 Found 464 hits with Last Name = 'baik' and Initial = 't'
Found 464 hits with Last Name = 'baik' and Initial = 't' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393357

(CHEMBL2152148)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(=O)(=O)Nc1ccc(F)cc1Cl Show InChI InChI=1S/C18H14Cl2FN5O3S/c1-23-18(27)16-17(22)24-8-14(25-16)9-2-4-11(19)15(6-9)30(28,29)26-13-5-3-10(21)7-12(13)20/h2-8,26H,1H3,(H2,22,24)(H,23,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393365
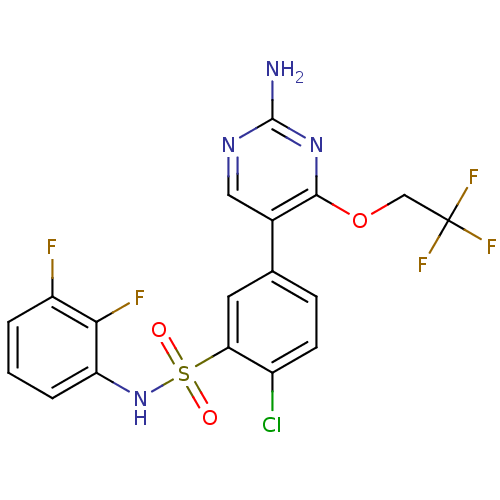
(CHEMBL2152257)Show SMILES Nc1ncc(-c2ccc(Cl)c(c2)S(=O)(=O)Nc2cccc(F)c2F)c(OCC(F)(F)F)n1 Show InChI InChI=1S/C18H12ClF5N4O3S/c19-11-5-4-9(10-7-26-17(25)27-16(10)31-8-18(22,23)24)6-14(11)32(29,30)28-13-3-1-2-12(20)15(13)21/h1-7,28H,8H2,(H2,25,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393359
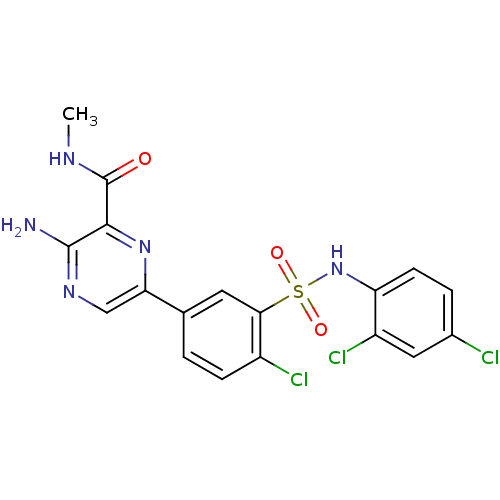
(CHEMBL2152150)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(=O)(=O)Nc1ccc(Cl)cc1Cl Show InChI InChI=1S/C18H14Cl3N5O3S/c1-23-18(27)16-17(22)24-8-14(25-16)9-2-4-11(20)15(6-9)30(28,29)26-13-5-3-10(19)7-12(13)21/h2-8,26H,1H3,(H2,22,24)(H,23,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393333
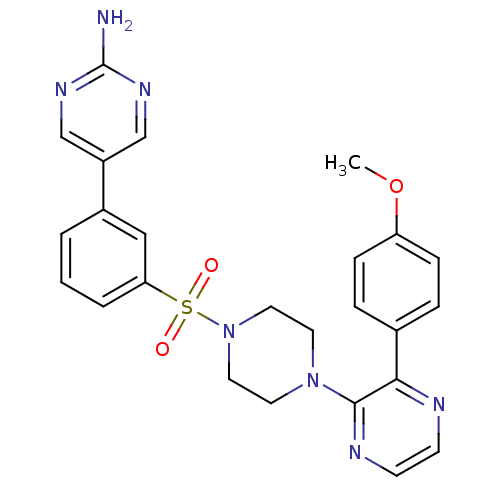
(CHEMBL2151050)Show SMILES COc1ccc(cc1)-c1nccnc1N1CCN(CC1)S(=O)(=O)c1cccc(c1)-c1cnc(N)nc1 Show InChI InChI=1S/C25H25N7O3S/c1-35-21-7-5-18(6-8-21)23-24(28-10-9-27-23)31-11-13-32(14-12-31)36(33,34)22-4-2-3-19(15-22)20-16-29-25(26)30-17-20/h2-10,15-17H,11-14H2,1H3,(H2,26,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393348
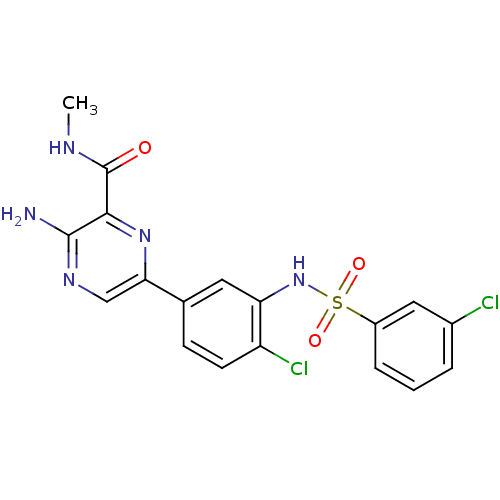
(CHEMBL2152139)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(NS(=O)(=O)c2cccc(Cl)c2)c1 Show InChI InChI=1S/C18H15Cl2N5O3S/c1-22-18(26)16-17(21)23-9-15(24-16)10-5-6-13(20)14(7-10)25-29(27,28)12-4-2-3-11(19)8-12/h2-9,25H,1H3,(H2,21,23)(H,22,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393339
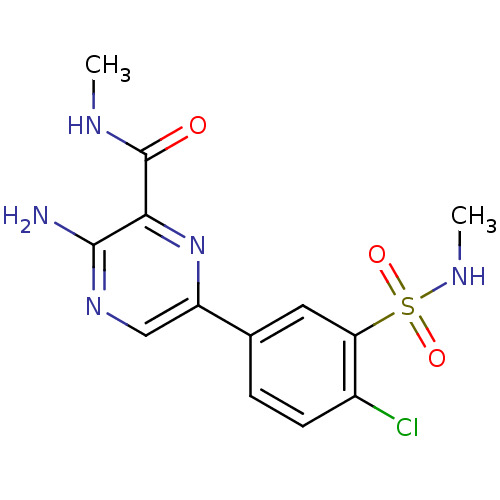
(CHEMBL2152131)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(=O)(=O)NC Show InChI InChI=1S/C13H14ClN5O3S/c1-16-13(20)11-12(15)18-6-9(19-11)7-3-4-8(14)10(5-7)23(21,22)17-2/h3-6,17H,1-2H3,(H2,15,18)(H,16,20) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393353
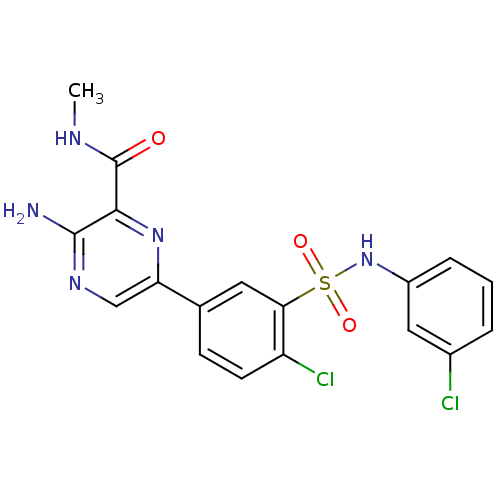
(CHEMBL2152144)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(=O)(=O)Nc1cccc(Cl)c1 Show InChI InChI=1S/C18H15Cl2N5O3S/c1-22-18(26)16-17(21)23-9-14(24-16)10-5-6-13(20)15(7-10)29(27,28)25-12-4-2-3-11(19)8-12/h2-9,25H,1H3,(H2,21,23)(H,22,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393342

(CHEMBL2152252)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(=O)(=O)Nc1cccc(F)c1F Show InChI InChI=1S/C18H14ClF2N5O3S/c1-23-18(27)16-17(22)24-8-13(25-16)9-5-6-10(19)14(7-9)30(28,29)26-12-4-2-3-11(20)15(12)21/h2-8,26H,1H3,(H2,22,24)(H,23,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393335
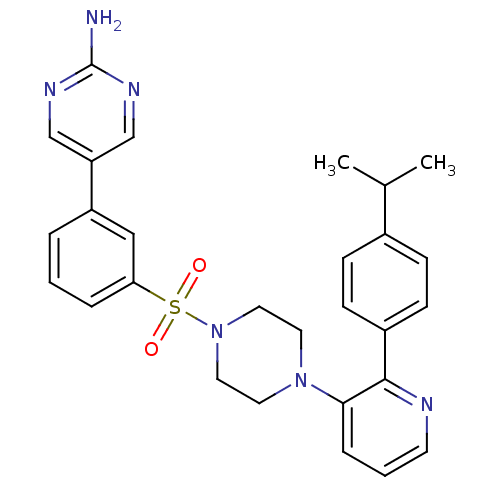
(CHEMBL2152127)Show SMILES CC(C)c1ccc(cc1)-c1ncccc1N1CCN(CC1)S(=O)(=O)c1cccc(c1)-c1cnc(N)nc1 Show InChI InChI=1S/C28H30N6O2S/c1-20(2)21-8-10-22(11-9-21)27-26(7-4-12-30-27)33-13-15-34(16-14-33)37(35,36)25-6-3-5-23(17-25)24-18-31-28(29)32-19-24/h3-12,17-20H,13-16H2,1-2H3,(H2,29,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393334
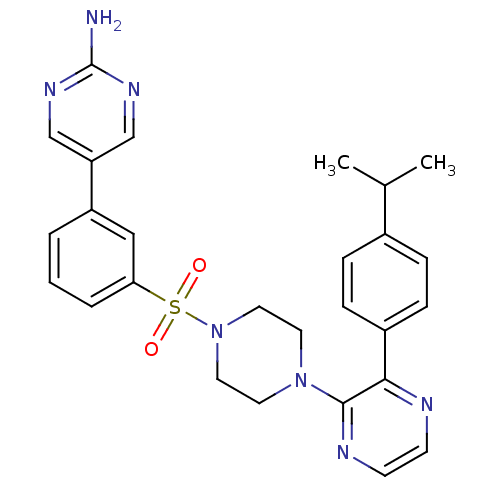
(CHEMBL2152126)Show SMILES CC(C)c1ccc(cc1)-c1nccnc1N1CCN(CC1)S(=O)(=O)c1cccc(c1)-c1cnc(N)nc1 Show InChI InChI=1S/C27H29N7O2S/c1-19(2)20-6-8-21(9-7-20)25-26(30-11-10-29-25)33-12-14-34(15-13-33)37(35,36)24-5-3-4-22(16-24)23-17-31-27(28)32-18-23/h3-11,16-19H,12-15H2,1-2H3,(H2,28,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393360
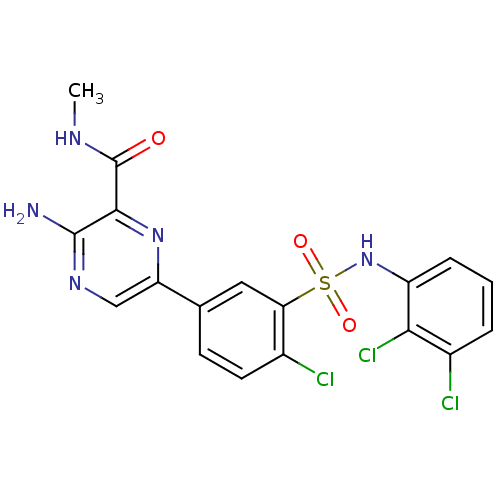
(CHEMBL2152250)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(=O)(=O)Nc1cccc(Cl)c1Cl Show InChI InChI=1S/C18H14Cl3N5O3S/c1-23-18(27)16-17(22)24-8-13(25-16)9-5-6-10(19)14(7-9)30(28,29)26-12-4-2-3-11(20)15(12)21/h2-8,26H,1H3,(H2,22,24)(H,23,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 18 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393364
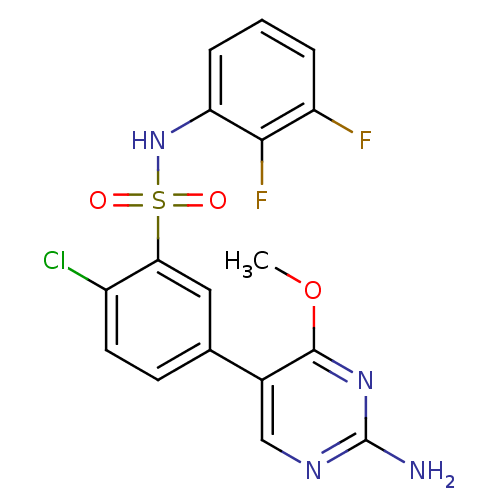
(CHEMBL2152256)Show SMILES COc1nc(N)ncc1-c1ccc(Cl)c(c1)S(=O)(=O)Nc1cccc(F)c1F Show InChI InChI=1S/C17H13ClF2N4O3S/c1-27-16-10(8-22-17(21)23-16)9-5-6-11(18)14(7-9)28(25,26)24-13-4-2-3-12(19)15(13)20/h2-8,24H,1H3,(H2,21,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393354
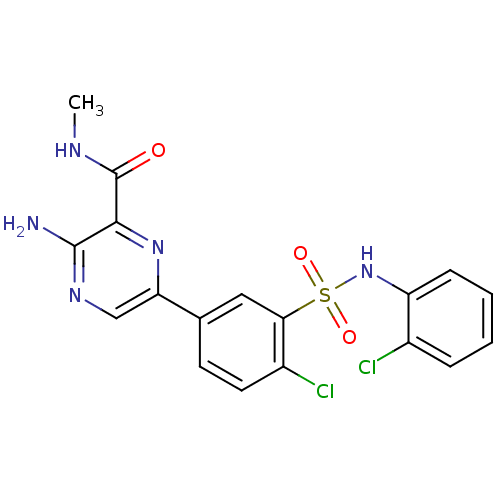
(CHEMBL2152145)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(=O)(=O)Nc1ccccc1Cl Show InChI InChI=1S/C18H15Cl2N5O3S/c1-22-18(26)16-17(21)23-9-14(24-16)10-6-7-12(20)15(8-10)29(27,28)25-13-5-3-2-4-11(13)19/h2-9,25H,1H3,(H2,21,23)(H,22,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50486034
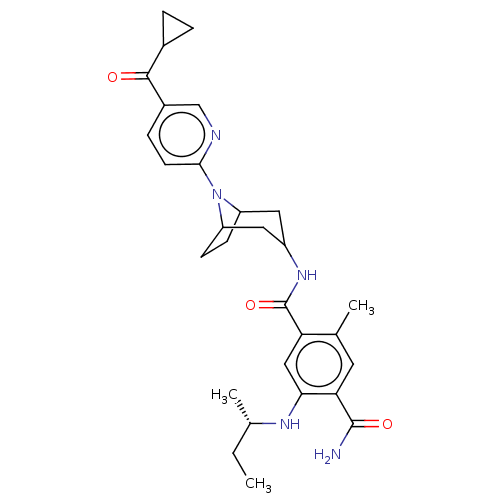
(CHEMBL2204501)Show SMILES CC[C@H](C)Nc1cc(C(=O)NC2CC3CCC(C2)N3c2ccc(cn2)C(=O)C2CC2)c(C)cc1C(N)=O |r,TLB:19:18:14.15:12.11.17,10:11:14.15:18| Show InChI InChI=1S/C29H37N5O3/c1-4-17(3)32-25-14-23(16(2)11-24(25)28(30)36)29(37)33-20-12-21-8-9-22(13-20)34(21)26-10-7-19(15-31-26)27(35)18-5-6-18/h7,10-11,14-15,17-18,20-22,32H,4-6,8-9,12-13H2,1-3H3,(H2,30,36)(H,33,37)/t17-,20?,21?,22?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis Inc
Curated by ChEMBL
| Assay Description
Displacement of biotinylated geldanamycin from human His-tagged Hsp90 by TR-FRET assay |
Bioorg Med Chem Lett 22: 5396-404 (2012)
Article DOI: 10.1016/j.bmcl.2012.07.052
BindingDB Entry DOI: 10.7270/Q25M68KK |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393332
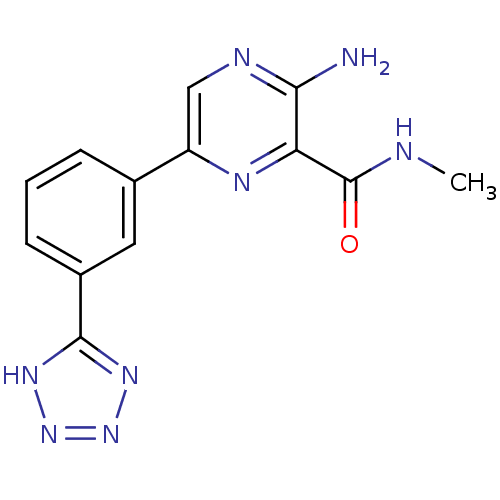
(CHEMBL2152125)Show InChI InChI=1S/C13H12N8O/c1-15-13(22)10-11(14)16-6-9(17-10)7-3-2-4-8(5-7)12-18-20-21-19-12/h2-6H,1H3,(H2,14,16)(H,15,22)(H,18,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393358
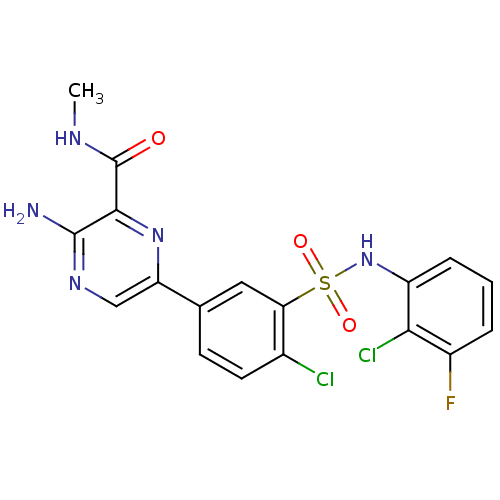
(CHEMBL2152149)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(=O)(=O)Nc1cccc(F)c1Cl Show InChI InChI=1S/C18H14Cl2FN5O3S/c1-23-18(27)16-17(22)24-8-13(25-16)9-5-6-10(19)14(7-9)30(28,29)26-12-4-2-3-11(21)15(12)20/h2-8,26H,1H3,(H2,22,24)(H,23,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50486035
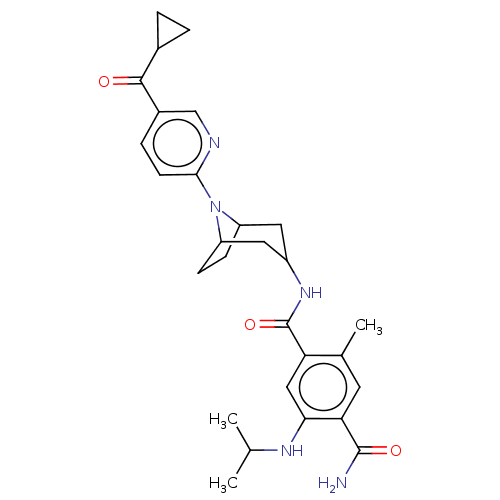
(CHEMBL2204498)Show SMILES CC(C)Nc1cc(C(=O)NC2CC3CCC(C2)N3c2ccc(cn2)C(=O)C2CC2)c(C)cc1C(N)=O |TLB:18:17:13.14:11.10.16,9:10:13.14:17| Show InChI InChI=1S/C28H35N5O3/c1-15(2)31-24-13-22(16(3)10-23(24)27(29)35)28(36)32-19-11-20-7-8-21(12-19)33(20)25-9-6-18(14-30-25)26(34)17-4-5-17/h6,9-10,13-15,17,19-21,31H,4-5,7-8,11-12H2,1-3H3,(H2,29,35)(H,32,36) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis Inc
Curated by ChEMBL
| Assay Description
Displacement of biotinylated geldanamycin from human His-tagged Hsp90 by TR-FRET assay |
Bioorg Med Chem Lett 22: 5396-404 (2012)
Article DOI: 10.1016/j.bmcl.2012.07.052
BindingDB Entry DOI: 10.7270/Q25M68KK |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50486032
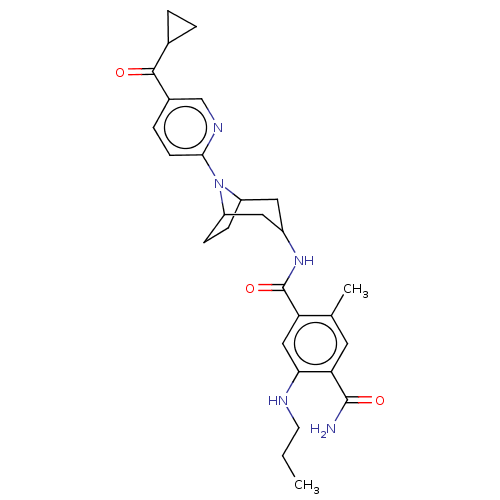
(CHEMBL2204497)Show SMILES CCCNc1cc(C(=O)NC2CC3CCC(C2)N3c2ccc(cn2)C(=O)C2CC2)c(C)cc1C(N)=O |TLB:18:17:13.14:11.10.16,9:10:13.14:17| Show InChI InChI=1S/C28H35N5O3/c1-3-10-30-24-14-22(16(2)11-23(24)27(29)35)28(36)32-19-12-20-7-8-21(13-19)33(20)25-9-6-18(15-31-25)26(34)17-4-5-17/h6,9,11,14-15,17,19-21,30H,3-5,7-8,10,12-13H2,1-2H3,(H2,29,35)(H,32,36) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis Inc
Curated by ChEMBL
| Assay Description
Displacement of biotinylated geldanamycin from human His-tagged Hsp90 by TR-FRET assay |
Bioorg Med Chem Lett 22: 5396-404 (2012)
Article DOI: 10.1016/j.bmcl.2012.07.052
BindingDB Entry DOI: 10.7270/Q25M68KK |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50486033

(CHEMBL2204502)Show SMILES CC[C@@H](C)Nc1cc(C(=O)NC2CC3CCC(C2)N3c2ccc(cn2)C(=O)C2CC2)c(C)cc1C(N)=O |r,TLB:19:18:14.15:12.11.17,10:11:14.15:18| Show InChI InChI=1S/C29H37N5O3/c1-4-17(3)32-25-14-23(16(2)11-24(25)28(30)36)29(37)33-20-12-21-8-9-22(13-20)34(21)26-10-7-19(15-31-26)27(35)18-5-6-18/h7,10-11,14-15,17-18,20-22,32H,4-6,8-9,12-13H2,1-3H3,(H2,30,36)(H,33,37)/t17-,20?,21?,22?/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis Inc
Curated by ChEMBL
| Assay Description
Displacement of biotinylated geldanamycin from human His-tagged Hsp90 by TR-FRET assay |
Bioorg Med Chem Lett 22: 5396-404 (2012)
Article DOI: 10.1016/j.bmcl.2012.07.052
BindingDB Entry DOI: 10.7270/Q25M68KK |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50486041

(CHEMBL2204500)Show SMILES Cc1cc(C(N)=O)c(NCC2CC2)cc1C(=O)NC1CC2CCC(C1)N2c1ccc(cn1)C(=O)C1CC1 |TLB:26:25:21.22:19.18.24,17:18:21.22:25| Show InChI InChI=1S/C29H35N5O3/c1-16-10-24(28(30)36)25(31-14-17-2-3-17)13-23(16)29(37)33-20-11-21-7-8-22(12-20)34(21)26-9-6-19(15-32-26)27(35)18-4-5-18/h6,9-10,13,15,17-18,20-22,31H,2-5,7-8,11-12,14H2,1H3,(H2,30,36)(H,33,37) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis Inc
Curated by ChEMBL
| Assay Description
Displacement of biotinylated geldanamycin from human His-tagged Hsp90 by TR-FRET assay |
Bioorg Med Chem Lett 22: 5396-404 (2012)
Article DOI: 10.1016/j.bmcl.2012.07.052
BindingDB Entry DOI: 10.7270/Q25M68KK |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50486036
(CHEMBL2204496)Show SMILES CCC(CC)Nc1cc(C(=O)NC2CC3CCC(C2)N3c2ccc(cn2)C(=O)C2CC2)c(C)cc1C(N)=O |TLB:20:19:15.16:13.12.18,11:12:15.16:19| Show InChI InChI=1S/C30H39N5O3/c1-4-20(5-2)33-26-15-24(17(3)12-25(26)29(31)37)30(38)34-21-13-22-9-10-23(14-21)35(22)27-11-8-19(16-32-27)28(36)18-6-7-18/h8,11-12,15-16,18,20-23,33H,4-7,9-10,13-14H2,1-3H3,(H2,31,37)(H,34,38) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis Inc
Curated by ChEMBL
| Assay Description
Displacement of biotinylated geldanamycin from human His-tagged Hsp90 by TR-FRET assay |
Bioorg Med Chem Lett 22: 5396-404 (2012)
Article DOI: 10.1016/j.bmcl.2012.07.052
BindingDB Entry DOI: 10.7270/Q25M68KK |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393355
(CHEMBL2152146)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(=O)(=O)Nc1ccc(Cl)cc1 Show InChI InChI=1S/C18H15Cl2N5O3S/c1-22-18(26)16-17(21)23-9-14(24-16)10-2-7-13(20)15(8-10)29(27,28)25-12-5-3-11(19)4-6-12/h2-9,25H,1H3,(H2,21,23)(H,22,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393340

(CHEMBL2152132)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(N)(=O)=O Show InChI InChI=1S/C12H12ClN5O3S/c1-16-12(19)10-11(14)17-5-8(18-10)6-2-3-7(13)9(4-6)22(15,20)21/h2-5H,1H3,(H2,14,17)(H,16,19)(H2,15,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 26 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50486042

(CHEMBL2204499)Show SMILES Cc1cc(C(N)=O)c(NC2CCCC2)cc1C(=O)NC1CC2CCC(C1)N2c1ccc(cn1)C(=O)C1CC1 |TLB:27:26:22.23:20.19.25,18:19:22.23:26| Show InChI InChI=1S/C30H37N5O3/c1-17-12-25(29(31)37)26(33-20-4-2-3-5-20)15-24(17)30(38)34-21-13-22-9-10-23(14-21)35(22)27-11-8-19(16-32-27)28(36)18-6-7-18/h8,11-12,15-16,18,20-23,33H,2-7,9-10,13-14H2,1H3,(H2,31,37)(H,34,38) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis Inc
Curated by ChEMBL
| Assay Description
Displacement of biotinylated geldanamycin from human His-tagged Hsp90 by TR-FRET assay |
Bioorg Med Chem Lett 22: 5396-404 (2012)
Article DOI: 10.1016/j.bmcl.2012.07.052
BindingDB Entry DOI: 10.7270/Q25M68KK |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50486043

(CHEMBL2204504)Show SMILES CCC(CC)Nc1cc(C(=O)NC2CC3CCC(C2)N3c2ccc(cn2)C(C)=O)c(C)cc1C(N)=O |TLB:20:19:15.16:13.12.18,11:12:15.16:19| Show InChI InChI=1S/C28H37N5O3/c1-5-19(6-2)31-25-14-23(16(3)11-24(25)27(29)35)28(36)32-20-12-21-8-9-22(13-20)33(21)26-10-7-18(15-30-26)17(4)34/h7,10-11,14-15,19-22,31H,5-6,8-9,12-13H2,1-4H3,(H2,29,35)(H,32,36) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis Inc
Curated by ChEMBL
| Assay Description
Displacement of biotinylated geldanamycin from human His-tagged Hsp90 by TR-FRET assay |
Bioorg Med Chem Lett 22: 5396-404 (2012)
Article DOI: 10.1016/j.bmcl.2012.07.052
BindingDB Entry DOI: 10.7270/Q25M68KK |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393351
(CHEMBL2152142)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(=O)(=O)Nc1cccc(OC)c1 Show InChI InChI=1S/C19H18ClN5O4S/c1-22-19(26)17-18(21)23-10-15(24-17)11-6-7-14(20)16(8-11)30(27,28)25-12-4-3-5-13(9-12)29-2/h3-10,25H,1-2H3,(H2,21,23)(H,22,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393329
(CHEMBL2152253)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(=O)(=O)Nc1cccc(F)c1C Show InChI InChI=1S/C19H17ClFN5O3S/c1-10-13(21)4-3-5-14(10)26-30(28,29)16-8-11(6-7-12(16)20)15-9-24-18(22)17(25-15)19(27)23-2/h3-9,26H,1-2H3,(H2,22,24)(H,23,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50486037
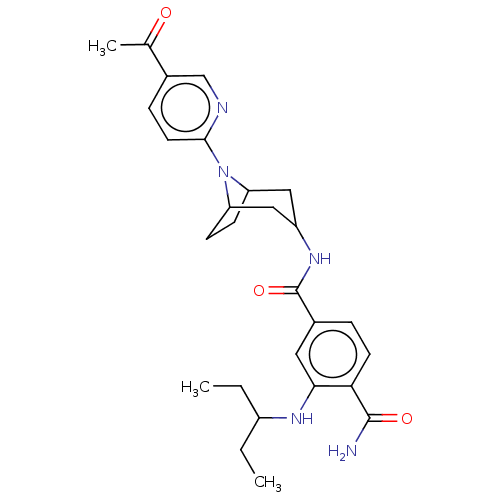
(CHEMBL2204503)Show SMILES CCC(CC)Nc1cc(ccc1C(N)=O)C(=O)NC1CC2CCC(C1)N2c1ccc(cn1)C(C)=O |TLB:26:25:21.22:19.18.24,17:18:21.22:25| Show InChI InChI=1S/C27H35N5O3/c1-4-19(5-2)30-24-12-17(6-10-23(24)26(28)34)27(35)31-20-13-21-8-9-22(14-20)32(21)25-11-7-18(15-29-25)16(3)33/h6-7,10-12,15,19-22,30H,4-5,8-9,13-14H2,1-3H3,(H2,28,34)(H,31,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis Inc
Curated by ChEMBL
| Assay Description
Displacement of biotinylated geldanamycin from human His-tagged Hsp90 by TR-FRET assay |
Bioorg Med Chem Lett 22: 5396-404 (2012)
Article DOI: 10.1016/j.bmcl.2012.07.052
BindingDB Entry DOI: 10.7270/Q25M68KK |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50393333

(CHEMBL2151050)Show SMILES COc1ccc(cc1)-c1nccnc1N1CCN(CC1)S(=O)(=O)c1cccc(c1)-c1cnc(N)nc1 Show InChI InChI=1S/C25H25N7O3S/c1-35-21-7-5-18(6-8-21)23-24(28-10-9-27-23)31-11-13-32(14-12-31)36(33,34)22-4-2-3-19(15-22)20-16-29-25(26)30-17-20/h2-10,15-17H,11-14H2,1H3,(H2,26,29,30) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kalpha expressed in sf9 cells coexpressing p85alpha assessed as amount of ATP consumed by luciferase-luciferin chemiluminescen... |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform
(Homo sapiens (Human)) | BDBM50393365

(CHEMBL2152257)Show SMILES Nc1ncc(-c2ccc(Cl)c(c2)S(=O)(=O)Nc2cccc(F)c2F)c(OCC(F)(F)F)n1 Show InChI InChI=1S/C18H12ClF5N4O3S/c19-11-5-4-9(10-7-26-17(25)27-16(10)31-8-18(22,23)24)6-14(11)32(29,30)28-13-3-1-2-12(20)15(13)21/h1-7,28H,8H2,(H2,25,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kbeta expressed in sf9 cells coexpressing p85alpha assessed as amount of ATP consumed by luciferase-luciferin chemiluminescenc... |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50393338
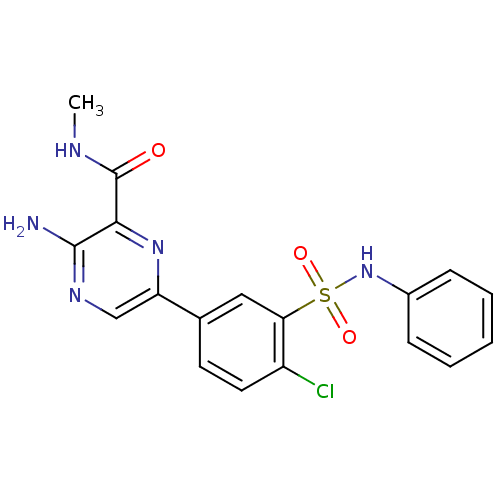
(CHEMBL2152130)Show SMILES CNC(=O)c1nc(cnc1N)-c1ccc(Cl)c(c1)S(=O)(=O)Nc1ccccc1 Show InChI InChI=1S/C18H16ClN5O3S/c1-21-18(25)16-17(20)22-10-14(23-16)11-7-8-13(19)15(9-11)28(26,27)24-12-5-3-2-4-6-12/h2-10,24H,1H3,(H2,20,22)(H,21,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis
Curated by ChEMBL
| Assay Description
Inhibition of human PI3Kgamma expressed in sf9 cells assessed as amount of ATP consumed by luciferase-luciferin chemiluminescence assay |
J Med Chem 55: 5467-82 (2012)
Article DOI: 10.1021/jm300403a
BindingDB Entry DOI: 10.7270/Q2DF6S9N |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-alpha/90-beta
(Homo sapiens (Human)) | BDBM50486038

(CHEMBL2204525)Show SMILES CC(=O)c1ccc(nc1)N1C2CCC1CC(C2)NC(=O)c1ccc(C(N)=O)c(NCC2CC2)c1 |TLB:6:9:11.12:16.15.14,THB:17:15:11.12:9| Show InChI InChI=1S/C26H31N5O3/c1-15(32)18-5-9-24(29-14-18)31-20-6-7-21(31)12-19(11-20)30-26(34)17-4-8-22(25(27)33)23(10-17)28-13-16-2-3-16/h4-5,8-10,14,16,19-21,28H,2-3,6-7,11-13H2,1H3,(H2,27,33)(H,30,34) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Exelixis Inc
Curated by ChEMBL
| Assay Description
Displacement of biotinylated geldanamycin from human His-tagged Hsp90 by TR-FRET assay |
Bioorg Med Chem Lett 22: 5396-404 (2012)
Article DOI: 10.1016/j.bmcl.2012.07.052
BindingDB Entry DOI: 10.7270/Q25M68KK |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355869
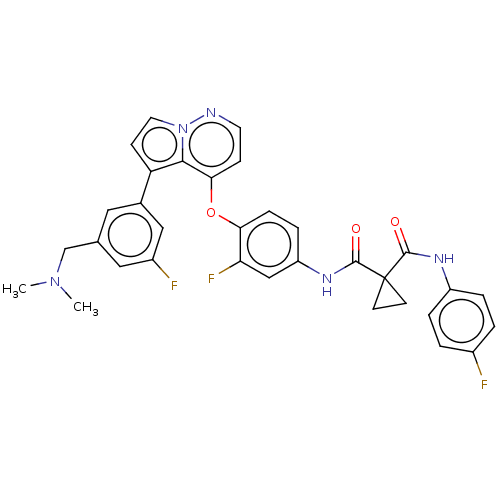
(N-(4-((5-(3-((dimethylamino)methyl)-5-fluorophenyl...)Show SMILES CN(C)Cc1cc(F)cc(c1)-c1ccn2nccc(Oc3ccc(NC(=O)C4(CC4)C(=O)Nc4ccc(F)cc4)cc3F)c12 Show InChI InChI=1S/C33H28F3N5O3/c1-40(2)19-20-15-21(17-23(35)16-20)26-10-14-41-30(26)29(9-13-37-41)44-28-8-7-25(18-27(28)36)39-32(43)33(11-12-33)31(42)38-24-5-3-22(34)4-6-24/h3-10,13-18H,11-12,19H2,1-2H3,(H,38,42)(H,39,43) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355855

(N-(3-fluoro-4-((5-(3-hydroxymethyl)phenyl)pyrrolo[...)Show SMILES OCc1cccc(c1)-c1ccn2nccc(Oc3ccc(NC(=O)c4cccn(-c5ccc(F)cc5)c4=O)cc3F)c12 Show InChI InChI=1S/C32H22F2N4O4/c33-22-6-9-24(10-7-22)37-15-2-5-26(32(37)41)31(40)36-23-8-11-28(27(34)18-23)42-29-12-14-35-38-16-13-25(30(29)38)21-4-1-3-20(17-21)19-39/h1-18,39H,19H2,(H,36,40) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355875
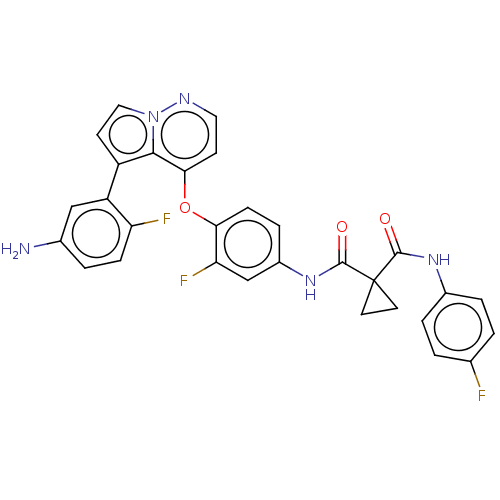
(N-(4-((5-(5-amino-2-fluoro)-5-fluorophenyl)pyrrolo...)Show SMILES Nc1ccc(F)c(c1)-c1ccn2nccc(Oc3ccc(NC(=O)C4(CC4)C(=O)Nc4ccc(F)cc4)cc3F)c12 Show InChI InChI=1S/C30H22F3N5O3/c31-17-1-4-19(5-2-17)36-28(39)30(11-12-30)29(40)37-20-6-8-25(24(33)16-20)41-26-9-13-35-38-14-10-21(27(26)38)22-15-18(34)3-7-23(22)32/h1-10,13-16H,11-12,34H2,(H,36,39)(H,37,40) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355874

(N-(4-((5-(3-amino-4-methoxy)-5-fluorophenyl)pyrrol...)Show SMILES COc1ccc(cc1N)-c1ccn2nccc(Oc3ccc(NC(=O)C4(CC4)C(=O)Nc4ccc(F)cc4)cc3F)c12 Show InChI InChI=1S/C31H25F2N5O4/c1-41-26-8-2-18(16-24(26)34)22-11-15-38-28(22)27(10-14-35-38)42-25-9-7-21(17-23(25)33)37-30(40)31(12-13-31)29(39)36-20-5-3-19(32)4-6-20/h2-11,14-17H,12-13,34H2,1H3,(H,36,39)(H,37,40) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355873

(N-(4-((5-(3-((ethylamino)methyl)phenyl)pyrrolo[1,2...)Show SMILES CCNCc1cccc(c1)-c1ccn2nccc(Oc3ccc(NC(=O)C4(CC4)C(=O)Nc4ccc(F)cc4)cc3F)c12 Show InChI InChI=1S/C33H29F2N5O3/c1-2-36-20-21-4-3-5-22(18-21)26-13-17-40-30(26)29(12-16-37-40)43-28-11-10-25(19-27(28)35)39-32(42)33(14-15-33)31(41)38-24-8-6-23(34)7-9-24/h3-13,16-19,36H,2,14-15,20H2,1H3,(H,38,41)(H,39,42) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355872
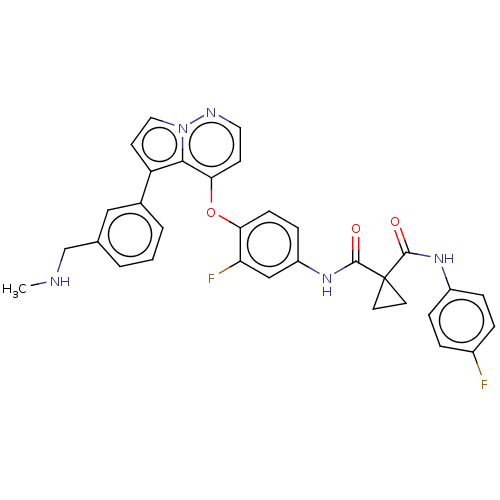
(N-(3-fluoro-4-((5-(3-((methylamino)methyl)phenyl)p...)Show SMILES CNCc1cccc(c1)-c1ccn2nccc(Oc3ccc(NC(=O)C4(CC4)C(=O)Nc4ccc(F)cc4)cc3F)c12 Show InChI InChI=1S/C32H27F2N5O3/c1-35-19-20-3-2-4-21(17-20)25-12-16-39-29(25)28(11-15-36-39)42-27-10-9-24(18-26(27)34)38-31(41)32(13-14-32)30(40)37-23-7-5-22(33)6-8-23/h2-12,15-18,35H,13-14,19H2,1H3,(H,37,40)(H,38,41) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355871
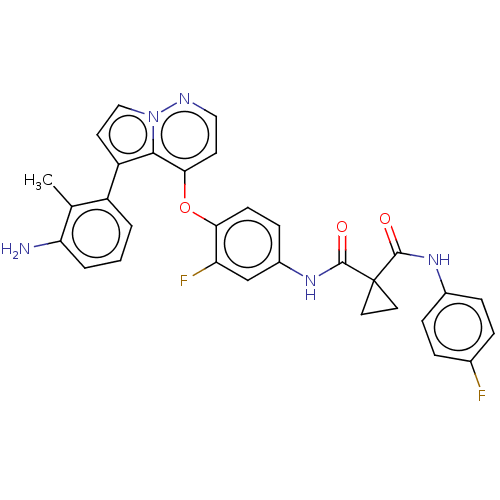
(N-(4-((5-(3-amino-2-methyl)-5-fluorophenyl)pyrrolo...)Show SMILES Cc1c(N)cccc1-c1ccn2nccc(Oc3ccc(NC(=O)C4(CC4)C(=O)Nc4ccc(F)cc4)cc3F)c12 Show InChI InChI=1S/C31H25F2N5O3/c1-18-22(3-2-4-25(18)34)23-12-16-38-28(23)27(11-15-35-38)41-26-10-9-21(17-24(26)33)37-30(40)31(13-14-31)29(39)36-20-7-5-19(32)6-8-20/h2-12,15-17H,13-14,34H2,1H3,(H,36,39)(H,37,40) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355870
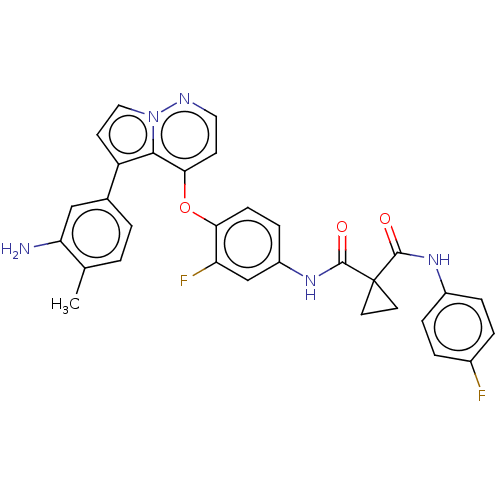
(N-(4-((5-(3-amino-4-methyl)-5-fluorophenyl)pyrrolo...)Show SMILES Cc1ccc(cc1N)-c1ccn2nccc(Oc3ccc(NC(=O)C4(CC4)C(=O)Nc4ccc(F)cc4)cc3F)c12 Show InChI InChI=1S/C31H25F2N5O3/c1-18-2-3-19(16-25(18)34)23-11-15-38-28(23)27(10-14-35-38)41-26-9-8-22(17-24(26)33)37-30(40)31(12-13-31)29(39)36-21-6-4-20(32)5-7-21/h2-11,14-17H,12-13,34H2,1H3,(H,36,39)(H,37,40) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355868
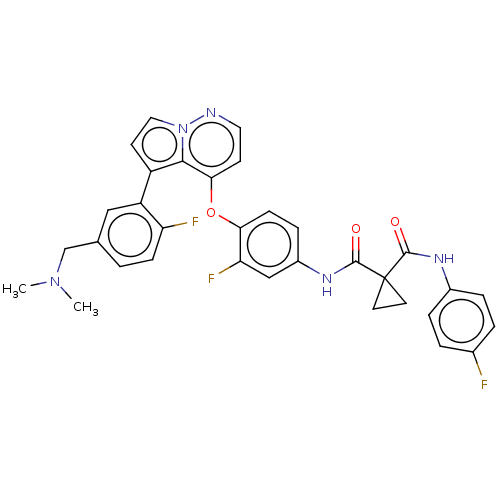
(N-(4-((5-(5-((dimethylamino)methyl)-2-fluorophenyl...)Show SMILES CN(C)Cc1ccc(F)c(c1)-c1ccn2nccc(Oc3ccc(NC(=O)C4(CC4)C(=O)Nc4ccc(F)cc4)cc3F)c12 Show InChI InChI=1S/C33H28F3N5O3/c1-40(2)19-20-3-9-26(35)25(17-20)24-12-16-41-30(24)29(11-15-37-41)44-28-10-8-23(18-27(28)36)39-32(43)33(13-14-33)31(42)38-22-6-4-21(34)5-7-22/h3-12,15-18H,13-14,19H2,1-2H3,(H,38,42)(H,39,43) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355876
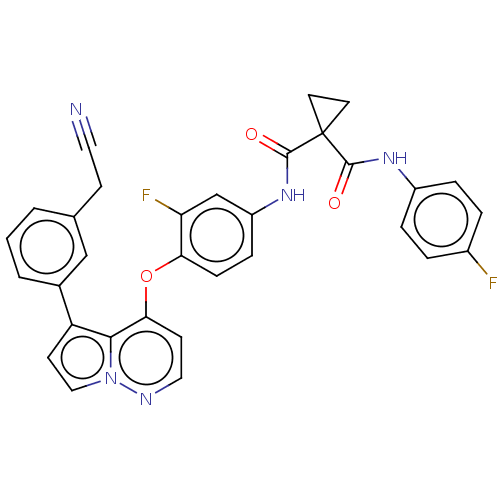
(N-(4-((5-(3-cyanomethylphenyl)pyrrolo[1,2-b]pyrida...)Show SMILES Fc1ccc(NC(=O)C2(CC2)C(=O)Nc2ccc(Oc3ccnn4ccc(-c5cccc(CC#N)c5)c34)c(F)c2)cc1 Show InChI InChI=1S/C32H23F2N5O3/c33-22-4-6-23(7-5-22)37-30(40)32(13-14-32)31(41)38-24-8-9-27(26(34)19-24)42-28-11-16-36-39-17-12-25(29(28)39)21-3-1-2-20(18-21)10-15-35/h1-9,11-12,16-19H,10,13-14H2,(H,37,40)(H,38,41) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355853

(4-ethoxy-N-(3-fluoro-4-((5-pyridin-4-yl)pyrrolo[1,...)Show SMILES CCOc1ccn(-c2ccc(F)cc2)c(=O)c1C(=O)Nc1ccc(Oc2ccnn3ccc(-c4ccncc4)c23)c(F)c1 Show InChI InChI=1S/C32H23F2N5O4/c1-2-42-27-13-17-38(23-6-3-21(33)4-7-23)32(41)29(27)31(40)37-22-5-8-26(25(34)19-22)43-28-11-16-36-39-18-12-24(30(28)39)20-9-14-35-15-10-20/h3-19H,2H2,1H3,(H,37,40) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
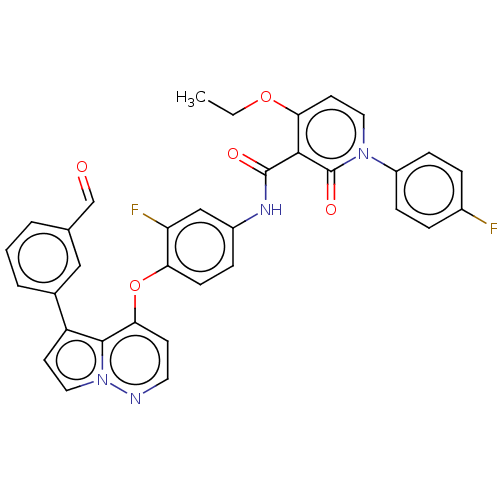
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355854
(4-ethoxy-N-(3-fluoro-4-((5-(3-formylphenyl)pyrrolo...)Show SMILES CCOc1ccn(-c2ccc(F)cc2)c(=O)c1C(=O)Nc1ccc(Oc2ccnn3ccc(-c4cccc(C=O)c4)c23)c(F)c1 Show InChI InChI=1S/C34H24F2N4O5/c1-2-44-29-14-16-39(25-9-6-23(35)7-10-25)34(43)31(29)33(42)38-24-8-11-28(27(36)19-24)45-30-12-15-37-40-17-13-26(32(30)40)22-5-3-4-21(18-22)20-41/h3-20H,2H2,1H3,(H,38,42) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355856

(4-ethoxy-N-(3-fluoro-4-((5-(3-(2-hydroxypropan-2-y...)Show SMILES CCOc1ccn(-c2ccc(F)cc2)c(=O)c1C(=O)Nc1ccc(Oc2ccnn3ccc(-c4cccc(c4)C(C)(C)O)c23)c(F)c1 Show InChI InChI=1S/C36H30F2N4O5/c1-4-46-30-16-18-41(26-11-8-24(37)9-12-26)35(44)32(30)34(43)40-25-10-13-29(28(38)21-25)47-31-14-17-39-42-19-15-27(33(31)42)22-6-5-7-23(20-22)36(2,3)45/h5-21,45H,4H2,1-3H3,(H,40,43) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355857

(4-ethoxy-N-(3-fluoro-4-((5-(3-(1-hydroxyethyl)phen...)Show SMILES CCOc1ccn(-c2ccc(F)cc2)c(=O)c1C(=O)Nc1ccc(Oc2ccnn3ccc(-c4cccc(c4)C(C)O)c23)c(F)c1 Show InChI InChI=1S/C35H28F2N4O5/c1-3-45-30-15-17-40(26-10-7-24(36)8-11-26)35(44)32(30)34(43)39-25-9-12-29(28(37)20-25)46-31-13-16-38-41-18-14-27(33(31)41)23-6-4-5-22(19-23)21(2)42/h4-21,42H,3H2,1-2H3,(H,39,43) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
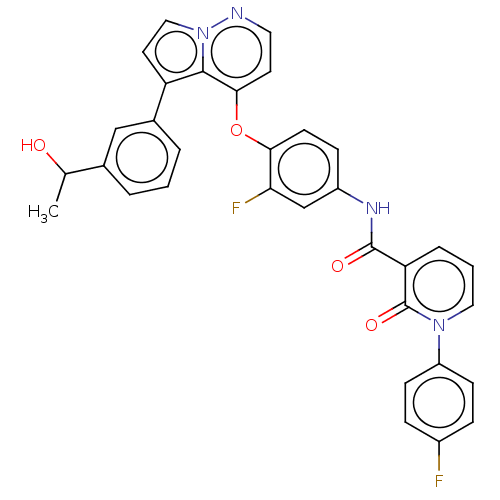
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355858
(N-(3-fluoro-4-((5-(3-(1-hydroxyethyl)phenyl)pyrrol...)Show SMILES CC(O)c1cccc(c1)-c1ccn2nccc(Oc3ccc(NC(=O)c4cccn(-c5ccc(F)cc5)c4=O)cc3F)c12 Show InChI InChI=1S/C33H24F2N4O4/c1-20(40)21-4-2-5-22(18-21)26-14-17-39-31(26)30(13-15-36-39)43-29-12-9-24(19-28(29)35)37-32(41)27-6-3-16-38(33(27)42)25-10-7-23(34)8-11-25/h2-20,40H,1H3,(H,37,41) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
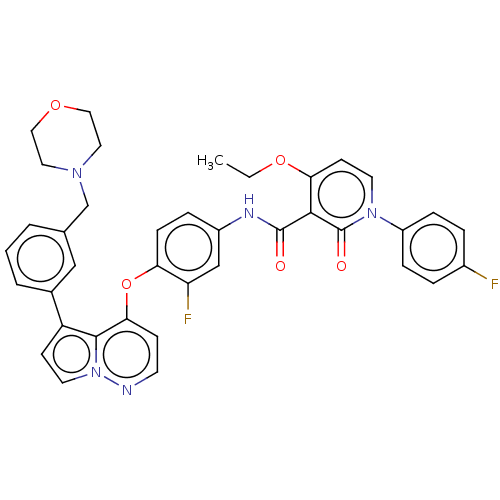
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355859
(4-ethoxy-N-(3-fluoro-4-((5-(pyrimidin-5-yl)pyrrolo...)Show SMILES CCOc1ccn(-c2ccc(F)cc2)c(=O)c1C(=O)Nc1ccc(Oc2ccnn3ccc(-c4cccc(CN5CCOCC5)c4)c23)c(F)c1 Show InChI InChI=1S/C38H33F2N5O5/c1-2-49-33-14-16-44(29-9-6-27(39)7-10-29)38(47)35(33)37(46)42-28-8-11-32(31(40)23-28)50-34-12-15-41-45-17-13-30(36(34)45)26-5-3-4-25(22-26)24-43-18-20-48-21-19-43/h3-17,22-23H,2,18-21,24H2,1H3,(H,42,46) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355860
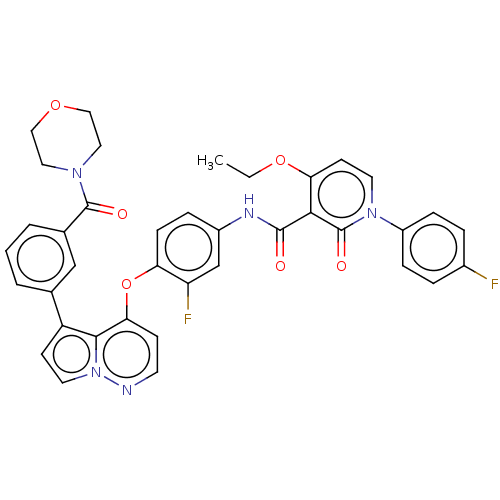
(4-ethoxy-N-(3-fluoro-4-((5-(3-morpholin-4-carbonyl...)Show SMILES CCOc1ccn(-c2ccc(F)cc2)c(=O)c1C(=O)Nc1ccc(Oc2ccnn3ccc(-c4cccc(c4)C(=O)N4CCOCC4)c23)c(F)c1 Show InChI InChI=1S/C38H31F2N5O6/c1-2-50-32-14-16-44(28-9-6-26(39)7-10-28)38(48)34(32)36(46)42-27-8-11-31(30(40)23-27)51-33-12-15-41-45-17-13-29(35(33)45)24-4-3-5-25(22-24)37(47)43-18-20-49-21-19-43/h3-17,22-23H,2,18-21H2,1H3,(H,42,46) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |
Hepatocyte growth factor receptor
(Homo sapiens (Human)) | BDBM355861
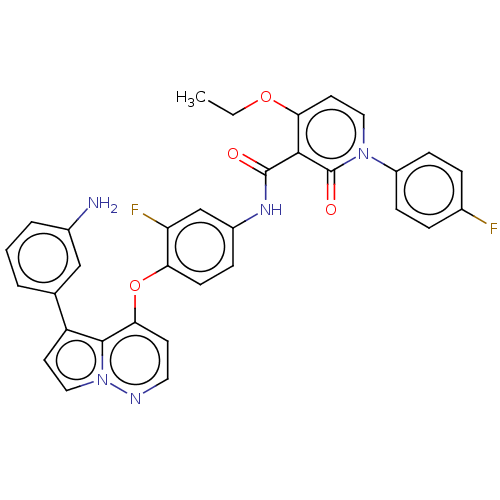
(4-ethoxy-N-(3-fluoro-4-((5-(3-aminophenyl)pyrrolo[...)Show SMILES CCOc1ccn(-c2ccc(F)cc2)c(=O)c1C(=O)Nc1ccc(Oc2ccnn3ccc(-c4cccc(N)c4)c23)c(F)c1 Show InChI InChI=1S/C33H25F2N5O4/c1-2-43-28-14-16-39(24-9-6-21(34)7-10-24)33(42)30(28)32(41)38-23-8-11-27(26(35)19-23)44-29-12-15-37-40-17-13-25(31(29)40)20-4-3-5-22(36)18-20/h3-19H,2,36H2,1H3,(H,38,41) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | <40 | n/a | n/a | n/a | n/a | n/a | n/a |
The Asan Foundation
US Patent
| Assay Description
The inhibitory effect of the compounds of the present invention on the activity of c-Met was confirmed as follows.Specifically, 250 μM G4Y1 pept... |
US Patent US9815840 (2017)
BindingDB Entry DOI: 10.7270/Q2862JQZ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data