 Found 1940 hits with Last Name = 'ota' and Initial = 'y'
Found 1940 hits with Last Name = 'ota' and Initial = 'y' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86812

(CAS_45263784 | NSC_45263784 | rac-2-(6-fluoro-5-(4...)Show SMILES [O-][N+](=O)c1ccc(cc1)-c1cc(cnc1F)C1CC2CCC1N2 |TLB:11:16:19.20:22| Show InChI InChI=1S/C17H16FN3O2/c18-17-15(10-1-4-13(5-2-10)21(22)23)7-11(9-19-17)14-8-12-3-6-16(14)20-12/h1-2,4-5,7,9,12,14,16,20H,3,6,8H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.00900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50162061

(2-(5-Ethynyl-pyridin-3-yl)-7-aza-bicyclo[2.2.1]hep...)Show SMILES C#Cc1cncc(c1)C1CC2CCC1N2 |THB:6:8:14:12.11| Show InChI InChI=1S/C13H14N2/c1-2-9-5-10(8-14-7-9)12-6-11-3-4-13(12)15-11/h1,5,7-8,11-13,15H,3-4,6H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Inhibition of [3H]epibatidine binding to Nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex |
J Med Chem 48: 1221-8 (2005)
Article DOI: 10.1021/jm040160b
BindingDB Entry DOI: 10.7270/Q26W9BV6 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50100717
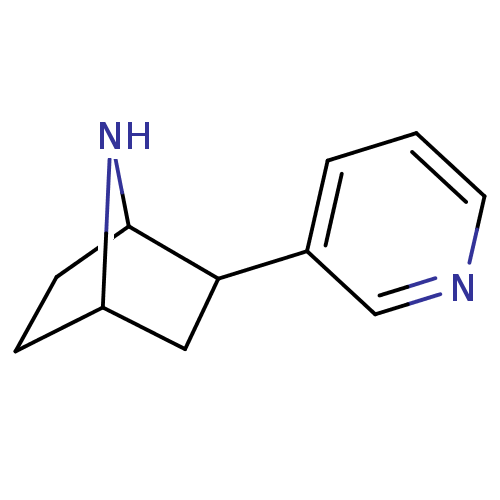
(2-(pyridin-3-yl)-7-aza-bicyclo[2.2.1]heptane | 2-P...)Show InChI InChI=1S/C11H14N2/c1-2-8(7-12-5-1)10-6-9-3-4-11(10)13-9/h1-2,5,7,9-11,13H,3-4,6H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50095523
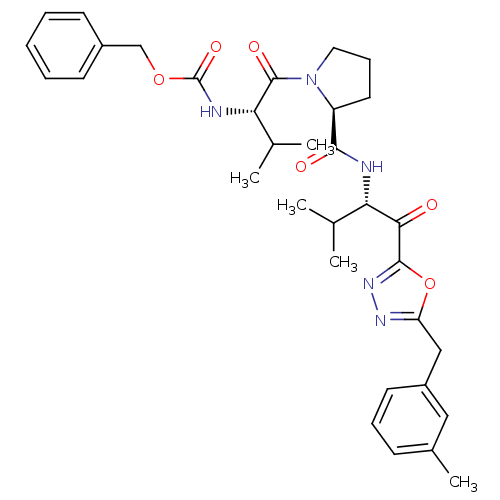
(CHEMBL285231 | [(S)-2-methyl-1-((S)-2-{(S)-2-methy...)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(=O)c1nnc(Cc2cccc(C)c2)o1 Show InChI InChI=1S/C33H41N5O6/c1-20(2)27(29(39)31-37-36-26(44-31)18-24-14-9-11-22(5)17-24)34-30(40)25-15-10-16-38(25)32(41)28(21(3)4)35-33(42)43-19-23-12-7-6-8-13-23/h6-9,11-14,17,20-21,25,27-28H,10,15-16,18-19H2,1-5H3,(H,34,40)(H,35,42)/t25-,27-,28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 0.0250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Colorado State University
Curated by ChEMBL
| Assay Description
Inhibition of human neutrophil elastase HNE, hydrolysis of MeO-Suc-Ala-Ala-Pro-Val-pNa |
J Med Chem 43: 4927-9 (2001)
BindingDB Entry DOI: 10.7270/Q25D8R3S |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50095523

(CHEMBL285231 | [(S)-2-methyl-1-((S)-2-{(S)-2-methy...)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(=O)c1nnc(Cc2cccc(C)c2)o1 Show InChI InChI=1S/C33H41N5O6/c1-20(2)27(29(39)31-37-36-26(44-31)18-24-14-9-11-22(5)17-24)34-30(40)25-15-10-16-38(25)32(41)28(21(3)4)35-33(42)43-19-23-12-7-6-8-13-23/h6-9,11-14,17,20-21,25,27-28H,10,15-16,18-19H2,1-5H3,(H,34,40)(H,35,42)/t25-,27-,28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 0.0250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cortech, Inc.
Curated by ChEMBL
| Assay Description
Binding constant derived from inhibition of elastase catalyzed hydrolysis of synthetic substrate |
J Med Chem 44: 1268-85 (2001)
BindingDB Entry DOI: 10.7270/Q2B858VW |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50049757

(()-2-(6-Chloro-pyridin-3-yl)-7-aza-bicyclo[2.2.1]h...)Show InChI InChI=1S/C11H13ClN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.0260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Inhibition of [3H]epibatidine binding to Nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex |
J Med Chem 48: 1221-8 (2005)
Article DOI: 10.1021/jm040160b
BindingDB Entry DOI: 10.7270/Q26W9BV6 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50049757

(()-2-(6-Chloro-pyridin-3-yl)-7-aza-bicyclo[2.2.1]h...)Show InChI InChI=1S/C11H13ClN2/c12-11-4-1-7(6-13-11)9-5-8-2-3-10(9)14-8/h1,4,6,8-10,14H,2-3,5H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.0260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86815
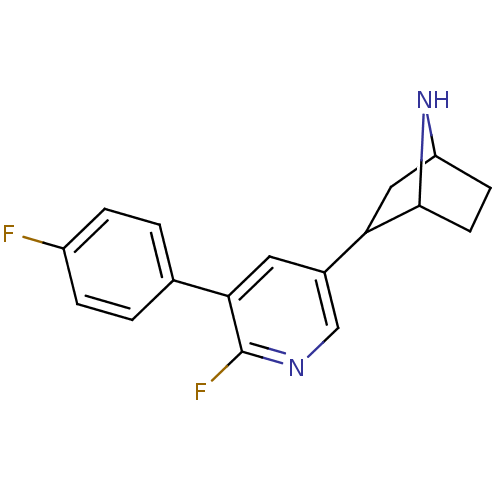
(CAS_45266019 | NSC_45266019 | rac-2-(6-fluoro-5-(4...)Show SMILES Fc1ccc(cc1)-c1cc(cnc1F)C1CC2CCC1N2 |TLB:9:14:17.18:20| Show InChI InChI=1S/C17H16F2N2/c18-12-3-1-10(2-4-12)15-7-11(9-20-17(15)19)14-8-13-5-6-16(14)21-13/h1-4,7,9,13-14,16,21H,5-6,8H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50162062

(2-(5-Iodo-pyridin-3-yl)-7-methyl-7-aza-bicyclo[2.2...)Show InChI InChI=1S/C12H15IN2/c1-15-10-2-3-12(15)11(5-10)8-4-9(13)7-14-6-8/h4,6-7,10-12H,2-3,5H2,1H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Inhibition of [3H]epibatidine binding to Nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex |
J Med Chem 48: 1221-8 (2005)
Article DOI: 10.1021/jm040160b
BindingDB Entry DOI: 10.7270/Q26W9BV6 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50162063

(2-(5-Fluoro-pyridin-3-yl)-7-aza-bicyclo[2.2.1]hept...)Show InChI InChI=1S/C11H13FN2/c12-8-3-7(5-13-6-8)10-4-9-1-2-11(10)14-9/h3,5-6,9-11,14H,1-2,4H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Inhibition of [3H]epibatidine binding to Nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex |
J Med Chem 48: 1221-8 (2005)
Article DOI: 10.1021/jm040160b
BindingDB Entry DOI: 10.7270/Q26W9BV6 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86816

(CAS_45263769 | NSC_45263769 | rac-2-(5-(4-chloroph...)Show SMILES Fc1ncc(cc1-c1ccc(Cl)cc1)C1CC2CCC1N2 |TLB:4:14:17.18:20| Show InChI InChI=1S/C17H16ClFN2/c18-12-3-1-10(2-4-12)15-7-11(9-20-17(15)19)14-8-13-5-6-16(14)21-13/h1-4,7,9,13-14,16,21H,5-6,8H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50162064
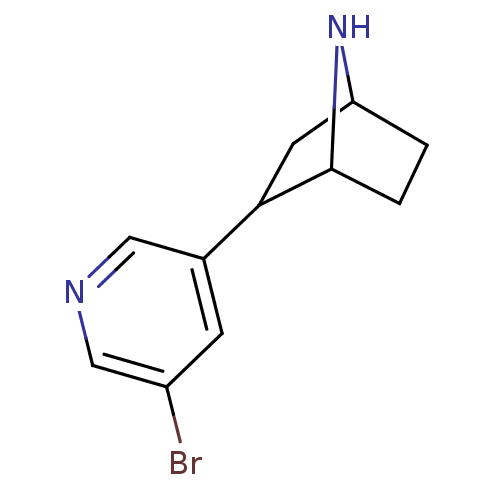
(2-(5-Bromo-pyridin-3-yl)-7-aza-bicyclo[2.2.1]hepta...)Show InChI InChI=1S/C11H13BrN2/c12-8-3-7(5-13-6-8)10-4-9-1-2-11(10)14-9/h3,5-6,9-11,14H,1-2,4H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Inhibition of [3H]epibatidine binding to Nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex |
J Med Chem 48: 1221-8 (2005)
Article DOI: 10.1021/jm040160b
BindingDB Entry DOI: 10.7270/Q26W9BV6 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86810
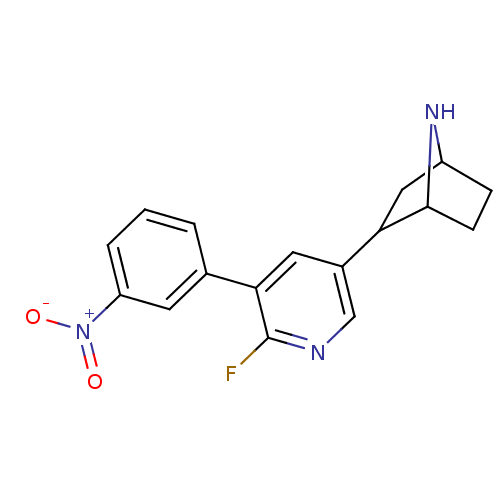
(CAS_45266065 | NSC_45266065 | rac-2-(6-fluoro-5-(3...)Show SMILES [O-][N+](=O)c1cccc(c1)-c1cc(cnc1F)C1CC2CCC1N2 |TLB:11:16:19.20:22| Show InChI InChI=1S/C17H16FN3O2/c18-17-15(10-2-1-3-13(6-10)21(22)23)7-11(9-19-17)14-8-12-4-5-16(14)20-12/h1-3,6-7,9,12,14,16,20H,4-5,8H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0530 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50162057
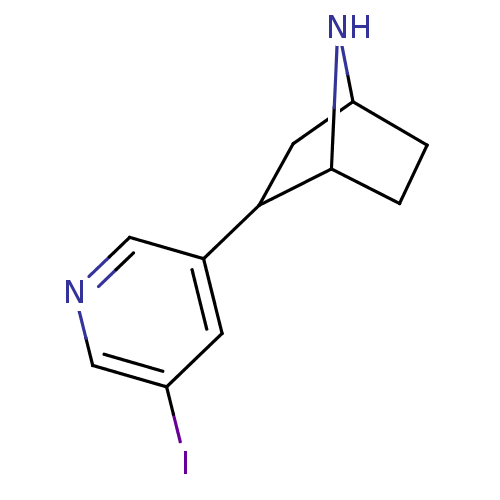
((+)2-(5-Iodo-pyridin-3-yl)-7-aza-bicyclo[2.2.1]hep...)Show InChI InChI=1S/C11H13IN2/c12-8-3-7(5-13-6-8)10-4-9-1-2-11(10)14-9/h3,5-6,9-11,14H,1-2,4H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Inhibition of [3H]epibatidine binding to Nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex |
J Med Chem 48: 1221-8 (2005)
Article DOI: 10.1021/jm040160b
BindingDB Entry DOI: 10.7270/Q26W9BV6 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50162056
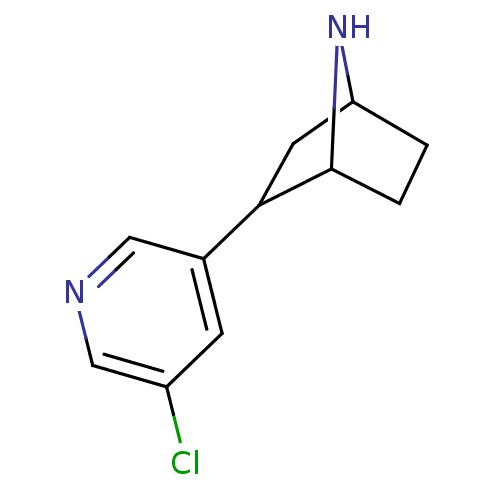
(2-(5-Chloro-pyridin-3-yl)-7-aza-bicyclo[2.2.1]hept...)Show InChI InChI=1S/C11H13ClN2/c12-8-3-7(5-13-6-8)10-4-9-1-2-11(10)14-9/h3,5-6,9-11,14H,1-2,4H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Inhibition of [3H]epibatidine binding to Nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex |
J Med Chem 48: 1221-8 (2005)
Article DOI: 10.1021/jm040160b
BindingDB Entry DOI: 10.7270/Q26W9BV6 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86805
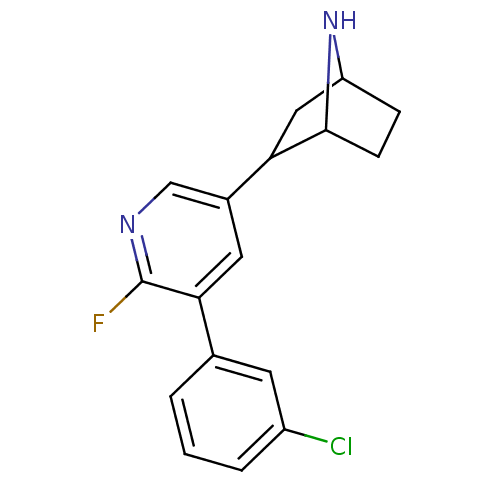
(CAS_45263788 | NSC_45263788 | US9150581, RTI-7527-...)Show SMILES Fc1ncc(cc1-c1cccc(Cl)c1)C1CC2CCC1N2 |TLB:4:14:17.18:20| Show InChI InChI=1S/C17H16ClFN2/c18-12-3-1-2-10(6-12)15-7-11(9-20-17(15)19)14-8-13-4-5-16(14)21-13/h1-3,6-7,9,13-14,16,21H,4-5,8H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86819

(CAS_45263772 | NSC_45263772 | rac-2-(6-fluoro-5-(3...)Show SMILES Fc1cccc(c1)-c1cc(cnc1F)C1CC2CCC1N2 |TLB:9:14:17.18:20| Show InChI InChI=1S/C17H16F2N2/c18-12-3-1-2-10(6-12)15-7-11(9-20-17(15)19)14-8-13-4-5-16(14)21-13/h1-3,6-7,9,13-14,16,21H,4-5,8H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86817
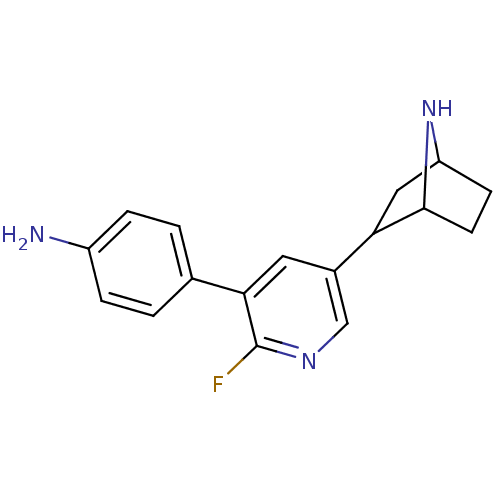
(CAS_45263779 | NSC_45263779 | rac-4-(5-(7-aza-bicy...)Show SMILES Nc1ccc(cc1)-c1cc(cnc1F)C1CC2CCC1N2 |TLB:9:14:17.18:20| Show InChI InChI=1S/C17H18FN3/c18-17-15(10-1-3-12(19)4-2-10)7-11(9-20-17)14-8-13-5-6-16(14)21-13/h1-4,7,9,13-14,16,21H,5-6,8,19H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0950 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50162060

(5-(7-Aza-bicyclo[2.2.1]hept-2-yl)-pyridin-3-ylamin...)Show InChI InChI=1S/C11H15N3/c12-8-3-7(5-13-6-8)10-4-9-1-2-11(10)14-9/h3,5-6,9-11,14H,1-2,4,12H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Inhibition of [3H]epibatidine binding to Nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex |
J Med Chem 48: 1221-8 (2005)
Article DOI: 10.1021/jm040160b
BindingDB Entry DOI: 10.7270/Q26W9BV6 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86807
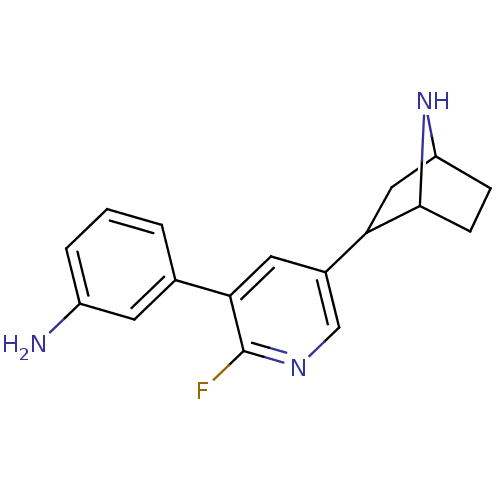
(CAS_45263775 | NSC_45263775 | rac-3-(5-(7-aza-bicy...)Show SMILES Nc1cccc(c1)-c1cc(cnc1F)C1CC2CCC1N2 |TLB:9:14:17.18:20| Show InChI InChI=1S/C17H18FN3/c18-17-15(10-2-1-3-12(19)6-10)7-11(9-20-17)14-8-13-4-5-16(14)21-13/h1-3,6-7,9,13-14,16,21H,4-5,8,19H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50166908
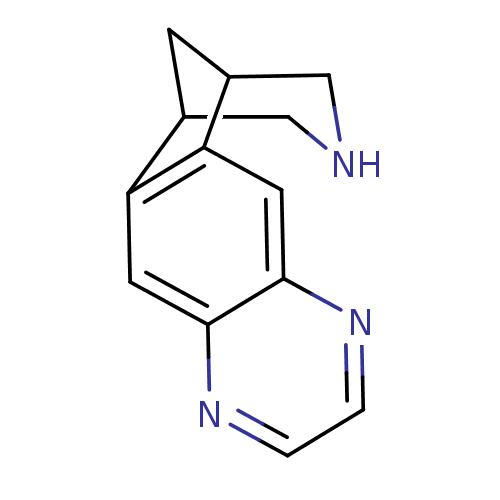
(5,8,14-triazatetracyclo[10.3.1.02,11.04,9]hexadeca...)Show InChI InChI=1S/C13H13N3/c1-2-16-13-5-11-9-3-8(6-14-7-9)10(11)4-12(13)15-1/h1-2,4-5,8-9,14H,3,6-7H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86818

(CAS_45266054 | NSC_45266054 | rac-2-(6-fluoro-5-(3...)Show SMILES COc1cccc(c1)-c1cc(cnc1F)C1CC2CCC1N2 |THB:10:15:18.19:21| Show InChI InChI=1S/C18H19FN2O/c1-22-14-4-2-3-11(7-14)16-8-12(10-20-18(16)19)15-9-13-5-6-17(15)21-13/h2-4,7-8,10,13,15,17,21H,5-6,9H2,1H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86809

(3'-(4-fluorophenyl)deschloroepibatidine | CAS_...)Show SMILES Fc1ccc(cc1)-c1cncc(c1)C1CC2CCC1N2 |TLB:11:13:16.17:19| Show InChI InChI=1S/C17H17FN2/c18-14-3-1-11(2-4-14)12-7-13(10-19-9-12)16-8-15-5-6-17(16)20-15/h1-4,7,9-10,15-17,20H,5-6,8H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86822

(CAS_45263767 | NSC_45263767 | rac-3'-(3-nitrop...)Show SMILES [O-][N+](=O)c1cccc(c1)-c1cncc(c1)C1CC2CCC1N2 |TLB:13:15:18.19:21| Show InChI InChI=1S/C17H17N3O2/c21-20(22)15-3-1-2-11(7-15)12-6-13(10-18-9-12)16-8-14-4-5-17(16)19-14/h1-3,6-7,9-10,14,16-17,19H,4-5,8H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86820

(CAS_45263774 | NSC_45263774 | rac-3'-(3-chloro...)Show SMILES Clc1cccc(c1)-c1cncc(c1)C1CC2CCC1N2 |TLB:11:13:16.17:19| Show InChI InChI=1S/C17H17ClN2/c18-14-3-1-2-11(7-14)12-6-13(10-19-9-12)16-8-15-4-5-17(16)20-15/h1-3,6-7,9-10,15-17,20H,4-5,8H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86821
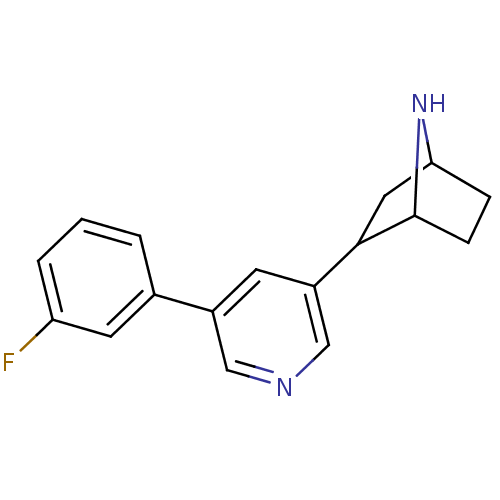
(CAS_45263766 | NSC_45263766 | rac-3'-(3-fluoro...)Show SMILES Fc1cccc(c1)-c1cncc(c1)C1CC2CCC1N2 |TLB:11:13:16.17:19| Show InChI InChI=1S/C17H17FN2/c18-14-3-1-2-11(7-14)12-6-13(10-19-9-12)16-8-15-4-5-17(16)20-15/h1-3,6-7,9-10,15-17,20H,4-5,8H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86811

(3-(4-chlorophenyl)deschloroepibatidine | CAS_44452...)Show SMILES Clc1ccc(cc1)-c1cncc(c1)C1CC2CCC1N2 |TLB:11:13:16.17:19| Show InChI InChI=1S/C17H17ClN2/c18-14-3-1-11(2-4-14)12-7-13(10-19-9-12)16-8-15-5-6-17(16)20-15/h1-4,7,9-10,15-17,20H,5-6,8H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86806
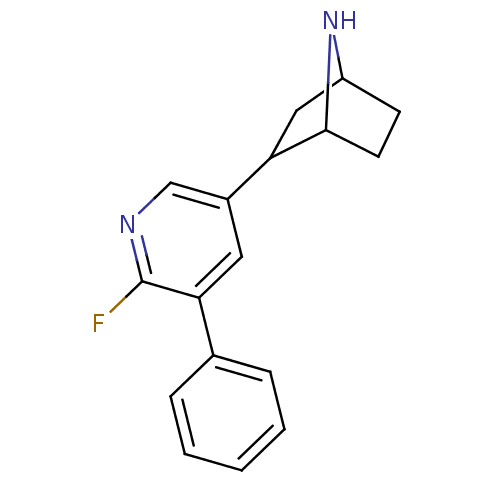
(CAS_45263762 | NSC_45263762 | US9150581, RTI-7527-...)Show SMILES Fc1ncc(cc1-c1ccccc1)C1CC2CCC1N2 |TLB:4:13:16.17:19| Show InChI InChI=1S/C17H17FN2/c18-17-15(11-4-2-1-3-5-11)8-12(10-19-17)14-9-13-6-7-16(14)20-13/h1-5,8,10,13-14,16,20H,6-7,9H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM50162059
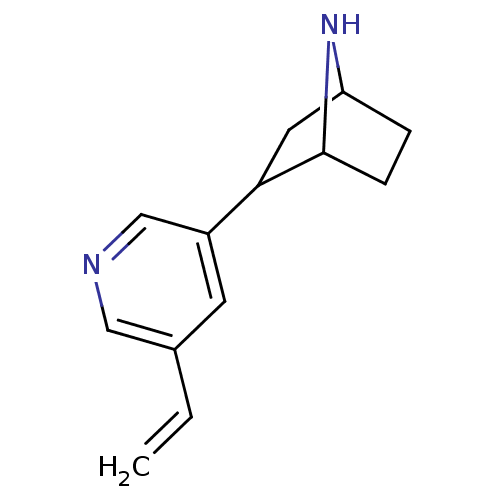
(2-(5-Vinyl-pyridin-3-yl)-7-aza-bicyclo[2.2.1]hepta...)Show SMILES C=Cc1cncc(c1)C1CC2CCC1N2 |THB:6:8:14:12.11| Show InChI InChI=1S/C13H16N2/c1-2-9-5-10(8-14-7-9)12-6-11-3-4-13(12)15-11/h2,5,7-8,11-13,15H,1,3-4,6H2 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Inhibition of [3H]epibatidine binding to Nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex |
J Med Chem 48: 1221-8 (2005)
Article DOI: 10.1021/jm040160b
BindingDB Entry DOI: 10.7270/Q26W9BV6 |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86813

(CAS_45266021 | NSC_45266021 | rac-3-(4-aminophenyl...)Show SMILES Nc1ccc(cc1)-c1cncc(c1)C1CC2CCC1N2 |TLB:11:13:16.17:19| Show InChI InChI=1S/C17H19N3/c18-14-3-1-11(2-4-14)12-7-13(10-19-9-12)16-8-15-5-6-17(16)20-15/h1-4,7,9-10,15-17,20H,5-6,8,18H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86814

(CAS_45266060 | NSC_45266060 | rac-3'-(4-nitrop...)Show SMILES [O-][N+](=O)c1ccc(cc1)-c1cncc(c1)C1CC2CCC1N2 |TLB:13:15:18.19:21| Show InChI InChI=1S/C17H17N3O2/c21-20(22)15-4-1-11(2-5-15)12-7-13(10-18-9-12)16-8-14-3-6-17(16)19-14/h1-2,4-5,7,9-10,14,16-17,19H,3,6,8H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86823

(CAS_45263792 | NSC_45263792 | rac-3-(3-methoxyphen...)Show SMILES COc1cccc(c1)-c1cncc(c1)C1CC2CCC1N2 |THB:12:14:17.18:20| Show InChI InChI=1S/C18H20N2O/c1-21-16-4-2-3-12(8-16)13-7-14(11-19-10-13)17-9-15-5-6-18(17)20-15/h2-4,7-8,10-11,15,17-18,20H,5-6,9H2,1H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50095526
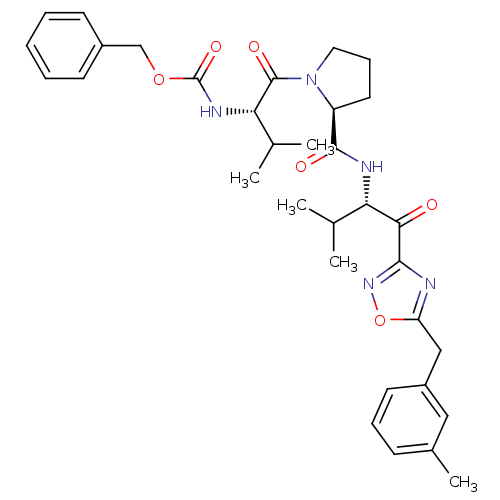
(CHEMBL24058 | [(S)-2-methyl-1-((S)-2-{(S)-2-methyl...)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(=O)c1noc(Cc2cccc(C)c2)n1 Show InChI InChI=1S/C33H41N5O6/c1-20(2)27(29(39)30-34-26(44-37-30)18-24-14-9-11-22(5)17-24)35-31(40)25-15-10-16-38(25)32(41)28(21(3)4)36-33(42)43-19-23-12-7-6-8-13-23/h6-9,11-14,17,20-21,25,27-28H,10,15-16,18-19H2,1-5H3,(H,35,40)(H,36,42)/t25-,27-,28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 0.490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Colorado State University
Curated by ChEMBL
| Assay Description
Inhibition of human neutrophil elastase HNE, hydrolysis of MeO-Suc-Ala-Ala-Pro-Val-pNa |
J Med Chem 43: 4927-9 (2001)
BindingDB Entry DOI: 10.7270/Q25D8R3S |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50095526

(CHEMBL24058 | [(S)-2-methyl-1-((S)-2-{(S)-2-methyl...)Show SMILES CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(=O)c1noc(Cc2cccc(C)c2)n1 Show InChI InChI=1S/C33H41N5O6/c1-20(2)27(29(39)30-34-26(44-37-30)18-24-14-9-11-22(5)17-24)35-31(40)25-15-10-16-38(25)32(41)28(21(3)4)36-33(42)43-19-23-12-7-6-8-13-23/h6-9,11-14,17,20-21,25,27-28H,10,15-16,18-19H2,1-5H3,(H,35,40)(H,36,42)/t25-,27-,28-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| 0.490 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cortech, Inc.
Curated by ChEMBL
| Assay Description
Binding constant derived from inhibition of elastase catalyzed hydrolysis of synthetic substrate |
J Med Chem 44: 1268-85 (2001)
BindingDB Entry DOI: 10.7270/Q2B858VW |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86824
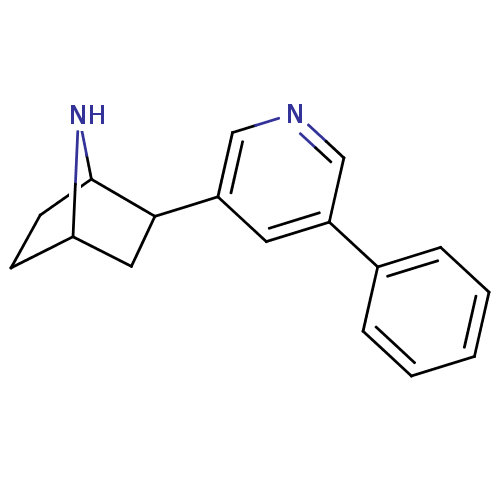
(CAS_45266048 | NSC_45266048 | rac-3-phenyldeschlor...)Show SMILES C1CC2NC1CC2c1cncc(c1)-c1ccccc1 |TLB:7:6:0.1:3| Show InChI InChI=1S/C17H18N2/c1-2-4-12(5-3-1)13-8-14(11-18-10-13)16-9-15-6-7-17(16)19-15/h1-5,8,10-11,15-17,19H,6-7,9H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50095530
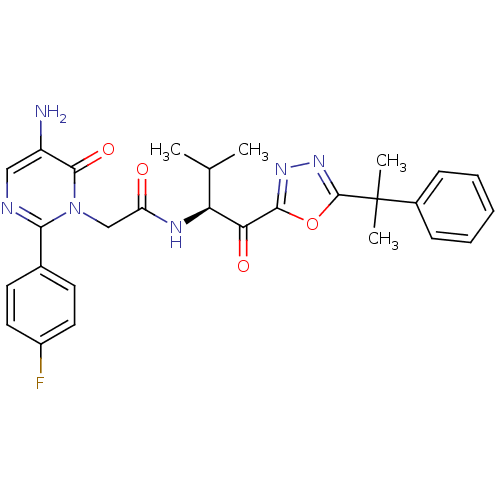
((S)-2-(5-amino-2-(4-fluorophenyl)-6-oxopyrimidin-1...)Show SMILES CC(C)[C@H](NC(=O)Cn1c(ncc(N)c1=O)-c1ccc(F)cc1)C(=O)c1nnc(o1)C(C)(C)c1ccccc1 Show InChI InChI=1S/C28H29FN6O4/c1-16(2)22(23(37)25-33-34-27(39-25)28(3,4)18-8-6-5-7-9-18)32-21(36)15-35-24(31-14-20(30)26(35)38)17-10-12-19(29)13-11-17/h5-14,16,22H,15,30H2,1-4H3,(H,32,36)/t22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cortech, Inc.
Curated by ChEMBL
| Assay Description
Binding constant derived from inhibition of elastase catalyzed hydrolysis of synthetic substrate |
J Med Chem 44: 1268-85 (2001)
BindingDB Entry DOI: 10.7270/Q2B858VW |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50095530

((S)-2-(5-amino-2-(4-fluorophenyl)-6-oxopyrimidin-1...)Show SMILES CC(C)[C@H](NC(=O)Cn1c(ncc(N)c1=O)-c1ccc(F)cc1)C(=O)c1nnc(o1)C(C)(C)c1ccccc1 Show InChI InChI=1S/C28H29FN6O4/c1-16(2)22(23(37)25-33-34-27(39-25)28(3,4)18-8-6-5-7-9-18)32-21(36)15-35-24(31-14-20(30)26(35)38)17-10-12-19(29)13-11-17/h5-14,16,22H,15,30H2,1-4H3,(H,32,36)/t22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Colorado State University
Curated by ChEMBL
| Assay Description
Inhibition of human neutrophil elastase HNE, hydrolysis of MeO-Suc-Ala-Ala-Pro-Val-pNa |
J Med Chem 43: 4927-9 (2001)
BindingDB Entry DOI: 10.7270/Q25D8R3S |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50095521
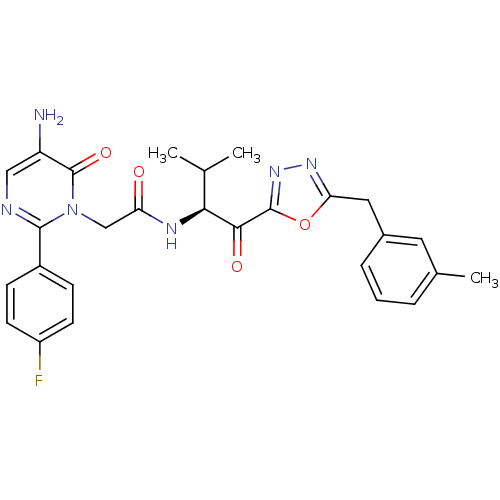
((S)-2-[5-Amino-2-(4-fluoro-phenyl)-6-oxo-6H-pyrimi...)Show SMILES CC(C)[C@H](NC(=O)Cn1c(ncc(N)c1=O)-c1ccc(F)cc1)C(=O)c1nnc(Cc2cccc(C)c2)o1 Show InChI InChI=1S/C27H27FN6O4/c1-15(2)23(24(36)26-33-32-22(38-26)12-17-6-4-5-16(3)11-17)31-21(35)14-34-25(30-13-20(29)27(34)37)18-7-9-19(28)10-8-18/h4-11,13,15,23H,12,14,29H2,1-3H3,(H,31,35)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cortech, Inc.
Curated by ChEMBL
| Assay Description
Binding constant derived from inhibition of elastase catalyzed hydrolysis of synthetic substrate |
J Med Chem 44: 1268-85 (2001)
BindingDB Entry DOI: 10.7270/Q2B858VW |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50095521

((S)-2-[5-Amino-2-(4-fluoro-phenyl)-6-oxo-6H-pyrimi...)Show SMILES CC(C)[C@H](NC(=O)Cn1c(ncc(N)c1=O)-c1ccc(F)cc1)C(=O)c1nnc(Cc2cccc(C)c2)o1 Show InChI InChI=1S/C27H27FN6O4/c1-15(2)23(24(36)26-33-32-22(38-26)12-17-6-4-5-16(3)11-17)31-21(35)14-34-25(30-13-20(29)27(34)37)18-7-9-19(28)10-8-18/h4-11,13,15,23H,12,14,29H2,1-3H3,(H,31,35)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.640 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Colorado State University
Curated by ChEMBL
| Assay Description
Inhibition of human neutrophil elastase HNE, hydrolysis of MeO-Suc-Ala-Ala-Pro-Val-pNa |
J Med Chem 43: 4927-9 (2001)
BindingDB Entry DOI: 10.7270/Q25D8R3S |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM86808
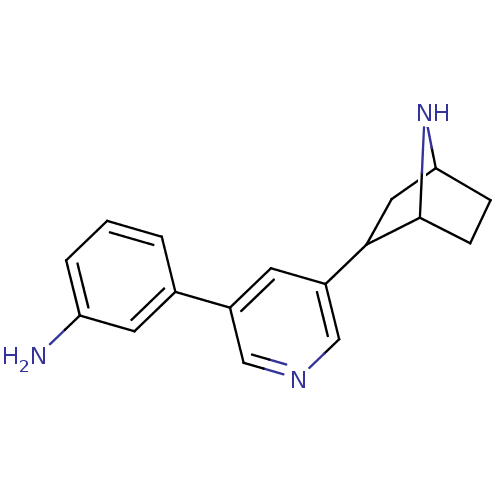
(CAS_45263778 | NSC_45263778 | rac-3'-(3-aminop...)Show SMILES Nc1cccc(c1)-c1cncc(c1)C1CC2CCC1N2 |TLB:11:13:16.17:19| Show InChI InChI=1S/C17H19N3/c18-14-3-1-2-11(7-14)12-6-13(10-19-9-12)16-8-15-4-5-17(16)20-15/h1-3,6-7,9-10,15-17,20H,4-5,8,18H2 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50095519
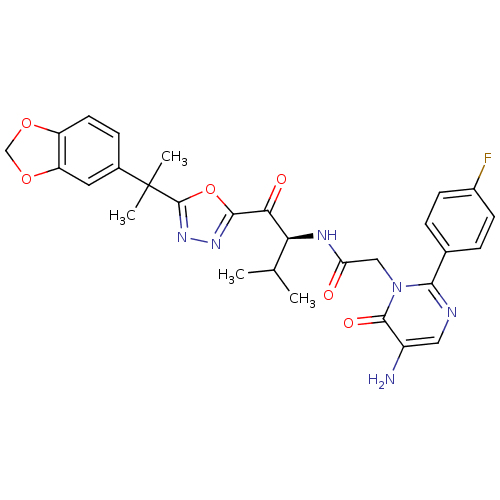
((S)-2-[5-Amino-2-(4-fluoro-phenyl)-6-oxo-6H-pyrimi...)Show SMILES CC(C)[C@H](NC(=O)Cn1c(ncc(N)c1=O)-c1ccc(F)cc1)C(=O)c1nnc(o1)C(C)(C)c1ccc2OCOc2c1 Show InChI InChI=1S/C29H29FN6O6/c1-15(2)23(33-22(37)13-36-25(32-12-19(31)27(36)39)16-5-8-18(30)9-6-16)24(38)26-34-35-28(42-26)29(3,4)17-7-10-20-21(11-17)41-14-40-20/h5-12,15,23H,13-14,31H2,1-4H3,(H,33,37)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cortech, Inc.
Curated by ChEMBL
| Assay Description
Binding constant derived from inhibition of elastase catalyzed hydrolysis of synthetic substrate |
J Med Chem 44: 1268-85 (2001)
BindingDB Entry DOI: 10.7270/Q2B858VW |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50095519

((S)-2-[5-Amino-2-(4-fluoro-phenyl)-6-oxo-6H-pyrimi...)Show SMILES CC(C)[C@H](NC(=O)Cn1c(ncc(N)c1=O)-c1ccc(F)cc1)C(=O)c1nnc(o1)C(C)(C)c1ccc2OCOc2c1 Show InChI InChI=1S/C29H29FN6O6/c1-15(2)23(33-22(37)13-36-25(32-12-19(31)27(36)39)16-5-8-18(30)9-6-16)24(38)26-34-35-28(42-26)29(3,4)17-7-10-20-21(11-17)41-14-40-20/h5-12,15,23H,13-14,31H2,1-4H3,(H,33,37)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Colorado State University
Curated by ChEMBL
| Assay Description
Inhibition of human neutrophil elastase HNE, hydrolysis of MeO-Suc-Ala-Ala-Pro-Val-pNa |
J Med Chem 43: 4927-9 (2001)
BindingDB Entry DOI: 10.7270/Q25D8R3S |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50095525
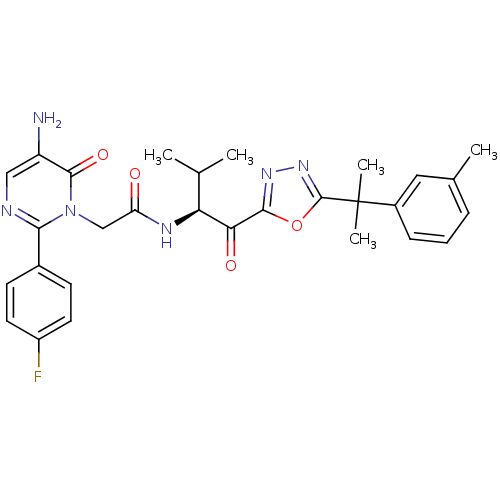
((S)-2-(5-amino-2-(4-fluorophenyl)-6-oxopyrimidin-1...)Show SMILES CC(C)[C@H](NC(=O)Cn1c(ncc(N)c1=O)-c1ccc(F)cc1)C(=O)c1nnc(o1)C(C)(C)c1cccc(C)c1 Show InChI InChI=1S/C29H31FN6O4/c1-16(2)23(24(38)26-34-35-28(40-26)29(4,5)19-8-6-7-17(3)13-19)33-22(37)15-36-25(32-14-21(31)27(36)39)18-9-11-20(30)12-10-18/h6-14,16,23H,15,31H2,1-5H3,(H,33,37)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Colorado State University
Curated by ChEMBL
| Assay Description
Inhibition of human neutrophil elastase HNE, hydrolysis of MeO-Suc-Ala-Ala-Pro-Val-pNa |
J Med Chem 43: 4927-9 (2001)
BindingDB Entry DOI: 10.7270/Q25D8R3S |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50095525

((S)-2-(5-amino-2-(4-fluorophenyl)-6-oxopyrimidin-1...)Show SMILES CC(C)[C@H](NC(=O)Cn1c(ncc(N)c1=O)-c1ccc(F)cc1)C(=O)c1nnc(o1)C(C)(C)c1cccc(C)c1 Show InChI InChI=1S/C29H31FN6O4/c1-16(2)23(24(38)26-34-35-28(40-26)29(4,5)19-8-6-7-17(3)13-19)33-22(37)15-36-25(32-14-21(31)27(36)39)18-9-11-20(30)12-10-18/h6-14,16,23H,15,31H2,1-5H3,(H,33,37)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cortech, Inc.
Curated by ChEMBL
| Assay Description
Binding constant derived from inhibition of elastase catalyzed hydrolysis of synthetic substrate |
J Med Chem 44: 1268-85 (2001)
BindingDB Entry DOI: 10.7270/Q2B858VW |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4
(Rattus norvegicus (Rat)) | BDBM50004108
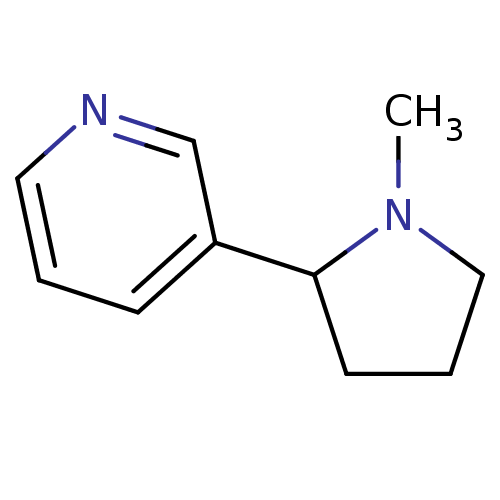
((+-)-nicotine | (R,S)-nicotine | (RS)-nicotine | 3...)Show InChI InChI=1S/C10H14N2/c1-12-7-3-5-10(12)9-4-2-6-11-8-9/h2,4,6,8,10H,3,5,7H2,1H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by PDSP Ki Database
| |
Bioorg Med Chem 16: 746-54 (2008)
Article DOI: 10.1016/j.bmc.2007.10.027
BindingDB Entry DOI: 10.7270/Q2H130KC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Rattus norvegicus (Rat)) | BDBM82070
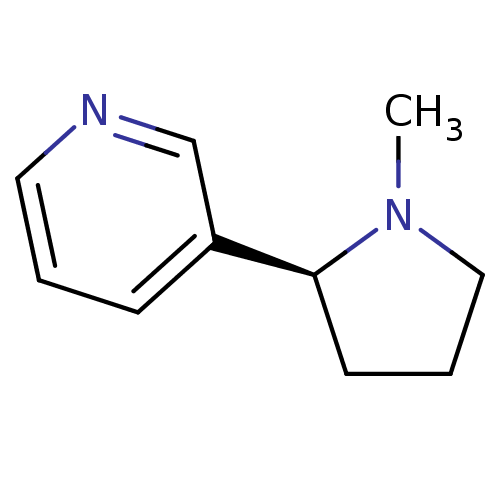
(CAS_29790-52-1 | NICOTINE-L (BASE) | Nicotine-D sa...)Show InChI InChI=1S/C10H14N2/c1-12-7-3-5-10(12)9-4-2-6-11-8-9/h2,4,6,8,10H,3,5,7H2,1H3/t10-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute
Curated by ChEMBL
| Assay Description
Inhibition of [3H]epibatidine binding to Nicotinic acetylcholine receptor alpha4-beta2 in rat cerebral cortex |
J Med Chem 48: 1221-8 (2005)
Article DOI: 10.1021/jm040160b
BindingDB Entry DOI: 10.7270/Q26W9BV6 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50098818
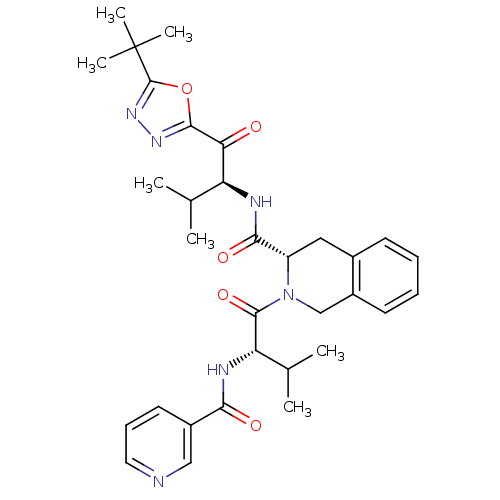
(2-{3-Methyl-2-[(pyridine-3-carbonyl)-amino]-butyry...)Show SMILES CC(C)[C@H](NC(=O)c1cccnc1)C(=O)N1Cc2ccccc2C[C@H]1C(=O)N[C@@H](C(C)C)C(=O)c1nnc(o1)C(C)(C)C Show InChI InChI=1S/C32H40N6O5/c1-18(2)24(26(39)29-36-37-31(43-29)32(5,6)7)34-28(41)23-15-20-11-8-9-12-22(20)17-38(23)30(42)25(19(3)4)35-27(40)21-13-10-14-33-16-21/h8-14,16,18-19,23-25H,15,17H2,1-7H3,(H,34,41)(H,35,40)/t23-,24-,25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.66 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cortech, Inc.
Curated by ChEMBL
| Assay Description
Binding constant derived from inhibition of elastase catalyzed hydrolysis of synthetic substrate |
J Med Chem 44: 1268-85 (2001)
BindingDB Entry DOI: 10.7270/Q2B858VW |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50098833
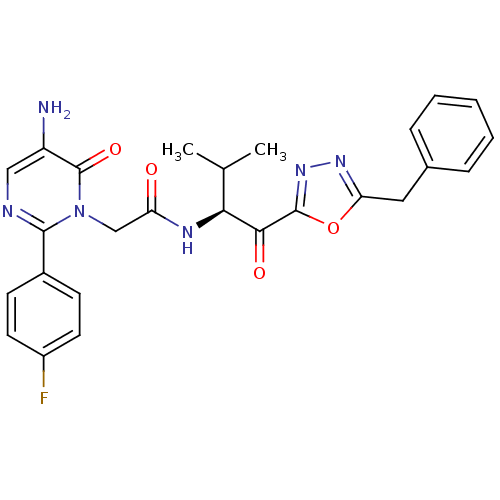
(2-[5-Amino-2-(4-fluoro-phenyl)-6-oxo-6H-pyrimidin-...)Show SMILES CC(C)[C@H](NC(=O)Cn1c(ncc(N)c1=O)-c1ccc(F)cc1)C(=O)c1nnc(Cc2ccccc2)o1 Show InChI InChI=1S/C26H25FN6O4/c1-15(2)22(23(35)25-32-31-21(37-25)12-16-6-4-3-5-7-16)30-20(34)14-33-24(29-13-19(28)26(33)36)17-8-10-18(27)11-9-17/h3-11,13,15,22H,12,14,28H2,1-2H3,(H,30,34)/t22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.25 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cortech, Inc.
Curated by ChEMBL
| Assay Description
Binding constant derived from inhibition of elastase catalyzed hydrolysis of synthetic substrate |
J Med Chem 44: 1268-85 (2001)
BindingDB Entry DOI: 10.7270/Q2B858VW |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50098827
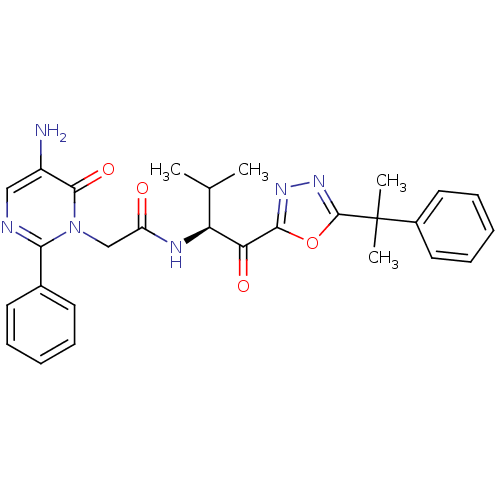
(2-(5-Amino-6-oxo-2-phenyl-6H-pyrimidin-1-yl)-N-{2-...)Show SMILES CC(C)[C@H](NC(=O)Cn1c(ncc(N)c1=O)-c1ccccc1)C(=O)c1nnc(o1)C(C)(C)c1ccccc1 Show InChI InChI=1S/C28H30N6O4/c1-17(2)22(23(36)25-32-33-27(38-25)28(3,4)19-13-9-6-10-14-19)31-21(35)16-34-24(18-11-7-5-8-12-18)30-15-20(29)26(34)37/h5-15,17,22H,16,29H2,1-4H3,(H,31,35)/t22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.55 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cortech, Inc.
Curated by ChEMBL
| Assay Description
Binding constant derived from inhibition of elastase catalyzed hydrolysis of synthetic substrate |
J Med Chem 44: 1268-85 (2001)
BindingDB Entry DOI: 10.7270/Q2B858VW |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50098817
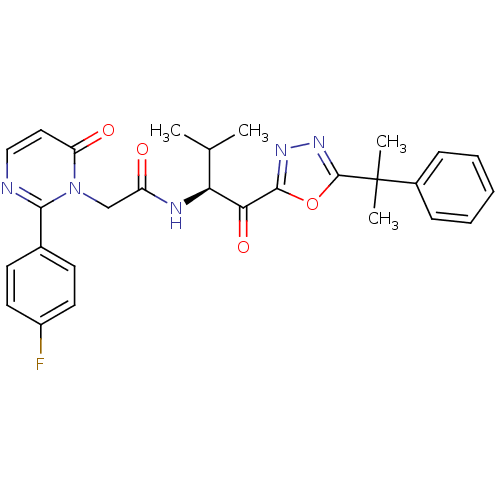
(2-[2-(4-Fluoro-phenyl)-6-oxo-6H-pyrimidin-1-yl]-N-...)Show SMILES CC(C)[C@H](NC(=O)Cn1c(nccc1=O)-c1ccc(F)cc1)C(=O)c1nnc(o1)C(C)(C)c1ccccc1 Show InChI InChI=1S/C28H28FN5O4/c1-17(2)23(24(37)26-32-33-27(38-26)28(3,4)19-8-6-5-7-9-19)31-21(35)16-34-22(36)14-15-30-25(34)18-10-12-20(29)13-11-18/h5-15,17,23H,16H2,1-4H3,(H,31,35)/t23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 3.35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Cortech, Inc.
Curated by ChEMBL
| Assay Description
Binding constant derived from inhibition of elastase catalyzed hydrolysis of synthetic substrate |
J Med Chem 44: 1268-85 (2001)
BindingDB Entry DOI: 10.7270/Q2B858VW |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data