 Found 1363 hits with Last Name = 'gould' and Initial = 'a'
Found 1363 hits with Last Name = 'gould' and Initial = 'a' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50550643
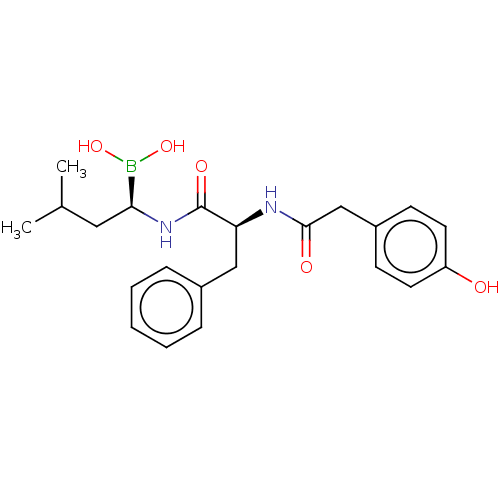
(CHEMBL4749207)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)Cc1ccc(O)cc1)B(O)O |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Binding affinity to human 20S constitutive proteasome beta 5 subunit assessed as equilibrium constant using fluorogenic peptide Ac-WLA-AMC as substra... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01161
BindingDB Entry DOI: 10.7270/Q2K077W3 |
More data for this
Ligand-Target Pair | |
Lethal factor
(Bacillus anthracis) | BDBM50236202
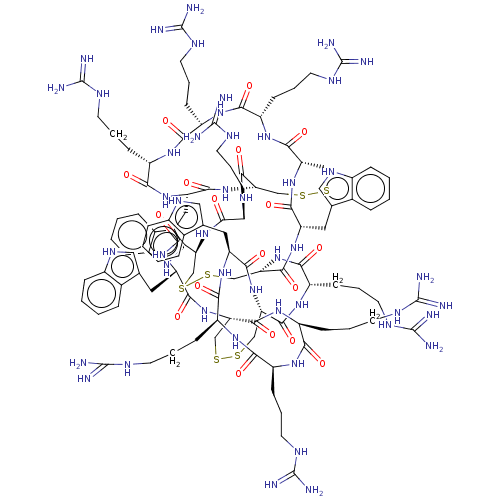
(CHEMBL4074171)Show SMILES NC(=N)NCCC[C@@H]1NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]2CSSC[C@H](NC(=O)[C@H](Cc3c[nH]c4ccccc34)NC(=O)[C@H](CCCNC(N)=N)NC1=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H]1CSSC[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]3CSSC[C@H](NC(=O)[C@H](Cc4c[nH]c5ccccc45)NC1=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c4ccccc14)C(=O)N3)C(=O)N[C@@H](Cc1c[nH]c3ccccc13)C(=O)N2 |r| Show InChI InChI=1S/C110H160N46O18S6/c111-103(112)127-33-9-25-67-85(157)141-71(29-13-37-131-107(119)120)89(161)147-75(41-55-45-135-63-21-5-1-17-59(55)63)93(165)153-81-51-177-175-49-79(97(169)143-69(87(159)139-67)27-11-35-129-105(115)116)155-95(167)77(43-57-47-137-65-23-7-3-19-61(57)65)150-102(174)84-54-180-179-53-83(151-91(163)73(145-99(81)171)31-15-39-133-109(123)124)101(173)149-78(44-58-48-138-66-24-8-4-20-62(58)66)96(168)156-80-50-176-178-52-82(100(172)146-74(92(164)152-84)32-16-40-134-110(125)126)154-94(166)76(42-56-46-136-64-22-6-2-18-60(56)64)148-90(162)72(30-14-38-132-108(121)122)142-86(158)68(26-10-34-128-104(113)114)140-88(160)70(144-98(80)170)28-12-36-130-106(117)118/h1-8,17-24,45-48,67-84,135-138H,9-16,25-44,49-54H2,(H,139,159)(H,140,160)(H,141,157)(H,142,158)(H,143,169)(H,144,170)(H,145,171)(H,146,172)(H,147,161)(H,148,162)(H,149,173)(H,150,174)(H,151,163)(H,152,164)(H,153,165)(H,154,166)(H,155,167)(H,156,168)(H4,111,112,127)(H4,113,114,128)(H4,115,116,129)(H4,117,118,130)(H4,119,120,131)(H4,121,122,132)(H4,123,124,133)(H4,125,126,134)/t67-,68-,69-,70-,71-,72-,73-,74-,75-,76-,77-,78-,79-,80-,81-,82-,83-,84-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
State University of New York
Curated by ChEMBL
| Assay Description
Inhibition of Bacillus anthracis lethal factor protease preincubated for 30 mins prior to addition of substrate containing fluorescent proteins CyPet... |
J Med Chem 60: 1916-1927 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01689
BindingDB Entry DOI: 10.7270/Q2377C05 |
More data for this
Ligand-Target Pair | |
Lethal factor
(Bacillus anthracis) | BDBM50236198
(CHEMBL4073105)Show SMILES CC[C@H](C)[C@@H]1NC(=O)[C@@H]2CSSC[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]3CSSC[C@H](NC1=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(=O)N[C@@H](Cc1ccccc1)C(=O)N3)C(=O)N[C@@H](CC(C)C)C(=O)N[C@H]1CSSC[C@H](NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC1=O)C(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N2 |r| Show InChI InChI=1S/C82H137N33O19S6/c1-8-41(6)60-76(133)112-56-38-140-136-34-52(110-68(125)50(30-43-17-10-9-11-18-43)100-57(117)31-98-63(120)45(20-13-25-94-79(85)86)105-77(134)61(42(7)116)115-74(56)131)70(127)103-47(22-15-27-96-81(89)90)65(122)107-53-35-137-139-37-55(73(130)114-60)108-66(123)48(23-16-28-97-82(91)92)104-71(128)54-36-138-135-33-51(109-67(124)49(29-39(2)3)106-72(53)129)69(126)102-46(21-14-26-95-80(87)88)64(121)101-44(19-12-24-93-78(83)84)62(119)99-32-58(118)113-59(40(4)5)75(132)111-54/h9-11,17-18,39-42,44-56,59-61,116H,8,12-16,19-38H2,1-7H3,(H,98,120)(H,99,119)(H,100,117)(H,101,121)(H,102,126)(H,103,127)(H,104,128)(H,105,134)(H,106,129)(H,107,122)(H,108,123)(H,109,124)(H,110,125)(H,111,132)(H,112,133)(H,113,118)(H,114,130)(H,115,131)(H4,83,84,93)(H4,85,86,94)(H4,87,88,95)(H4,89,90,96)(H4,91,92,97)/t41-,42+,44-,45-,46-,47-,48-,49-,50-,51-,52-,53-,54-,55-,56-,59-,60-,61-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 365 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
State University of New York
Curated by ChEMBL
| Assay Description
Inhibition of Bacillus anthracis lethal factor protease preincubated for 30 mins prior to addition of substrate containing fluorescent proteins CyPet... |
J Med Chem 60: 1916-1927 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01689
BindingDB Entry DOI: 10.7270/Q2377C05 |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-2
(Homo sapiens (Human)) | BDBM50550643

(CHEMBL4749207)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)Cc1ccc(O)cc1)B(O)O |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 487 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Binding affinity to human 20S constitutive proteasome beta 2 subunit assessed as equilibrium constant using fluorogenic peptide Ac-WLR-AMC as substra... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01161
BindingDB Entry DOI: 10.7270/Q2K077W3 |
More data for this
Ligand-Target Pair | |
Disintegrin and metalloproteinase domain-containing protein 17
(Homo sapiens (Human)) | BDBM50236202

(CHEMBL4074171)Show SMILES NC(=N)NCCC[C@@H]1NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]2CSSC[C@H](NC(=O)[C@H](Cc3c[nH]c4ccccc34)NC(=O)[C@H](CCCNC(N)=N)NC1=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H]1CSSC[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]3CSSC[C@H](NC(=O)[C@H](Cc4c[nH]c5ccccc45)NC1=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](Cc1c[nH]c4ccccc14)C(=O)N3)C(=O)N[C@@H](Cc1c[nH]c3ccccc13)C(=O)N2 |r| Show InChI InChI=1S/C110H160N46O18S6/c111-103(112)127-33-9-25-67-85(157)141-71(29-13-37-131-107(119)120)89(161)147-75(41-55-45-135-63-21-5-1-17-59(55)63)93(165)153-81-51-177-175-49-79(97(169)143-69(87(159)139-67)27-11-35-129-105(115)116)155-95(167)77(43-57-47-137-65-23-7-3-19-61(57)65)150-102(174)84-54-180-179-53-83(151-91(163)73(145-99(81)171)31-15-39-133-109(123)124)101(173)149-78(44-58-48-138-66-24-8-4-20-62(58)66)96(168)156-80-50-176-178-52-82(100(172)146-74(92(164)152-84)32-16-40-134-110(125)126)154-94(166)76(42-56-46-136-64-22-6-2-18-60(56)64)148-90(162)72(30-14-38-132-108(121)122)142-86(158)68(26-10-34-128-104(113)114)140-88(160)70(144-98(80)170)28-12-36-130-106(117)118/h1-8,17-24,45-48,67-84,135-138H,9-16,25-44,49-54H2,(H,139,159)(H,140,160)(H,141,157)(H,142,158)(H,143,169)(H,144,170)(H,145,171)(H,146,172)(H,147,161)(H,148,162)(H,149,173)(H,150,174)(H,151,163)(H,152,164)(H,153,165)(H,154,166)(H,155,167)(H,156,168)(H4,111,112,127)(H4,113,114,128)(H4,115,116,129)(H4,117,118,130)(H4,119,120,131)(H4,121,122,132)(H4,123,124,133)(H4,125,126,134)/t67-,68-,69-,70-,71-,72-,73-,74-,75-,76-,77-,78-,79-,80-,81-,82-,83-,84-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
State University of New York
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TACE extracellular domain (215 to 671 residues) assessed as inhibition of proteolytic activity preincubated for 30 mi... |
J Med Chem 60: 1916-1927 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01689
BindingDB Entry DOI: 10.7270/Q2377C05 |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM21489
((2S)-2-{[(1S)-1-carboxy-2-{1-[(3,5-dichlorophenyl)...)Show SMILES CC(C)C[C@H](N[C@@H](Cc1cncn1Cc1cc(Cl)cc(Cl)c1)C(O)=O)C(O)=O |r| Show InChI InChI=1S/C19H23Cl2N3O4/c1-11(2)3-16(18(25)26)23-17(19(27)28)7-15-8-22-10-24(15)9-12-4-13(20)6-14(21)5-12/h4-6,8,10-11,16-17,23H,3,7,9H2,1-2H3,(H,25,26)(H,27,28)/t16-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| n/a | n/a | 0.440 | n/a | n/a | n/a | n/a | 6.5 | 22 |
Millennium Pharmaceuticals
| Assay Description
Enzyme assay was conducted in 384-well microplates format. Activity was monitored by measuring increase in fluorescence (excitation @320 nm, emission... |
J Am Chem Soc 124: 11852-3 (2002)
Article DOI: 10.1021/ja0277226
BindingDB Entry DOI: 10.7270/Q2KP80FN |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM50339608
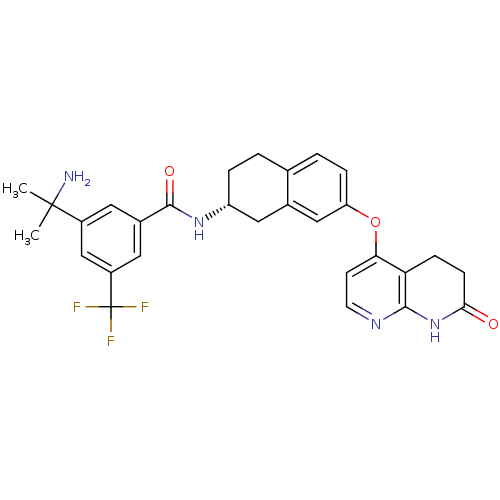
(3-(1-Amino-1-methylethyl)-N-{(2R)-7-[(7-oxo-5,6,7,...)Show SMILES CC(C)(N)c1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H29F3N4O3/c1-28(2,33)19-11-18(12-20(15-19)29(30,31)32)27(38)35-21-5-3-16-4-6-22(14-17(16)13-21)39-24-9-10-34-26-23(24)7-8-25(37)36-26/h4,6,9-12,14-15,21H,3,5,7-8,13,33H2,1-2H3,(H,35,38)(H,34,36,37)/t21-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of RET |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50339609
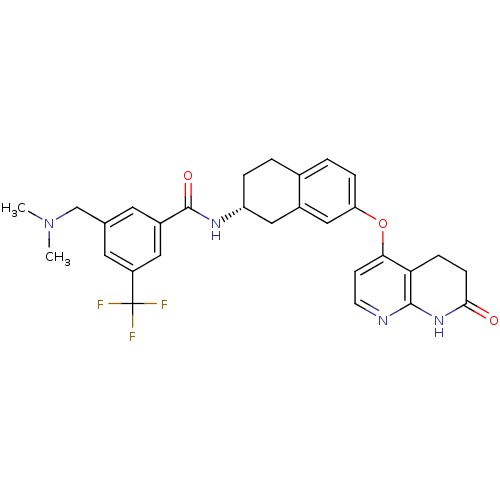
(3-[(Dimethylamino)methyl]-N-{(2R)-7-[(7-oxo-5,6,7,...)Show SMILES CN(C)Cc1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H29F3N4O3/c1-36(2)16-17-11-20(13-21(12-17)29(30,31)32)28(38)34-22-5-3-18-4-6-23(15-19(18)14-22)39-25-9-10-33-27-24(25)7-8-26(37)35-27/h4,6,9-13,15,22H,3,5,7-8,14,16H2,1-2H3,(H,34,38)(H,33,35,37)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Abl1 |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-8
(Homo sapiens (Human)) | BDBM50550643

(CHEMBL4749207)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)Cc1ccc(O)cc1)B(O)O |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human 20S immunoproteasome beta 5 subunit chymotrypsin-like activity using fluorogenic peptide Ac-WLA-AMC as substrate in presence of P... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01161
BindingDB Entry DOI: 10.7270/Q2K077W3 |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50550643

(CHEMBL4749207)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)Cc1ccc(O)cc1)B(O)O |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human 20S constitutive proteasome beta 5 subunit chymotrypsin-like activity using fluorogenic peptide Ac-WLA-AMC as substrate in presen... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01161
BindingDB Entry DOI: 10.7270/Q2K077W3 |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM21488
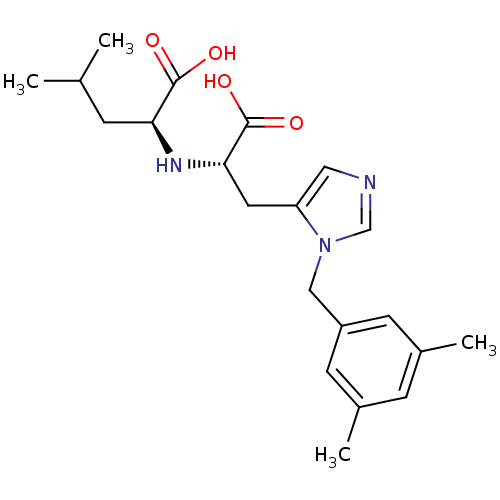
((2S)-2-{[(1S)-1-carboxy-2-{1-[(3,5-dimethylphenyl)...)Show SMILES CC(C)C[C@H](N[C@@H](Cc1cncn1Cc1cc(C)cc(C)c1)C(O)=O)C(O)=O |r| Show InChI InChI=1S/C21H29N3O4/c1-13(2)5-18(20(25)26)23-19(21(27)28)9-17-10-22-12-24(17)11-16-7-14(3)6-15(4)8-16/h6-8,10,12-13,18-19,23H,5,9,11H2,1-4H3,(H,25,26)(H,27,28)/t18-,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | 6.5 | 22 |
Millennium Pharmaceuticals
| Assay Description
Enzyme assay was conducted in 384-well microplates format. Activity was monitored by measuring increase in fluorescence (excitation @320 nm, emission... |
J Am Chem Soc 124: 11852-3 (2002)
Article DOI: 10.1021/ja0277226
BindingDB Entry DOI: 10.7270/Q2KP80FN |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50139157

(CHEMBL3764588)Show SMILES Cl.Cn1nccc1-c1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C30H26F3N5O3.ClH/c1-38-25(8-11-35-38)19-12-20(14-21(13-19)30(31,32)33)29(40)36-22-4-2-17-3-5-23(16-18(17)15-22)41-26-9-10-34-28-24(26)6-7-27(39)37-28;/h3,5,8-14,16,22H,2,4,6-7,15H2,1H3,(H,36,40)(H,34,37,39);1H/t22-;/m1./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-3... |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase receptor Ret
(Homo sapiens (Human)) | BDBM50339609

(3-[(Dimethylamino)methyl]-N-{(2R)-7-[(7-oxo-5,6,7,...)Show SMILES CN(C)Cc1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H29F3N4O3/c1-36(2)16-17-11-20(13-21(12-17)29(30,31)32)28(38)34-22-5-3-18-4-6-23(15-19(18)14-22)39-25-9-10-33-27-24(25)7-8-26(37)35-27/h4,6,9-13,15,22H,3,5,7-8,14,16H2,1-2H3,(H,34,38)(H,33,35,37)/t22-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of RET |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50139150
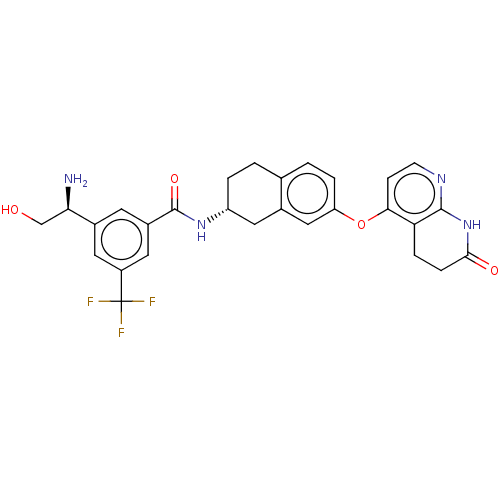
(CHEMBL3763999)Show SMILES Cl.N[C@H](CO)c1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C28H27F3N4O4.ClH/c29-28(30,31)19-10-17(23(32)14-36)9-18(11-19)27(38)34-20-3-1-15-2-4-21(13-16(15)12-20)39-24-7-8-33-26-22(24)5-6-25(37)35-26;/h2,4,7-11,13,20,23,36H,1,3,5-6,12,14,32H2,(H,34,38)(H,33,35,37);1H/t20-,23-;/m1./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-3... |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50139149

(CHEMBL3765012)Show SMILES Cl.Cl.NC1(CCC1)c1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C30H29F3N4O3.2ClH/c31-30(32,33)21-13-19(12-20(16-21)29(34)9-1-10-29)28(39)36-22-4-2-17-3-5-23(15-18(17)14-22)40-25-8-11-35-27-24(25)6-7-26(38)37-27;;/h3,5,8,11-13,15-16,22H,1-2,4,6-7,9-10,14,34H2,(H,36,39)(H,35,37,38);2*1H/t22-;;/m1../s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-3... |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50139147
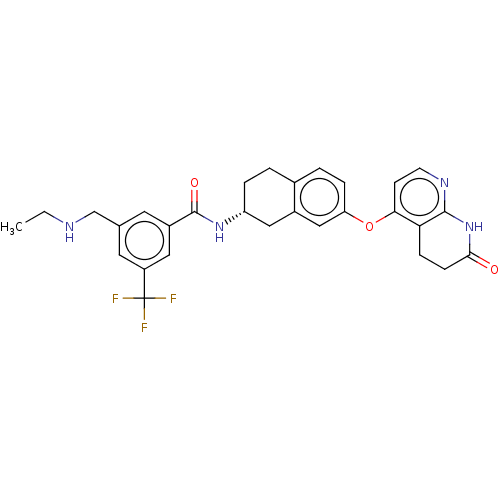
(CHEMBL3765509)Show SMILES Cl.Cl.CCNCc1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H29F3N4O3.2ClH/c1-2-33-16-17-11-20(13-21(12-17)29(30,31)32)28(38)35-22-5-3-18-4-6-23(15-19(18)14-22)39-25-9-10-34-27-24(25)7-8-26(37)36-27;;/h4,6,9-13,15,22,33H,2-3,5,7-8,14,16H2,1H3,(H,35,38)(H,34,36,37);2*1H/t22-;;/m1../s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-3... |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-8
(Homo sapiens (Human)) | BDBM50069989

((R)-3-methyl-1-((S)-3-phenyl-2-(pyrazine-2-carboxa...)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)c1cnccn1)B(O)O |r| Show InChI InChI=1S/C19H25BN4O4/c1-13(2)10-17(20(27)28)24-18(25)15(11-14-6-4-3-5-7-14)23-19(26)16-12-21-8-9-22-16/h3-9,12-13,15,17,27-28H,10-11H2,1-2H3,(H,23,26)(H,24,25)/t15-,17-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human 20S immunoproteasome beta 5 subunit chymotrypsin-like activity using fluorogenic peptide Ac-WLA-AMC as substrate in presence of P... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01161
BindingDB Entry DOI: 10.7270/Q2K077W3 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50139153
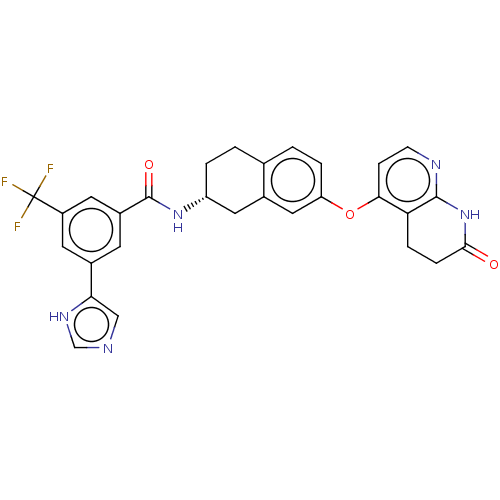
(CHEMBL3765355)Show SMILES FC(F)(F)c1cc(cc(c1)-c1cnc[nH]1)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H24F3N5O3/c30-29(31,32)20-10-18(24-14-33-15-35-24)9-19(11-20)28(39)36-21-3-1-16-2-4-22(13-17(16)12-21)40-25-7-8-34-27-23(25)5-6-26(38)37-27/h2,4,7-11,13-15,21H,1,3,5-6,12H2,(H,33,35)(H,36,39)(H,34,37,38)/t21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-3... |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50339608

(3-(1-Amino-1-methylethyl)-N-{(2R)-7-[(7-oxo-5,6,7,...)Show SMILES CC(C)(N)c1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H29F3N4O3/c1-28(2,33)19-11-18(12-20(15-19)29(30,31)32)27(38)35-21-5-3-16-4-6-22(14-17(16)13-21)39-24-9-10-34-26-23(24)7-8-25(37)36-26/h4,6,9-12,14-15,21H,3,5,7-8,13,33H2,1-2H3,(H,35,38)(H,34,36,37)/t21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-3... |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50139158
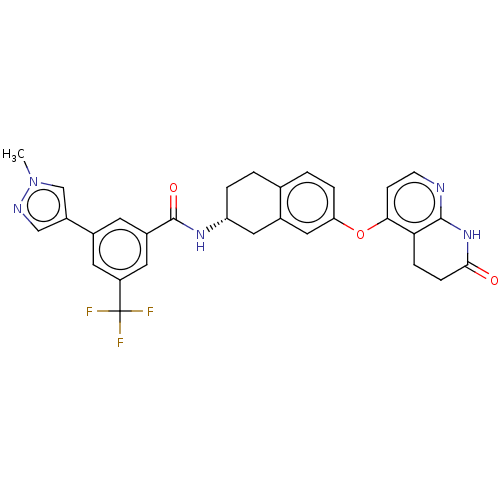
(CHEMBL3763646)Show SMILES Cl.Cn1cc(cn1)-c1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C30H26F3N5O3.ClH/c1-38-16-21(15-35-38)18-10-20(12-22(11-18)30(31,32)33)29(40)36-23-4-2-17-3-5-24(14-19(17)13-23)41-26-8-9-34-28-25(26)6-7-27(39)37-28;/h3,5,8-12,14-16,23H,2,4,6-7,13H2,1H3,(H,36,40)(H,34,37,39);1H/t23-;/m1./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-3... |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50139156
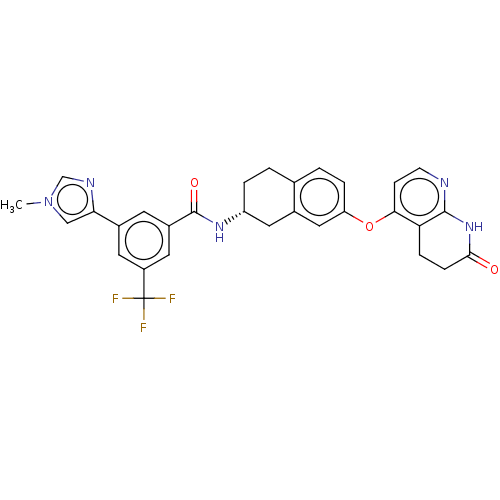
(CHEMBL3763694)Show SMILES Cl.Cl.Cn1cnc(c1)-c1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C30H26F3N5O3.2ClH/c1-38-15-25(35-16-38)19-10-20(12-21(11-19)30(31,32)33)29(40)36-22-4-2-17-3-5-23(14-18(17)13-22)41-26-8-9-34-28-24(26)6-7-27(39)37-28;;/h3,5,8-12,14-16,22H,2,4,6-7,13H2,1H3,(H,36,40)(H,34,37,39);2*1H/t22-;;/m1../s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-3... |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50139151

(CHEMBL3764870)Show SMILES Cl.Cl.OC1(CNC1)c1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H27F3N4O4.2ClH/c30-29(31,32)20-10-18(9-19(13-20)28(39)14-33-15-28)27(38)35-21-3-1-16-2-4-22(12-17(16)11-21)40-24-7-8-34-26-23(24)5-6-25(37)36-26;;/h2,4,7-10,12-13,21,33,39H,1,3,5-6,11,14-15H2,(H,35,38)(H,34,36,37);2*1H/t21-;;/m1../s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-3... |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50339609

(3-[(Dimethylamino)methyl]-N-{(2R)-7-[(7-oxo-5,6,7,...)Show SMILES CN(C)Cc1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H29F3N4O3/c1-36(2)16-17-11-20(13-21(12-17)29(30,31)32)28(38)34-22-5-3-18-4-6-23(15-19(18)14-22)39-25-9-10-33-27-24(25)7-8-26(37)35-27/h4,6,9-13,15,22H,3,5,7-8,14,16H2,1-2H3,(H,34,38)(H,33,35,37)/t22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of C-Raf |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50339621
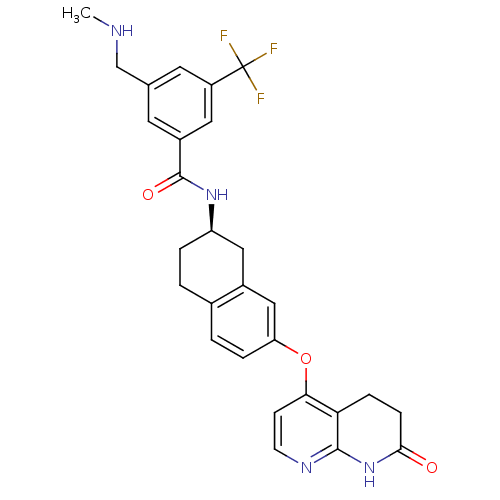
(3-[(methylamino)methyl]-N-{(2R)-7-[(7-oxo-5,6,7,8-...)Show SMILES CNCc1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C28H27F3N4O3/c1-32-15-16-10-19(12-20(11-16)28(29,30)31)27(37)34-21-4-2-17-3-5-22(14-18(17)13-21)38-24-8-9-33-26-23(24)6-7-25(36)35-26/h3,5,8-12,14,21,32H,2,4,6-7,13,15H2,1H3,(H,34,37)(H,33,35,36)/t21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50339608

(3-(1-Amino-1-methylethyl)-N-{(2R)-7-[(7-oxo-5,6,7,...)Show SMILES CC(C)(N)c1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H29F3N4O3/c1-28(2,33)19-11-18(12-20(15-19)29(30,31)32)27(38)35-21-5-3-16-4-6-22(14-17(16)13-21)39-24-9-10-34-26-23(24)7-8-25(37)36-26/h4,6,9-12,14-15,21H,3,5,7-8,13,33H2,1-2H3,(H,35,38)(H,34,36,37)/t21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of C-Raf |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
Discoidin domain-containing receptor 2
(Homo sapiens (Human)) | BDBM50339609

(3-[(Dimethylamino)methyl]-N-{(2R)-7-[(7-oxo-5,6,7,...)Show SMILES CN(C)Cc1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H29F3N4O3/c1-36(2)16-17-11-20(13-21(12-17)29(30,31)32)28(38)34-22-5-3-18-4-6-23(15-19(18)14-22)39-25-9-10-33-27-24(25)7-8-26(37)35-27/h4,6,9-13,15,22H,3,5,7-8,14,16H2,1-2H3,(H,34,38)(H,33,35,37)/t22-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of DDR2 |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50339609

(3-[(Dimethylamino)methyl]-N-{(2R)-7-[(7-oxo-5,6,7,...)Show SMILES CN(C)Cc1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H29F3N4O3/c1-36(2)16-17-11-20(13-21(12-17)29(30,31)32)28(38)34-22-5-3-18-4-6-23(15-19(18)14-22)39-25-9-10-33-27-24(25)7-8-26(37)35-27/h4,6,9-13,15,22H,3,5,7-8,14,16H2,1-2H3,(H,34,38)(H,33,35,37)/t22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50139148
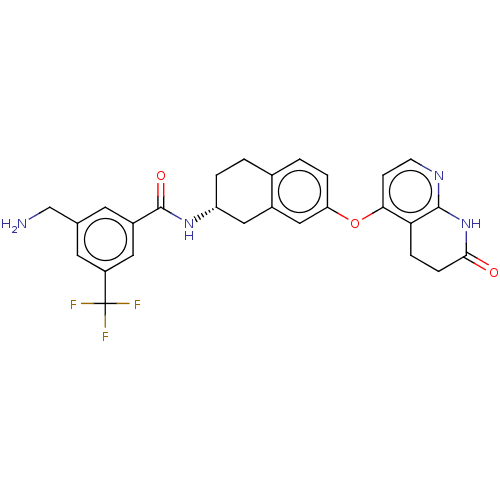
(CHEMBL3764101)Show SMILES Cl.Cl.NCc1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C27H25F3N4O3.2ClH/c28-27(29,30)19-10-15(14-31)9-18(11-19)26(36)33-20-3-1-16-2-4-21(13-17(16)12-20)37-23-7-8-32-25-22(23)5-6-24(35)34-25;;/h2,4,7-11,13,20H,1,3,5-6,12,14,31H2,(H,33,36)(H,32,34,35);2*1H/t20-;;/m1../s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-3... |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50139155
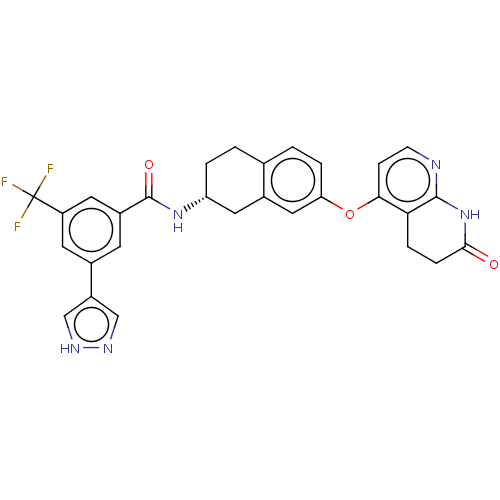
(CHEMBL3765708)Show SMILES Cl.FC(F)(F)c1cc(cc(c1)-c1cn[nH]c1)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H24F3N5O3.ClH/c30-29(31,32)21-10-17(20-14-34-35-15-20)9-19(11-21)28(39)36-22-3-1-16-2-4-23(13-18(16)12-22)40-25-7-8-33-27-24(25)5-6-26(38)37-27;/h2,4,7-11,13-15,22H,1,3,5-6,12H2,(H,34,35)(H,36,39)(H,33,37,38);1H/t22-;/m1./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-3... |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50139159
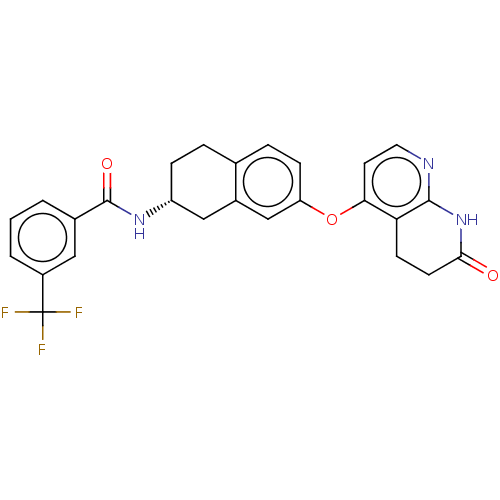
(CHEMBL3765596)Show SMILES FC(F)(F)c1cccc(c1)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C26H22F3N3O3/c27-26(28,29)18-3-1-2-16(12-18)25(34)31-19-6-4-15-5-7-20(14-17(15)13-19)35-22-10-11-30-24-21(22)8-9-23(33)32-24/h1-3,5,7,10-12,14,19H,4,6,8-9,13H2,(H,31,34)(H,30,32,33)/t19-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50339615

((2S)-7-({2-[(Cyclopropylcarbonyl)amino]pyridin-4-y...)Show SMILES CN(C)Cc1cc(NC(=O)[C@H]2CCc3ccc(Oc4ccnc(NC(=O)C5CC5)c4)cc3C2)cc(c1)C(F)(F)F |r| Show InChI InChI=1S/C30H31F3N4O3/c1-37(2)17-18-11-23(30(31,32)33)15-24(12-18)35-29(39)21-6-3-19-7-8-25(14-22(19)13-21)40-26-9-10-34-27(16-26)36-28(38)20-4-5-20/h7-12,14-16,20-21H,3-6,13,17H2,1-2H3,(H,35,39)(H,34,36,38)/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of C-Raf assessed as reduction in [33P]ATP incorporation into biotinylated substrate peptide after 3 hrs by flash plate assay |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50322823

((S)-N-(4-(3-chloro-4-fluorophenylamino)-7-(tetrahy...)Show SMILES CN(C)C\C=C\C(=O)Nc1cc2c(Nc3ccc(F)c(Cl)c3)ncnc2cc1O[C@H]1CCOC1 |r| Show InChI InChI=1S/C24H25ClFN5O3/c1-31(2)8-3-4-23(32)30-21-11-17-20(12-22(21)34-16-7-9-33-13-16)27-14-28-24(17)29-15-5-6-19(26)18(25)10-15/h3-6,10-12,14,16H,7-9,13H2,1-2H3,(H,30,32)(H,27,28,29)/b4-3+/t16-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
|
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Disintegrin and metalloproteinase domain-containing protein 17
(Homo sapiens (Human)) | BDBM50236205

(CHEMBL4064688)Show SMILES CC[C@H](C)[C@@H]1NC(=O)[C@@H]2CSSC[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]3CSSC[C@H](NC1=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)NCC(=O)N[C@@H](Cc1ccccc1)C(=O)N3)C(=O)N[C@@H](CC(C)C)C(=O)N[C@H]1CSSC[C@H](NC(=O)[C@H](Cc3c[nH]c4ccccc34)NC(=O)CNC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC1=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N2 |r| Show InChI InChI=1S/C88H138N34O19S6/c1-6-44(4)66-82(140)120-63-42-147-144-38-59(118-74(132)56(32-46-17-8-7-9-18-46)107-64(124)35-106-69(127)51(22-13-27-100-85(91)92)113-83(141)67(45(5)123)122-81(63)139)77(135)111-53(24-15-29-102-87(95)96)71(129)115-61-40-145-146-41-62(80(138)121-66)116-72(130)54(25-16-30-103-88(97)98)112-78(136)60-39-143-142-37-58(117-73(131)55(31-43(2)3)114-79(61)137)76(134)110-52(23-14-28-101-86(93)94)70(128)109-50(21-12-26-99-84(89)90)68(126)105-36-65(125)108-57(75(133)119-60)33-47-34-104-49-20-11-10-19-48(47)49/h7-11,17-20,34,43-45,50-63,66-67,104,123H,6,12-16,21-33,35-42H2,1-5H3,(H,105,126)(H,106,127)(H,107,124)(H,108,125)(H,109,128)(H,110,134)(H,111,135)(H,112,136)(H,113,141)(H,114,137)(H,115,129)(H,116,130)(H,117,131)(H,118,132)(H,119,133)(H,120,140)(H,121,138)(H,122,139)(H4,89,90,99)(H4,91,92,100)(H4,93,94,101)(H4,95,96,102)(H4,97,98,103)/t44-,45+,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,66-,67-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.80 | n/a | n/a | n/a | n/a | n/a | n/a |
State University of New York
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human TACE extracellular domain (215 to 671 residues) assessed as inhibition of proteolytic activity preincubated for 30 mi... |
J Med Chem 60: 1916-1927 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01689
BindingDB Entry DOI: 10.7270/Q2377C05 |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50139152

(CHEMBL3764275)Show SMILES OCC1COCCN1Cc1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C32H33F3N4O5/c33-32(34,35)23-12-19(16-39-9-10-43-18-25(39)17-40)11-22(13-23)31(42)37-24-3-1-20-2-4-26(15-21(20)14-24)44-28-7-8-36-30-27(28)5-6-29(41)38-30/h2,4,7-8,11-13,15,24-25,40H,1,3,5-6,9-10,14,16-18H2,(H,37,42)(H,36,38,41)/t24-,25?/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceuticals International Co. 40 Landsdowne Street, Cambridge, MA 02139, United States.
Curated by ChEMBL
| Assay Description
Inhibition of CDK2/Cyclin E (unknown origin) expressed in baculoviral infected insect Sf9 cells using histone H1 as substrate in presence of [gamma-3... |
Bioorg Med Chem Lett 26: 1156-60 (2016)
Article DOI: 10.1016/j.bmcl.2016.01.049
BindingDB Entry DOI: 10.7270/Q29P33HD |
More data for this
Ligand-Target Pair | |
Histone deacetylase 6
(Homo sapiens (Human)) | BDBM22449

(CHEMBL356769 | N-(4-{(2R,4R,6S)-4-{[(4,5-diphenyl-...)Show SMILES OCc1ccc(cc1)[C@@H]1C[C@H](CSc2nc(c(o2)-c2ccccc2)-c2ccccc2)O[C@@H](O1)c1ccc(NC(=O)CCCCCCC(=O)NO)cc1 |r| Show InChI InChI=1S/C41H43N3O7S/c45-26-28-17-19-29(20-18-28)35-25-34(27-52-41-43-38(30-11-5-3-6-12-30)39(51-41)31-13-7-4-8-14-31)49-40(50-35)32-21-23-33(24-22-32)42-36(46)15-9-1-2-10-16-37(47)44-48/h3-8,11-14,17-24,34-35,40,45,48H,1-2,9-10,15-16,25-27H2,(H,42,46)(H,44,47)/t34-,35+,40+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of recombinant N-GST-tagged HDAC6 (unknown origin) assessed as reduction in deacetylation of Ac-Arg-Gly-Lys(Ac)-AMC substrate by fluoresce... |
Bioorg Med Chem Lett 24: 5450-4 (2015)
Article DOI: 10.1016/j.bmcl.2014.10.022
BindingDB Entry DOI: 10.7270/Q2B859QH |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50339608

(3-(1-Amino-1-methylethyl)-N-{(2R)-7-[(7-oxo-5,6,7,...)Show SMILES CC(C)(N)c1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H29F3N4O3/c1-28(2,33)19-11-18(12-20(15-19)29(30,31)32)27(38)35-21-5-3-16-4-6-22(14-17(16)13-21)39-24-9-10-34-26-23(24)7-8-25(37)36-26/h4,6,9-12,14-15,21H,3,5,7-8,13,33H2,1-2H3,(H,35,38)(H,34,36,37)/t21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of wild-type B-Raf |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
Angiotensin-converting enzyme 2
(Homo sapiens (Human)) | BDBM21486

((2S)-2-{[(1S)-1-carboxy-2-{1-[(3-methylphenyl)meth...)Show SMILES CC(C)C[C@H](N[C@@H](Cc1cncn1Cc1cccc(C)c1)C(O)=O)C(O)=O |r| Show InChI InChI=1S/C20H27N3O4/c1-13(2)7-17(19(24)25)22-18(20(26)27)9-16-10-21-12-23(16)11-15-6-4-5-14(3)8-15/h4-6,8,10,12-13,17-18,22H,7,9,11H2,1-3H3,(H,24,25)(H,26,27)/t17-,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | 6.5 | 22 |
Millennium Pharmaceuticals
| Assay Description
Enzyme assay was conducted in 384-well microplates format. Activity was monitored by measuring increase in fluorescence (excitation @320 nm, emission... |
J Am Chem Soc 124: 11852-3 (2002)
Article DOI: 10.1021/ja0277226
BindingDB Entry DOI: 10.7270/Q2KP80FN |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50339613

((2S)-N-[3-(aminomethyl)-5-(trifluoromethyl)phenyl]...)Show SMILES NCc1cc(NC(=O)[C@H]2CCc3ccc(Oc4ccnc(NC(=O)C5CC5)c4)cc3C2)cc(c1)C(F)(F)F |r| Show InChI InChI=1S/C28H27F3N4O3/c29-28(30,31)21-9-16(15-32)10-22(13-21)34-27(37)19-4-1-17-5-6-23(12-20(17)11-19)38-24-7-8-33-25(14-24)35-26(36)18-2-3-18/h5-10,12-14,18-19H,1-4,11,15,32H2,(H,34,37)(H,33,35,36)/t19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of C-Raf assessed as reduction in [33P]ATP incorporation into biotinylated substrate peptide after 3 hrs by flash plate assay |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50339616

(7-({2-[(Cyclopropylcarbonyl)amino]pyridin-4-yl}oxy...)Show SMILES CC(C)NCc1cc(NC(=O)C2CCc3ccc(Oc4ccnc(NC(=O)C5CC5)c4)cc3C2)cc(c1)C(F)(F)F Show InChI InChI=1S/C31H33F3N4O3/c1-18(2)36-17-19-11-24(31(32,33)34)15-25(12-19)37-30(40)22-6-3-20-7-8-26(14-23(20)13-22)41-27-9-10-35-28(16-27)38-29(39)21-4-5-21/h7-12,14-16,18,21-22,36H,3-6,13,17H2,1-2H3,(H,37,40)(H,35,38,39) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of C-Raf assessed as reduction in [33P]ATP incorporation into biotinylated substrate peptide after 3 hrs by flash plate assay |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase B-raf
(Homo sapiens (Human)) | BDBM50339609

(3-[(Dimethylamino)methyl]-N-{(2R)-7-[(7-oxo-5,6,7,...)Show SMILES CN(C)Cc1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H29F3N4O3/c1-36(2)16-17-11-20(13-21(12-17)29(30,31)32)28(38)34-22-5-3-18-4-6-23(15-19(18)14-22)39-25-9-10-33-27-24(25)7-8-26(37)35-27/h4,6,9-13,15,22H,3,5,7-8,14,16H2,1-2H3,(H,34,38)(H,33,35,37)/t22-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of wild-type B-Raf |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530873

(CHEMBL4456215)Show SMILES CN(C1CCCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1)S(C)(=O)=O Show InChI InChI=1S/C22H30N8O3S2/c1-28(35(2,31)32)16-4-3-5-29(13-16)14-17-10-18-19(34-17)21(30-6-8-33-9-7-30)27-20(26-18)15-11-24-22(23)25-12-15/h10-12,16H,3-9,13-14H2,1-2H3,(H2,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-9
(Homo sapiens (Human)) | BDBM50069989

((R)-3-methyl-1-((S)-3-phenyl-2-(pyrazine-2-carboxa...)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)c1cnccn1)B(O)O |r| Show InChI InChI=1S/C19H25BN4O4/c1-13(2)10-17(20(27)28)24-18(25)15(11-14-6-4-3-5-7-14)23-19(26)16-12-21-8-9-22-16/h3-9,12-13,15,17,27-28H,10-11H2,1-2H3,(H,23,26)(H,24,25)/t15-,17-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human 20S immunoproteasome beta 1 subunit caspase-like activity using fluorogenic peptide Ac-nLPnLD-AMC as substrate in presence of PA2... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01161
BindingDB Entry DOI: 10.7270/Q2K077W3 |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-8
(Homo sapiens (Human)) | BDBM50550642
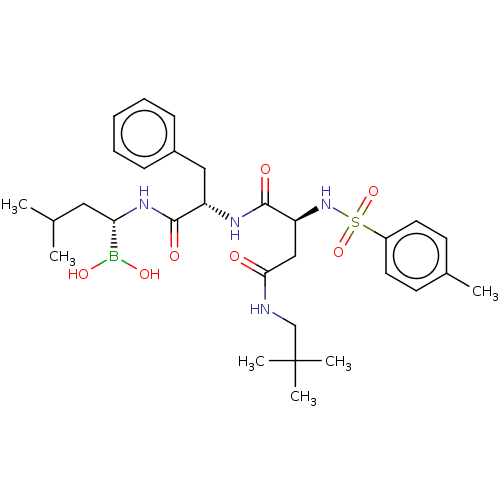
(CHEMBL4787081)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(=O)NCC(C)(C)C)NS(=O)(=O)c1ccc(C)cc1)B(O)O |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human 20S immunoproteasome beta 5 subunit chymotrypsin-like activity using fluorogenic peptide Ac-WLA-AMC as substrate in presence of P... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01161
BindingDB Entry DOI: 10.7270/Q2K077W3 |
More data for this
Ligand-Target Pair | |
Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit delta isoform
(Homo sapiens (Human)) | BDBM50530873

(CHEMBL4456215)Show SMILES CN(C1CCCN(Cc2cc3nc(nc(N4CCOCC4)c3s2)-c2cnc(N)nc2)C1)S(C)(=O)=O Show InChI InChI=1S/C22H30N8O3S2/c1-28(35(2,31)32)16-4-3-5-29(13-16)14-17-10-18-19(34-17)21(30-6-8-33-9-7-30)27-20(26-18)15-11-24-22(23)25-12-15/h10-12,16H,3-9,13-14H2,1-2H3,(H2,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Nottingham
Curated by ChEMBL
| Assay Description
Inhibition of PI3Kdelta (unknown origin) using biotin-PIP3 as substrate preincubated for 15 mins followed by substrate addition and measured after 60... |
J Med Chem 62: 10402-10422 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01499
BindingDB Entry DOI: 10.7270/Q27H1P12 |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-9
(Homo sapiens (Human)) | BDBM50550643

(CHEMBL4749207)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)Cc1ccc(O)cc1)B(O)O |r| | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human 20S immunoproteasome beta 1 subunit caspase-like activity using fluorogenic peptide Ac-nLPnLD-AMC as substrate in presence of PA2... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01161
BindingDB Entry DOI: 10.7270/Q2K077W3 |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50339611
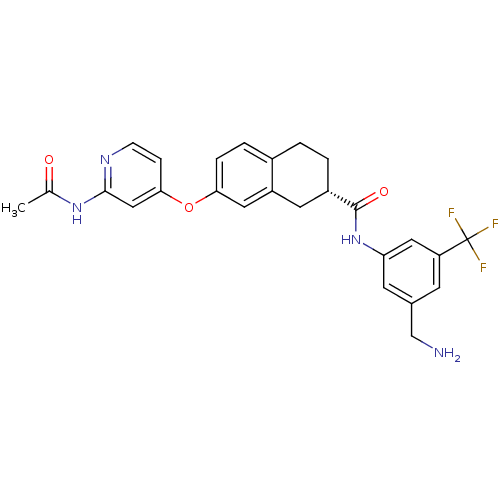
((2S)-7-{[2-(acetylamino)pyridin-4-yl]oxy}-N-[3-(am...)Show SMILES CC(=O)Nc1cc(Oc2ccc3CC[C@@H](Cc3c2)C(=O)Nc2cc(CN)cc(c2)C(F)(F)F)ccn1 |r| Show InChI InChI=1S/C26H25F3N4O3/c1-15(34)32-24-13-23(6-7-31-24)36-22-5-4-17-2-3-18(10-19(17)11-22)25(35)33-21-9-16(14-30)8-20(12-21)26(27,28)29/h4-9,11-13,18H,2-3,10,14,30H2,1H3,(H,33,35)(H,31,32,34)/t18-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of C-Raf assessed as reduction in [33P]ATP incorporation into biotinylated substrate peptide after 3 hrs by flash plate assay |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM16673

(4-[4-({[4-chloro-3-(trifluoromethyl)phenyl]carbamo...)Show SMILES CNC(=O)c1cc(Oc2ccc(NC(=O)Nc3ccc(Cl)c(c3)C(F)(F)F)cc2)ccn1 Show InChI InChI=1S/C21H16ClF3N4O3/c1-26-19(30)18-11-15(8-9-27-18)32-14-5-2-12(3-6-14)28-20(31)29-13-4-7-17(22)16(10-13)21(23,24)25/h2-11H,1H3,(H,26,30)(H2,28,29,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of C-Raf |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50339614

((2R)-N-[3-(aminomethyl)-5-(trifluoromethyl)phenyl]...)Show SMILES NCc1cc(NC(=O)[C@@H]2CCc3ccc(Oc4ccnc(NC(=O)C5CC5)c4)cc3C2)cc(c1)C(F)(F)F |r| Show InChI InChI=1S/C28H27F3N4O3/c29-28(30,31)21-9-16(15-32)10-22(13-21)34-27(37)19-4-1-17-5-6-23(12-20(17)11-19)38-24-7-8-33-25(14-24)35-26(36)18-2-3-18/h5-10,12-14,18-19H,1-4,11,15,32H2,(H,34,37)(H,33,35,36)/t19-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of C-Raf assessed as reduction in [33P]ATP incorporation into biotinylated substrate peptide after 3 hrs by flash plate assay |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |
Proteasome subunit beta type-5
(Homo sapiens (Human)) | BDBM50069989

((R)-3-methyl-1-((S)-3-phenyl-2-(pyrazine-2-carboxa...)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)c1cnccn1)B(O)O |r| Show InChI InChI=1S/C19H25BN4O4/c1-13(2)10-17(20(27)28)24-18(25)15(11-14-6-4-3-5-7-14)23-19(26)16-12-21-8-9-22-16/h3-9,12-13,15,17,27-28H,10-11H2,1-2H3,(H,23,26)(H,24,25)/t15-,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of human 20S constitutive proteasome beta 5 subunit chymotrypsin-like activity using fluorogenic peptide Ac-WLA-AMC as substrate in presen... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.8b01161
BindingDB Entry DOI: 10.7270/Q2K077W3 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Vascular endothelial growth factor receptor 2
(Homo sapiens (Human)) | BDBM50339608

(3-(1-Amino-1-methylethyl)-N-{(2R)-7-[(7-oxo-5,6,7,...)Show SMILES CC(C)(N)c1cc(cc(c1)C(F)(F)F)C(=O)N[C@@H]1CCc2ccc(Oc3ccnc4NC(=O)CCc34)cc2C1 |r| Show InChI InChI=1S/C29H29F3N4O3/c1-28(2,33)19-11-18(12-20(15-19)29(30,31)32)27(38)35-21-5-3-16-4-6-22(14-17(16)13-21)39-24-9-10-34-26-23(24)7-8-25(37)36-26/h4,6,9-12,14-15,21H,3,5,7-8,13,33H2,1-2H3,(H,35,38)(H,34,36,37)/t21-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Millennium Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibition of KDR |
J Med Chem 54: 1836-46 (2011)
Article DOI: 10.1021/jm101479y
BindingDB Entry DOI: 10.7270/Q2DB824F |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data