

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50208985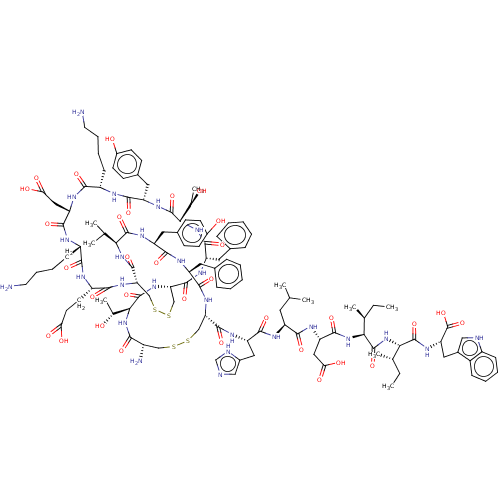 (CHEMBL3885244) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a |
Jagiellonian University Medical College Curated by ChEMBL | Assay Description Displacement of [125I]endothelin-1 from human recombinant ETB receptor expressed in CHO cells measured after 120 mins by scintillation counting metho... | Bioorg Med Chem 25: 471-482 (2017) Article DOI: 10.1016/j.bmc.2016.11.014 BindingDB Entry DOI: 10.7270/Q2CF9S3S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50155245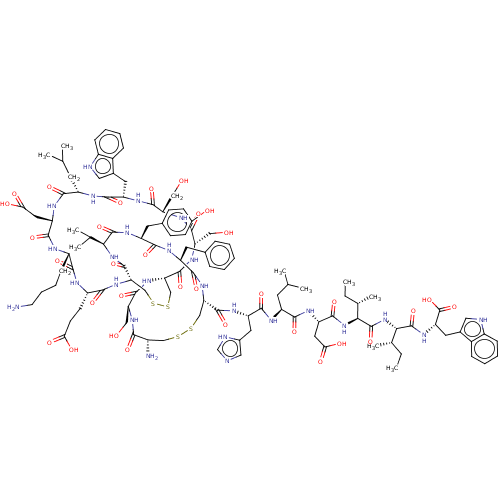 (CHEMBL3774897) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a |
Jagiellonian University Medical College Curated by ChEMBL | Assay Description Displacement of [125I]Endothelin-1 from human recombinant Endothelin-B receptor expressed in CHO cells | Bioorg Med Chem 24: 1793-810 (2016) Article DOI: 10.1016/j.bmc.2016.03.006 BindingDB Entry DOI: 10.7270/Q2J67JS7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50197953 (CHEMBL3980643) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Perugia Curated by ChEMBL | Assay Description Agonist activity at human recombinant ETB receptor expressed in CHOK1 cells assessed as induction of Ca2+ mobilization by fluorimetric method | Eur J Med Chem 121: 658-670 (2016) Article DOI: 10.1016/j.ejmech.2016.06.006 BindingDB Entry DOI: 10.7270/Q2T155MF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50061086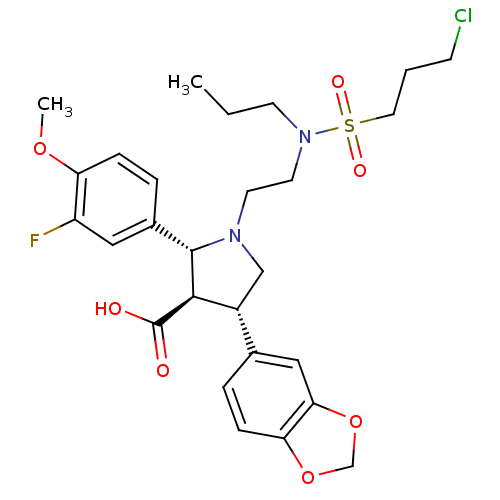 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-{2-[(3-chloro...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.140 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding assay performed using human Endothelin B receptor (hETB) expressed in chinese hamster ovary cells(CHO) | J Med Chem 40: 3217-27 (1997) Article DOI: 10.1021/jm970101g BindingDB Entry DOI: 10.7270/Q2C24VJR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50061078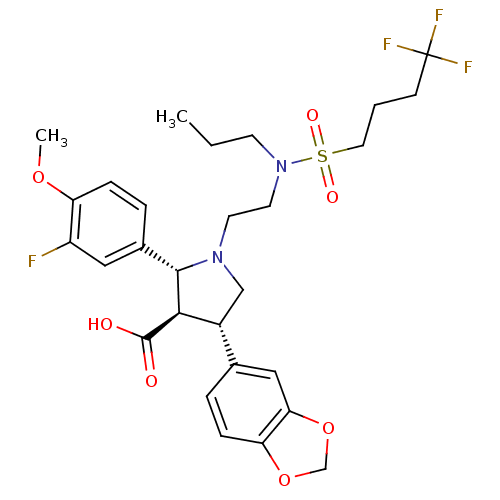 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-2-(3-fluoro-4-m...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding assay performed using human Endothelin B receptor (hETB) expressed in chinese hamster ovary cells(CHO) | J Med Chem 40: 3217-27 (1997) Article DOI: 10.1021/jm970101g BindingDB Entry DOI: 10.7270/Q2C24VJR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50000558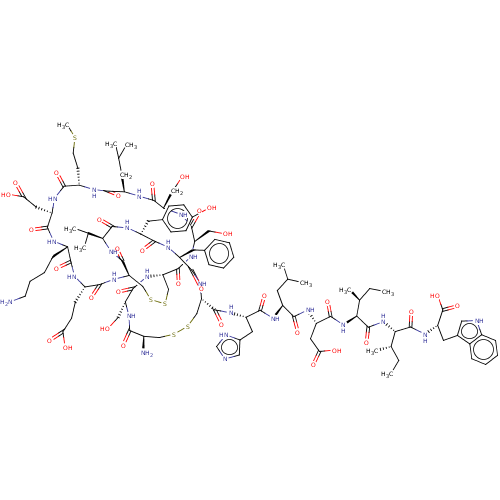 (CHEMBL437472 | ET-1 | Endothelin -1 | Endothelin 1...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Receptor binding affinity was determined against [125I]ET1 with membranes prepared from human placenta for ETB receptor | Bioorg Med Chem Lett 7: 2223-2228 (1997) Article DOI: 10.1016/S0960-894X(97)00400-9 BindingDB Entry DOI: 10.7270/Q2WQ049M | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50395673 (CHEMBL2165327) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.208 | n/a | n/a | n/a | n/a | n/a | n/a |
Actelion Pharmaceuticals Ltd. Curated by ChEMBL | Assay Description Displacement of [I125]ET1 from recombinant ETB receptor expressed in CHO cells after 2 hrs by TopCount analysis | J Med Chem 55: 7849-61 (2012) Article DOI: 10.1021/jm3009103 BindingDB Entry DOI: 10.7270/Q21C1XZT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50061074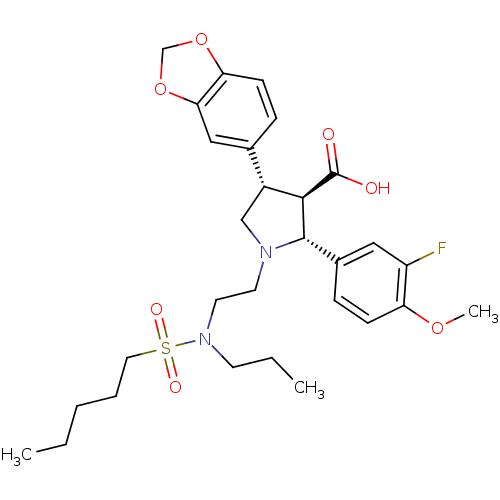 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-2-(3-fluoro-4-m...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding assay performed using human Endothelin B receptor (hETB) expressed in chinese hamster ovary cells(CHO) | J Med Chem 40: 3217-27 (1997) Article DOI: 10.1021/jm970101g BindingDB Entry DOI: 10.7270/Q2C24VJR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50197955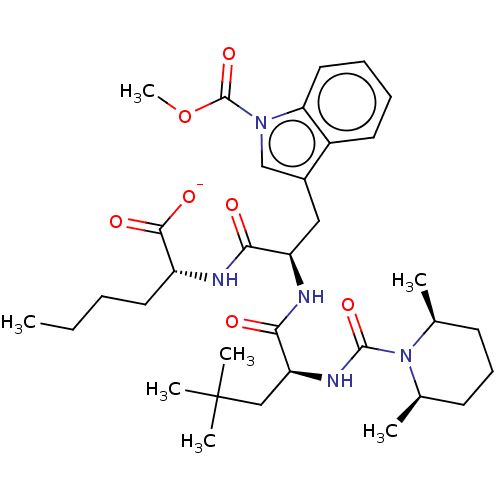 (CHEMBL1515091) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 0.410 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Perugia Curated by ChEMBL | Assay Description Antagonist activity at ETB receptor in human BSMC assessed as inhibition of endothelin-1 or 3-mediated Ca2+ mobilization preincubated for 5 mins foll... | Eur J Med Chem 121: 658-670 (2016) Article DOI: 10.1016/j.ejmech.2016.06.006 BindingDB Entry DOI: 10.7270/Q2T155MF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50061096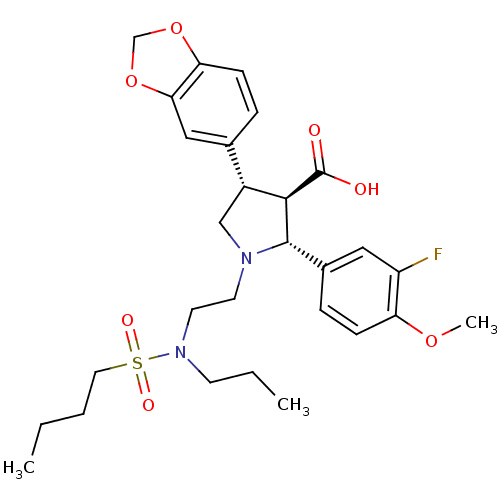 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-{2-[(butane-1...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.470 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding assay performed using human Endothelin B receptor (hETB) expressed in chinese hamster ovary cells(CHO) | J Med Chem 40: 3217-27 (1997) Article DOI: 10.1021/jm970101g BindingDB Entry DOI: 10.7270/Q2C24VJR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50061085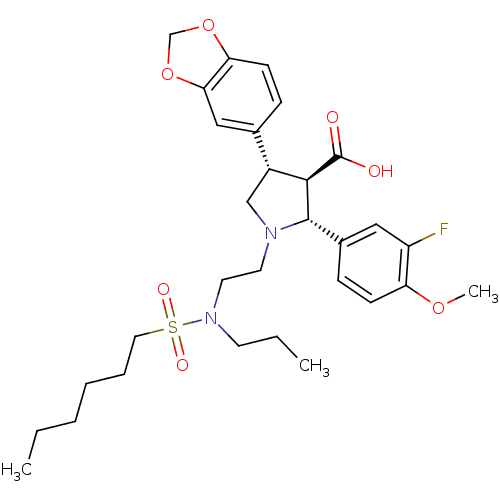 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-2-(3-fluoro-4-m...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.480 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding assay performed using human Endothelin B receptor (hETB) expressed in chinese hamster ovary cells(CHO) | J Med Chem 40: 3217-27 (1997) Article DOI: 10.1021/jm970101g BindingDB Entry DOI: 10.7270/Q2C24VJR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50061096 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-{2-[(butane-1...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Ability of the compound to block the ET-1-induced hydrolysis of inositol phosphate in Endothelin B receptor of chinese hamster ovary(CHO) cells. | J Med Chem 40: 3217-27 (1997) Article DOI: 10.1021/jm970101g BindingDB Entry DOI: 10.7270/Q2C24VJR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080623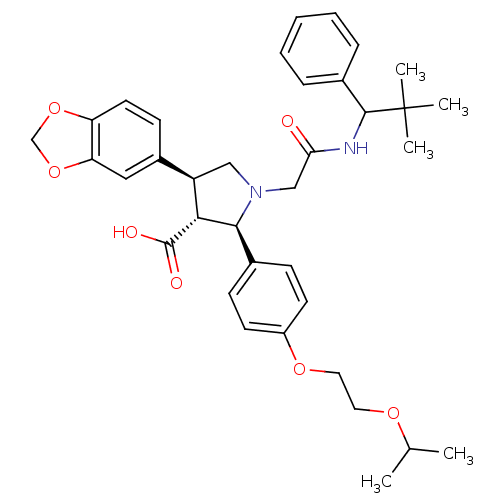 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(2,2-dimethy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.710 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50284769 ((S)-2-((R)-3-(2-Bromo-1H-indol-3-yl)-2-{(S)-2-cycl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Functional inhibition of ET-1 induced [Ca2+] increase in human Girardi heart (hGH) cells, which express Endothelin B receptor | Bioorg Med Chem Lett 5: 1483-1488 (1995) Article DOI: 10.1016/0960-894X(95)00237-N BindingDB Entry DOI: 10.7270/Q27D2VM3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080589 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(2,6-diethyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding affinity towards human Endothelin B receptor (hET -B) | J Med Chem 42: 3668-78 (1999) Article DOI: 10.1021/jm990170q BindingDB Entry DOI: 10.7270/Q24T6HK7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50061086 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-{2-[(3-chloro...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.830 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Ability of the compound to block the ET-1-induced hydrolysis of inositol phosphate in Endothelin B receptor of chinese hamster ovary(CHO) cells. | J Med Chem 40: 3217-27 (1997) Article DOI: 10.1021/jm970101g BindingDB Entry DOI: 10.7270/Q2C24VJR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080628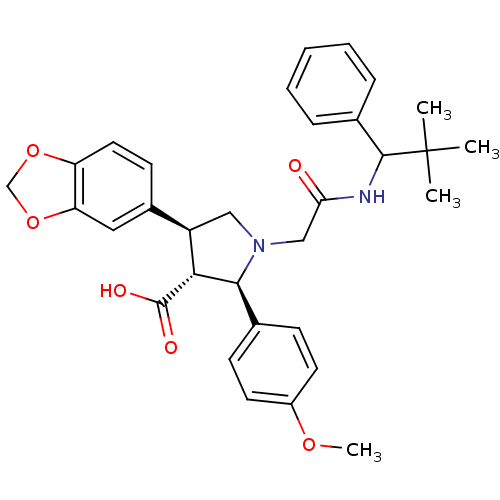 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(2,2-dimethy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.940 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080619 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(di-o-tolylm...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080597 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(2,6-diethyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding affinity towards human Endothelin B receptor (hET -B) | J Med Chem 42: 3668-78 (1999) Article DOI: 10.1021/jm990170q BindingDB Entry DOI: 10.7270/Q24T6HK7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50051385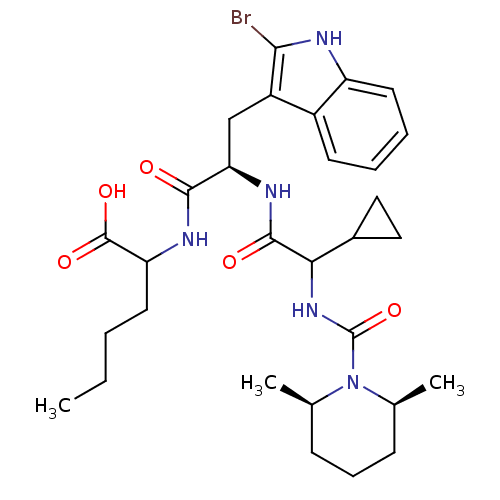 (2-((R)-3-(2-Bromo-1H-indol-3-yl)-2-{2-cyclopropyl-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute Curated by ChEMBL | Assay Description Binding affinity towards Endothelin B receptor in human girardi heart cell membranes | J Med Chem 39: 2313-30 (1996) Article DOI: 10.1021/jm9600914 BindingDB Entry DOI: 10.7270/Q2XS5W2G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080616 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(2-ethyl-1-p...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080597 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(2,6-diethyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding affinity towards human Endothelin B receptor (hET -B) | J Med Chem 42: 3668-78 (1999) Article DOI: 10.1021/jm990170q BindingDB Entry DOI: 10.7270/Q24T6HK7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50061085 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-2-(3-fluoro-4-m...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.12 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Ability of the compound to block the ET-1-induced hydrolysis of inositol phosphate in Endothelin B receptor of chinese hamster ovary(CHO) cells. | J Med Chem 40: 3217-27 (1997) Article DOI: 10.1021/jm970101g BindingDB Entry DOI: 10.7270/Q2C24VJR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080593 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(2,6-diethyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding affinity towards human Endothelin B receptor (hET -B) | J Med Chem 42: 3668-78 (1999) Article DOI: 10.1021/jm990170q BindingDB Entry DOI: 10.7270/Q24T6HK7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50286614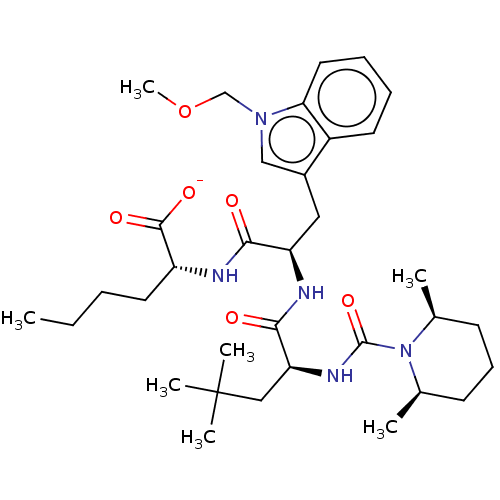 (CHEMBL155943 | CHEMBL3143473 | Sodium; 2-[2-{2-[(2...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Antagonistic activity against endothelin B (ETB) receptor using human girardi heart cells. | Bioorg Med Chem Lett 5: 621-626 (1995) Article DOI: 10.1016/0960-894X(95)00084-7 BindingDB Entry DOI: 10.7270/Q27081DM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50534382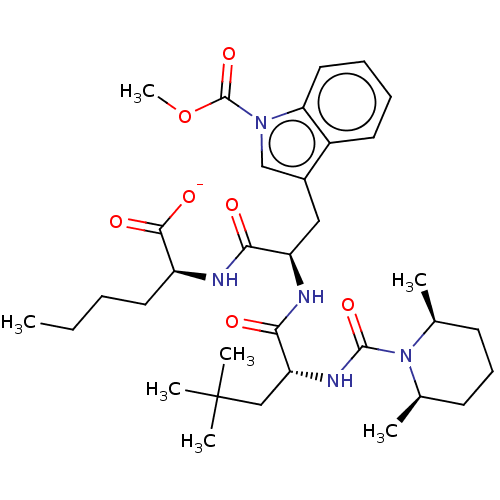 (CHEMBL4568795) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Centre Hospitalier Universitaire Vaudois (CHUV) Curated by ChEMBL | Assay Description Displacement of [125I]ET-1 from ET-B receptor in human Girardi heart cells incubated for 4 hrs by gamma counting method | J Med Chem 59: 8168-88 (2016) Article DOI: 10.1021/acs.jmedchem.5b01781 BindingDB Entry DOI: 10.7270/Q22N55RM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50249565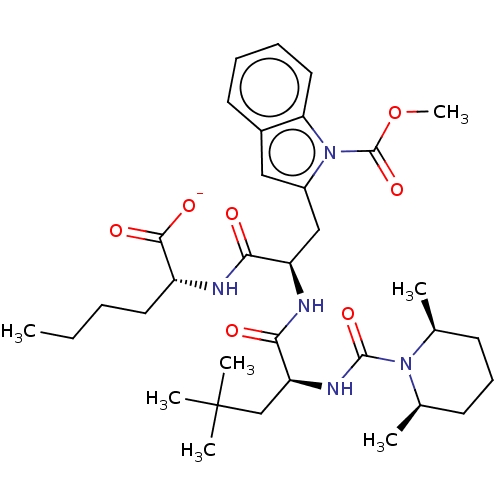 (CHEMBL4075238) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
St. John's University Curated by ChEMBL | Assay Description Antagonist activity at ETB receptor (unknown origin) expressed in HEK293T cells measured after 30 mins by CCF4-AM dye based GeneBlazer FRET assay | Bioorg Med Chem Lett 27: 2281-2285 (2017) Article DOI: 10.1016/j.bmcl.2017.04.049 BindingDB Entry DOI: 10.7270/Q2Q52S21 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080588 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(2,6-diethyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding affinity towards human Endothelin B receptor (hET -B) | J Med Chem 42: 3668-78 (1999) Article DOI: 10.1021/jm990170q BindingDB Entry DOI: 10.7270/Q24T6HK7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080588 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(2,6-diethyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding affinity towards human Endothelin B receptor (hET -B) | J Med Chem 42: 3668-78 (1999) Article DOI: 10.1021/jm990170q BindingDB Entry DOI: 10.7270/Q24T6HK7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50061074 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-2-(3-fluoro-4-m...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.28 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Ability of the compound to block the ET-1-induced hydrolysis of inositol phosphate in Endothelin B receptor of chinese hamster ovary(CHO) cells. | J Med Chem 40: 3217-27 (1997) Article DOI: 10.1021/jm970101g BindingDB Entry DOI: 10.7270/Q2C24VJR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50071433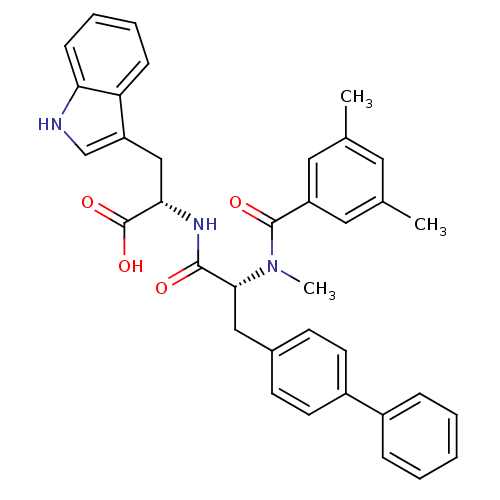 ((S)-2-{(R)-3-Biphenyl-4-yl-2-[(3,5-dimethyl-benzoy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Centre Hospitalier Universitaire Vaudois (CHUV) Curated by ChEMBL | Assay Description Displacement of [125I]ET-1 from human ET-B receptor expressed in CHO cell membrane incubated for 2 hrs | J Med Chem 59: 8168-88 (2016) Article DOI: 10.1021/acs.jmedchem.5b01781 BindingDB Entry DOI: 10.7270/Q22N55RM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080592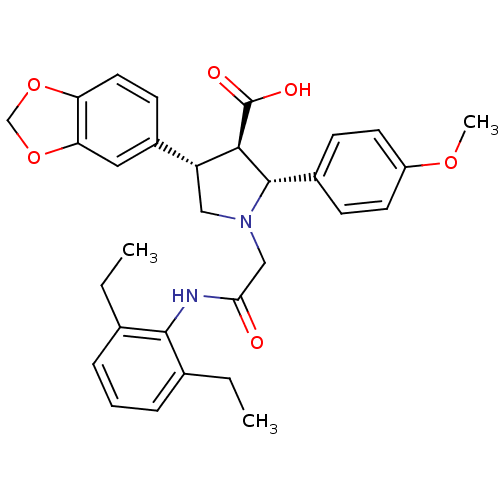 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(2,6-diethyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding affinity towards human Endothelin B receptor (hET -B) | J Med Chem 42: 3668-78 (1999) Article DOI: 10.1021/jm990170q BindingDB Entry DOI: 10.7270/Q24T6HK7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080592 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(2,6-diethyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding affinity towards human Endothelin B receptor (hET -B) | J Med Chem 42: 3668-78 (1999) Article DOI: 10.1021/jm990170q BindingDB Entry DOI: 10.7270/Q24T6HK7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50061078 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-2-(3-fluoro-4-m...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Ability of the compound to block the ET-1-induced hydrolysis of inositol phosphate in Endothelin B receptor of chinese hamster ovary(CHO) cells. | J Med Chem 40: 3217-27 (1997) Article DOI: 10.1021/jm970101g BindingDB Entry DOI: 10.7270/Q2C24VJR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080602 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-2-(4-methoxy-ph...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding affinity towards human Endothelin B receptor (hET -B) | J Med Chem 42: 3668-78 (1999) Article DOI: 10.1021/jm990170q BindingDB Entry DOI: 10.7270/Q24T6HK7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080614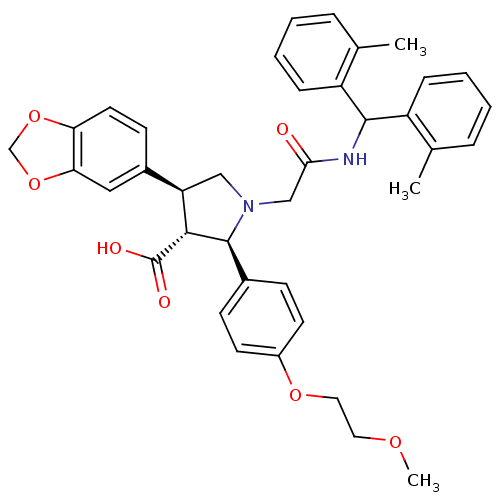 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(di-o-tolylm...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080617 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(di-o-tolylm...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080617 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(di-o-tolylm...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50124419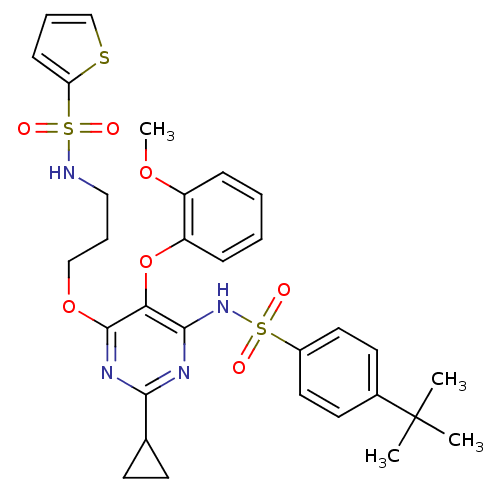 (CHEMBL172297 | Thiophene-2-sulfonic acid {3-[6-(4-...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Actelion Pharmaceuticals Ltd Curated by ChEMBL | Assay Description Inhibitory concentration required against [125I]-ET-1 binding to membranes of CHO cells expressing human ETB receptor | Bioorg Med Chem Lett 13: 951-4 (2003) BindingDB Entry DOI: 10.7270/Q2D50M9J | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080622 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-2-(4-methoxy-ph...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080624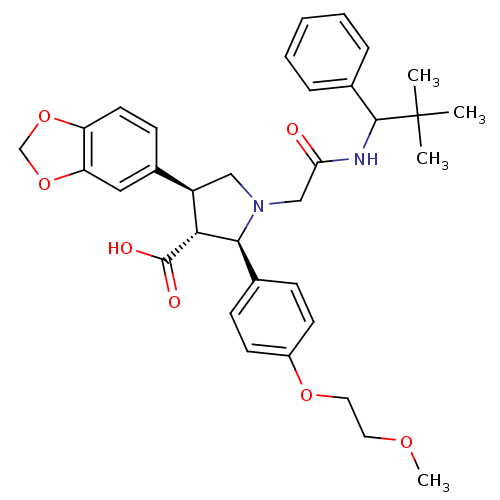 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-[(2,2-dimethy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080610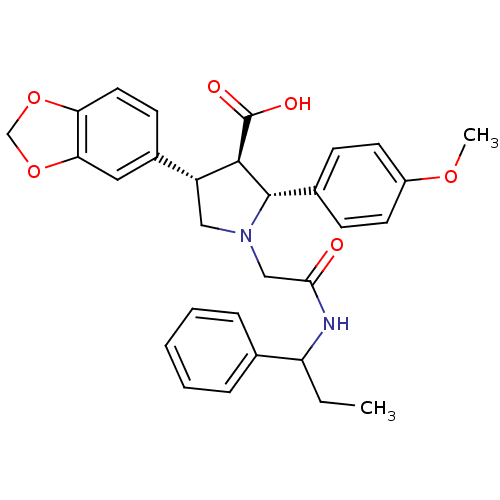 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-2-(4-methoxy-ph...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080627 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-1-({[(2-ethyl-p...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080615 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-2-(4-methoxy-ph...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50080620 ((2R,3R,4S)-4-Benzo[1,3]dioxol-5-yl-2-(4-methoxy-ph...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories Curated by ChEMBL | Assay Description Binding ability determined by the displacement of [125 I]ET-3 from the human Endothelin B receptor | J Med Chem 42: 3679-89 (1999) Article DOI: 10.1021/jm990171i BindingDB Entry DOI: 10.7270/Q2125RVT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50532557 (CHEMBL4435789) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Actelion Pharmaceuticals Ltd Curated by ChEMBL | Assay Description Displacement of [125I]-ET-1 from human ETB receptor expressed in CHO cell membranes after 2 hrs by scintillation counting | Bioorg Med Chem Lett 26: 3381-94 (2016) Article DOI: 10.1016/j.bmcl.2016.06.014 BindingDB Entry DOI: 10.7270/Q26113SR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50532557 (CHEMBL4435789) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Actelion Pharmaceuticals Ltd Curated by ChEMBL | Assay Description Displacement of [125I]-ET-1 from human ETB receptor expressed in CHO cell membranes after 2 hrs by scintillation counting | Bioorg Med Chem Lett 26: 3381-94 (2016) Article DOI: 10.1016/j.bmcl.2016.06.014 BindingDB Entry DOI: 10.7270/Q26113SR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50051406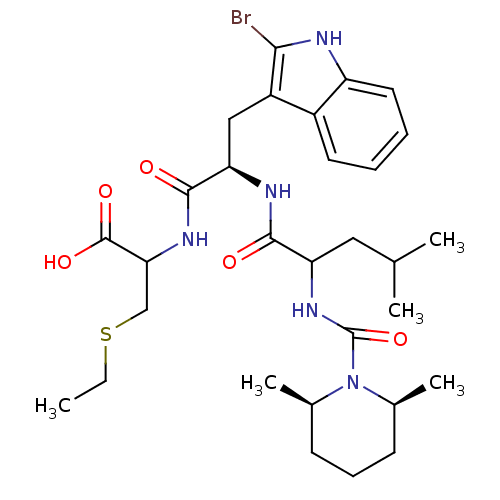 (2-((R)-3-(2-Bromo-1H-indol-3-yl)-2-{2-[((2S,6R)-2,...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute Curated by ChEMBL | Assay Description Binding affinity rowards Endothelin B receptor in human girardi heart cell membranes | J Med Chem 39: 2313-30 (1996) Article DOI: 10.1021/jm9600914 BindingDB Entry DOI: 10.7270/Q2XS5W2G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50075421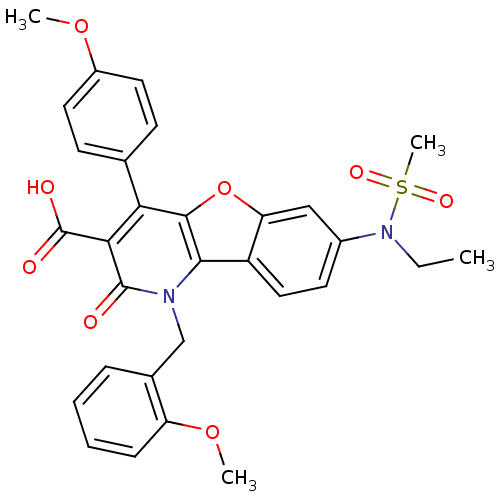 (7-(Ethyl-methanesulfonyl-amino)-1-(2-methoxy-benzy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck KGaA Curated by ChEMBL | Assay Description Ability to displace [125I]-Endothelin-1 from endothelin B receptor in porcine kidney membranes | Bioorg Med Chem Lett 9: 619-22 (1999) BindingDB Entry DOI: 10.7270/Q2DB811P | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Endothelin receptor type B (Homo sapiens (Human)) | BDBM50104987 (CHEMBL367445 | sodium salt of 4-tert-Butyl-N-{6-[2...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 3.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Tanabe Seiyaku Co., Ltd. Curated by ChEMBL | Assay Description In vitro for specific binding of [125I]-ET-1 to GHcell expressed in Endothelin B receptor | J Med Chem 44: 3355-68 (2001) BindingDB Entry DOI: 10.7270/Q2QN662B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 1260 total ) | Next | Last >> |