30,31) | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 354 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CSNK1D (unknown origin) using CSNK1D/Ser/Thr 11 peptide incubated for 1 hr by FRET based Z'-LYTE assay |
ACS Med Chem Lett 10: 487-492 (2019)
Article DOI: 10.1021/acsmedchemlett.8b00523
BindingDB Entry DOI: 10.7270/Q289195J |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50593722
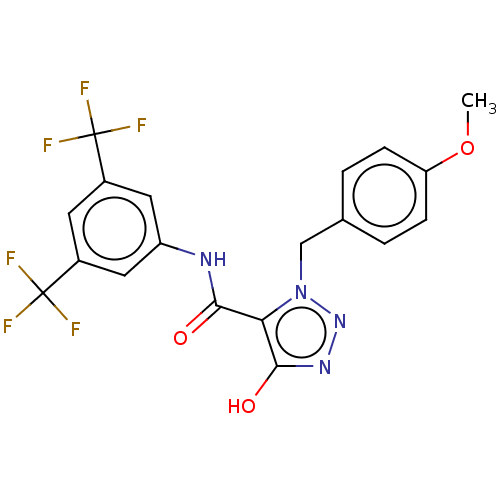
(CHEMBL5191752)Show SMILES COc1ccc(Cn2nnc(O)c2C(=O)Nc2cc(cc(c2)C(F)(F)F)C(F)(F)F)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50593723
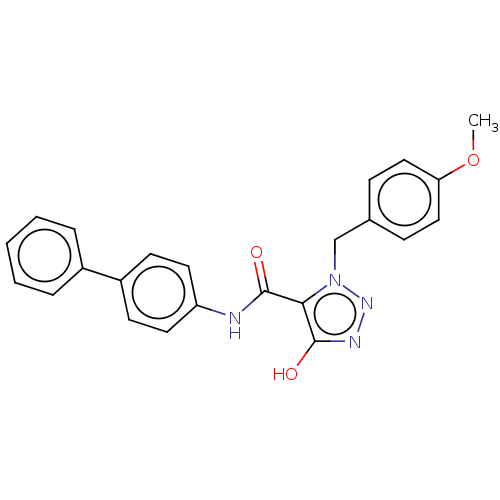
(CHEMBL5187723)Show SMILES COc1ccc(Cn2nnc(O)c2C(=O)Nc2ccc(cc2)-c2ccccc2)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 470 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Casein kinase I isoform delta
(Homo sapiens (Human)) | BDBM50509983

(CHEMBL3940534)Show SMILES Fc1ccc(cc1)-c1n[nH]c2ccc(cc12)-c1n[nH]c(CN2CCCC2)n1 Show InChI InChI=1S/C20H19FN6/c21-15-6-3-13(4-7-15)19-16-11-14(5-8-17(16)23-25-19)20-22-18(24-26-20)12-27-9-1-2-10-27/h3-8,11H,1-2,9-10,12H2,(H,23,25)(H,22,24,26) | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 848 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CSNK1D (unknown origin) using CSNK1D/Ser/Thr 11 peptide incubated for 1 hr by FRET based Z'-LYTE assay |
ACS Med Chem Lett 10: 487-492 (2019)
Article DOI: 10.1021/acsmedchemlett.8b00523
BindingDB Entry DOI: 10.7270/Q289195J |
More data for this
Ligand-Target Pair | |
Casein kinase I isoform delta
(Homo sapiens (Human)) | BDBM50509982

(CHEMBL4519384)Show SMILES C[C@@H](NC(=O)c1ccc2[nH]nc(-c3cccc(NS(C)(=O)=O)c3)c2c1)c1ccccc1 |r| Show InChI InChI=1S/C23H22N4O3S/c1-15(16-7-4-3-5-8-16)24-23(28)18-11-12-21-20(14-18)22(26-25-21)17-9-6-10-19(13-17)27-31(2,29)30/h3-15,27H,1-2H3,(H,24,28)(H,25,26)/t15-/m1/s1 | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.03E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of CSNK1D (unknown origin) using CSNK1D/Ser/Thr 11 peptide incubated for 1 hr by FRET based Z'-LYTE assay |
ACS Med Chem Lett 10: 487-492 (2019)
Article DOI: 10.1021/acsmedchemlett.8b00523
BindingDB Entry DOI: 10.7270/Q289195J |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50593731
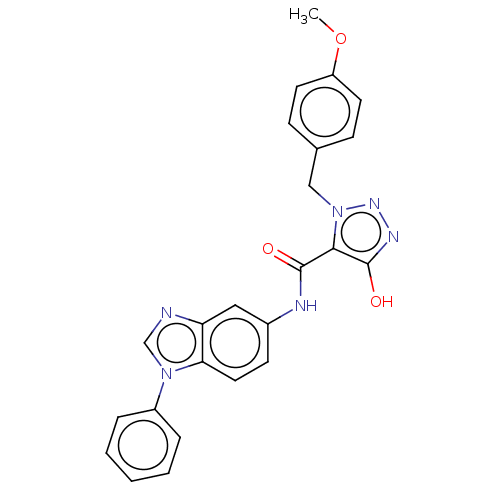
(CHEMBL5183881)Show SMILES COc1ccc(Cn2nnc(O)c2C(=O)Nc2ccc3n(cnc3c2)-c2ccccc2)cc1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.63E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C2
(Homo sapiens (Human)) | BDBM50509767

(CHEMBL4513510)Show SMILES COc1ccc2[nH]c(cc2c1)C(=O)N1CCC(CC(C)(C)O)CC1 Show InChI InChI=1S/C19H26N2O3/c1-19(2,23)12-13-6-8-21(9-7-13)18(22)17-11-14-10-15(24-3)4-5-16(14)20-17/h4-5,10-11,13,20,23H,6-9,12H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.ejmech.2022.114366
BindingDB Entry DOI: 10.7270/Q2TB1BWP |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423697
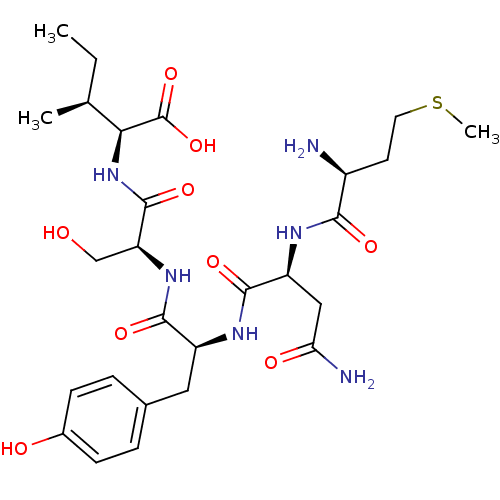
(CHEMBL568986)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CO)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(O)=O |r| Show InChI InChI=1S/C27H42N6O9S/c1-4-14(2)22(27(41)42)33-26(40)20(13-34)32-24(38)18(11-15-5-7-16(35)8-6-15)31-25(39)19(12-21(29)36)30-23(37)17(28)9-10-43-3/h5-8,14,17-20,22,34-35H,4,9-13,28H2,1-3H3,(H2,29,36)(H,30,37)(H,31,39)(H,32,38)(H,33,40)(H,41,42)/t14-,17-,18-,19-,20-,22-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 6.03E+4 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423698
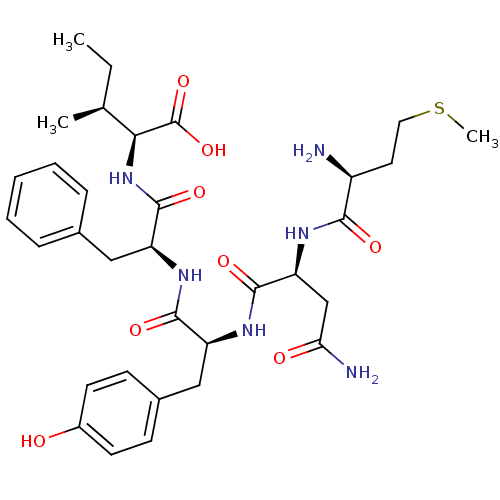
(CHEMBL568987)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(O)=O |r| Show InChI InChI=1S/C33H46N6O8S/c1-4-19(2)28(33(46)47)39-32(45)25(16-20-8-6-5-7-9-20)38-30(43)24(17-21-10-12-22(40)13-11-21)37-31(44)26(18-27(35)41)36-29(42)23(34)14-15-48-3/h5-13,19,23-26,28,40H,4,14-18,34H2,1-3H3,(H2,35,41)(H,36,42)(H,37,44)(H,38,43)(H,39,45)(H,46,47)/t19-,23-,24-,25-,26-,28-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.91E+5 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Thymidylate synthase
(Homo sapiens (Human)) | BDBM50590310

(CHEMBL5195943)Show SMILES CC(C)C[C@H](NC(=O)CC[C@H](NC(=O)c1ccc(NCc2cnc3nc(N)[nH]c(=O)c3n2)cc1)C(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)N[C@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | 1.20E+5 | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02107
BindingDB Entry DOI: 10.7270/Q2VH5SS8 |
More data for this
Ligand-Target Pair | |
Thymidylate synthase
(Homo sapiens (Human)) | BDBM50590309

(CHEMBL5196526)Show SMILES CC(C)C[C@H](NC(=O)CC[C@H](NC(=O)c1ccc(NCc2cnc3nc(N)[nH]c(=O)c3n2)cc1)C(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O |r| | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | 4.40E+4 | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c02107
BindingDB Entry DOI: 10.7270/Q2VH5SS8 |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423699
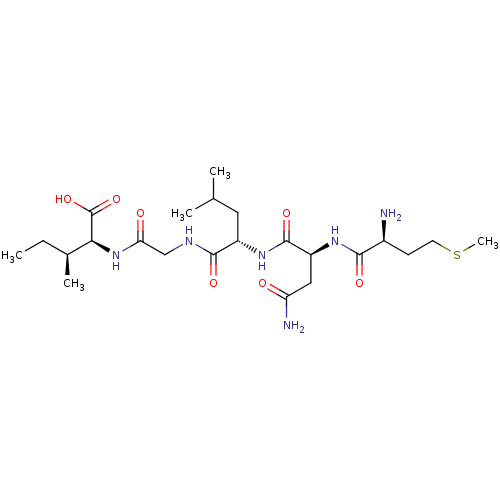
(CHEMBL571766)Show SMILES CC[C@H](C)[C@H](NC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(O)=O |r| Show InChI InChI=1S/C23H42N6O7S/c1-6-13(4)19(23(35)36)29-18(31)11-26-21(33)15(9-12(2)3)28-22(34)16(10-17(25)30)27-20(32)14(24)7-8-37-5/h12-16,19H,6-11,24H2,1-5H3,(H2,25,30)(H,26,33)(H,27,32)(H,28,34)(H,29,31)(H,35,36)/t13-,14-,15-,16-,19-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 5.75E+5 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423700
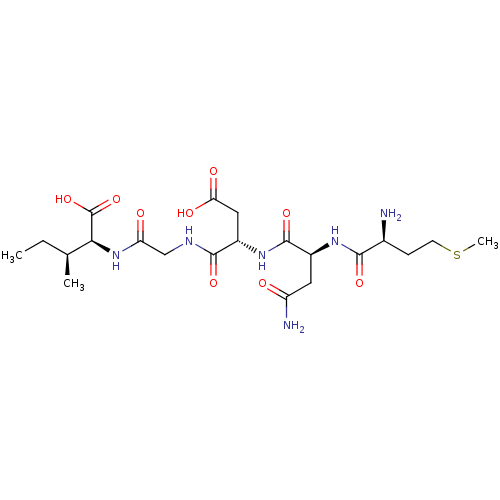
(CHEMBL568812)Show SMILES CC[C@H](C)[C@H](NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(O)=O |r| Show InChI InChI=1S/C21H36N6O9S/c1-4-10(2)17(21(35)36)27-15(29)9-24-19(33)13(8-16(30)31)26-20(34)12(7-14(23)28)25-18(32)11(22)5-6-37-3/h10-13,17H,4-9,22H2,1-3H3,(H2,23,28)(H,24,33)(H,25,32)(H,26,34)(H,27,29)(H,30,31)(H,35,36)/t10-,11-,12-,13-,17-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.02E+6 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423701
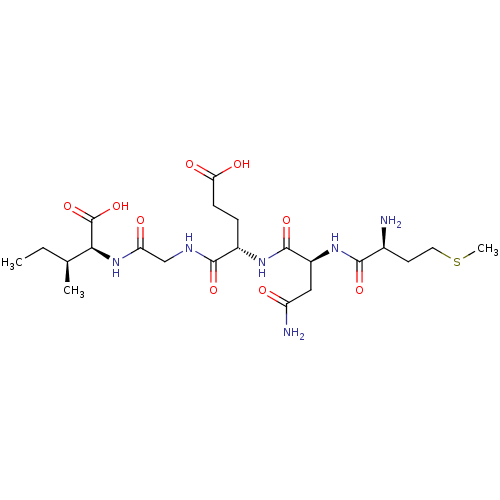
(CHEMBL576268)Show SMILES CC[C@H](C)[C@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(O)=O |r| Show InChI InChI=1S/C22H38N6O9S/c1-4-11(2)18(22(36)37)28-16(30)10-25-20(34)13(5-6-17(31)32)26-21(35)14(9-15(24)29)27-19(33)12(23)7-8-38-3/h11-14,18H,4-10,23H2,1-3H3,(H2,24,29)(H,25,34)(H,26,35)(H,27,33)(H,28,30)(H,31,32)(H,36,37)/t11-,12-,13-,14-,18-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 2.29E+6 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423702
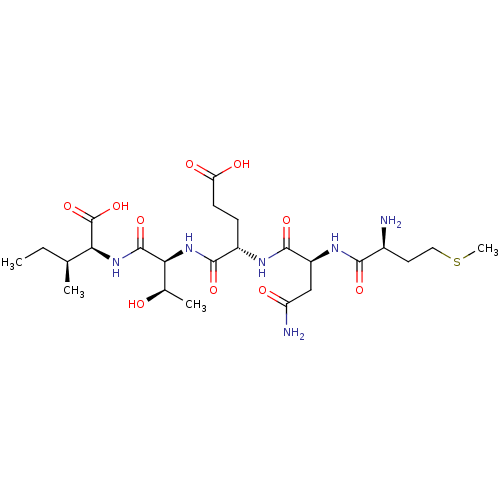
(CHEMBL585737)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)[C@@H](C)O)C(O)=O |r| Show InChI InChI=1S/C24H42N6O10S/c1-5-11(2)18(24(39)40)29-23(38)19(12(3)31)30-21(36)14(6-7-17(33)34)27-22(37)15(10-16(26)32)28-20(35)13(25)8-9-41-4/h11-15,18-19,31H,5-10,25H2,1-4H3,(H2,26,32)(H,27,37)(H,28,35)(H,29,38)(H,30,36)(H,33,34)(H,39,40)/t11-,12+,13-,14-,15-,18-,19-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 3.39E+6 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423703
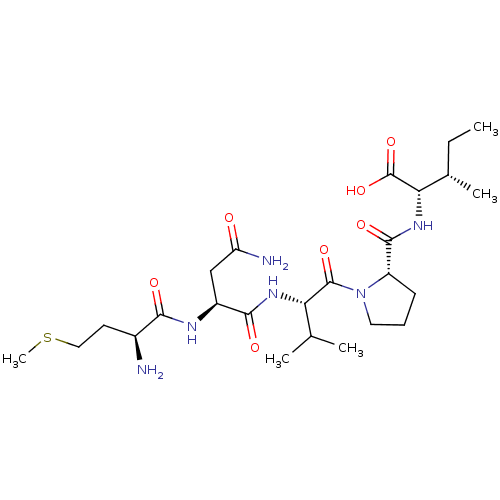
(CHEMBL582831)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(C)C)C(O)=O |r| Show InChI InChI=1S/C25H44N6O7S/c1-6-14(4)20(25(37)38)30-23(35)17-8-7-10-31(17)24(36)19(13(2)3)29-22(34)16(12-18(27)32)28-21(33)15(26)9-11-39-5/h13-17,19-20H,6-12,26H2,1-5H3,(H2,27,32)(H,28,33)(H,29,34)(H,30,35)(H,37,38)/t14-,15-,16-,17-,19-,20-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 3.31E+6 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423704

(CHEMBL567022)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](Cc1cnc[nH]1)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(O)=O |r| Show InChI InChI=1S/C26H42N8O7S/c1-4-14(2)21(26(40)41)33-23(37)17(10-15-12-29-13-30-15)31-24(38)19-6-5-8-34(19)25(39)18(11-20(28)35)32-22(36)16(27)7-9-42-3/h12-14,16-19,21H,4-11,27H2,1-3H3,(H2,28,35)(H,29,30)(H,31,38)(H,32,36)(H,33,37)(H,40,41)/t14-,16-,17-,18-,19-,21-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 7.08E+6 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423705

(CHEMBL569309)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCCN)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(C)C)C(O)=O |r| Show InChI InChI=1S/C26H49N7O7S/c1-6-15(4)21(26(39)40)33-25(38)20(14(2)3)32-23(36)17(9-7-8-11-27)30-24(37)18(13-19(29)34)31-22(35)16(28)10-12-41-5/h14-18,20-21H,6-13,27-28H2,1-5H3,(H2,29,34)(H,30,37)(H,31,35)(H,32,36)(H,33,38)(H,39,40)/t15-,16-,17-,18-,20-,21-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.32E+7 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423706
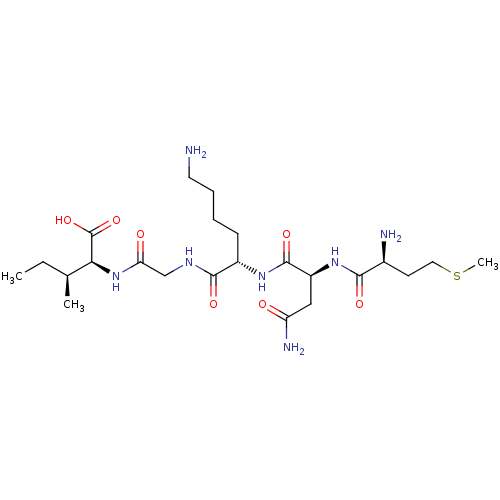
(CHEMBL570451)Show SMILES CC[C@H](C)[C@H](NC(=O)CNC(=O)[C@H](CCCCN)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(O)=O |r| Show InChI InChI=1S/C23H43N7O7S/c1-4-13(2)19(23(36)37)30-18(32)12-27-21(34)15(7-5-6-9-24)28-22(35)16(11-17(26)31)29-20(33)14(25)8-10-38-3/h13-16,19H,4-12,24-25H2,1-3H3,(H2,26,31)(H,27,34)(H,28,35)(H,29,33)(H,30,32)(H,36,37)/t13-,14-,15-,16-,19-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 1.51E+7 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423707
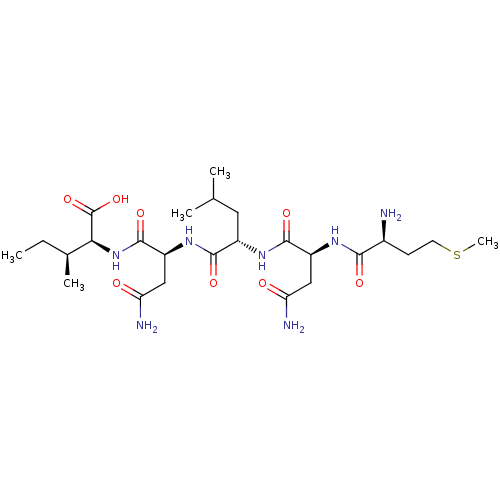
(CHEMBL571997)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(O)=O |r| Show InChI InChI=1S/C25H45N7O8S/c1-6-13(4)20(25(39)40)32-24(38)17(11-19(28)34)31-22(36)15(9-12(2)3)30-23(37)16(10-18(27)33)29-21(35)14(26)7-8-41-5/h12-17,20H,6-11,26H2,1-5H3,(H2,27,33)(H2,28,34)(H,29,35)(H,30,37)(H,31,36)(H,32,38)(H,39,40)/t13-,14-,15-,16-,17-,20-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 4.37E+4 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423709

(CHEMBL568900)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1c[nH]c2ccccc12)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(O)=O |r| Show InChI InChI=1S/C30H44N8O8S/c1-4-15(2)25(30(45)46)38-29(44)22(13-24(33)40)37-27(42)20(11-16-14-34-19-8-6-5-7-17(16)19)36-28(43)21(12-23(32)39)35-26(41)18(31)9-10-47-3/h5-8,14-15,18,20-22,25,34H,4,9-13,31H2,1-3H3,(H2,32,39)(H2,33,40)(H,35,41)(H,36,43)(H,37,42)(H,38,44)(H,45,46)/t15-,18-,20-,21-,22-,25-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 2.51E+4 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423710

(CHEMBL569308)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(O)=O |r| Show InChI InChI=1S/C24H41N7O10S/c1-4-11(2)19(24(40)41)31-23(39)15(10-17(27)33)30-21(37)13(5-6-18(34)35)28-22(38)14(9-16(26)32)29-20(36)12(25)7-8-42-3/h11-15,19H,4-10,25H2,1-3H3,(H2,26,32)(H2,27,33)(H,28,38)(H,29,36)(H,30,37)(H,31,39)(H,34,35)(H,40,41)/t11-,12-,13-,14-,15-,19-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 3.89E+4 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |
Cysteine synthase
(Haemophilus influenzae (strain ATCC 51907 / DSM 11...) | BDBM50423708
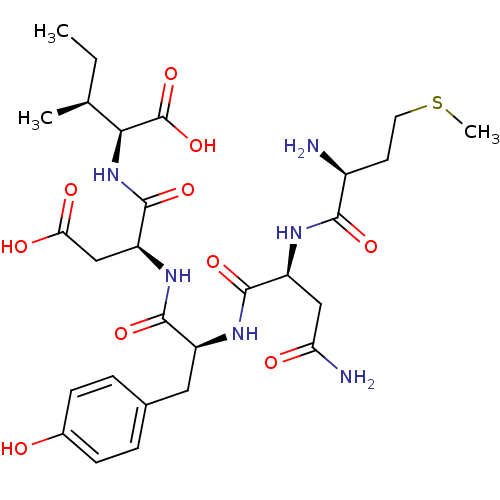
(CHEMBL568828)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CCSC)C(O)=O |r| Show InChI InChI=1S/C28H42N6O10S/c1-4-14(2)23(28(43)44)34-27(42)20(13-22(37)38)33-25(40)18(11-15-5-7-16(35)8-6-15)32-26(41)19(12-21(30)36)31-24(39)17(29)9-10-45-3/h5-8,14,17-20,23,35H,4,9-13,29H2,1-3H3,(H2,30,36)(H,31,39)(H,32,41)(H,33,40)(H,34,42)(H,37,38)(H,43,44)/t14-,17-,18-,19-,20-,23-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | n/a | 2.57E+4 | n/a | n/a | n/a | n/a | n/a |
University of Parma
Curated by ChEMBL
| Assay Description
Binding affinity to Haemophilus influenzae O-acetylserine sulfhydrylase expressed in Escherichia coli BL21 (DE3) by fluorescence emission spectra ana... |
J Med Chem 53: 345-56 (2010)
Article DOI: 10.1021/jm901325e
BindingDB Entry DOI: 10.7270/Q2QZ2C8P |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data