 Found 683 hits with Last Name = 'desrayaud' and Initial = 's'
Found 683 hits with Last Name = 'desrayaud' and Initial = 's' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Metabotropic glutamate receptor 5
(Homo sapiens (Human)) | BDBM50336921
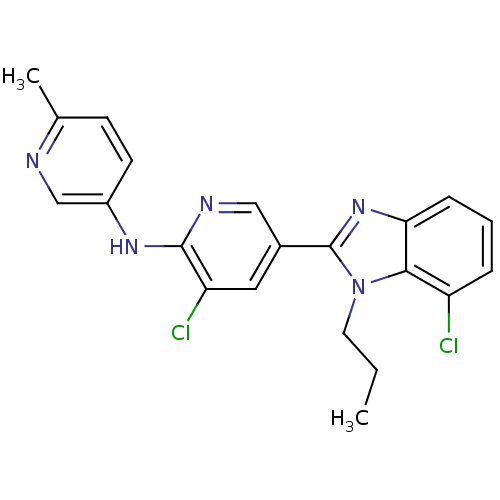
(3-chloro-5-(7-chloro-1-propyl-1H-benzo[d]imidazol-...)Show SMILES CCCn1c(nc2cccc(Cl)c12)-c1cnc(Nc2ccc(C)nc2)c(Cl)c1 Show InChI InChI=1S/C21H19Cl2N5/c1-3-9-28-19-16(22)5-4-6-18(19)27-21(28)14-10-17(23)20(25-11-14)26-15-8-7-13(2)24-12-15/h4-8,10-12H,3,9H2,1-2H3,(H,25,26) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]ABP688 from human recombinant mGlu5 receptor |
ACS Med Chem Lett 2: 58-62 (2011)
Article DOI: 10.1021/ml100215b
BindingDB Entry DOI: 10.7270/Q22R3RZM |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 5
(Rattus norvegicus (Rat)) | BDBM50305056
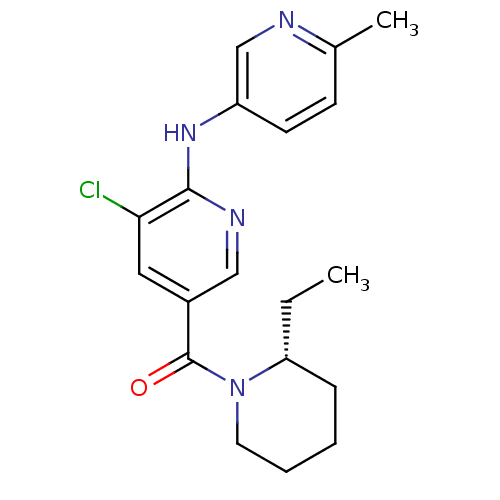
((R)-(5-chloro-6-(6-methylpyridin-3-ylamino)pyridin...)Show SMILES CC[C@@H]1CCCCN1C(=O)c1cnc(Nc2ccc(C)nc2)c(Cl)c1 |r| Show InChI InChI=1S/C19H23ClN4O/c1-3-16-6-4-5-9-24(16)19(25)14-10-17(20)18(22-11-14)23-15-8-7-13(2)21-12-15/h7-8,10-12,16H,3-6,9H2,1-2H3,(H,22,23)/t16-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Displacement of [3H]ABP688 from mGluR5 in rat brain cortex |
Bioorg Med Chem Lett 20: 184-8 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.001
BindingDB Entry DOI: 10.7270/Q2QN66VN |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 5
(Rattus norvegicus (Rat)) | BDBM50336921

(3-chloro-5-(7-chloro-1-propyl-1H-benzo[d]imidazol-...)Show SMILES CCCn1c(nc2cccc(Cl)c12)-c1cnc(Nc2ccc(C)nc2)c(Cl)c1 Show InChI InChI=1S/C21H19Cl2N5/c1-3-9-28-19-16(22)5-4-6-18(19)27-21(28)14-10-17(23)20(25-11-14)26-15-8-7-13(2)24-12-15/h4-8,10-12H,3,9H2,1-2H3,(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Displacement of [3H]ABP688 from mGlu5 receptor in rat brain tissue |
ACS Med Chem Lett 2: 58-62 (2011)
Article DOI: 10.1021/ml100215b
BindingDB Entry DOI: 10.7270/Q22R3RZM |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 5
(Homo sapiens (Human)) | BDBM50305056

((R)-(5-chloro-6-(6-methylpyridin-3-ylamino)pyridin...)Show SMILES CC[C@@H]1CCCCN1C(=O)c1cnc(Nc2ccc(C)nc2)c(Cl)c1 |r| Show InChI InChI=1S/C19H23ClN4O/c1-3-16-6-4-5-9-24(16)19(25)14-10-17(20)18(22-11-14)23-15-8-7-13(2)21-12-15/h7-8,10-12,16H,3-6,9H2,1-2H3,(H,22,23)/t16-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Displacement of [3H]ABP688 from human recombinant mGluR5 expressed in CHO cells |
Bioorg Med Chem Lett 20: 184-8 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.001
BindingDB Entry DOI: 10.7270/Q2QN66VN |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 5
(Homo sapiens (Human)) | BDBM50392323
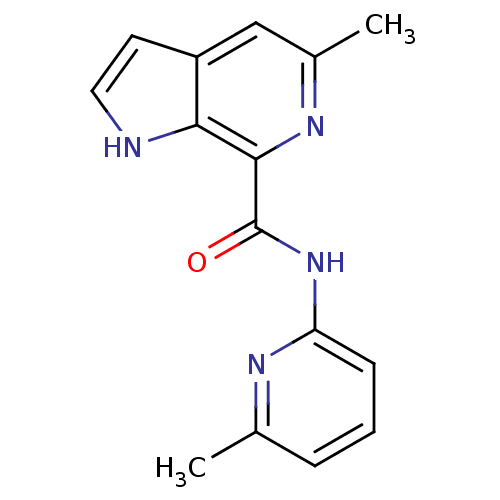
(CHEMBL2153782)Show InChI InChI=1S/C15H14N4O/c1-9-4-3-5-12(17-9)19-15(20)14-13-11(6-7-16-13)8-10(2)18-14/h3-8,16H,1-2H3,(H,17,19,20) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 43 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Displacement of [3H]ABP688 from human mGluR5a transmembrane region expressed in mouse L(tk-) cells |
Bioorg Med Chem Lett 22: 6454-9 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.053
BindingDB Entry DOI: 10.7270/Q2765GDX |
More data for this
Ligand-Target Pair | |
Cathepsin E
(Homo sapiens (Human)) | BDBM50305527
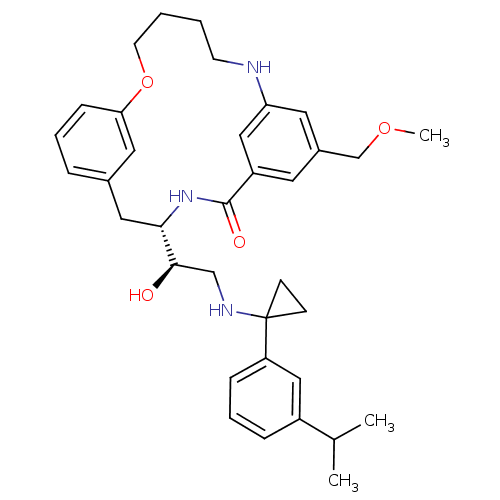
((4S)-4-[(1R)-1-hydroxy-2-({1-[3-(1-methylethyl)phe...)Show SMILES COCc1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c1)c2)[C@H](O)CNC1(CC1)c1cccc(c1)C(C)C)c3 |r| Show InChI InChI=1S/C35H45N3O4/c1-24(2)27-9-7-10-29(20-27)35(12-13-35)37-22-33(39)32-19-25-8-6-11-31(18-25)42-15-5-4-14-36-30-17-26(23-41-3)16-28(21-30)34(40)38-32/h6-11,16-18,20-21,24,32-33,36-37,39H,4-5,12-15,19,22-23H2,1-3H3,(H,38,40)/t32-,33+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.400 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin E |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305542
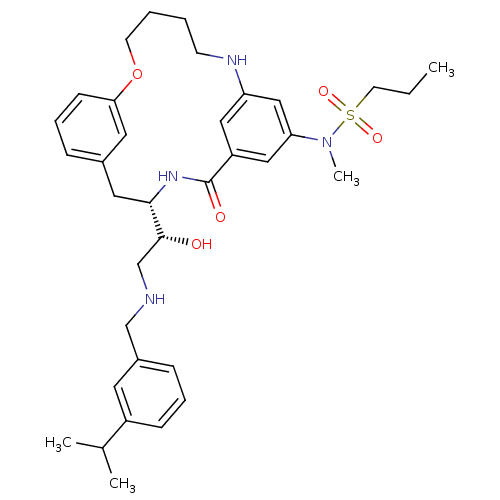
(CHEMBL595016 | Propane-1-sulfonic acid{(S)-4-[(R)-...)Show SMILES CCCS(=O)(=O)N(C)c1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c2)c1)[C@H](O)CNCc1cccc(c1)C(C)C)c3 |r| Show InChI InChI=1S/C35H48N4O5S/c1-5-16-45(42,43)39(4)31-21-29-20-30(22-31)37-14-6-7-15-44-32-13-9-10-26(18-32)19-33(38-35(29)41)34(40)24-36-23-27-11-8-12-28(17-27)25(2)3/h8-13,17-18,20-22,25,33-34,36-37,40H,5-7,14-16,19,23-24H2,1-4H3,(H,38,41)/t33-,34+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305544
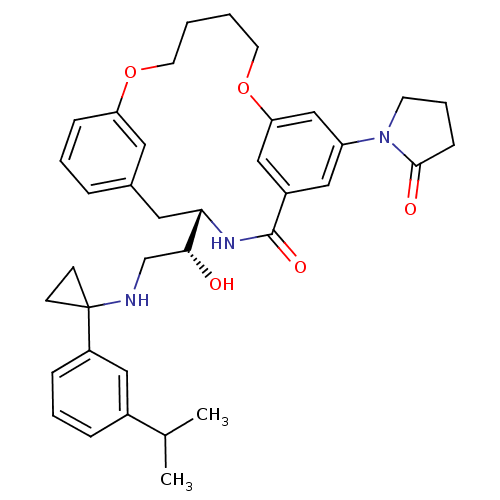
((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES CC(C)c1cccc(c1)C1(CC1)NC[C@@H](O)[C@@H]1Cc2cccc(OCCCCOc3cc(cc(c3)C(=O)N1)N1CCCC1=O)c2 |r| Show InChI InChI=1S/C37H45N3O5/c1-25(2)27-9-6-10-29(20-27)37(13-14-37)38-24-34(41)33-19-26-8-5-11-31(18-26)44-16-3-4-17-45-32-22-28(36(43)39-33)21-30(23-32)40-15-7-12-35(40)42/h5-6,8-11,18,20-23,25,33-34,38,41H,3-4,7,12-17,19,24H2,1-2H3,(H,39,43)/t33-,34+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50305527

((4S)-4-[(1R)-1-hydroxy-2-({1-[3-(1-methylethyl)phe...)Show SMILES COCc1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c1)c2)[C@H](O)CNC1(CC1)c1cccc(c1)C(C)C)c3 |r| Show InChI InChI=1S/C35H45N3O4/c1-24(2)27-9-7-10-29(20-27)35(12-13-35)37-22-33(39)32-19-25-8-6-11-31(18-25)42-15-5-4-14-36-30-17-26(23-41-3)16-28(21-30)34(40)38-32/h6-11,16-18,20-21,24,32-33,36-37,39H,4-5,12-15,19,22-23H2,1-3H3,(H,38,40)/t32-,33+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin D |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50294218
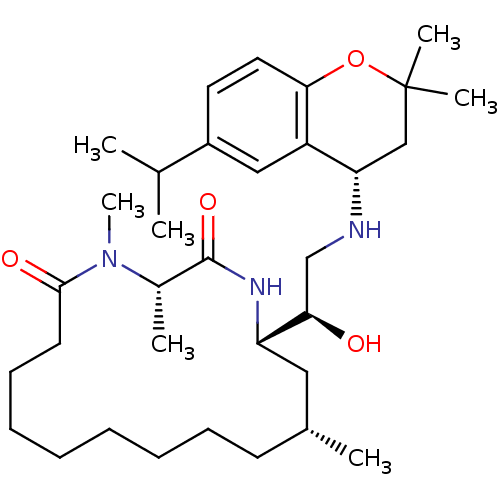
((3S,14R,16S)-16-((R)-1-hydroxy-2-((S)-6-isopropyl-...)Show SMILES CC(C)c1ccc2OC(C)(C)C[C@H](NC[C@@H](O)[C@@H]3C[C@H](C)CCCCCCCCC(=O)N(C)[C@@H](C)C(=O)N3)c2c1 |r| Show InChI InChI=1S/C33H55N3O4/c1-22(2)25-16-17-30-26(19-25)28(20-33(5,6)40-30)34-21-29(37)27-18-23(3)14-12-10-8-9-11-13-15-31(38)36(7)24(4)32(39)35-27/h16-17,19,22-24,27-29,34,37H,8-15,18,20-21H2,1-7H3,(H,35,39)/t23-,24+,27+,28+,29-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human cathepsin D |
Bioorg Med Chem Lett 19: 1366-70 (2009)
Article DOI: 10.1016/j.bmcl.2009.01.055
BindingDB Entry DOI: 10.7270/Q2SB45S3 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase mTOR
(Homo sapiens (Human)) | BDBM92862
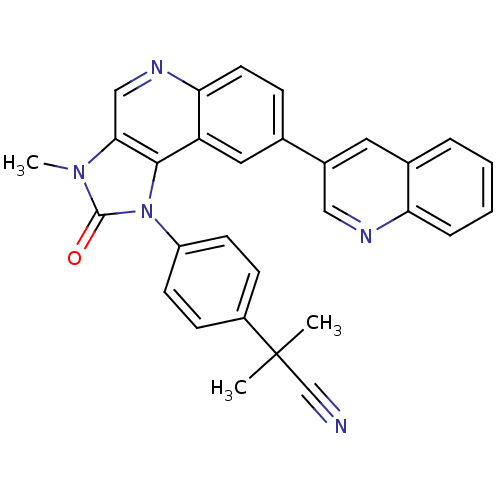
(US9284315, BEZ-235 | mTOR Inhibitor, BEZ235)Show SMILES Cn1c2cnc3ccc(cc3c2n(-c2ccc(cc2)C(C)(C)C#N)c1=O)-c1cnc2ccccc2c1 Show InChI InChI=1S/C30H23N5O/c1-30(2,18-31)22-9-11-23(12-10-22)35-28-24-15-19(21-14-20-6-4-5-7-25(20)32-16-21)8-13-26(24)33-17-27(28)34(3)29(35)36/h4-17H,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00267
BindingDB Entry DOI: 10.7270/Q28P64KJ |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50386487
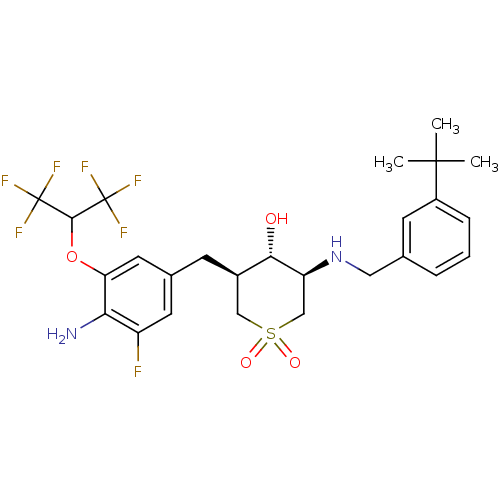
(CHEMBL2048047)Show SMILES CC(C)(C)c1cccc(CN[C@H]2CS(=O)(=O)C[C@@H](Cc3cc(F)c(N)c(OC(C(F)(F)F)C(F)(F)F)c3)[C@@H]2O)c1 |r| Show InChI InChI=1S/C26H31F7N2O4S/c1-24(2,3)17-6-4-5-14(8-17)11-35-19-13-40(37,38)12-16(22(19)36)7-15-9-18(27)21(34)20(10-15)39-23(25(28,29)30)26(31,32)33/h4-6,8-10,16,19,22-23,35-36H,7,11-13,34H2,1-3H3/t16-,19+,22+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 ectodomain after 1 hr by fluorescence analysis |
J Med Chem 55: 3364-86 (2012)
Article DOI: 10.1021/jm300069y
BindingDB Entry DOI: 10.7270/Q2P55PJW |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50386513
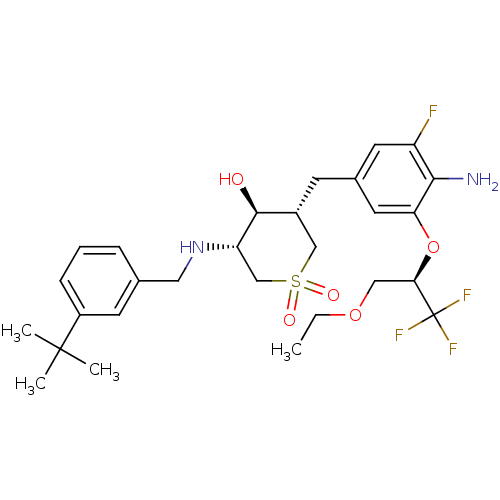
(CHEMBL2048051)Show SMILES CCOC[C@@H](Oc1cc(C[C@@H]2CS(=O)(=O)C[C@H](NCc3cccc(c3)C(C)(C)C)[C@H]2O)cc(F)c1N)C(F)(F)F |r| Show InChI InChI=1S/C28H38F4N2O5S/c1-5-38-14-24(28(30,31)32)39-23-12-18(11-21(29)25(23)33)9-19-15-40(36,37)16-22(26(19)35)34-13-17-7-6-8-20(10-17)27(2,3)4/h6-8,10-12,19,22,24,26,34-35H,5,9,13-16,33H2,1-4H3/t19-,22+,24-,26+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 ectodomain after 1 hr by fluorescence analysis |
J Med Chem 55: 3364-86 (2012)
Article DOI: 10.1021/jm300069y
BindingDB Entry DOI: 10.7270/Q2P55PJW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50386515
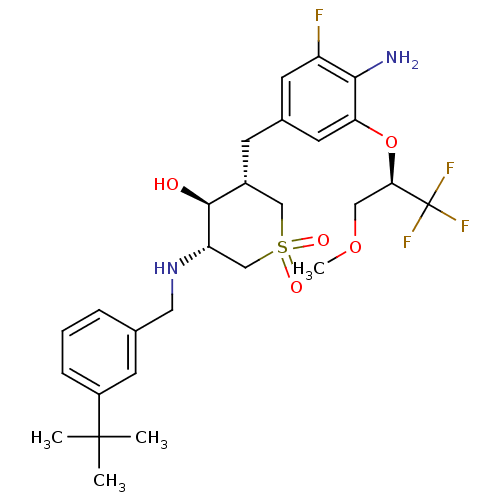
(CHEMBL2048053)Show SMILES COC[C@@H](Oc1cc(C[C@@H]2CS(=O)(=O)C[C@H](NCc3cccc(c3)C(C)(C)C)[C@H]2O)cc(F)c1N)C(F)(F)F |r| Show InChI InChI=1S/C27H36F4N2O5S/c1-26(2,3)19-7-5-6-16(9-19)12-33-21-15-39(35,36)14-18(25(21)34)8-17-10-20(28)24(32)22(11-17)38-23(13-37-4)27(29,30)31/h5-7,9-11,18,21,23,25,33-34H,8,12-15,32H2,1-4H3/t18-,21+,23-,25+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 ectodomain after 1 hr by fluorescence analysis |
J Med Chem 55: 3364-86 (2012)
Article DOI: 10.1021/jm300069y
BindingDB Entry DOI: 10.7270/Q2P55PJW |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50305531
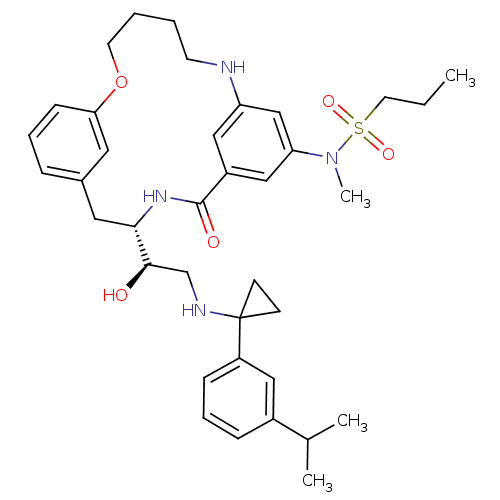
(CHEMBL595136 | Propane-1-sulfonic acid((S)-4-{(R)-...)Show SMILES CCCS(=O)(=O)N(C)c1cc2NCCCCOc3cccc(C[C@H](NC(=O)c(c2)c1)[C@H](O)CNC1(CC1)c1cccc(c1)C(C)C)c3 |r| Show InChI InChI=1S/C37H50N4O5S/c1-5-18-47(44,45)41(4)32-23-29-22-31(24-32)38-16-6-7-17-46-33-13-8-10-27(19-33)20-34(40-36(29)43)35(42)25-39-37(14-15-37)30-12-9-11-28(21-30)26(2)3/h8-13,19,21-24,26,34-35,38-39,42H,5-7,14-18,20,25H2,1-4H3,(H,40,43)/t34-,35+/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 expressed in Escherichia coli |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50386481
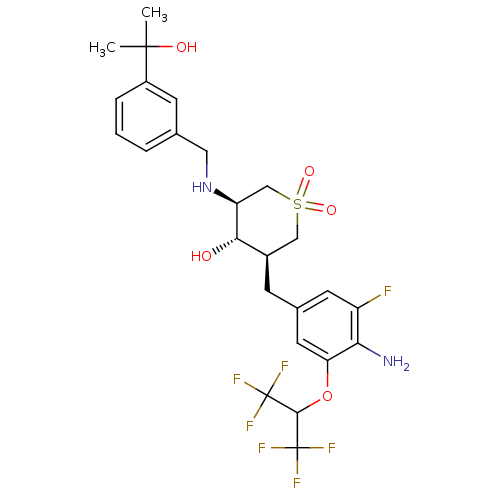
(CHEMBL2048058)Show SMILES CC(C)(O)c1cccc(CN[C@H]2CS(=O)(=O)C[C@@H](Cc3cc(F)c(N)c(OC(C(F)(F)F)C(F)(F)F)c3)[C@@H]2O)c1 |r| Show InChI InChI=1S/C25H29F7N2O5S/c1-23(2,36)16-5-3-4-13(7-16)10-34-18-12-40(37,38)11-15(21(18)35)6-14-8-17(26)20(33)19(9-14)39-22(24(27,28)29)25(30,31)32/h3-5,7-9,15,18,21-22,34-36H,6,10-12,33H2,1-2H3/t15-,18+,21+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 ectodomain after 1 hr by fluorescence analysis |
J Med Chem 55: 3364-86 (2012)
Article DOI: 10.1021/jm300069y
BindingDB Entry DOI: 10.7270/Q2P55PJW |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM16047
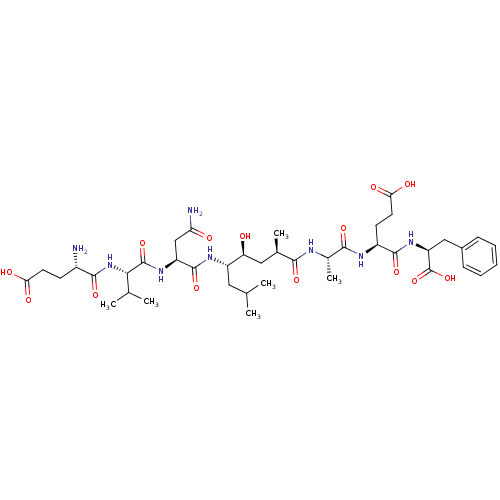
((4S)-4-[(2S)-2-[(2R,4S,5S)-5-[(2S)-2-[(2S)-2-[(4S)...)Show SMILES CC(C)C[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](NC(=O)[C@@H](N)CCC(O)=O)C(C)C)[C@@H](O)C[C@@H](C)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](Cc1ccccc1)C(O)=O |r| Show InChI InChI=1S/C41H64N8O14/c1-20(2)16-27(46-39(60)28(19-31(43)51)47-40(61)34(21(3)4)49-37(58)25(42)12-14-32(52)53)30(50)17-22(5)35(56)44-23(6)36(57)45-26(13-15-33(54)55)38(59)48-29(41(62)63)18-24-10-8-7-9-11-24/h7-11,20-23,25-30,34,50H,12-19,42H2,1-6H3,(H2,43,51)(H,44,56)(H,45,57)(H,46,60)(H,47,61)(H,48,59)(H,49,58)(H,52,53)(H,54,55)(H,62,63)/t22-,23+,25+,26+,27+,28+,29+,30+,34+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 ectodomain after 1 hr by fluorescence analysis |
J Med Chem 55: 3364-86 (2012)
Article DOI: 10.1021/jm300069y
BindingDB Entry DOI: 10.7270/Q2P55PJW |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50294218

((3S,14R,16S)-16-((R)-1-hydroxy-2-((S)-6-isopropyl-...)Show SMILES CC(C)c1ccc2OC(C)(C)C[C@H](NC[C@@H](O)[C@@H]3C[C@H](C)CCCCCCCCC(=O)N(C)[C@@H](C)C(=O)N3)c2c1 |r| Show InChI InChI=1S/C33H55N3O4/c1-22(2)25-16-17-30-26(19-25)28(20-33(5,6)40-30)34-21-29(37)27-18-23(3)14-12-10-8-9-11-13-15-31(38)36(7)24(4)32(39)35-27/h16-17,19,22-24,27-29,34,37H,8-15,18,20-21H2,1-7H3,(H,35,39)/t23-,24+,27+,28+,29-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 |
Bioorg Med Chem Lett 19: 1366-70 (2009)
Article DOI: 10.1016/j.bmcl.2009.01.055
BindingDB Entry DOI: 10.7270/Q2SB45S3 |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50386482
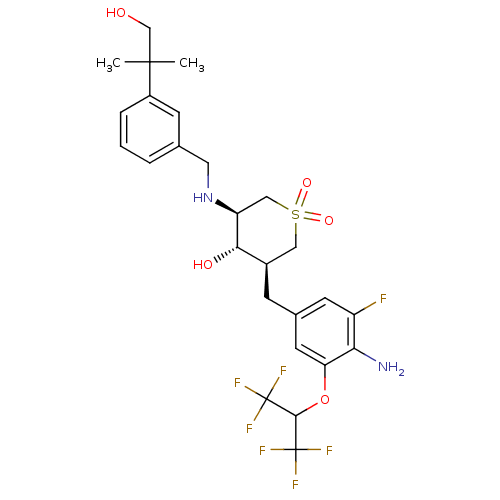
(CHEMBL2048059)Show SMILES CC(C)(CO)c1cccc(CN[C@H]2CS(=O)(=O)C[C@@H](Cc3cc(F)c(N)c(OC(C(F)(F)F)C(F)(F)F)c3)[C@@H]2O)c1 |r| Show InChI InChI=1S/C26H31F7N2O5S/c1-24(2,13-36)17-5-3-4-14(7-17)10-35-19-12-41(38,39)11-16(22(19)37)6-15-8-18(27)21(34)20(9-15)40-23(25(28,29)30)26(31,32)33/h3-5,7-9,16,19,22-23,35-37H,6,10-13,34H2,1-2H3/t16-,19+,22+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 ectodomain after 1 hr by fluorescence analysis |
J Med Chem 55: 3364-86 (2012)
Article DOI: 10.1021/jm300069y
BindingDB Entry DOI: 10.7270/Q2P55PJW |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50386512

(CHEMBL2048050)Show SMILES C[C@@H](Oc1cc(C[C@@H]2CS(=O)(=O)C[C@H](NCc3cccc(c3)C(C)(C)C)[C@H]2O)cc(F)c1N)C(F)(F)F |r| Show InChI InChI=1S/C26H34F4N2O4S/c1-15(26(28,29)30)36-22-11-17(10-20(27)23(22)31)8-18-13-37(34,35)14-21(24(18)33)32-12-16-6-5-7-19(9-16)25(2,3)4/h5-7,9-11,15,18,21,24,32-33H,8,12-14,31H2,1-4H3/t15-,18-,21+,24+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 ectodomain after 1 hr by fluorescence analysis |
J Med Chem 55: 3364-86 (2012)
Article DOI: 10.1021/jm300069y
BindingDB Entry DOI: 10.7270/Q2P55PJW |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50437055
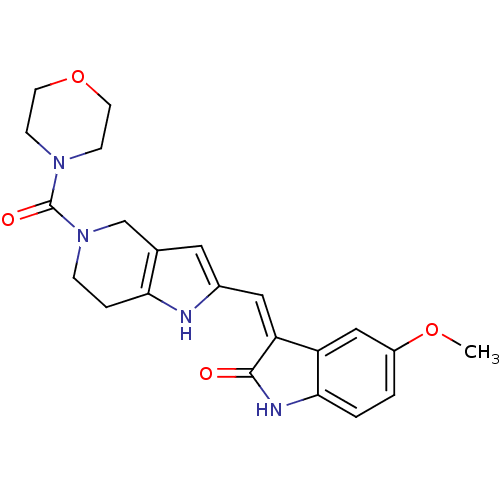
(CHEMBL2403370)Show SMILES COc1ccc2NC(=O)\C(=C/c3cc4CN(CCc4[nH]3)C(=O)N3CCOCC3)c2c1 Show InChI InChI=1S/C22H24N4O4/c1-29-16-2-3-20-17(12-16)18(21(27)24-20)11-15-10-14-13-26(5-4-19(14)23-15)22(28)25-6-8-30-9-7-25/h2-3,10-12,23H,4-9,13H2,1H3,(H,24,27)/b18-11- | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human LRRK2 (1885 to 2132) using 5-Fluo-Ahx-RLGRDKYKTLRQIRQGNTK-OH as substrate after 60 mins by fluorescence assay |
Bioorg Med Chem Lett 23: 4085-90 (2013)
Article DOI: 10.1016/j.bmcl.2013.05.054
BindingDB Entry DOI: 10.7270/Q2SB474D |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50305536
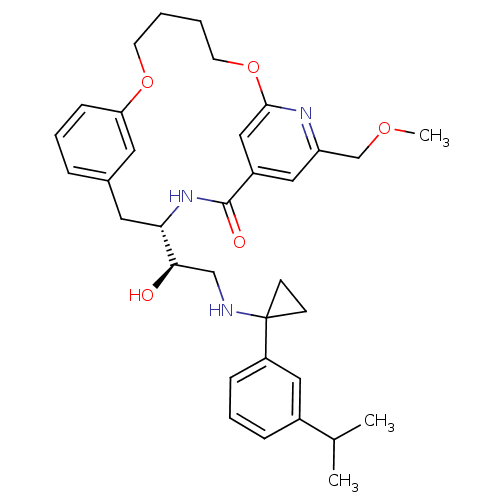
((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES COCc1cc2cc(OCCCCOc3cccc(C[C@H](NC2=O)[C@H](O)CNC2(CC2)c2cccc(c2)C(C)C)c3)n1 |r| Show InChI InChI=1S/C34H43N3O5/c1-23(2)25-9-7-10-27(18-25)34(12-13-34)35-21-31(38)30-17-24-8-6-11-29(16-24)41-14-4-5-15-42-32-20-26(33(39)37-30)19-28(36-32)22-40-3/h6-11,16,18-20,23,30-31,35,38H,4-5,12-15,17,21-22H2,1-3H3,(H,37,39)/t30-,31+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin D |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50386516
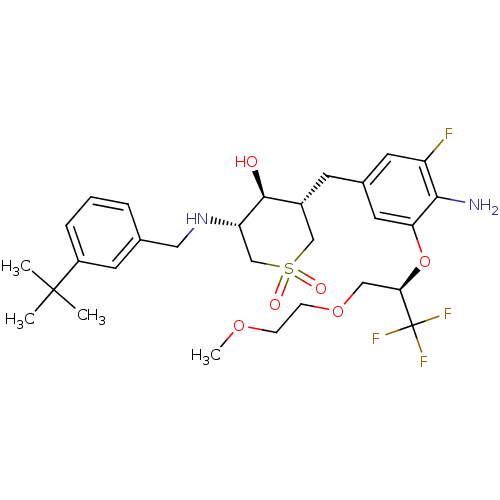
(CHEMBL2048054)Show SMILES COCCOC[C@@H](Oc1cc(C[C@@H]2CS(=O)(=O)C[C@H](NCc3cccc(c3)C(C)(C)C)[C@H]2O)cc(F)c1N)C(F)(F)F |r| Show InChI InChI=1S/C29H40F4N2O6S/c1-28(2,3)21-7-5-6-18(11-21)14-35-23-17-42(37,38)16-20(27(23)36)10-19-12-22(30)26(34)24(13-19)41-25(29(31,32)33)15-40-9-8-39-4/h5-7,11-13,20,23,25,27,35-36H,8-10,14-17,34H2,1-4H3/t20-,23+,25-,27+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 ectodomain after 1 hr by fluorescence analysis |
J Med Chem 55: 3364-86 (2012)
Article DOI: 10.1021/jm300069y
BindingDB Entry DOI: 10.7270/Q2P55PJW |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50112822
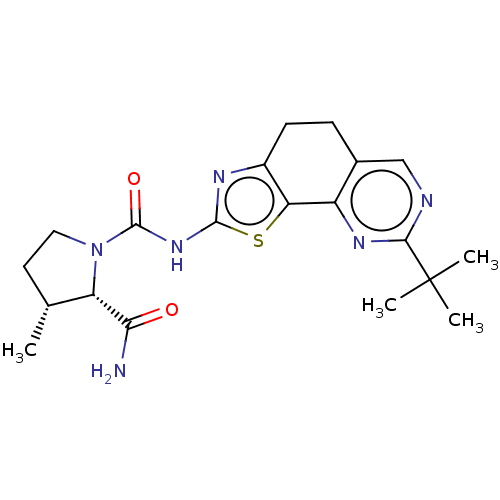
(CHEMBL3609525)Show SMILES C[C@@H]1CCN([C@@H]1C(N)=O)C(=O)Nc1nc2CCc3cnc(nc3-c2s1)C(C)(C)C |r| Show InChI InChI=1S/C20H26N6O2S/c1-10-7-8-26(14(10)16(21)27)19(28)25-18-23-12-6-5-11-9-22-17(20(2,3)4)24-13(11)15(12)29-18/h9-10,14H,5-8H2,1-4H3,(H2,21,27)(H,23,25,28)/t10-,14+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of N-terminal myristoylated human PI3Kalpha expressed in Rat1 cells assessed as inhibition of Akt phosphorylatuion at Ser473 by ELISA |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cathepsin E
(Homo sapiens (Human)) | BDBM50305537
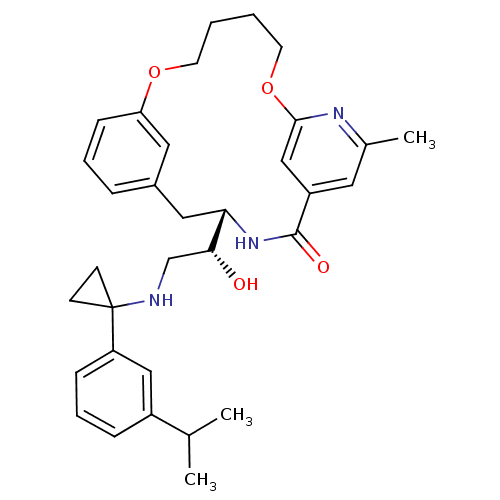
((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES CC(C)c1cccc(c1)C1(CC1)NC[C@@H](O)[C@@H]1Cc2cccc(OCCCCOc3cc(cc(C)n3)C(=O)N1)c2 |r| Show InChI InChI=1S/C33H41N3O4/c1-22(2)25-9-7-10-27(19-25)33(12-13-33)34-21-30(37)29-18-24-8-6-11-28(17-24)39-14-4-5-15-40-31-20-26(32(38)36-29)16-23(3)35-31/h6-11,16-17,19-20,22,29-30,34,37H,4-5,12-15,18,21H2,1-3H3,(H,36,38)/t29-,30+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin E |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 5
(Homo sapiens (Human)) | BDBM50392324
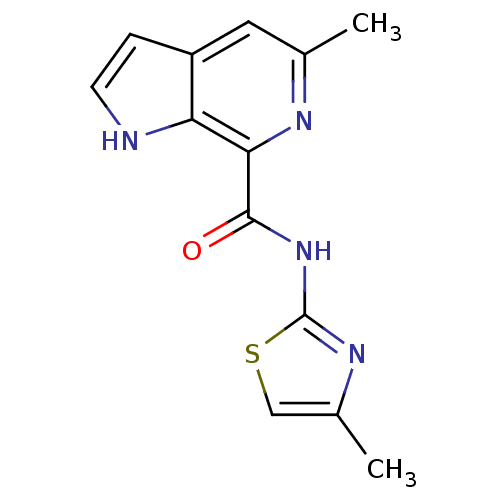
(CHEMBL2153783)Show InChI InChI=1S/C13H12N4OS/c1-7-5-9-3-4-14-10(9)11(15-7)12(18)17-13-16-8(2)6-19-13/h3-6,14H,1-2H3,(H,16,17,18) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Antagonist activity at human mGluR5a expressed in mouse L(tk-) cells assessed as inhibition of glutamate-induced Ca2+ influx by FLIPR assay |
Bioorg Med Chem Lett 22: 6454-9 (2012)
Article DOI: 10.1016/j.bmcl.2012.08.053
BindingDB Entry DOI: 10.7270/Q2765GDX |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50112823

(CHEMBL3609526)Show SMILES C[C@@H]1CCN([C@@H]1C(N)=O)C(=O)Nc1nc2CCc3cnc(nc3-c2s1)C(C)(C)C(F)(F)F |r| Show InChI InChI=1S/C20H23F3N6O2S/c1-9-6-7-29(13(9)15(24)30)18(31)28-17-26-11-5-4-10-8-25-16(19(2,3)20(21,22)23)27-12(10)14(11)32-17/h8-9,13H,4-7H2,1-3H3,(H2,24,30)(H,26,28,31)/t9-,13+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of N-terminal myristoylated human PI3Kalpha expressed in Rat1 cells assessed as inhibition of Akt phosphorylatuion at Ser473 by ELISA |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50112820
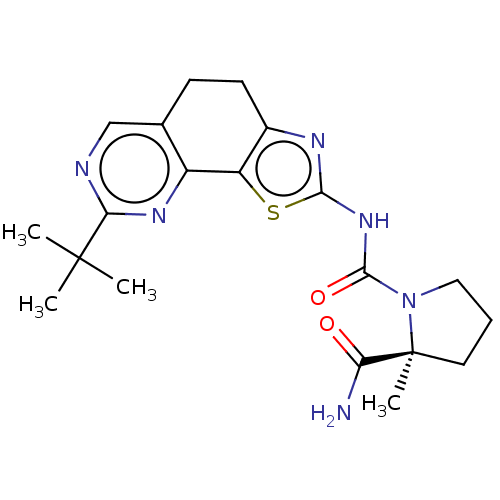
(CHEMBL3609523)Show SMILES CC(C)(C)c1ncc2CCc3nc(NC(=O)N4CCC[C@@]4(C)C(N)=O)sc3-c2n1 |r| Show InChI InChI=1S/C20H26N6O2S/c1-19(2,3)16-22-10-11-6-7-12-14(13(11)24-16)29-17(23-12)25-18(28)26-9-5-8-20(26,4)15(21)27/h10H,5-9H2,1-4H3,(H2,21,27)(H,23,25,28)/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of N-terminal myristoylated human PI3Kalpha expressed in Rat1 cells assessed as inhibition of Akt phosphorylatuion at Ser473 by ELISA |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50112826
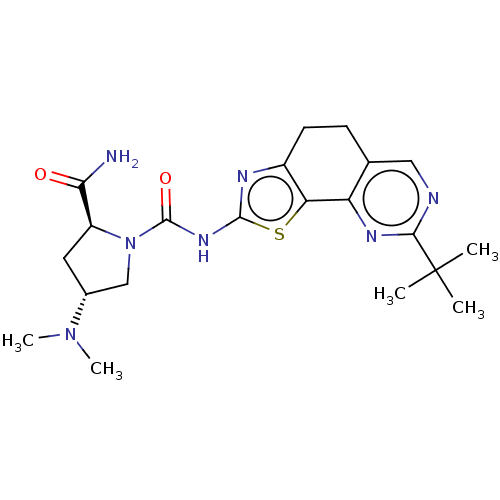
(CHEMBL3609529)Show SMILES CN(C)[C@@H]1C[C@H](N(C1)C(=O)Nc1nc2CCc3cnc(nc3-c2s1)C(C)(C)C)C(N)=O |r| Show InChI InChI=1S/C21H29N7O2S/c1-21(2,3)18-23-9-11-6-7-13-16(15(11)25-18)31-19(24-13)26-20(30)28-10-12(27(4)5)8-14(28)17(22)29/h9,12,14H,6-8,10H2,1-5H3,(H2,22,29)(H,24,26,30)/t12-,14+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) using phosphatidylinositol as substrate preincubated for 15 mins followed by ATP addition measured after 1 h... |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50437051

(CHEMBL2403375)Show SMILES CC(C)Oc1ccc2NC(=O)\C(=C/c3cc4CN(CCc4[nH]3)C(C)=O)c2c1 Show InChI InChI=1S/C21H23N3O3/c1-12(2)27-16-4-5-20-17(10-16)18(21(26)23-20)9-15-8-14-11-24(13(3)25)7-6-19(14)22-15/h4-5,8-10,12,22H,6-7,11H2,1-3H3,(H,23,26)/b18-9- | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human LRRK2 (1885 to 2132) using 5-Fluo-Ahx-RLGRDKYKTLRQIRQGNTK-OH as substrate after 60 mins by fluorescence assay |
Bioorg Med Chem Lett 23: 4085-90 (2013)
Article DOI: 10.1016/j.bmcl.2013.05.054
BindingDB Entry DOI: 10.7270/Q2SB474D |
More data for this
Ligand-Target Pair | |
Leucine-rich repeat serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50437052
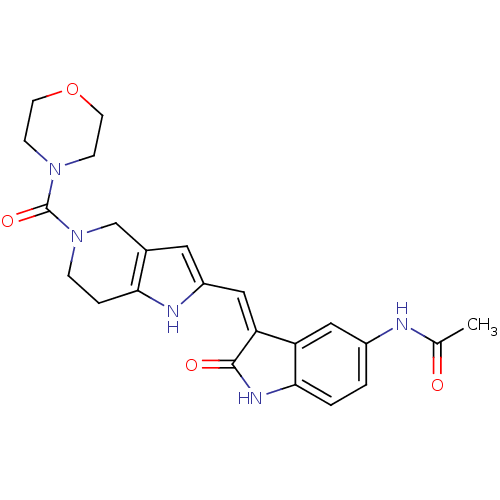
(CHEMBL2403374)Show SMILES CC(=O)Nc1ccc2NC(=O)\C(=C/c3cc4CN(CCc4[nH]3)C(=O)N3CCOCC3)c2c1 Show InChI InChI=1S/C23H25N5O4/c1-14(29)24-16-2-3-21-18(11-16)19(22(30)26-21)12-17-10-15-13-28(5-4-20(15)25-17)23(31)27-6-8-32-9-7-27/h2-3,10-12,25H,4-9,13H2,1H3,(H,24,29)(H,26,30)/b19-12- | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of human LRRK2 (1885 to 2132) using 5-Fluo-Ahx-RLGRDKYKTLRQIRQGNTK-OH as substrate after 60 mins by fluorescence assay |
Bioorg Med Chem Lett 23: 4085-90 (2013)
Article DOI: 10.1016/j.bmcl.2013.05.054
BindingDB Entry DOI: 10.7270/Q2SB474D |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM50437061

(CHEMBL2403377)Show SMILES CC(=O)N1CCc2[nH]c(\C=C3/C(=O)Nc4ccc(cc34)C#N)cc2C1 Show InChI InChI=1S/C19H16N4O2/c1-11(24)23-5-4-17-13(10-23)7-14(21-17)8-16-15-6-12(9-20)2-3-18(15)22-19(16)25/h2-3,6-8,21H,4-5,10H2,1H3,(H,22,25)/b16-8- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of RET (unknown origin) |
Bioorg Med Chem Lett 23: 4085-90 (2013)
Article DOI: 10.1016/j.bmcl.2013.05.054
BindingDB Entry DOI: 10.7270/Q2SB474D |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM50437059
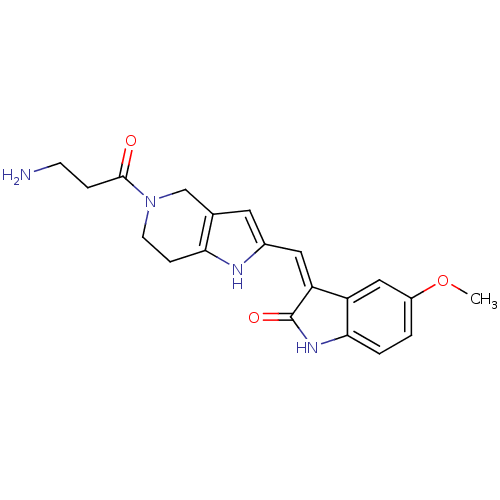
(CHEMBL2403366)Show SMILES COc1ccc2NC(=O)\C(=C/c3cc4CN(CCc4[nH]3)C(=O)CCN)c2c1 Show InChI InChI=1S/C20H22N4O3/c1-27-14-2-3-18-15(10-14)16(20(26)23-18)9-13-8-12-11-24(19(25)4-6-21)7-5-17(12)22-13/h2-3,8-10,22H,4-7,11,21H2,1H3,(H,23,26)/b16-9- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of RET (unknown origin) |
Bioorg Med Chem Lett 23: 4085-90 (2013)
Article DOI: 10.1016/j.bmcl.2013.05.054
BindingDB Entry DOI: 10.7270/Q2SB474D |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50112819

(CHEMBL3609522)Show SMILES CC(C)(C)c1ncc2CCc3nc(NC(=O)N4CCC[C@H]4C(N)=O)sc3-c2n1 |r| Show InChI InChI=1S/C19H24N6O2S/c1-19(2,3)16-21-9-10-6-7-11-14(13(10)23-16)28-17(22-11)24-18(27)25-8-4-5-12(25)15(20)26/h9,12H,4-8H2,1-3H3,(H2,20,26)(H,22,24,27)/t12-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) using phosphatidylinositol as substrate preincubated for 15 mins followed by ATP addition measured after 1 h... |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | |
Cathepsin E
(Homo sapiens (Human)) | BDBM50305536

((S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyc...)Show SMILES COCc1cc2cc(OCCCCOc3cccc(C[C@H](NC2=O)[C@H](O)CNC2(CC2)c2cccc(c2)C(C)C)c3)n1 |r| Show InChI InChI=1S/C34H43N3O5/c1-23(2)25-9-7-10-27(18-25)34(12-13-34)35-21-31(38)30-17-24-8-6-11-29(16-24)41-14-4-5-15-42-32-20-26(33(39)37-30)19-28(36-32)22-40-3/h6-11,16,18-20,23,30-31,35,38H,4-5,12-15,17,21-22H2,1-3H3,(H,37,39)/t30-,31+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedicalResearch
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin E |
Bioorg Med Chem Lett 20: 603-7 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.092
BindingDB Entry DOI: 10.7270/Q2H1324C |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50112817
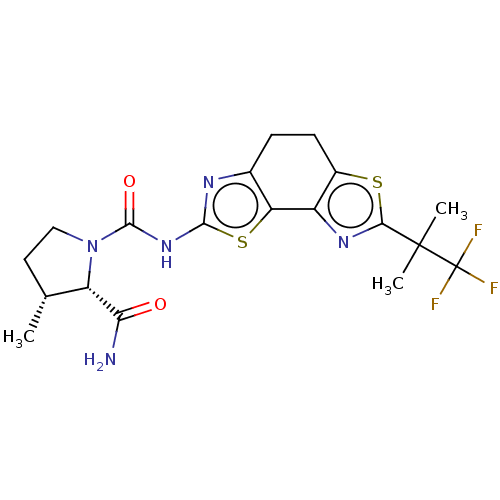
(CHEMBL3609520)Show SMILES C[C@@H]1CCN([C@@H]1C(N)=O)C(=O)Nc1nc2CCc3sc(nc3-c2s1)C(C)(C)C(F)(F)F |r| Show InChI InChI=1S/C19H22F3N5O2S2/c1-8-6-7-27(12(8)14(23)28)17(29)26-16-24-9-4-5-10-11(13(9)31-16)25-15(30-10)18(2,3)19(20,21)22/h8,12H,4-7H2,1-3H3,(H2,23,28)(H,24,26,29)/t8-,12+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of N-terminal myristoylated human PI3Kalpha expressed in Rat1 cells assessed as inhibition of Akt phosphorylatuion at Ser473 by ELISA |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 5
(Homo sapiens (Human)) | BDBM50305054
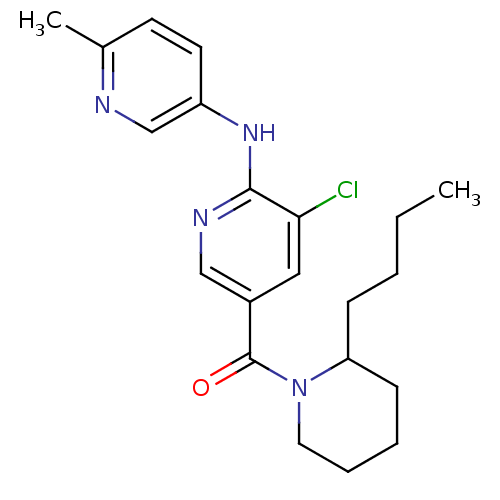
(CHEMBL594767 | rac-(2-butylpiperidin-1-yl)(5-chlor...)Show SMILES CCCCC1CCCCN1C(=O)c1cnc(Nc2ccc(C)nc2)c(Cl)c1 Show InChI InChI=1S/C21H27ClN4O/c1-3-4-7-18-8-5-6-11-26(18)21(27)16-12-19(22)20(24-13-16)25-17-10-9-15(2)23-14-17/h9-10,12-14,18H,3-8,11H2,1-2H3,(H,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for Biomedical Research
Curated by ChEMBL
| Assay Description
Antagonist activity at human mGluR5 assessed as inhibition of quisqualate-induced intracellular inositol phosphate accumulation |
Bioorg Med Chem Lett 20: 184-8 (2010)
Article DOI: 10.1016/j.bmcl.2009.11.001
BindingDB Entry DOI: 10.7270/Q2QN66VN |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50112820

(CHEMBL3609523)Show SMILES CC(C)(C)c1ncc2CCc3nc(NC(=O)N4CCC[C@@]4(C)C(N)=O)sc3-c2n1 |r| Show InChI InChI=1S/C20H26N6O2S/c1-19(2,3)16-22-10-11-6-7-12-14(13(11)24-16)29-17(23-12)25-18(28)26-9-5-8-20(26,4)15(21)27/h10H,5-9H2,1-4H3,(H2,21,27)(H,23,25,28)/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) using phosphatidylinositol as substrate preincubated for 15 mins followed by ATP addition measured after 1 h... |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50112824
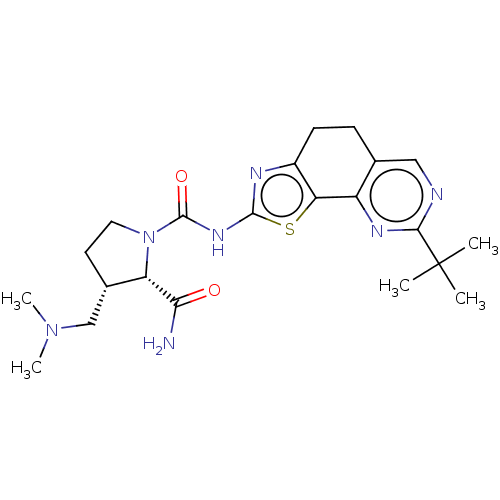
(CHEMBL3609527)Show SMILES CN(C)C[C@@H]1CCN([C@@H]1C(N)=O)C(=O)Nc1nc2CCc3cnc(nc3-c2s1)C(C)(C)C |r| Show InChI InChI=1S/C22H31N7O2S/c1-22(2,3)19-24-10-12-6-7-14-17(15(12)26-19)32-20(25-14)27-21(31)29-9-8-13(11-28(4)5)16(29)18(23)30/h10,13,16H,6-9,11H2,1-5H3,(H2,23,30)(H,25,27,31)/t13-,16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) using phosphatidylinositol as substrate preincubated for 15 mins followed by ATP addition measured after 1 h... |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit gamma isoform
(Homo sapiens (Human)) | BDBM50112822

(CHEMBL3609525)Show SMILES C[C@@H]1CCN([C@@H]1C(N)=O)C(=O)Nc1nc2CCc3cnc(nc3-c2s1)C(C)(C)C |r| Show InChI InChI=1S/C20H26N6O2S/c1-10-7-8-26(14(10)16(21)27)19(28)25-18-23-12-6-5-11-9-22-17(20(2,3)4)24-13(11)15(12)29-18/h9-10,14H,5-8H2,1-4H3,(H2,21,27)(H,23,25,28)/t10-,14+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kgamma (unknown origin) using PI or PIP2:PS as substrate by TR-FRET assay |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50112823

(CHEMBL3609526)Show SMILES C[C@@H]1CCN([C@@H]1C(N)=O)C(=O)Nc1nc2CCc3cnc(nc3-c2s1)C(C)(C)C(F)(F)F |r| Show InChI InChI=1S/C20H23F3N6O2S/c1-9-6-7-29(13(9)15(24)30)18(31)28-17-26-11-5-4-10-8-25-16(19(2,3)20(21,22)23)27-12(10)14(11)32-17/h8-9,13H,4-7H2,1-3H3,(H2,24,30)(H,26,28,31)/t9-,13+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) using phosphatidylinositol as substrate preincubated for 15 mins followed by ATP addition measured after 1 h... |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM50437053
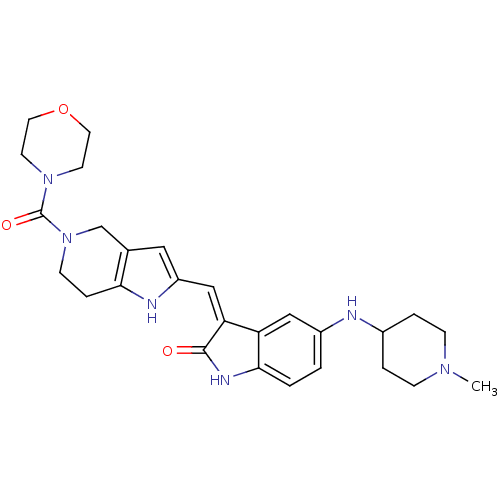
(CHEMBL2403373)Show SMILES CN1CCC(CC1)Nc1ccc2NC(=O)\C(=C/c3cc4CN(CCc4[nH]3)C(=O)N3CCOCC3)c2c1 Show InChI InChI=1S/C27H34N6O3/c1-31-7-4-19(5-8-31)28-20-2-3-25-22(15-20)23(26(34)30-25)16-21-14-18-17-33(9-6-24(18)29-21)27(35)32-10-12-36-13-11-32/h2-3,14-16,19,28-29H,4-13,17H2,1H3,(H,30,34)/b23-16- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of RET (unknown origin) |
Bioorg Med Chem Lett 23: 4085-90 (2013)
Article DOI: 10.1016/j.bmcl.2013.05.054
BindingDB Entry DOI: 10.7270/Q2SB474D |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Rattus norvegicus (Rat)) | BDBM92862

(US9284315, BEZ-235 | mTOR Inhibitor, BEZ235)Show SMILES Cn1c2cnc3ccc(cc3c2n(-c2ccc(cc2)C(C)(C)C#N)c1=O)-c1cnc2ccccc2c1 Show InChI InChI=1S/C30H23N5O/c1-30(2,18-31)22-9-11-23(12-10-22)35-28-24-15-19(21-14-20-6-4-5-7-25(20)32-16-21)8-13-26(24)33-17-27(28)34(3)29(35)36/h4-17H,1-3H3 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00267
BindingDB Entry DOI: 10.7270/Q28P64KJ |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50386483
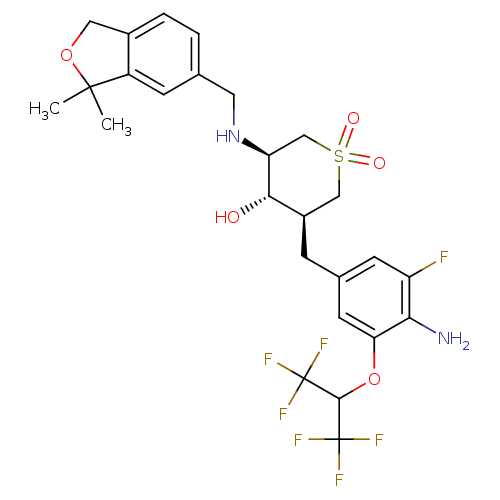
(CHEMBL2048060)Show SMILES CC1(C)OCc2ccc(CN[C@H]3CS(=O)(=O)C[C@@H](Cc4cc(F)c(N)c(OC(C(F)(F)F)C(F)(F)F)c4)[C@@H]3O)cc12 |r| Show InChI InChI=1S/C26H29F7N2O5S/c1-24(2)17-6-13(3-4-15(17)10-39-24)9-35-19-12-41(37,38)11-16(22(19)36)5-14-7-18(27)21(34)20(8-14)40-23(25(28,29)30)26(31,32)33/h3-4,6-8,16,19,22-23,35-36H,5,9-12,34H2,1-2H3/t16-,19+,22+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 ectodomain after 1 hr by fluorescence analysis |
J Med Chem 55: 3364-86 (2012)
Article DOI: 10.1021/jm300069y
BindingDB Entry DOI: 10.7270/Q2P55PJW |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50112819

(CHEMBL3609522)Show SMILES CC(C)(C)c1ncc2CCc3nc(NC(=O)N4CCC[C@H]4C(N)=O)sc3-c2n1 |r| Show InChI InChI=1S/C19H24N6O2S/c1-19(2,3)16-21-9-10-6-7-11-14(13(10)23-16)28-17(22-11)24-18(27)25-8-4-5-12(25)15(20)26/h9,12H,4-8H2,1-3H3,(H2,20,26)(H,22,24,27)/t12-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of N-terminal myristoylated human PI3Kalpha expressed in Rat1 cells assessed as inhibition of Akt phosphorylatuion at Ser473 by ELISA |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50112821

(CHEMBL3609524)Show SMILES [H][C@@]12C[C@@]1(N(CC2)C(=O)Nc1nc2CCc3cnc(nc3-c2s1)C(C)(C)C)C(N)=O |r| Show InChI InChI=1S/C20H24N6O2S/c1-19(2,3)16-22-9-10-4-5-12-14(13(10)24-16)29-17(23-12)25-18(28)26-7-6-11-8-20(11,26)15(21)27/h9,11H,4-8H2,1-3H3,(H2,21,27)(H,23,25,28)/t11-,20+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) using phosphatidylinositol as substrate preincubated for 15 mins followed by ATP addition measured after 1 h... |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM50437056

(CHEMBL2403369)Show SMILES COc1ccc2NC(=O)\C(=C/c3cc4CN(CCc4[nH]3)C(=O)N3CCN(C)CC3)c2c1 Show InChI InChI=1S/C23H27N5O3/c1-26-7-9-27(10-8-26)23(30)28-6-5-20-15(14-28)11-16(24-20)12-19-18-13-17(31-2)3-4-21(18)25-22(19)29/h3-4,11-13,24H,5-10,14H2,1-2H3,(H,25,29)/b19-12- | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of RET (unknown origin) |
Bioorg Med Chem Lett 23: 4085-90 (2013)
Article DOI: 10.1016/j.bmcl.2013.05.054
BindingDB Entry DOI: 10.7270/Q2SB474D |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50386508
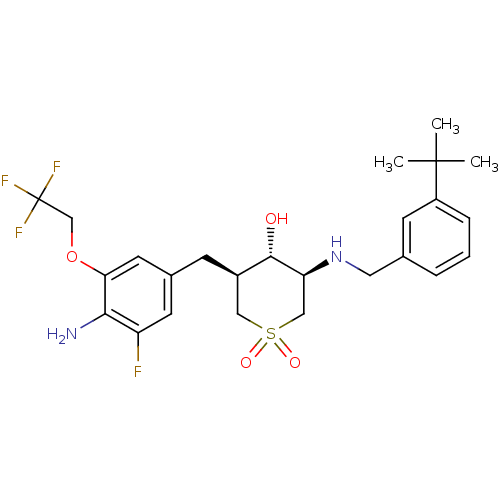
(CHEMBL2048045)Show SMILES CC(C)(C)c1cccc(CN[C@H]2CS(=O)(=O)C[C@@H](Cc3cc(F)c(N)c(OCC(F)(F)F)c3)[C@@H]2O)c1 |r| Show InChI InChI=1S/C25H32F4N2O4S/c1-24(2,3)18-6-4-5-15(8-18)11-31-20-13-36(33,34)12-17(23(20)32)7-16-9-19(26)22(30)21(10-16)35-14-25(27,28)29/h4-6,8-10,17,20,23,31-32H,7,11-14,30H2,1-3H3/t17-,20+,23+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 ectodomain after 1 hr by fluorescence analysis |
J Med Chem 55: 3364-86 (2012)
Article DOI: 10.1021/jm300069y
BindingDB Entry DOI: 10.7270/Q2P55PJW |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50386507
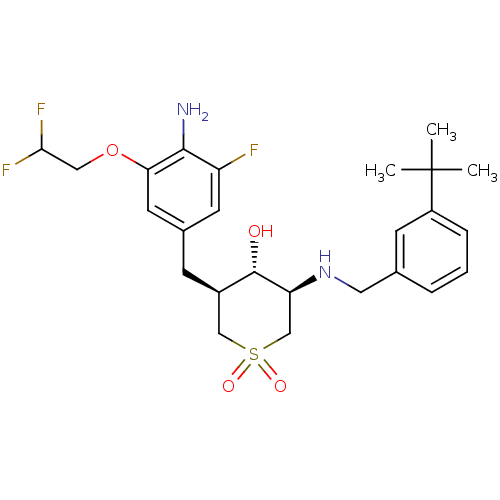
(CHEMBL2048044)Show SMILES CC(C)(C)c1cccc(CN[C@H]2CS(=O)(=O)C[C@@H](Cc3cc(F)c(N)c(OCC(F)F)c3)[C@@H]2O)c1 |r| Show InChI InChI=1S/C25H33F3N2O4S/c1-25(2,3)18-6-4-5-15(8-18)11-30-20-14-35(32,33)13-17(24(20)31)7-16-9-19(26)23(29)21(10-16)34-12-22(27)28/h4-6,8-10,17,20,22,24,30-31H,7,11-14,29H2,1-3H3/t17-,20+,24+/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Pharma AG
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant BACE1 ectodomain after 1 hr by fluorescence analysis |
J Med Chem 55: 3364-86 (2012)
Article DOI: 10.1021/jm300069y
BindingDB Entry DOI: 10.7270/Q2P55PJW |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit alpha isoform
(Homo sapiens (Human)) | BDBM50112822

(CHEMBL3609525)Show SMILES C[C@@H]1CCN([C@@H]1C(N)=O)C(=O)Nc1nc2CCc3cnc(nc3-c2s1)C(C)(C)C |r| Show InChI InChI=1S/C20H26N6O2S/c1-10-7-8-26(14(10)16(21)27)19(28)25-18-23-12-6-5-11-9-22-17(20(2,3)4)24-13(11)15(12)29-18/h9-10,14H,5-8H2,1-4H3,(H2,21,27)(H,23,25,28)/t10-,14+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institutes for BioMedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kalpha (unknown origin) using phosphatidylinositol as substrate preincubated for 15 mins followed by ATP addition measured after 1 h... |
Bioorg Med Chem Lett 25: 3575-81 (2015)
Article DOI: 10.1016/j.bmcl.2015.06.067
BindingDB Entry DOI: 10.7270/Q2DF6SZ5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data