 Found 1592 hits with Last Name = 'demartino' and Initial = 'j'
Found 1592 hits with Last Name = 'demartino' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50204512
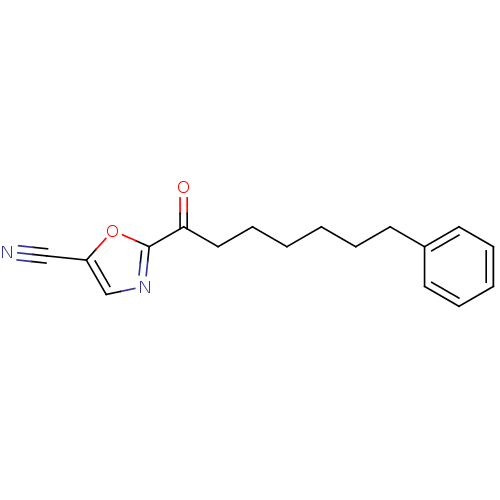
(2-(7-phenylheptanoyl)oxazole-5-carbonitrile | CHEM...)Show InChI InChI=1S/C17H18N2O2/c18-12-15-13-19-17(21-15)16(20)11-7-2-1-4-8-14-9-5-3-6-10-14/h3,5-6,9-10,13H,1-2,4,7-8,11H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247136

(2-(7-phenylheptanoyl)oxazole-4-carbonitrile | CHEM...)Show InChI InChI=1S/C17H18N2O2/c18-12-15-13-21-17(19-15)16(20)11-7-2-1-4-8-14-9-5-3-6-10-14/h3,5-6,9-10,13H,1-2,4,7-8,11H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50204483
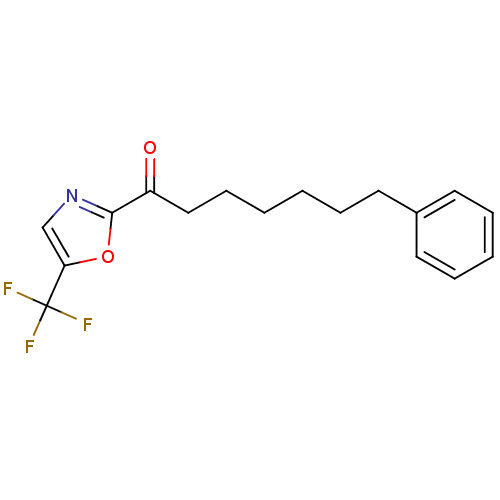
(7-phenyl-1-(5-(trifluoromethyl)oxazol-2-yl)heptan-...)Show InChI InChI=1S/C17H18F3NO2/c18-17(19,20)15-12-21-16(23-15)14(22)11-7-2-1-4-8-13-9-5-3-6-10-13/h3,5-6,9-10,12H,1-2,4,7-8,11H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
C-C chemokine receptor type 5
(Homo sapiens (Human)) | BDBM50088301
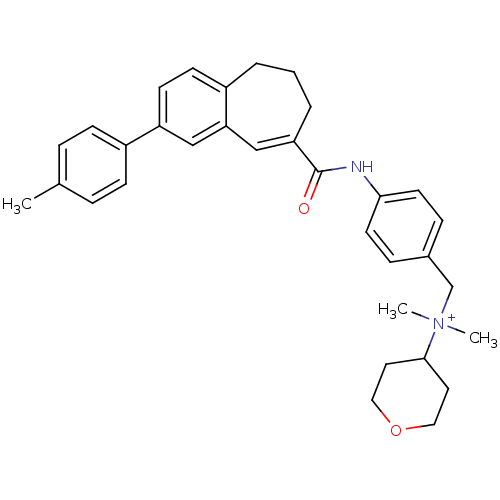
((E)-N,N-dimethyl-N-(4-(2-p-tolyl-6,7-dihydro-5H-be...)Show SMILES Cc1ccc(cc1)-c1ccc2CCCC(=Cc2c1)C(=O)Nc1ccc(C[N+](C)(C)C2CCOCC2)cc1 |c:15| Show InChI InChI=1S/C33H38N2O2/c1-24-7-11-27(12-8-24)28-14-13-26-5-4-6-29(22-30(26)21-28)33(36)34-31-15-9-25(10-16-31)23-35(2,3)32-17-19-37-20-18-32/h7-16,21-22,32H,4-6,17-20,23H2,1-3H3/p+1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against specific binding of [125I]-MIP-1 alpha to human CCR5 receptor |
Bioorg Med Chem Lett 11: 265-70 (2001)
BindingDB Entry DOI: 10.7270/Q2668CFZ |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247103

(7-phenyl-1-(4-(pyridin-4-yl)oxazol-2-yl)heptan-1-o...)Show InChI InChI=1S/C21H22N2O2/c24-20(11-7-2-1-4-8-17-9-5-3-6-10-17)21-23-19(16-25-21)18-12-14-22-15-13-18/h3,5-6,9-10,12-16H,1-2,4,7-8,11H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247106

(2-(7-phenylheptanoyl)oxazole-4-carboxamide | CHEMB...)Show InChI InChI=1S/C17H20N2O3/c18-16(21)14-12-22-17(19-14)15(20)11-7-2-1-4-8-13-9-5-3-6-10-13/h3,5-6,9-10,12H,1-2,4,7-8,11H2,(H2,18,21) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247107

(CHEMBL460493 | N-methyl-2-(7-phenylheptanoyl)oxazo...)Show InChI InChI=1S/C18H22N2O3/c1-19-17(22)15-13-23-18(20-15)16(21)12-8-3-2-5-9-14-10-6-4-7-11-14/h4,6-7,10-11,13H,2-3,5,8-9,12H2,1H3,(H,19,22) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247074
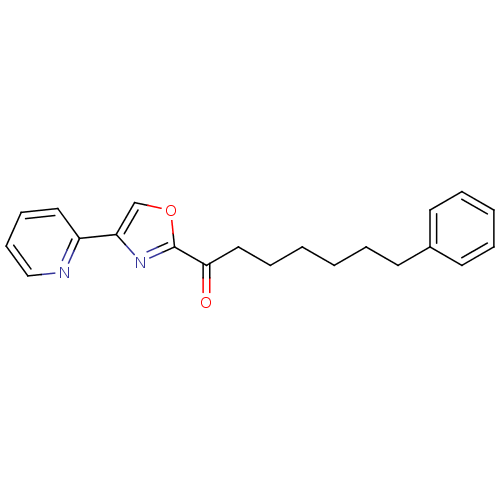
(7-phenyl-1-(4-(pyridin-2-yl)oxazol-2-yl)heptan-1-o...)Show InChI InChI=1S/C21H22N2O2/c24-20(14-7-2-1-4-10-17-11-5-3-6-12-17)21-23-19(16-25-21)18-13-8-9-15-22-18/h3,5-6,8-9,11-13,15-16H,1-2,4,7,10,14H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247139
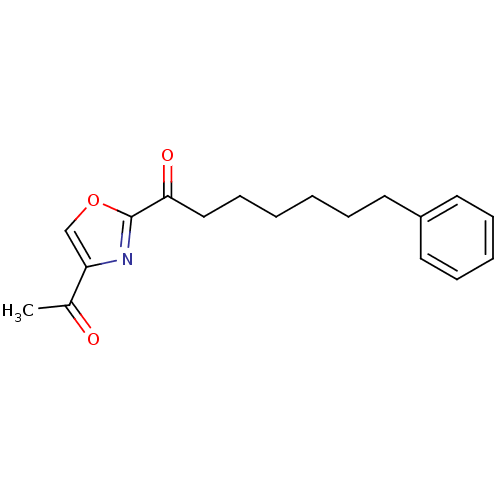
(1-(4-acetyloxazol-2-yl)-7-phenylheptan-1-one | CHE...)Show InChI InChI=1S/C18H21NO3/c1-14(20)16-13-22-18(19-16)17(21)12-8-3-2-5-9-15-10-6-4-7-11-15/h4,6-7,10-11,13H,2-3,5,8-9,12H2,1H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247035

(1-(4-bromooxazol-2-yl)-7-phenylheptan-1-one | CHEM...)Show InChI InChI=1S/C16H18BrNO2/c17-15-12-20-16(18-15)14(19)11-7-2-1-4-8-13-9-5-3-6-10-13/h3,5-6,9-10,12H,1-2,4,7-8,11H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247073
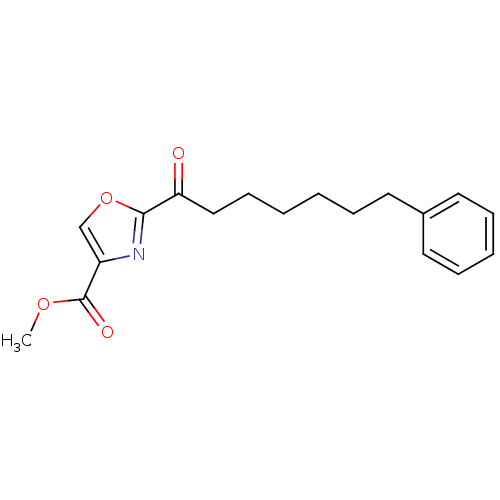
(CHEMBL461539 | methyl 2-(7-phenylheptanoyl)oxazole...)Show InChI InChI=1S/C18H21NO4/c1-22-18(21)15-13-23-17(19-15)16(20)12-8-3-2-5-9-14-10-6-4-7-11-14/h4,6-7,10-11,13H,2-3,5,8-9,12H2,1H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247137
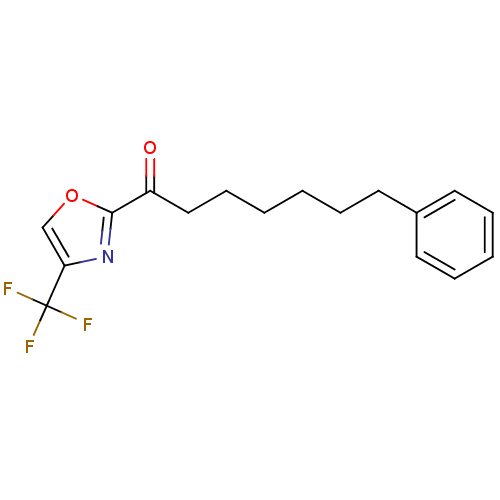
(7-phenyl-1-(4-(trifluoromethyl)oxazol-2-yl)heptan-...)Show InChI InChI=1S/C17H18F3NO2/c18-17(19,20)15-12-23-16(21-15)14(22)11-7-2-1-4-8-13-9-5-3-6-10-13/h3,5-6,9-10,12H,1-2,4,7-8,11H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247036
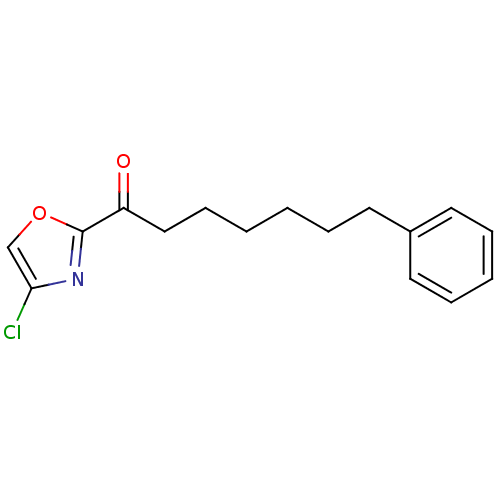
(1-(4-chlorooxazol-2-yl)-7-phenylheptan-1-one | CHE...)Show InChI InChI=1S/C16H18ClNO2/c17-15-12-20-16(18-15)14(19)11-7-2-1-4-8-13-9-5-3-6-10-13/h3,5-6,9-10,12H,1-2,4,7-8,11H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM23120
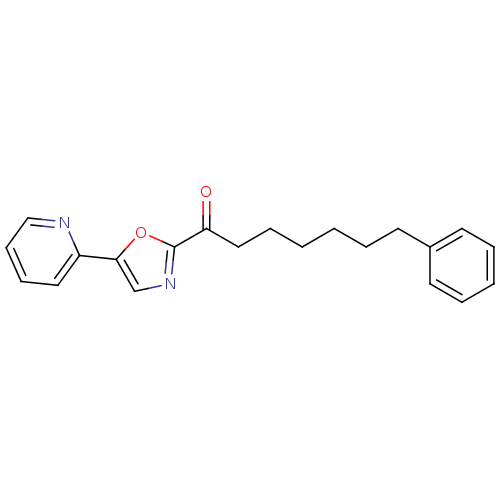
(7-phenyl-1-[5-(pyridin-2-yl)-1,3-oxazol-2-yl]hepta...)Show InChI InChI=1S/C21H22N2O2/c24-19(14-7-2-1-4-10-17-11-5-3-6-12-17)21-23-16-20(25-21)18-13-8-9-15-22-18/h3,5-6,8-9,11-13,15-16H,1-2,4,7,10,14H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 4.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247037
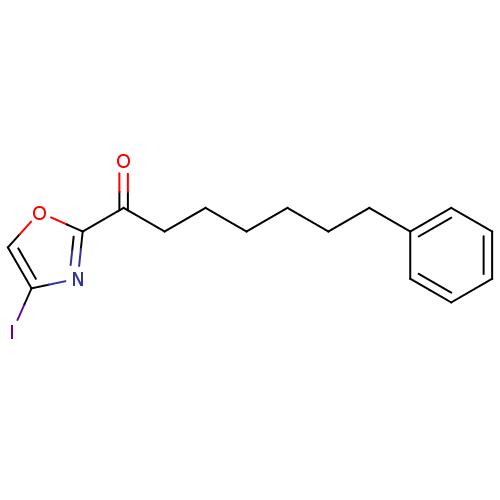
(1-(4-iodooxazol-2-yl)-7-phenylheptan-1-one | CHEMB...)Show InChI InChI=1S/C16H18INO2/c17-15-12-20-16(18-15)14(19)11-7-2-1-4-8-13-9-5-3-6-10-13/h3,5-6,9-10,12H,1-2,4,7-8,11H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 6.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247075
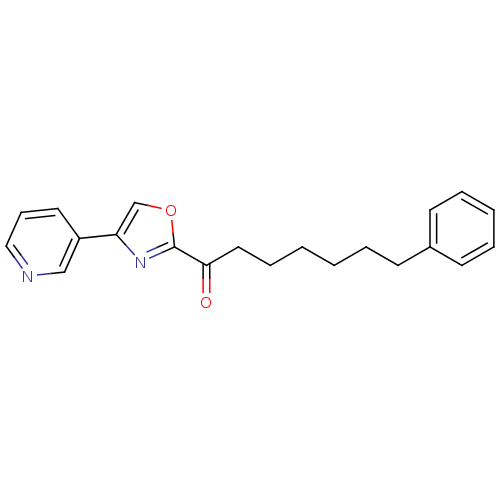
(7-phenyl-1-(4-(pyridin-3-yl)oxazol-2-yl)heptan-1-o...)Show InChI InChI=1S/C21H22N2O2/c24-20(13-7-2-1-4-9-17-10-5-3-6-11-17)21-23-19(16-25-21)18-12-8-14-22-15-18/h3,5-6,8,10-12,14-16H,1-2,4,7,9,13H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247070

(1-(4-(methylthio)oxazol-2-yl)-7-phenylheptan-1-one...)Show InChI InChI=1S/C17H21NO2S/c1-21-16-13-20-17(18-16)15(19)12-8-3-2-5-9-14-10-6-4-7-11-14/h4,6-7,10-11,13H,2-3,5,8-9,12H2,1H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 29 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24693
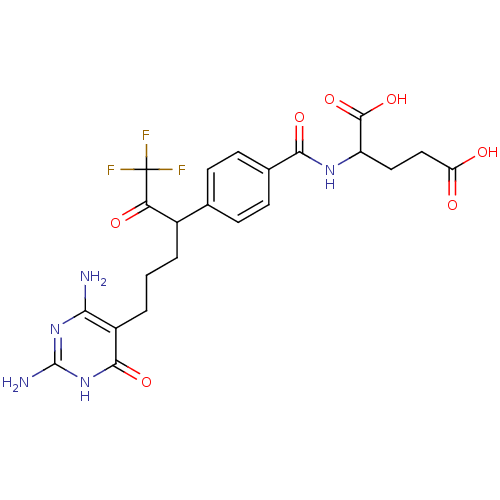
(10-CF3CO-DDACTHF (5) | 10-CF3CO-DDACTHF, 2 | 2-({4...)Show SMILES Nc1nc(N)c(CCCC(C(=O)C(F)(F)F)c2ccc(cc2)C(=O)NC(CCC(O)=O)C(O)=O)c(=O)[nH]1 Show InChI InChI=1S/C22H24F3N5O7/c23-22(24,25)16(33)12(2-1-3-13-17(26)29-21(27)30-19(13)35)10-4-6-11(7-5-10)18(34)28-14(20(36)37)8-9-15(31)32/h4-7,12,14H,1-3,8-9H2,(H,28,34)(H,31,32)(H,36,37)(H5,26,27,29,30,35) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
| Assay Description
Enzyme activity assays of recombinant hGAR Tfase and recombinant hAICAR Tfase were performed as previously described. Kinetics of the enzyme reaction... |
Biochemistry 52: 5133-44 (2013)
Article DOI: 10.1021/bi4005182
BindingDB Entry DOI: 10.7270/Q2K93669 |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24693

(10-CF3CO-DDACTHF (5) | 10-CF3CO-DDACTHF, 2 | 2-({4...)Show SMILES Nc1nc(N)c(CCCC(C(=O)C(F)(F)F)c2ccc(cc2)C(=O)NC(CCC(O)=O)C(O)=O)c(=O)[nH]1 Show InChI InChI=1S/C22H24F3N5O7/c23-22(24,25)16(33)12(2-1-3-13-17(26)29-21(27)30-19(13)35)10-4-6-11(7-5-10)18(34)28-14(20(36)37)8-9-15(31)32/h4-7,12,14H,1-3,8-9H2,(H,28,34)(H,31,32)(H,36,37)(H5,26,27,29,30,35) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 30 | -43.1 | n/a | n/a | n/a | n/a | n/a | 7.5 | 26 |
The Scripps Research Institute
| Assay Description
Assays were initiated by the addition of GAR to the reaction mixture containing GAR Tfase, test compounds, and cofactor. The assay monitors the defor... |
J Med Chem 51: 5441-8 (2008)
Article DOI: 10.1021/jm800555h
BindingDB Entry DOI: 10.7270/Q2WD3XVB |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24693

(10-CF3CO-DDACTHF (5) | 10-CF3CO-DDACTHF, 2 | 2-({4...)Show SMILES Nc1nc(N)c(CCCC(C(=O)C(F)(F)F)c2ccc(cc2)C(=O)NC(CCC(O)=O)C(O)=O)c(=O)[nH]1 Show InChI InChI=1S/C22H24F3N5O7/c23-22(24,25)16(33)12(2-1-3-13-17(26)29-21(27)30-19(13)35)10-4-6-11(7-5-10)18(34)28-14(20(36)37)8-9-15(31)32/h4-7,12,14H,1-3,8-9H2,(H,28,34)(H,31,32)(H,36,37)(H5,26,27,29,30,35) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant GAR Tfase |
J Med Chem 49: 2998-3002 (2006)
Article DOI: 10.1021/jm0601147
BindingDB Entry DOI: 10.7270/Q2VX0H9X |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50204506
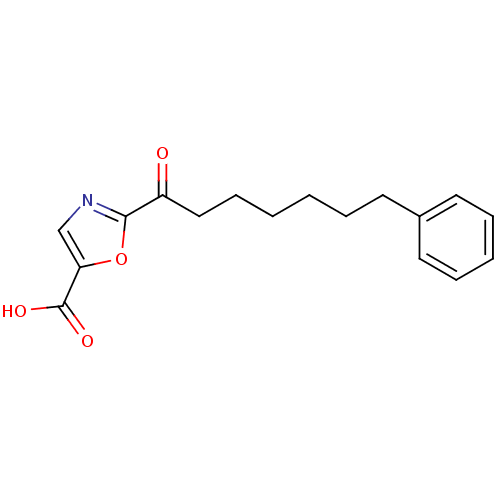
(2-(7-phenylheptanoyl)oxazole-5-carboxylic acid | C...)Show InChI InChI=1S/C17H19NO4/c19-14(16-18-12-15(22-16)17(20)21)11-7-2-1-4-8-13-9-5-3-6-10-13/h3,5-6,9-10,12H,1-2,4,7-8,11H2,(H,20,21) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247108
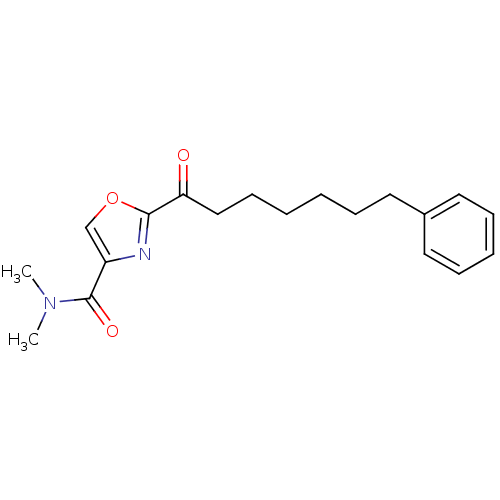
(CHEMBL462589 | N,N-dimethyl-2-(7-phenylheptanoyl)o...)Show InChI InChI=1S/C19H24N2O3/c1-21(2)19(23)16-14-24-18(20-16)17(22)13-9-4-3-6-10-15-11-7-5-8-12-15/h5,7-8,11-12,14H,3-4,6,9-10,13H2,1-2H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50204493
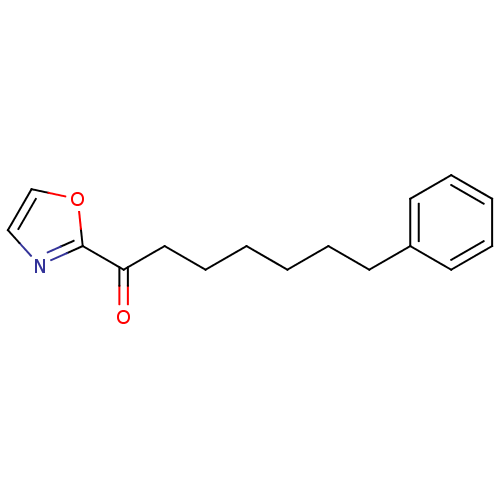
(1-(oxazol-2-yl)-7-phenylheptan-1-one | CHEMBL22012...)Show InChI InChI=1S/C16H19NO2/c18-15(16-17-12-13-19-16)11-7-2-1-4-8-14-9-5-3-6-10-14/h3,5-6,9-10,12-13H,1-2,4,7-8,11H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 48 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247105
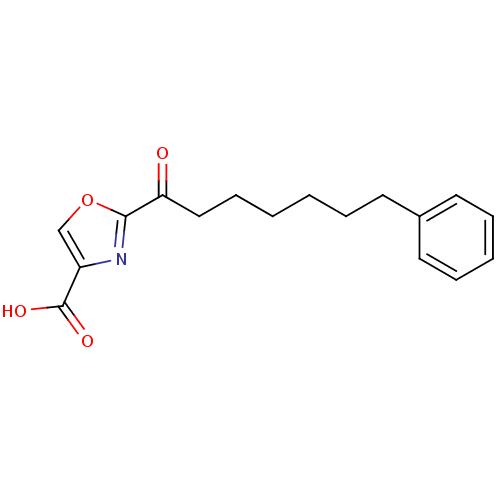
(2-(7-phenylheptanoyl)oxazole-4-carboxylic acid | C...)Show InChI InChI=1S/C17H19NO4/c19-15(16-18-14(12-22-16)17(20)21)11-7-2-1-4-8-13-9-5-3-6-10-13/h3,5-6,9-10,12H,1-2,4,7-8,11H2,(H,20,21) | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 53 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247071

(2-(7-phenylheptanoyl)oxazole-4-carbaldehyde | CHEM...)Show InChI InChI=1S/C17H19NO3/c19-12-15-13-21-17(18-15)16(20)11-7-2-1-4-8-14-9-5-3-6-10-14/h3,5-6,9-10,12-13H,1-2,4,7-8,11H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM50005518
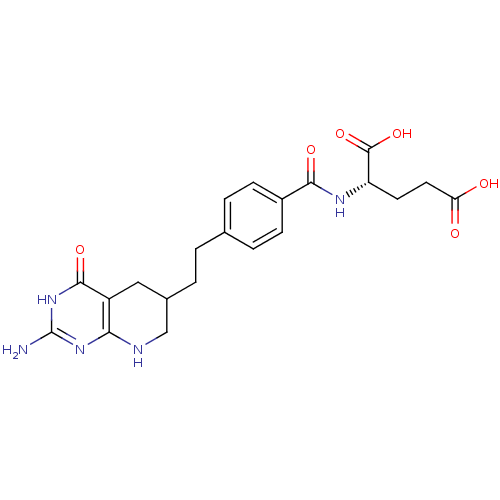
((S)-2-(4-(2-((R)-2-amino-4-oxo-1,4,5,6,7,8-hexahyd...)Show SMILES Nc1nc2NCC(CCc3ccc(cc3)C(=O)N[C@@H](CCC(O)=O)C(O)=O)Cc2c(=O)[nH]1 |r| Show InChI InChI=1S/C21H25N5O6/c22-21-25-17-14(19(30)26-21)9-12(10-23-17)2-1-11-3-5-13(6-4-11)18(29)24-15(20(31)32)7-8-16(27)28/h3-6,12,15H,1-2,7-10H2,(H,24,29)(H,27,28)(H,31,32)(H4,22,23,25,26,30)/t12?,15-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant GAR Tfase |
J Med Chem 49: 2998-3002 (2006)
Article DOI: 10.1021/jm0601147
BindingDB Entry DOI: 10.7270/Q2VX0H9X |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM22590
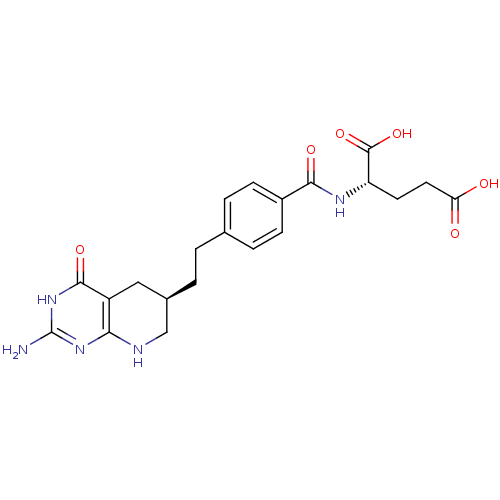
((2S)-2-[(4-{2-[(6R)-2-amino-4-oxo-1H,4H,5H,6H,7H,8...)Show SMILES Nc1nc2NC[C@H](CCc3ccc(cc3)C(=O)N[C@@H](CCC(O)=O)C(O)=O)Cc2c(=O)[nH]1 Show InChI InChI=1S/C21H25N5O6/c22-21-25-17-14(19(30)26-21)9-12(10-23-17)2-1-11-3-5-13(6-4-11)18(29)24-15(20(31)32)7-8-16(27)28/h3-6,12,15H,1-2,7-10H2,(H,24,29)(H,27,28)(H,31,32)(H4,22,23,25,26,30)/t12-,15+/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 60 | -41.4 | n/a | n/a | n/a | n/a | n/a | 7.5 | 26 |
The Scripps Research Institute
| Assay Description
Assays were initiated by the addition of GAR to the reaction mixture containing GAR Tfase, test compounds, and cofactor. The assay monitors the defor... |
J Med Chem 51: 5441-8 (2008)
Article DOI: 10.1021/jm800555h
BindingDB Entry DOI: 10.7270/Q2WD3XVB |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247104

(7-phenyl-1-(4-phenyloxazol-2-yl)heptan-1-one | CHE...)Show InChI InChI=1S/C22H23NO2/c24-21(16-10-2-1-5-11-18-12-6-3-7-13-18)22-23-20(17-25-22)19-14-8-4-9-15-19/h3-4,6-9,12-15,17H,1-2,5,10-11,16H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM50186739
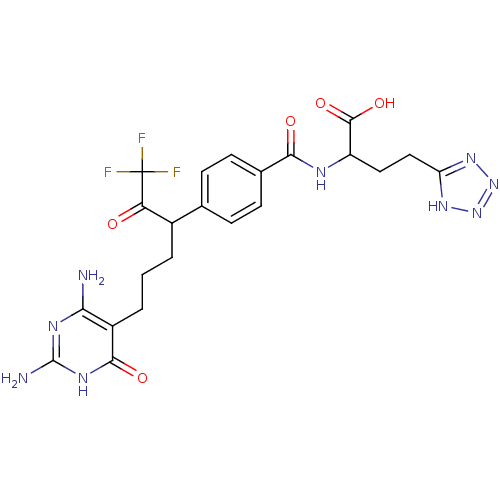
((2S)-2-(4-(6-(2,4-diamino-6-oxo-1,6-dihydropyrimid...)Show SMILES Nc1nc(N)c(CCCC(C(=O)C(F)(F)F)c2ccc(cc2)C(=O)NC(CCc2nnn[nH]2)C(O)=O)c(=O)[nH]1 Show InChI InChI=1S/C22H24F3N9O5/c23-22(24,25)16(35)12(2-1-3-13-17(26)29-21(27)30-19(13)37)10-4-6-11(7-5-10)18(36)28-14(20(38)39)8-9-15-31-33-34-32-15/h4-7,12,14H,1-3,8-9H2,(H,28,36)(H,38,39)(H,31,32,33,34)(H5,26,27,29,30,37) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant GAR Tfase |
J Med Chem 49: 2998-3002 (2006)
Article DOI: 10.1021/jm0601147
BindingDB Entry DOI: 10.7270/Q2VX0H9X |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24685
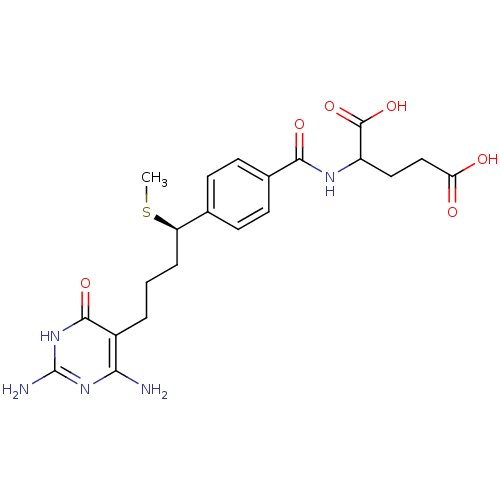
(10-thiomethyl-DDACTHF, 10R-3 | 10R (8) | 2-({4-[(1...)Show SMILES CS[C@H](CCCc1c(N)nc(N)[nH]c1=O)c1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O |r| Show InChI InChI=1S/C21H27N5O6S/c1-33-15(4-2-3-13-17(22)25-21(23)26-19(13)30)11-5-7-12(8-6-11)18(29)24-14(20(31)32)9-10-16(27)28/h5-8,14-15H,2-4,9-10H2,1H3,(H,24,29)(H,27,28)(H,31,32)(H5,22,23,25,26,30)/t14?,15-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
| Assay Description
Enzyme activity assays of recombinant hGAR Tfase and recombinant hAICAR Tfase were performed as previously described. Kinetics of the enzyme reaction... |
Biochemistry 52: 5133-44 (2013)
Article DOI: 10.1021/bi4005182
BindingDB Entry DOI: 10.7270/Q2K93669 |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24686
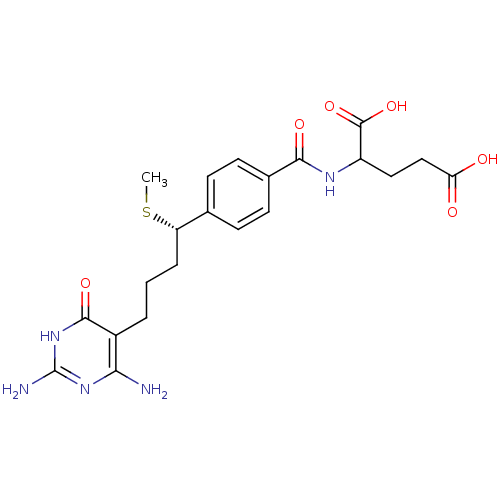
(10-thiomethyl-DDACTHF, 10S-3 | 10S (7) | 2-({4-[(1...)Show SMILES CS[C@@H](CCCc1c(N)nc(N)[nH]c1=O)c1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O |r| Show InChI InChI=1S/C21H27N5O6S/c1-33-15(4-2-3-13-17(22)25-21(23)26-19(13)30)11-5-7-12(8-6-11)18(29)24-14(20(31)32)9-10-16(27)28/h5-8,14-15H,2-4,9-10H2,1H3,(H,24,29)(H,27,28)(H,31,32)(H5,22,23,25,26,30)/t14?,15-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 180 | -38.6 | n/a | n/a | n/a | n/a | n/a | 7.5 | 26 |
The Scripps Research Institute
| Assay Description
Assays were initiated by the addition of GAR to the reaction mixture containing GAR Tfase, test compounds, and cofactor. The assay monitors the defor... |
J Med Chem 51: 5441-8 (2008)
Article DOI: 10.1021/jm800555h
BindingDB Entry DOI: 10.7270/Q2WD3XVB |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24686

(10-thiomethyl-DDACTHF, 10S-3 | 10S (7) | 2-({4-[(1...)Show SMILES CS[C@@H](CCCc1c(N)nc(N)[nH]c1=O)c1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O |r| Show InChI InChI=1S/C21H27N5O6S/c1-33-15(4-2-3-13-17(22)25-21(23)26-19(13)30)11-5-7-12(8-6-11)18(29)24-14(20(31)32)9-10-16(27)28/h5-8,14-15H,2-4,9-10H2,1H3,(H,24,29)(H,27,28)(H,31,32)(H5,22,23,25,26,30)/t14?,15-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
| Assay Description
Enzyme activity assays of recombinant hGAR Tfase and recombinant hAICAR Tfase were performed as previously described. Kinetics of the enzyme reaction... |
Biochemistry 52: 5133-44 (2013)
Article DOI: 10.1021/bi4005182
BindingDB Entry DOI: 10.7270/Q2K93669 |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24685

(10-thiomethyl-DDACTHF, 10R-3 | 10R (8) | 2-({4-[(1...)Show SMILES CS[C@H](CCCc1c(N)nc(N)[nH]c1=O)c1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O |r| Show InChI InChI=1S/C21H27N5O6S/c1-33-15(4-2-3-13-17(22)25-21(23)26-19(13)30)11-5-7-12(8-6-11)18(29)24-14(20(31)32)9-10-16(27)28/h5-8,14-15H,2-4,9-10H2,1H3,(H,24,29)(H,27,28)(H,31,32)(H5,22,23,25,26,30)/t14?,15-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 210 | -38.2 | n/a | n/a | n/a | n/a | n/a | 7.5 | 26 |
The Scripps Research Institute
| Assay Description
Assays were initiated by the addition of GAR to the reaction mixture containing GAR Tfase, test compounds, and cofactor. The assay monitors the defor... |
J Med Chem 51: 5441-8 (2008)
Article DOI: 10.1021/jm800555h
BindingDB Entry DOI: 10.7270/Q2WD3XVB |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50141931

((R)-[(2S,3S)-3-[4-(5-Biphenyl-4-ylmethyl-2-ethyl-2...)Show SMILES CCn1nc(Cc2ccc(cc2)-c2ccccc2)cc1C1CCN(C[C@H]2CN(C[C@@H]2c2cccc(F)c2)[C@H](C2CCCCC2)C(O)=O)CC1 Show InChI InChI=1S/C42H51FN4O2/c1-2-47-40(26-38(44-47)24-30-16-18-32(19-17-30)31-10-5-3-6-11-31)33-20-22-45(23-21-33)27-36-28-46(29-39(36)35-14-9-15-37(43)25-35)41(42(48)49)34-12-7-4-8-13-34/h3,5-6,9-11,14-19,25-26,33-34,36,39,41H,2,4,7-8,12-13,20-24,27-29H2,1H3,(H,48,49)/t36-,39+,41+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against hERG Voltage-gated potassium channel subunit Kv11.1 |
Bioorg Med Chem Lett 14: 947-52 (2004)
Article DOI: 10.1016/j.bmcl.2003.12.006
BindingDB Entry DOI: 10.7270/Q2F18Z5V |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247072

(7-phenyl-1-(4-(2,2,2-trifluoroacetyl)oxazol-2-yl)h...)Show InChI InChI=1S/C18H18F3NO3/c19-18(20,21)16(24)14-12-25-17(22-14)15(23)11-7-2-1-4-8-13-9-5-3-6-10-13/h3,5-6,9-10,12H,1-2,4,7-8,11H2 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247038

(1-(4-methyloxazol-2-yl)-7-phenylheptan-1-one | CHE...)Show InChI InChI=1S/C17H21NO2/c1-14-13-20-17(18-14)16(19)12-8-3-2-5-9-15-10-6-4-7-11-15/h4,6-7,10-11,13H,2-3,5,8-9,12H2,1H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Fatty-acid amide hydrolase 1 [30-579]
(Rattus norvegicus (rat)) | BDBM50247138

(1-(4-methoxyoxazol-2-yl)-7-phenylheptan-1-one | CH...)Show InChI InChI=1S/C17H21NO3/c1-20-16-13-21-17(18-16)15(19)12-8-3-2-5-9-14-10-6-4-7-11-14/h4,6-7,10-11,13H,2-3,5,8-9,12H2,1H3 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
Curated by ChEMBL
| Assay Description
Inhibition of rat FAAH |
Bioorg Med Chem Lett 18: 5842-6 (2008)
Article DOI: 10.1016/j.bmcl.2008.06.084
BindingDB Entry DOI: 10.7270/Q2DN44V8 |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24689
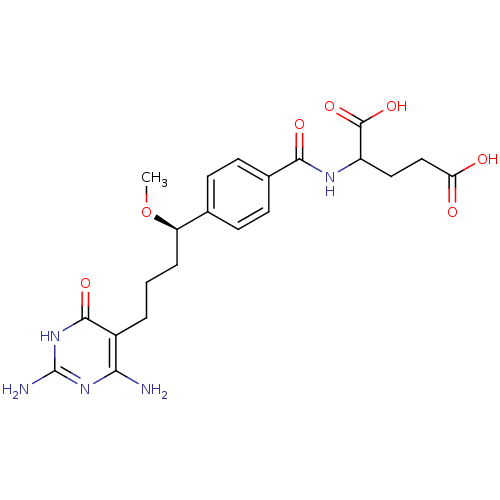
(10-methoxy-DDACTHF, 10R-8 | 2-({4-[(1R)-4-(2,4-dia...)Show SMILES CO[C@H](CCCc1c(N)nc(N)[nH]c1=O)c1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O |r| Show InChI InChI=1S/C21H27N5O7/c1-33-15(4-2-3-13-17(22)25-21(23)26-19(13)30)11-5-7-12(8-6-11)18(29)24-14(20(31)32)9-10-16(27)28/h5-8,14-15H,2-4,9-10H2,1H3,(H,24,29)(H,27,28)(H,31,32)(H5,22,23,25,26,30)/t14?,15-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 850 | -34.8 | n/a | n/a | n/a | n/a | n/a | 7.5 | 26 |
The Scripps Research Institute
| Assay Description
Assays were initiated by the addition of GAR to the reaction mixture containing GAR Tfase, test compounds, and cofactor. The assay monitors the defor... |
J Med Chem 51: 5441-8 (2008)
Article DOI: 10.1021/jm800555h
BindingDB Entry DOI: 10.7270/Q2WD3XVB |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50141911

((R)-2-[(2S,3S)-3-{4-[5-(4-Cyano-benzyl)-2-ethyl-2H...)Show SMILES CCn1nc(Cc2ccc(cc2)C#N)cc1C1CCN(C[C@H]2CN(C[C@@H]2c2cccc(F)c2)[C@@H](C(O)=O)C(C)(C)C)CC1 Show InChI InChI=1S/C35H44FN5O2/c1-5-41-32(19-30(38-41)17-24-9-11-25(20-37)12-10-24)26-13-15-39(16-14-26)21-28-22-40(33(34(42)43)35(2,3)4)23-31(28)27-7-6-8-29(36)18-27/h6-12,18-19,26,28,31,33H,5,13-17,21-23H2,1-4H3,(H,42,43)/t28-,31+,33-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against hERG Voltage-gated potassium channel subunit Kv11.1 |
Bioorg Med Chem Lett 14: 947-52 (2004)
Article DOI: 10.1016/j.bmcl.2003.12.006
BindingDB Entry DOI: 10.7270/Q2F18Z5V |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24687
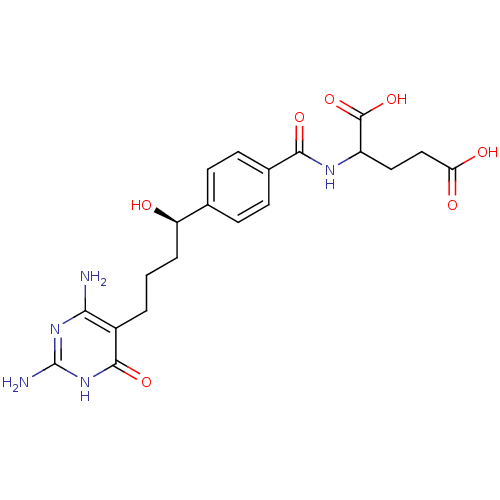
(10-hydroxy-DDACTHF, 10R-7 | 2-({4-[(1R)-4-(2,4-dia...)Show SMILES Nc1nc(N)c(CCC[C@@H](O)c2ccc(cc2)C(=O)NC(CCC(O)=O)C(O)=O)c(=O)[nH]1 |r| Show InChI InChI=1S/C20H25N5O7/c21-16-12(18(30)25-20(22)24-16)2-1-3-14(26)10-4-6-11(7-5-10)17(29)23-13(19(31)32)8-9-15(27)28/h4-7,13-14,26H,1-3,8-9H2,(H,23,29)(H,27,28)(H,31,32)(H5,21,22,24,25,30)/t13?,14-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10E+3 | -34.1 | n/a | n/a | n/a | n/a | n/a | 7.5 | 26 |
The Scripps Research Institute
| Assay Description
Assays were initiated by the addition of GAR to the reaction mixture containing GAR Tfase, test compounds, and cofactor. The assay monitors the defor... |
J Med Chem 51: 5441-8 (2008)
Article DOI: 10.1021/jm800555h
BindingDB Entry DOI: 10.7270/Q2WD3XVB |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24691
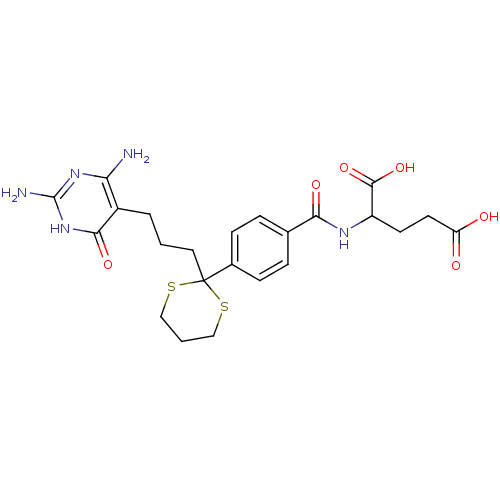
(2-[(4-{2-[3-(2,4-diamino-6-oxo-1,6-dihydropyrimidi...)Show SMILES Nc1nc(N)c(CCCC2(SCCCS2)c2ccc(cc2)C(=O)NC(CCC(O)=O)C(O)=O)c(=O)[nH]1 Show InChI InChI=1S/C23H29N5O6S2/c24-18-15(20(32)28-22(25)27-18)3-1-10-23(35-11-2-12-36-23)14-6-4-13(5-7-14)19(31)26-16(21(33)34)8-9-17(29)30/h4-7,16H,1-3,8-12H2,(H,26,31)(H,29,30)(H,33,34)(H5,24,25,27,28,32) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10E+3 | -34.1 | n/a | n/a | n/a | n/a | n/a | 7.5 | 26 |
The Scripps Research Institute
| Assay Description
Assays were initiated by the addition of GAR to the reaction mixture containing GAR Tfase, test compounds, and cofactor. The assay monitors the defor... |
J Med Chem 51: 5441-8 (2008)
Article DOI: 10.1021/jm800555h
BindingDB Entry DOI: 10.7270/Q2WD3XVB |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24690
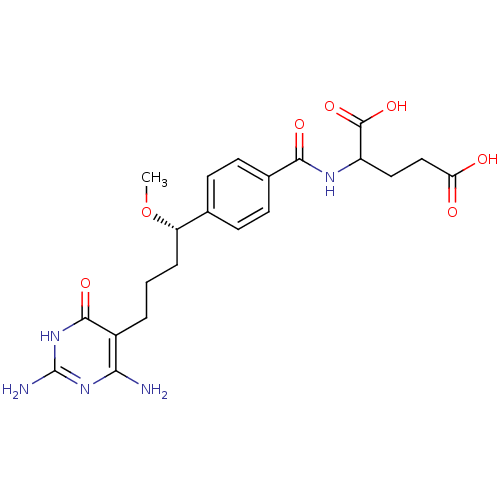
(10-methoxy-DDACTHF, 10S-8 | 2-({4-[(1S)-4-(2,4-dia...)Show SMILES CO[C@@H](CCCc1c(N)nc(N)[nH]c1=O)c1ccc(cc1)C(=O)NC(CCC(O)=O)C(O)=O |r| Show InChI InChI=1S/C21H27N5O7/c1-33-15(4-2-3-13-17(22)25-21(23)26-19(13)30)11-5-7-12(8-6-11)18(29)24-14(20(31)32)9-10-16(27)28/h5-8,14-15H,2-4,9-10H2,1H3,(H,24,29)(H,27,28)(H,31,32)(H5,22,23,25,26,30)/t14?,15-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20E+3 | -33.9 | n/a | n/a | n/a | n/a | n/a | 7.5 | 26 |
The Scripps Research Institute
| Assay Description
Assays were initiated by the addition of GAR to the reaction mixture containing GAR Tfase, test compounds, and cofactor. The assay monitors the defor... |
J Med Chem 51: 5441-8 (2008)
Article DOI: 10.1021/jm800555h
BindingDB Entry DOI: 10.7270/Q2WD3XVB |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24688
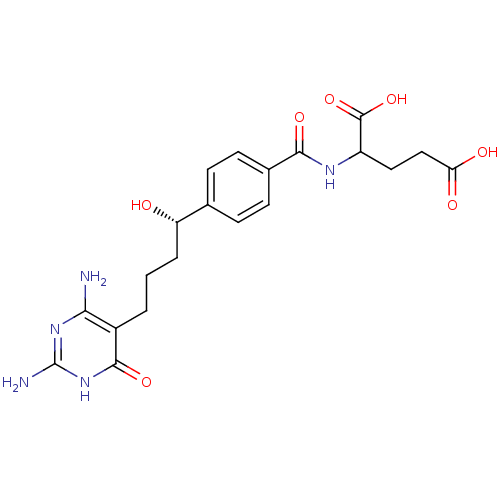
(10-hydroxy-DDACTHF, 10S-7 | 2-({4-[(1S)-4-(2,4-dia...)Show SMILES Nc1nc(N)c(CCC[C@H](O)c2ccc(cc2)C(=O)NC(CCC(O)=O)C(O)=O)c(=O)[nH]1 |r| Show InChI InChI=1S/C20H25N5O7/c21-16-12(18(30)25-20(22)24-16)2-1-3-14(26)10-4-6-11(7-5-10)17(29)23-13(19(31)32)8-9-15(27)28/h4-7,13-14,26H,1-3,8-9H2,(H,23,29)(H,27,28)(H,31,32)(H5,21,22,24,25,30)/t13?,14-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40E+3 | -33.5 | n/a | n/a | n/a | n/a | n/a | 7.5 | 26 |
The Scripps Research Institute
| Assay Description
Assays were initiated by the addition of GAR to the reaction mixture containing GAR Tfase, test compounds, and cofactor. The assay monitors the defor... |
J Med Chem 51: 5441-8 (2008)
Article DOI: 10.1021/jm800555h
BindingDB Entry DOI: 10.7270/Q2WD3XVB |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24692
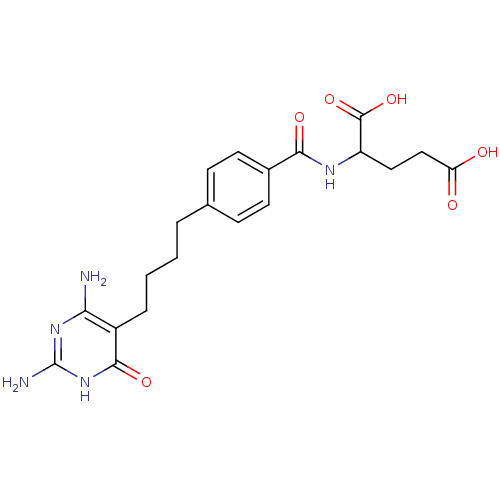
(2-({4-[4-(2,4-diamino-6-oxo-1,6-dihydropyrimidin-5...)Show SMILES Nc1nc(N)c(CCCCc2ccc(cc2)C(=O)NC(CCC(O)=O)C(O)=O)c(=O)[nH]1 Show InChI InChI=1S/C20H25N5O6/c21-16-13(18(29)25-20(22)24-16)4-2-1-3-11-5-7-12(8-6-11)17(28)23-14(19(30)31)9-10-15(26)27/h5-8,14H,1-4,9-10H2,(H,23,28)(H,26,27)(H,30,31)(H5,21,22,24,25,29) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Scripps Research Institute
| Assay Description
Enzyme activity assays of recombinant hGAR Tfase and recombinant hAICAR Tfase were performed as previously described. Kinetics of the enzyme reaction... |
Biochemistry 52: 5133-44 (2013)
Article DOI: 10.1021/bi4005182
BindingDB Entry DOI: 10.7270/Q2K93669 |
More data for this
Ligand-Target Pair | |
Trifunctional purine biosynthetic protein adenosine-3
(Homo sapiens (Human)) | BDBM24692

(2-({4-[4-(2,4-diamino-6-oxo-1,6-dihydropyrimidin-5...)Show SMILES Nc1nc(N)c(CCCCc2ccc(cc2)C(=O)NC(CCC(O)=O)C(O)=O)c(=O)[nH]1 Show InChI InChI=1S/C20H25N5O6/c21-16-13(18(29)25-20(22)24-16)4-2-1-3-11-5-7-12(8-6-11)17(28)23-14(19(30)31)9-10-15(26)27/h5-8,14H,1-4,9-10H2,(H,23,28)(H,26,27)(H,30,31)(H5,21,22,24,25,29) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.70E+3 | -33.0 | n/a | n/a | n/a | n/a | n/a | 7.5 | 26 |
The Scripps Research Institute
| Assay Description
Assays were initiated by the addition of GAR to the reaction mixture containing GAR Tfase, test compounds, and cofactor. The assay monitors the defor... |
J Med Chem 51: 5441-8 (2008)
Article DOI: 10.1021/jm800555h
BindingDB Entry DOI: 10.7270/Q2WD3XVB |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50141908

((R)-2-[(2S,3S)-3-{4-[5-(4-tert-Butyl-benzyl)-2-eth...)Show SMILES CCn1nc(Cc2ccc(cc2)C(C)(C)C)cc1C1CCN(C[C@H]2CN(C[C@@H]2c2cccc(F)c2)[C@@H](C(O)=O)C(C)(C)C)CC1 Show InChI InChI=1S/C38H53FN4O2/c1-8-43-34(22-32(40-43)20-26-12-14-30(15-13-26)37(2,3)4)27-16-18-41(19-17-27)23-29-24-42(35(36(44)45)38(5,6)7)25-33(29)28-10-9-11-31(39)21-28/h9-15,21-22,27,29,33,35H,8,16-20,23-25H2,1-7H3,(H,44,45)/t29-,33+,35-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against hERG Voltage-gated potassium channel subunit Kv11.1 |
Bioorg Med Chem Lett 14: 947-52 (2004)
Article DOI: 10.1016/j.bmcl.2003.12.006
BindingDB Entry DOI: 10.7270/Q2F18Z5V |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50141905
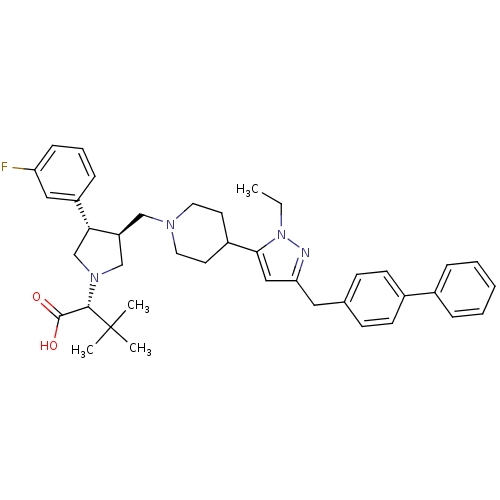
((R)-2-[(2S,3S)-3-[4-(5-Biphenyl-4-ylmethyl-2-ethyl...)Show SMILES CCn1nc(Cc2ccc(cc2)-c2ccccc2)cc1C1CCN(C[C@H]2CN(C[C@@H]2c2cccc(F)c2)[C@@H](C(O)=O)C(C)(C)C)CC1 Show InChI InChI=1S/C40H49FN4O2/c1-5-45-37(24-35(42-45)22-28-14-16-30(17-15-28)29-10-7-6-8-11-29)31-18-20-43(21-19-31)25-33-26-44(38(39(46)47)40(2,3)4)27-36(33)32-12-9-13-34(41)23-32/h6-17,23-24,31,33,36,38H,5,18-22,25-27H2,1-4H3,(H,46,47)/t33-,36+,38-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against hERG Voltage-gated potassium channel subunit Kv11.1 |
Bioorg Med Chem Lett 14: 947-52 (2004)
Article DOI: 10.1016/j.bmcl.2003.12.006
BindingDB Entry DOI: 10.7270/Q2F18Z5V |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50141978
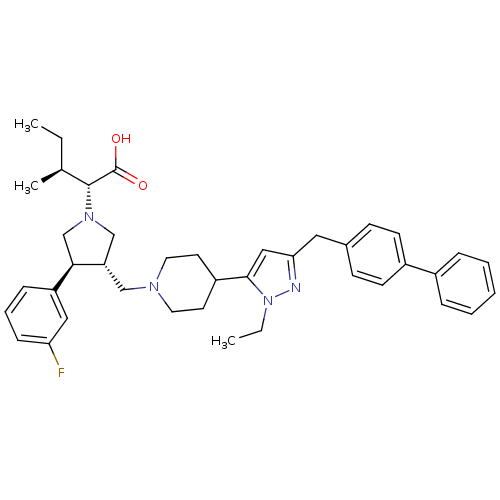
((2R,4S)-2-[(2S,3S)-3-[4-(5-Biphenyl-4-ylmethyl-2-e...)Show SMILES CC[C@H](C)[C@@H](N1C[C@H](CN2CCC(CC2)c2cc(Cc3ccc(cc3)-c3ccccc3)nn2CC)[C@H](C1)c1cccc(F)c1)C(O)=O Show InChI InChI=1S/C40H49FN4O2/c1-4-28(3)39(40(46)47)44-26-34(37(27-44)33-12-9-13-35(41)23-33)25-43-20-18-32(19-21-43)38-24-36(42-45(38)5-2)22-29-14-16-31(17-15-29)30-10-7-6-8-11-30/h6-17,23-24,28,32,34,37,39H,4-5,18-22,25-27H2,1-3H3,(H,46,47)/t28-,34-,37+,39+/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against hERG Voltage-gated potassium channel subunit Kv11.1 |
Bioorg Med Chem Lett 14: 947-52 (2004)
Article DOI: 10.1016/j.bmcl.2003.12.006
BindingDB Entry DOI: 10.7270/Q2F18Z5V |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50141935
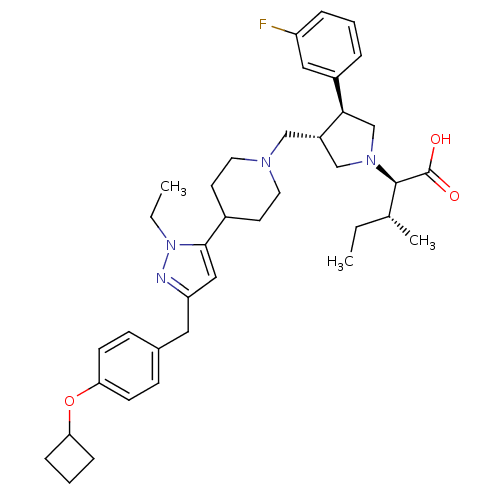
((2R,4R)-2-[(2S,3S)-3-{4-[5-(4-Cyclobutoxy-benzyl)-...)Show SMILES CC[C@@H](C)[C@@H](N1C[C@H](CN2CCC(CC2)c2cc(Cc3ccc(OC4CCC4)cc3)nn2CC)[C@H](C1)c1cccc(F)c1)C(O)=O Show InChI InChI=1S/C38H51FN4O3/c1-4-26(3)37(38(44)45)42-24-30(35(25-42)29-8-6-9-31(39)21-29)23-41-18-16-28(17-19-41)36-22-32(40-43(36)5-2)20-27-12-14-34(15-13-27)46-33-10-7-11-33/h6,8-9,12-15,21-22,26,28,30,33,35,37H,4-5,7,10-11,16-20,23-25H2,1-3H3,(H,44,45)/t26-,30+,35-,37-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against hERG Voltage-gated potassium channel subunit Kv11.1 |
Bioorg Med Chem Lett 14: 947-52 (2004)
Article DOI: 10.1016/j.bmcl.2003.12.006
BindingDB Entry DOI: 10.7270/Q2F18Z5V |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50141951
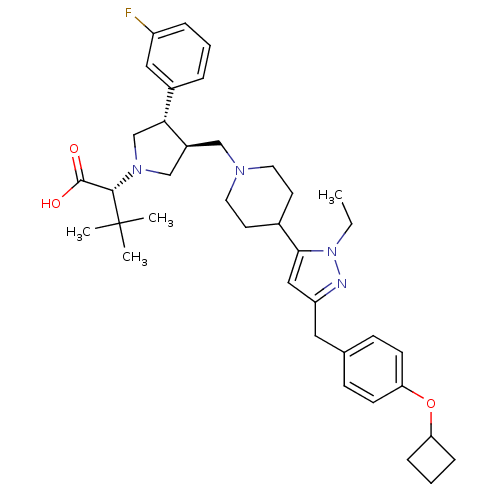
((R)-2-[(2S,3S)-3-{4-[5-(4-Cyclobutoxy-benzyl)-2-et...)Show SMILES CCn1nc(Cc2ccc(OC3CCC3)cc2)cc1C1CCN(C[C@H]2CN(C[C@@H]2c2cccc(F)c2)[C@@H](C(O)=O)C(C)(C)C)CC1 Show InChI InChI=1S/C38H51FN4O3/c1-5-43-35(22-31(40-43)20-26-12-14-33(15-13-26)46-32-10-7-11-32)27-16-18-41(19-17-27)23-29-24-42(36(37(44)45)38(2,3)4)25-34(29)28-8-6-9-30(39)21-28/h6,8-9,12-15,21-22,27,29,32,34,36H,5,7,10-11,16-20,23-25H2,1-4H3,(H,44,45)/t29-,34+,36-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Research Laboratories
Curated by ChEMBL
| Assay Description
Binding affinity against hERG Voltage-gated potassium channel subunit Kv11.1 |
Bioorg Med Chem Lett 14: 947-52 (2004)
Article DOI: 10.1016/j.bmcl.2003.12.006
BindingDB Entry DOI: 10.7270/Q2F18Z5V |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data