 Found 2270 hits with Last Name = 'ryan' and Initial = 'j'
Found 2270 hits with Last Name = 'ryan' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Mu-type opioid receptor
(MOUSE) | BDBM21865
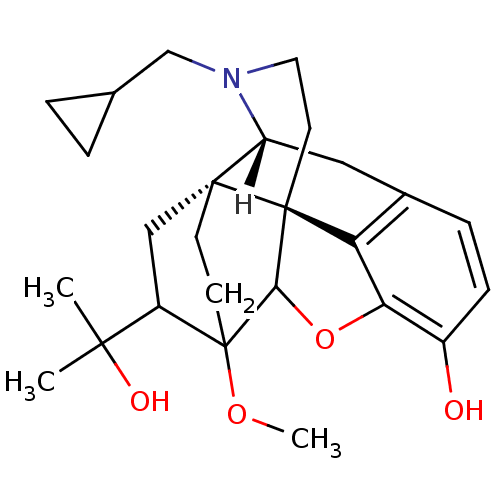
((14beta)-17-(cyclopropylmethyl)-18-(1-hydroxy-1-me...)Show SMILES [H][C@]12Cc3ccc(O)c4OC5[C@](CCN1CC1CC1)(c34)[C@@]21CCC5(OC)C(C1)C(C)(C)O |TLB:28:26:11.10:21.22,8:19:20:14.12.13,4:3:20:14.12.13| Show InChI InChI=1S/C26H35NO4/c1-23(2,29)18-13-24-8-9-26(18,30-3)22-25(24)10-11-27(14-15-4-5-15)19(24)12-16-6-7-17(28)21(31-22)20(16)25/h6-7,15,18-19,22,28-29H,4-5,8-14H2,1-3H3/t18?,19-,22?,24-,25+,26?/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50189257
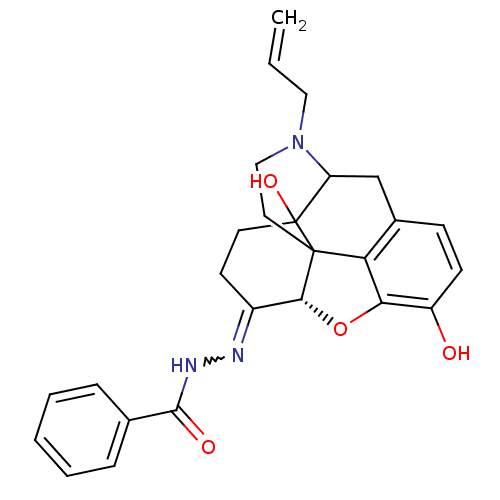
(CHEMBL378753 | NalBzOH)Show SMILES Oc1ccc2CC3N(CC=C)CCC45[C@H](Oc1c24)C(CCC35O)=NNC(=O)c1ccccc1 |w:23.28,TLB:22:21:7.11.12:17.5.4,THB:8:7:21:17.5.4,16:17:21:7.11.12| Show InChI InChI=1S/C26H27N3O4/c1-2-13-29-14-12-25-21-17-8-9-19(30)22(21)33-23(25)18(10-11-26(25,32)20(29)15-17)27-28-24(31)16-6-4-3-5-7-16/h2-9,20,23,30,32H,1,10-15H2,(H,28,31)/t20?,23-,25?,26?/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM21865

((14beta)-17-(cyclopropylmethyl)-18-(1-hydroxy-1-me...)Show SMILES [H][C@]12Cc3ccc(O)c4OC5[C@](CCN1CC1CC1)(c34)[C@@]21CCC5(OC)C(C1)C(C)(C)O |TLB:28:26:11.10:21.22,8:19:20:14.12.13,4:3:20:14.12.13| Show InChI InChI=1S/C26H35NO4/c1-23(2,29)18-13-24-8-9-26(18,30-3)22-25(24)10-11-27(14-15-4-5-15)19(24)12-16-6-7-17(28)21(31-22)20(16)25/h6-7,15,18-19,22,28-29H,4-5,8-14H2,1-3H3/t18?,19-,22?,24-,25+,26?/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
DrugBank
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50189257

(CHEMBL378753 | NalBzOH)Show SMILES Oc1ccc2CC3N(CC=C)CCC45[C@H](Oc1c24)C(CCC35O)=NNC(=O)c1ccccc1 |w:23.28,TLB:22:21:7.11.12:17.5.4,THB:8:7:21:17.5.4,16:17:21:7.11.12| Show InChI InChI=1S/C26H27N3O4/c1-2-13-29-14-12-25-21-17-8-9-19(30)22(21)33-23(25)18(10-11-26(25,32)20(29)15-17)27-28-24(31)16-6-4-3-5-7-16/h2-9,20,23,30,32H,1,10-15H2,(H,28,31)/t20?,23-,25?,26?/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50095150
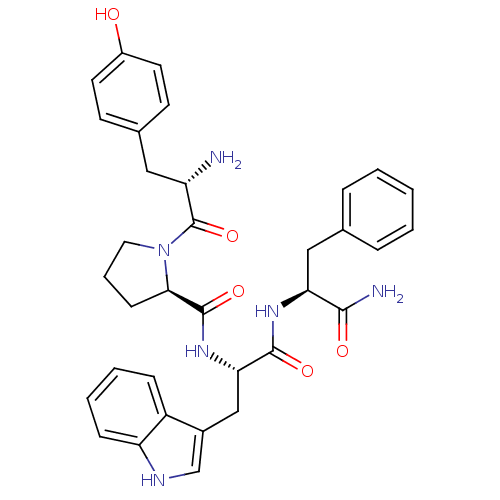
((R)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C34H38N6O5/c35-26(17-22-12-14-24(41)15-13-22)34(45)40-16-6-11-30(40)33(44)39-29(19-23-20-37-27-10-5-4-9-25(23)27)32(43)38-28(31(36)42)18-21-7-2-1-3-8-21/h1-5,7-10,12-15,20,26,28-30,37,41H,6,11,16-19,35H2,(H2,36,42)(H,38,43)(H,39,44)/t26-,28-,29-,30+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50000788
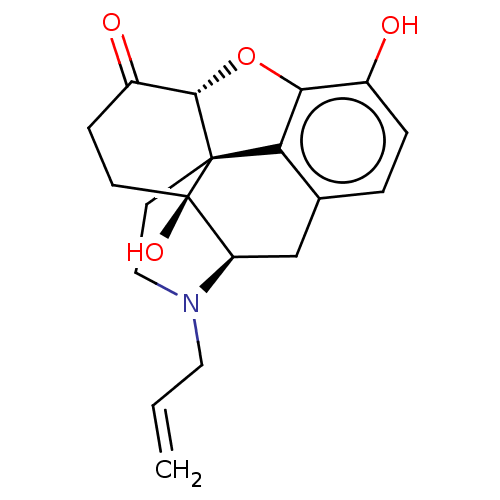
((morphine)4-methyl-(1S,5R,13R,14S,17R)-12-oxa-4-az...)Show SMILES Oc1ccc2C[C@H]3N(CC=C)CC[C@@]45[C@@H](Oc1c24)C(=O)CC[C@@]35O |r| Show InChI InChI=1S/C19H21NO4/c1-2-8-20-9-7-18-15-11-3-4-12(21)16(15)24-17(18)13(22)5-6-19(18,23)14(20)10-11/h2-4,14,17,21,23H,1,5-10H2/t14-,17+,18+,19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50067740
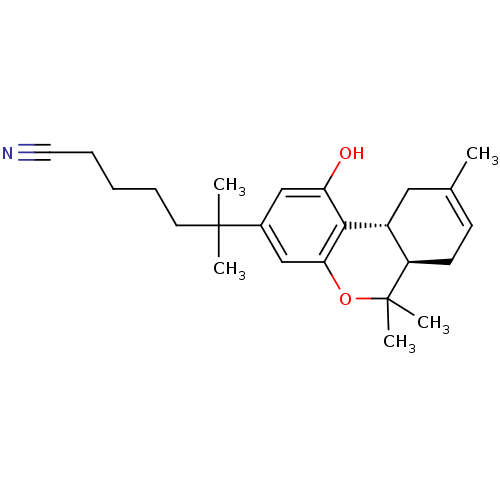
(6-((6aR,10aR)-1-Hydroxy-6,6,9-trimethyl-6a,7,10,10...)Show SMILES CC1=CC[C@@H]2[C@@H](C1)c1c(O)cc(cc1OC2(C)C)C(C)(C)CCCCC#N |t:1| Show InChI InChI=1S/C24H33NO2/c1-16-9-10-19-18(13-16)22-20(26)14-17(15-21(22)27-24(19,4)5)23(2,3)11-7-6-8-12-25/h9,14-15,18-19,26H,6-8,10-11,13H2,1-5H3/t18-,19-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organix, Inc.
Curated by ChEMBL
| Assay Description
Evaluated for its binding affinity towards Cannabinoid receptor 1 (CB1) |
J Med Chem 41: 4400-7 (1998)
Article DOI: 10.1021/jm9803875
BindingDB Entry DOI: 10.7270/Q2M909CZ |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(CALF) | BDBM50106276
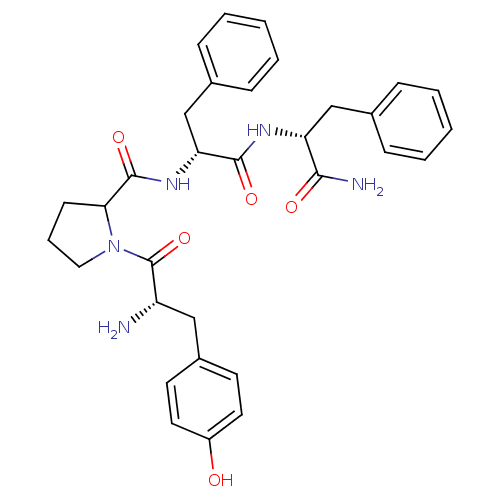
(1-[2-Amino-3-(4-hydroxy-phenyl)-propionyl]-pyrroli...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCCC1C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C32H37N5O5/c33-25(18-23-13-15-24(38)16-14-23)32(42)37-17-7-12-28(37)31(41)36-27(20-22-10-5-2-6-11-22)30(40)35-26(29(34)39)19-21-8-3-1-4-9-21/h1-6,8-11,13-16,25-28,38H,7,12,17-20,33H2,(H2,34,39)(H,35,40)(H,36,41)/t25-,26+,27+,28?/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50000092

((-)-(etorphine) | (-)-morphine | (1S,5R,13R,14S)-1...)Show SMILES CN1CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4O)ccc5O |r,c:16,TLB:13:12:8.9.10:3.2.1| Show InChI InChI=1S/C17H19NO3/c1-18-7-6-17-10-3-5-13(20)16(17)21-15-12(19)4-2-9(14(15)17)8-11(10)18/h2-5,10-11,13,16,19-20H,6-8H2,1H3/t10-,11+,13-,16-,17-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 0.450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mu-type opioid receptor
(MOUSE) | BDBM50008984
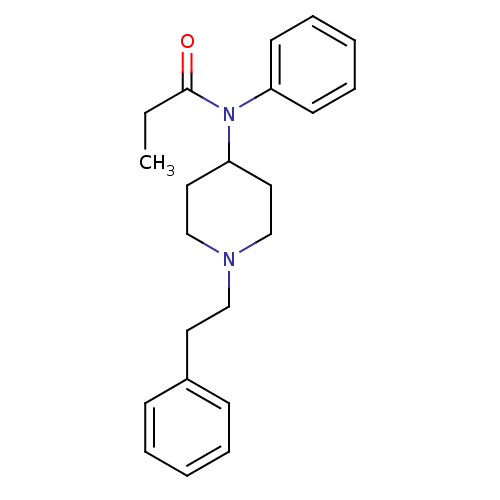
(4-(4-Chloro-benzyl)-2-(1-methyl-azepan-4-yl)-2H-ph...)Show InChI InChI=1S/C22H28N2O/c1-2-22(25)24(20-11-7-4-8-12-20)21-14-17-23(18-15-21)16-13-19-9-5-3-6-10-19/h3-12,21H,2,13-18H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 0.450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50106276

(1-[2-Amino-3-(4-hydroxy-phenyl)-propionyl]-pyrroli...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCCC1C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C32H37N5O5/c33-25(18-23-13-15-24(38)16-14-23)32(42)37-17-7-12-28(37)31(41)36-27(20-22-10-5-2-6-11-22)30(40)35-26(29(34)39)19-21-8-3-1-4-9-21/h1-6,8-11,13-16,25-28,38H,7,12,17-20,33H2,(H2,34,39)(H,35,40)(H,36,41)/t25-,26+,27+,28?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50067742

(7-((6aR,10aR)-1-Hydroxy-6,6,9-trimethyl-6a,7,10,10...)Show SMILES CC1=CC[C@@H]2[C@@H](C1)c1c(O)cc(cc1OC2(C)C)C(C)(C)CCCCCC#N |t:1| Show InChI InChI=1S/C25H35NO2/c1-17-10-11-20-19(14-17)23-21(27)15-18(16-22(23)28-25(20,4)5)24(2,3)12-8-6-7-9-13-26/h10,15-16,19-20,27H,6-9,11-12,14H2,1-5H3/t19-,20-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organix, Inc.
Curated by ChEMBL
| Assay Description
Evaluated for its binding affinity towards Cannabinoid receptor 1 (CB1) |
J Med Chem 41: 4400-7 (1998)
Article DOI: 10.1021/jm9803875
BindingDB Entry DOI: 10.7270/Q2M909CZ |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM21015

((2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydroxyphenyl...)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N(C)[C@@H](Cc1ccccc1)C(=O)NCCO Show InChI InChI=1S/C26H35N5O6/c1-17(30-25(36)21(27)14-19-8-10-20(33)11-9-19)24(35)29-16-23(34)31(2)22(26(37)28-12-13-32)15-18-6-4-3-5-7-18/h3-11,17,21-22,32-33H,12-16,27H2,1-2H3,(H,28,37)(H,29,35)(H,30,36)/t17-,21+,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM21015

((2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydroxyphenyl...)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N(C)[C@@H](Cc1ccccc1)C(=O)NCCO Show InChI InChI=1S/C26H35N5O6/c1-17(30-25(36)21(27)14-19-8-10-20(33)11-9-19)24(35)29-16-23(34)31(2)22(26(37)28-12-13-32)15-18-6-4-3-5-7-18/h3-11,17,21-22,32-33H,12-16,27H2,1-2H3,(H,28,37)(H,29,35)(H,30,36)/t17-,21+,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50095150

((R)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C34H38N6O5/c35-26(17-22-12-14-24(41)15-13-22)34(45)40-16-6-11-30(40)33(44)39-29(19-23-20-37-27-10-5-4-9-25(23)27)32(43)38-28(31(36)42)18-21-7-2-1-3-8-21/h1-5,7-10,12-15,20,26,28-30,37,41H,6,11,16-19,35H2,(H2,36,42)(H,38,43)(H,39,44)/t26-,28-,29-,30+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.660 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50000788

((morphine)4-methyl-(1S,5R,13R,14S,17R)-12-oxa-4-az...)Show SMILES Oc1ccc2C[C@H]3N(CC=C)CC[C@@]45[C@@H](Oc1c24)C(=O)CC[C@@]35O |r| Show InChI InChI=1S/C19H21NO4/c1-2-8-20-9-7-18-15-11-3-4-12(21)16(15)24-17(18)13(22)5-6-19(18,23)14(20)10-11/h2-4,14,17,21,23H,1,5-10H2/t14-,17+,18+,19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.670 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(CALF) | BDBM50095150

((R)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C34H38N6O5/c35-26(17-22-12-14-24(41)15-13-22)34(45)40-16-6-11-30(40)33(44)39-29(19-23-20-37-27-10-5-4-9-25(23)27)32(43)38-28(31(36)42)18-21-7-2-1-3-8-21/h1-5,7-10,12-15,20,26,28-30,37,41H,6,11,16-19,35H2,(H2,36,42)(H,38,43)(H,39,44)/t26-,28-,29-,30+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.670 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50095150

((R)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C34H38N6O5/c35-26(17-22-12-14-24(41)15-13-22)34(45)40-16-6-11-30(40)33(44)39-29(19-23-20-37-27-10-5-4-9-25(23)27)32(43)38-28(31(36)42)18-21-7-2-1-3-8-21/h1-5,7-10,12-15,20,26,28-30,37,41H,6,11,16-19,35H2,(H2,36,42)(H,38,43)(H,39,44)/t26-,28-,29-,30+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Type-1 angiotensin II receptor A
(RAT) | BDBM82259

(CAS_123856 | L-158,809 | NSC_123856)Show SMILES CCc1nc2c(C)cc(C)nc2n1-c1ccc(cc1)-c1ccccc1-c1nnn[nH]1 Show InChI InChI=1S/C23H21N7/c1-4-20-25-21-14(2)13-15(3)24-23(21)30(20)17-11-9-16(10-12-17)18-7-5-6-8-19(18)22-26-28-29-27-22/h5-13H,4H2,1-3H3,(H,26,27,28,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Parke-Davis Pharmaceutical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 272: 963-9 (1995)
BindingDB Entry DOI: 10.7270/Q2J38R2N |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50067735
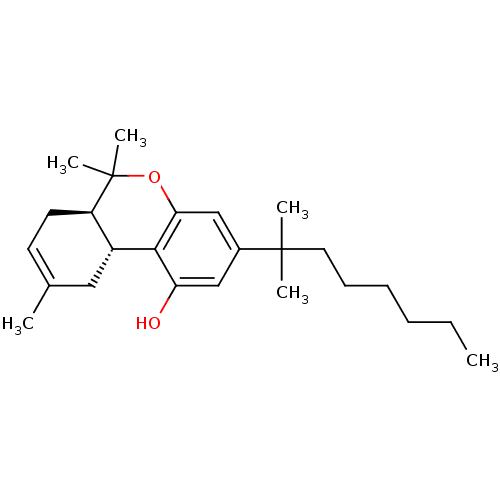
((6aR,10aR)-3-(1,1-Dimethyl-heptyl)-6,6,9-trimethyl...)Show SMILES CCCCCCC(C)(C)c1cc(O)c2[C@@H]3CC(C)=CC[C@H]3C(C)(C)Oc2c1 |c:17| Show InChI InChI=1S/C25H38O2/c1-7-8-9-10-13-24(3,4)18-15-21(26)23-19-14-17(2)11-12-20(19)25(5,6)27-22(23)16-18/h11,15-16,19-20,26H,7-10,12-14H2,1-6H3/t19-,20-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.770 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organix, Inc.
Curated by ChEMBL
| Assay Description
Evaluated for its binding affinity towards Cannabinoid receptor 1 (CB1) |
J Med Chem 41: 4400-7 (1998)
Article DOI: 10.1021/jm9803875
BindingDB Entry DOI: 10.7270/Q2M909CZ |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000092

((-)-(etorphine) | (-)-morphine | (1S,5R,13R,14S)-1...)Show SMILES CN1CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4O)ccc5O |r,c:16,TLB:13:12:8.9.10:3.2.1| Show InChI InChI=1S/C17H19NO3/c1-18-7-6-17-10-3-5-13(20)16(17)21-15-12(19)4-2-9(14(15)17)8-11(10)18/h2-5,10-11,13,16,19-20H,6-8H2,1H3/t10-,11+,13-,16-,17-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 0.820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mu-type opioid receptor
(MOUSE) | BDBM21015

((2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydroxyphenyl...)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N(C)[C@@H](Cc1ccccc1)C(=O)NCCO Show InChI InChI=1S/C26H35N5O6/c1-17(30-25(36)21(27)14-19-8-10-20(33)11-9-19)24(35)29-16-23(34)31(2)22(26(37)28-12-13-32)15-18-6-4-3-5-7-18/h3-11,17,21-22,32-33H,12-16,27H2,1-2H3,(H,28,37)(H,29,35)(H,30,36)/t17-,21+,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.820 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50067741
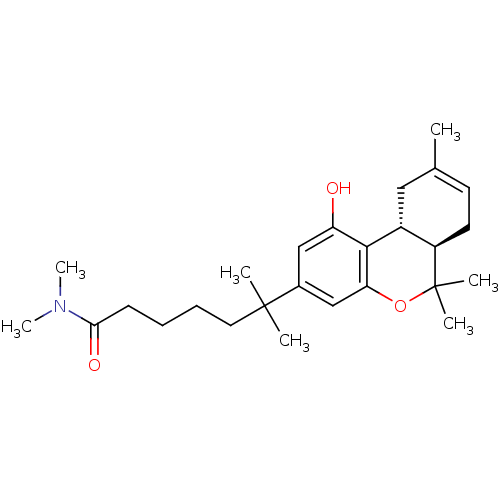
(6-((6aR,10aR)-1-Hydroxy-6,6,9-trimethyl-6a,7,10,10...)Show SMILES CN(C)C(=O)CCCCC(C)(C)c1cc(O)c2[C@@H]3CC(C)=CC[C@H]3C(C)(C)Oc2c1 |c:20| Show InChI InChI=1S/C26H39NO3/c1-17-11-12-20-19(14-17)24-21(28)15-18(16-22(24)30-26(20,4)5)25(2,3)13-9-8-10-23(29)27(6)7/h11,15-16,19-20,28H,8-10,12-14H2,1-7H3/t19-,20-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.860 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organix, Inc.
Curated by ChEMBL
| Assay Description
Evaluated for its binding affinity towards Cannabinoid receptor 1 (CB1) |
J Med Chem 41: 4400-7 (1998)
Article DOI: 10.1021/jm9803875
BindingDB Entry DOI: 10.7270/Q2M909CZ |
More data for this
Ligand-Target Pair | |
E3 ubiquitin-protein ligase Mdm2
(Homo sapiens (Human)) | BDBM50433561
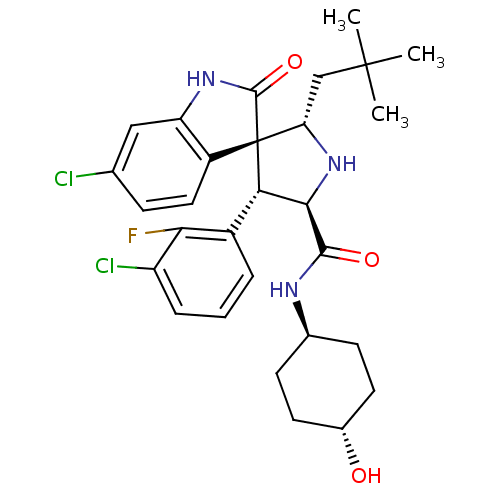
(CHEMBL2381408)Show SMILES CC(C)(C)C[C@@H]1N[C@H]([C@H](c2cccc(Cl)c2F)[C@]11C(=O)Nc2cc(Cl)ccc12)C(=O)N[C@H]1CC[C@H](O)CC1 |r,wU:8.8,5.4,31.34,wD:17.29,7.31,34.38,(40.88,-57.18,;39.34,-57.18,;38.57,-58.51,;40.11,-58.5,;38.57,-55.84,;37.04,-55.84,;37.52,-54.37,;36.27,-53.47,;35.03,-54.37,;33.51,-54.09,;32.52,-55.27,;31.01,-54.99,;30.49,-53.53,;31.49,-52.36,;30.98,-50.9,;33,-52.64,;34.01,-51.48,;35.49,-55.84,;36.38,-57.1,;37.93,-57.11,;35.48,-58.34,;34.01,-57.86,;32.69,-58.61,;31.36,-57.84,;30.03,-58.6,;31.37,-56.3,;32.71,-55.54,;34.03,-56.32,;36.26,-51.93,;34.93,-51.16,;37.6,-51.15,;37.6,-49.61,;36.26,-48.86,;36.26,-47.31,;37.6,-46.54,;37.6,-45,;38.93,-47.32,;38.93,-48.85,)| Show InChI InChI=1S/C29H34Cl2FN3O3/c1-28(2,3)14-22-29(19-12-7-15(30)13-21(19)34-27(29)38)23(18-5-4-6-20(31)24(18)32)25(35-22)26(37)33-16-8-10-17(36)11-9-16/h4-7,12-13,16-17,22-23,25,35-36H,8-11,14H2,1-3H3,(H,33,37)(H,34,38)/t16-,17-,22-,23-,25+,29+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 0.880 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Binding affinity to recombinant human His6-tagged HDM2 (1 to 118 residues) assessed as reduction in PMDM6-F binding incubated for 15 to 30 mins by fl... |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.1c01524
BindingDB Entry DOI: 10.7270/Q2Z03D1V |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mu-type opioid receptor
(MOUSE) | BDBM50008984

(4-(4-Chloro-benzyl)-2-(1-methyl-azepan-4-yl)-2H-ph...)Show InChI InChI=1S/C22H28N2O/c1-2-22(25)24(20-11-7-4-8-12-20)21-14-17-23(18-15-21)16-13-19-9-5-3-6-10-19/h3-12,21H,2,13-18H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 0.940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mu-type opioid receptor
(MOUSE) | BDBM50095150

((R)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C34H38N6O5/c35-26(17-22-12-14-24(41)15-13-22)34(45)40-16-6-11-30(40)33(44)39-29(19-23-20-37-27-10-5-4-9-25(23)27)32(43)38-28(31(36)42)18-21-7-2-1-3-8-21/h1-5,7-10,12-15,20,26,28-30,37,41H,6,11,16-19,35H2,(H2,36,42)(H,38,43)(H,39,44)/t26-,28-,29-,30+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.940 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50016846
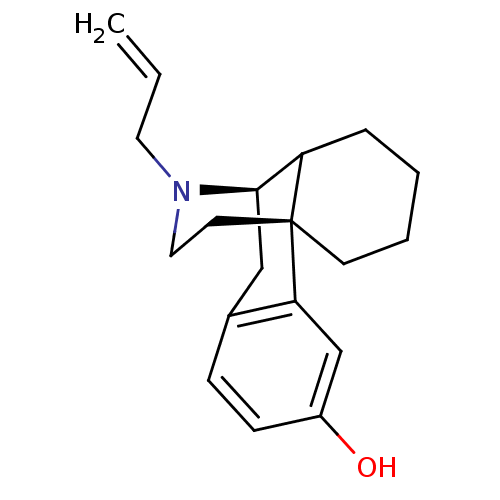
((levallorphan)17-allyl-(1R,9R)-17-azatetracyclo[7....)Show SMILES Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC=C)c2c1 |TLB:8:7:19.4.5:15.13.14| Show InChI InChI=1S/C19H25NO/c1-2-10-20-11-9-19-8-4-3-5-16(19)18(20)12-14-6-7-15(21)13-17(14)19/h2,6-7,13,16,18,21H,1,3-5,8-12H2/t16?,18-,19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.960 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50016846

((levallorphan)17-allyl-(1R,9R)-17-azatetracyclo[7....)Show SMILES Oc1ccc2C[C@@H]3C4CCCC[C@]4(CCN3CC=C)c2c1 |TLB:8:7:19.4.5:15.13.14| Show InChI InChI=1S/C19H25NO/c1-2-10-20-11-9-19-8-4-3-5-16(19)18(20)12-14-6-7-15(21)13-17(14)19/h2,6-7,13,16,18,21H,1,3-5,8-12H2/t16?,18-,19-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.970 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Mus musculus (Mouse)) | BDBM50060612
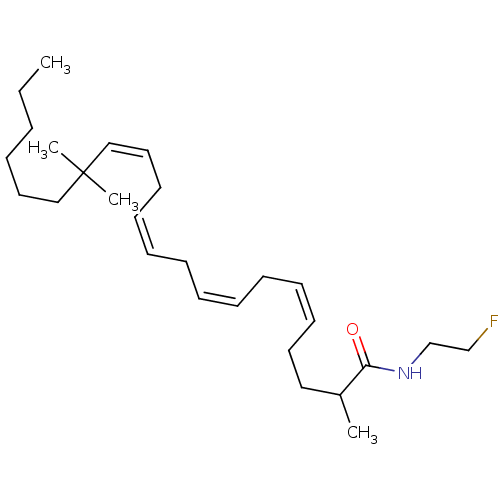
((5Z,8Z,11Z,14Z)-2,16,16-Trimethyl-docosa-5,8,11,14...)Show SMILES CCCCCCC(C)(C)\C=C/C\C=C/C\C=C/C\C=C/CCC(C)C(=O)NCCF Show InChI InChI=1S/C27H46FNO/c1-5-6-7-18-21-27(3,4)22-19-16-14-12-10-8-9-11-13-15-17-20-25(2)26(30)29-24-23-28/h8-9,12-15,19,22,25H,5-7,10-11,16-18,20-21,23-24H2,1-4H3,(H,29,30)/b9-8-,14-12-,15-13-,22-19- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organix, Inc.
Curated by ChEMBL
| Assay Description
Binding affinity for Cannabinoid receptor 1 in presence of phenylmethylsulfonyl fluoride (PMSF) |
J Med Chem 40: 3617-25 (1997)
Article DOI: 10.1021/jm970212f
BindingDB Entry DOI: 10.7270/Q2D50NNF |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50000092

((-)-(etorphine) | (-)-morphine | (1S,5R,13R,14S)-1...)Show SMILES CN1CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4O)ccc5O |r,c:16,TLB:13:12:8.9.10:3.2.1| Show InChI InChI=1S/C17H19NO3/c1-18-7-6-17-10-3-5-13(20)16(17)21-15-12(19)4-2-9(14(15)17)8-11(10)18/h2-5,10-11,13,16,19-20H,6-8H2,1H3/t10-,11+,13-,16-,17-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 1.04 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM50106276

(1-[2-Amino-3-(4-hydroxy-phenyl)-propionyl]-pyrroli...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCCC1C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C32H37N5O5/c33-25(18-23-13-15-24(38)16-14-23)32(42)37-17-7-12-28(37)31(41)36-27(20-22-10-5-2-6-11-22)30(40)35-26(29(34)39)19-21-8-3-1-4-9-21/h1-6,8-11,13-16,25-28,38H,7,12,17-20,33H2,(H2,34,39)(H,35,40)(H,36,41)/t25-,26+,27+,28?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50067730

(6-((6aR,10aR)-1-Hydroxy-6,6,9-trimethyl-6a,7,10,10...)Show SMILES CC1=CC[C@@H]2[C@@H](C1)c1c(O)cc(cc1OC2(C)C)C(C)(C)CCCCC(=O)NN1CCCCC1 |t:1| Show InChI InChI=1S/C29H44N2O3/c1-20-12-13-23-22(17-20)27-24(32)18-21(19-25(27)34-29(23,4)5)28(2,3)14-8-7-11-26(33)30-31-15-9-6-10-16-31/h12,18-19,22-23,32H,6-11,13-17H2,1-5H3,(H,30,33)/t22-,23-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organix, Inc.
Curated by ChEMBL
| Assay Description
Evaluated for its binding affinity towards Cannabinoid receptor 1 (CB1) |
J Med Chem 41: 4400-7 (1998)
Article DOI: 10.1021/jm9803875
BindingDB Entry DOI: 10.7270/Q2M909CZ |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50019056
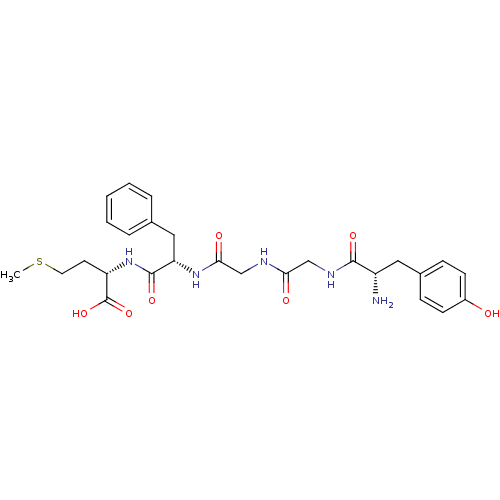
((S)-2-[(S)-2-(2-{2-[(S)-2-Amino-3-(4-hydroxy-pheny...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(O)=O Show InChI InChI=1S/C27H35N5O7S/c1-40-12-11-21(27(38)39)32-26(37)22(14-17-5-3-2-4-6-17)31-24(35)16-29-23(34)15-30-25(36)20(28)13-18-7-9-19(33)10-8-18/h2-10,20-22,33H,11-16,28H2,1H3,(H,29,34)(H,30,36)(H,31,35)(H,32,37)(H,38,39)/t20-,21-,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50019056

((S)-2-[(S)-2-(2-{2-[(S)-2-Amino-3-(4-hydroxy-pheny...)Show SMILES CSCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(O)=O Show InChI InChI=1S/C27H35N5O7S/c1-40-12-11-21(27(38)39)32-26(37)22(14-17-5-3-2-4-6-17)31-24(35)16-29-23(34)15-30-25(36)20(28)13-18-7-9-19(33)10-8-18/h2-10,20-22,33H,11-16,28H2,1H3,(H,29,34)(H,30,36)(H,31,35)(H,32,37)(H,38,39)/t20-,21-,22-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.28 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 3
(Homo sapiens (Human)) | BDBM50062522
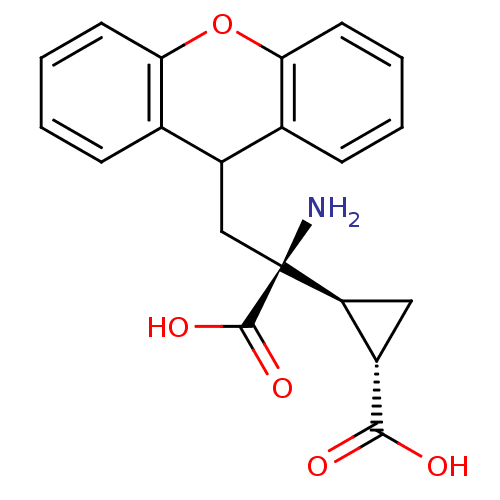
((1S,2S)-2-((S)-1-amino-1-carboxy-2-(9H-xanthen-9-y...)Show SMILES N[C@@](CC1c2ccccc2Oc2ccccc12)([C@H]1C[C@@H]1C(O)=O)C(O)=O Show InChI InChI=1S/C20H19NO5/c21-20(19(24)25,15-9-13(15)18(22)23)10-14-11-5-1-3-7-16(11)26-17-8-4-2-6-12(14)17/h1-8,13-15H,9-10,21H2,(H,22,23)(H,24,25)/t13-,15-,20-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-LY459477 from recombinant human mGlu3 receptor expressed in hamster AV12 cell membranes co-expressing rat EAAT1 incubated for 90... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2611419 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mu-type opioid receptor
(MOUSE) | BDBM50106276

(1-[2-Amino-3-(4-hydroxy-phenyl)-propionyl]-pyrroli...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCCC1C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C32H37N5O5/c33-25(18-23-13-15-24(38)16-14-23)32(42)37-17-7-12-28(37)31(41)36-27(20-22-10-5-2-6-11-22)30(40)35-26(29(34)39)19-21-8-3-1-4-9-21/h1-6,8-11,13-16,25-28,38H,7,12,17-20,33H2,(H2,34,39)(H,35,40)(H,36,41)/t25-,26+,27+,28?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(Rattus norvegicus (rat)) | BDBM21015

((2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydroxyphenyl...)Show SMILES C[C@@H](NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(=O)NCC(=O)N(C)[C@@H](Cc1ccccc1)C(=O)NCCO Show InChI InChI=1S/C26H35N5O6/c1-17(30-25(36)21(27)14-19-8-10-20(33)11-9-19)24(35)29-16-23(34)31(2)22(26(37)28-12-13-32)15-18-6-4-3-5-7-18/h3-11,17,21-22,32-33H,12-16,27H2,1-2H3,(H,28,37)(H,29,35)(H,30,36)/t17-,21+,22+/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.49 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50106276

(1-[2-Amino-3-(4-hydroxy-phenyl)-propionyl]-pyrroli...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCCC1C(=O)N[C@H](Cc1ccccc1)C(=O)N[C@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C32H37N5O5/c33-25(18-23-13-15-24(38)16-14-23)32(42)37-17-7-12-28(37)31(41)36-27(20-22-10-5-2-6-11-22)30(40)35-26(29(34)39)19-21-8-3-1-4-9-21/h1-6,8-11,13-16,25-28,38H,7,12,17-20,33H2,(H2,34,39)(H,35,40)(H,36,41)/t25-,26+,27+,28?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.57 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Type-2 angiotensin II receptor
(RAT) | BDBM50009338
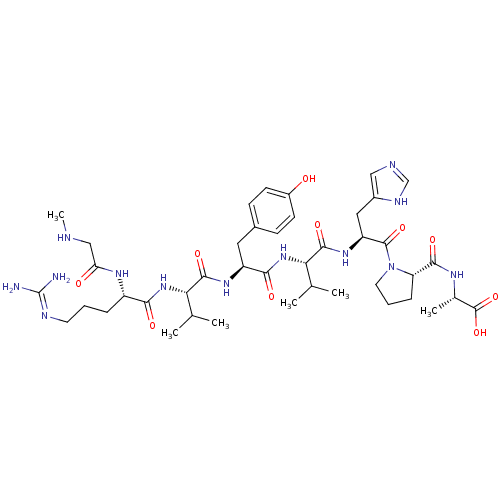
((S)-2-((S)-1-((S)-2-((S)-2-((S)-2-((S)-2-((S)-5-(d...)Show SMILES CNCC(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C)C(O)=O |r,wU:60.63,wD:24.23,36.36,43.43,56.59,17.16,6.5,(6.84,-31.4,;5.3,-31.4,;4.53,-30.07,;2.99,-30.07,;2.22,-31.4,;2.22,-28.73,;2.99,-27.4,;4.53,-27.4,;5.3,-26.07,;4.53,-24.73,;5.3,-23.4,;6.84,-23.4,;7.61,-24.73,;7.61,-22.07,;2.22,-26.07,;2.99,-24.73,;.68,-26.07,;-.09,-24.73,;.68,-23.4,;2.22,-23.4,;-.09,-22.07,;-1.63,-24.73,;-2.4,-23.4,;-2.4,-26.07,;-3.94,-26.07,;-4.71,-24.73,;-6.25,-24.73,;-7.02,-23.4,;-8.56,-23.4,;-9.33,-24.73,;-10.87,-24.73,;-8.56,-26.07,;-7.02,-26.07,;-4.71,-27.4,;-6.25,-27.4,;-3.94,-28.73,;-4.71,-30.07,;-6.25,-30.07,;-7.02,-28.73,;-7.02,-31.4,;-3.94,-31.4,;-2.4,-31.4,;-4.71,-32.73,;-3.94,-34.07,;-4.71,-35.4,;-6.25,-35.4,;-7.15,-34.16,;-8.62,-34.63,;-8.62,-36.17,;-7.15,-36.65,;-2.4,-34.07,;-1.63,-32.73,;-1.63,-35.4,;-2.26,-36.81,;-1.12,-37.84,;.22,-37.07,;-.1,-35.56,;.93,-34.42,;.45,-32.95,;2.42,-34.82,;3.5,-33.73,;3.11,-32.24,;5,-34.13,;6.09,-33.04,;5.39,-35.61,)| Show InChI InChI=1S/C42H65N13O10/c1-22(2)33(53-35(58)28(50-32(57)20-45-6)9-7-15-47-42(43)44)38(61)51-29(17-25-11-13-27(56)14-12-25)36(59)54-34(23(3)4)39(62)52-30(18-26-19-46-21-48-26)40(63)55-16-8-10-31(55)37(60)49-24(5)41(64)65/h11-14,19,21-24,28-31,33-34,45,56H,7-10,15-18,20H2,1-6H3,(H,46,48)(H,49,60)(H,50,57)(H,51,61)(H,52,62)(H,53,58)(H,54,59)(H,64,65)(H4,43,44,47)/t24-,28-,29-,30-,31-,33-,34-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Parke-Davis Pharmaceutical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 272: 963-9 (1995)
BindingDB Entry DOI: 10.7270/Q2J38R2N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM21025
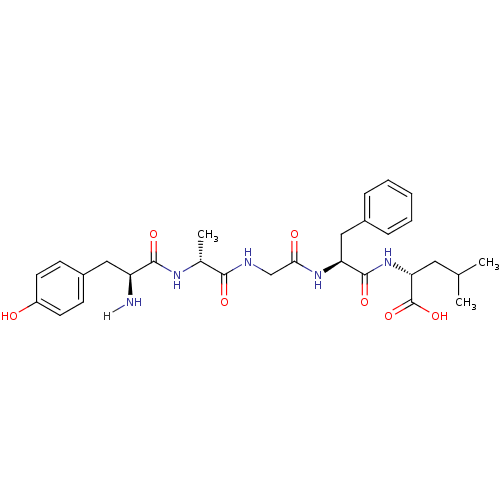
((2R)-2-[(2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydro...)Show SMILES CC(C)C[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(O)=O Show InChI InChI=1S/C29H39N5O7/c1-17(2)13-24(29(40)41)34-28(39)23(15-19-7-5-4-6-8-19)33-25(36)16-31-26(37)18(3)32-27(38)22(30)14-20-9-11-21(35)12-10-20/h4-12,17-18,22-24,35H,13-16,30H2,1-3H3,(H,31,37)(H,32,38)(H,33,36)(H,34,39)(H,40,41)/t18-,22+,23+,24-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50000092

((-)-(etorphine) | (-)-morphine | (1S,5R,13R,14S)-1...)Show SMILES CN1CC[C@@]23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4O)ccc5O |r,c:16,TLB:13:12:8.9.10:3.2.1| Show InChI InChI=1S/C17H19NO3/c1-18-7-6-17-10-3-5-13(20)16(17)21-15-12(19)4-2-9(14(15)17)8-11(10)18/h2-5,10-11,13,16,19-20H,6-8H2,1H3/t10-,11+,13-,16-,17-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 1.68 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Type-1 angiotensin II receptor A
(RAT) | BDBM50009338

((S)-2-((S)-1-((S)-2-((S)-2-((S)-2-((S)-2-((S)-5-(d...)Show SMILES CNCC(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](Cc1ccc(O)cc1)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](Cc1cnc[nH]1)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C)C(O)=O |r,wU:60.63,wD:24.23,36.36,43.43,56.59,17.16,6.5,(6.84,-31.4,;5.3,-31.4,;4.53,-30.07,;2.99,-30.07,;2.22,-31.4,;2.22,-28.73,;2.99,-27.4,;4.53,-27.4,;5.3,-26.07,;4.53,-24.73,;5.3,-23.4,;6.84,-23.4,;7.61,-24.73,;7.61,-22.07,;2.22,-26.07,;2.99,-24.73,;.68,-26.07,;-.09,-24.73,;.68,-23.4,;2.22,-23.4,;-.09,-22.07,;-1.63,-24.73,;-2.4,-23.4,;-2.4,-26.07,;-3.94,-26.07,;-4.71,-24.73,;-6.25,-24.73,;-7.02,-23.4,;-8.56,-23.4,;-9.33,-24.73,;-10.87,-24.73,;-8.56,-26.07,;-7.02,-26.07,;-4.71,-27.4,;-6.25,-27.4,;-3.94,-28.73,;-4.71,-30.07,;-6.25,-30.07,;-7.02,-28.73,;-7.02,-31.4,;-3.94,-31.4,;-2.4,-31.4,;-4.71,-32.73,;-3.94,-34.07,;-4.71,-35.4,;-6.25,-35.4,;-7.15,-34.16,;-8.62,-34.63,;-8.62,-36.17,;-7.15,-36.65,;-2.4,-34.07,;-1.63,-32.73,;-1.63,-35.4,;-2.26,-36.81,;-1.12,-37.84,;.22,-37.07,;-.1,-35.56,;.93,-34.42,;.45,-32.95,;2.42,-34.82,;3.5,-33.73,;3.11,-32.24,;5,-34.13,;6.09,-33.04,;5.39,-35.61,)| Show InChI InChI=1S/C42H65N13O10/c1-22(2)33(53-35(58)28(50-32(57)20-45-6)9-7-15-47-42(43)44)38(61)51-29(17-25-11-13-27(56)14-12-25)36(59)54-34(23(3)4)39(62)52-30(18-26-19-46-21-48-26)40(63)55-16-8-10-31(55)37(60)49-24(5)41(64)65/h11-14,19,21-24,28-31,33-34,45,56H,7-10,15-18,20H2,1-6H3,(H,46,48)(H,49,60)(H,50,57)(H,51,61)(H,52,62)(H,53,58)(H,54,59)(H,64,65)(H4,43,44,47)/t24-,28-,29-,30-,31-,33-,34-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Parke-Davis Pharmaceutical Research
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 272: 963-9 (1995)
BindingDB Entry DOI: 10.7270/Q2J38R2N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM82083
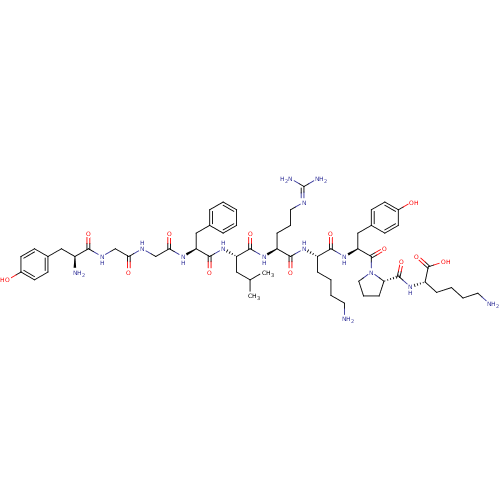
(Alpha-Neoendorphin | CAS_69671-17-6 | CB0427223)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-c1ccccc1)-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6]-[#7]-[#6](=O)-[#6@@H](-[#7])-[#6]-c1ccc(-[#8])cc1)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-c1ccc(-[#8])cc1)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](-[#8])=O Show InChI InChI=1S/C60H89N15O13/c1-36(2)30-46(73-56(84)47(32-37-12-4-3-5-13-37)69-51(79)35-67-50(78)34-68-52(80)42(63)31-38-18-22-40(76)23-19-38)55(83)71-44(16-10-28-66-60(64)65)53(81)70-43(14-6-8-26-61)54(82)74-48(33-39-20-24-41(77)25-21-39)58(86)75-29-11-17-49(75)57(85)72-45(59(87)88)15-7-9-27-62/h3-5,12-13,18-25,36,42-49,76-77H,6-11,14-17,26-35,61-63H2,1-2H3,(H,67,78)(H,68,80)(H,69,79)(H,70,81)(H,71,83)(H,72,85)(H,73,84)(H,74,82)(H,87,88)(H4,64,65,66)/t42-,43-,44-,45-,46-,47-,48-,49-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 2.15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50125682
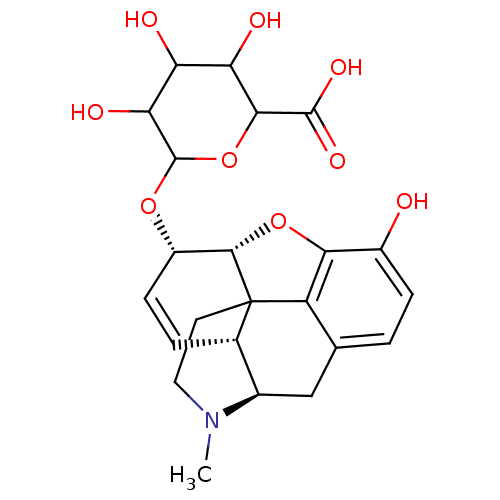
(3,4,5-trihydroxy-6-[10-hydroxy-4-methyl-(5R,13R,14...)Show SMILES CN1CCC23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4OC1OC(C(O)C(O)C1O)C(O)=O)ccc5O |c:16,THB:0:1:12:8.9.10| Show InChI InChI=1S/C23H27NO9/c1-24-7-6-23-10-3-5-13(31-22-17(28)15(26)16(27)19(33-22)21(29)30)20(23)32-18-12(25)4-2-9(14(18)23)8-11(10)24/h2-5,10-11,13,15-17,19-20,22,25-28H,6-8H2,1H3,(H,29,30)/t10-,11+,13-,15?,16?,17?,19?,20-,22?,23?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.35 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM50125682

(3,4,5-trihydroxy-6-[10-hydroxy-4-methyl-(5R,13R,14...)Show SMILES CN1CCC23[C@H]4Oc5c2c(C[C@@H]1[C@@H]3C=C[C@@H]4OC1OC(C(O)C(O)C1O)C(O)=O)ccc5O |c:16,THB:0:1:12:8.9.10| Show InChI InChI=1S/C23H27NO9/c1-24-7-6-23-10-3-5-13(31-22-17(28)15(26)16(27)19(33-22)21(29)30)20(23)32-18-12(25)4-2-9(14(18)23)8-11(10)24/h2-5,10-11,13,15-17,19-20,22,25-28H,6-8H2,1H3,(H,29,30)/t10-,11+,13-,15?,16?,17?,19?,20-,22?,23?/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 2.47 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(MOUSE) | BDBM21025

((2R)-2-[(2S)-2-{2-[(2R)-2-[(2S)-2-amino-3-(4-hydro...)Show SMILES CC(C)C[C@@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)[C@@H](C)NC(=O)[C@@H](N)Cc1ccc(O)cc1)C(O)=O Show InChI InChI=1S/C29H39N5O7/c1-17(2)13-24(29(40)41)34-28(39)23(15-19-7-5-4-6-8-19)33-25(36)16-31-26(37)18(3)32-27(38)22(30)14-20-9-11-21(35)12-10-20/h4-12,17-18,22-24,35H,13-16,30H2,1-3H3,(H,31,37)(H,32,38)(H,33,36)(H,34,39)(H,40,41)/t18-,22+,23+,24-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 2.49 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 3
(Homo sapiens (Human)) | BDBM50558766
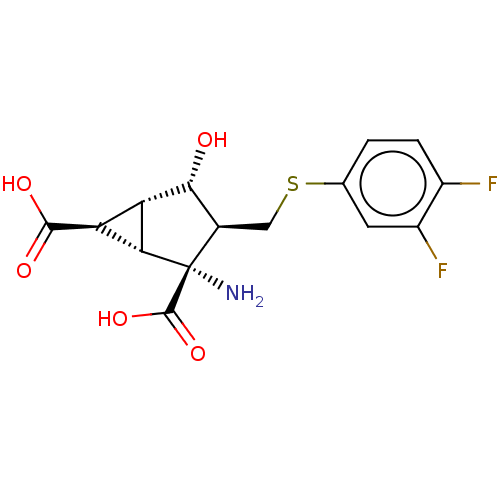
(CHEMBL4751065)Show SMILES Cl.[H][C@]12[C@H](C(O)=O)[C@@]1([H])[C@@](N)([C@H](CSc1ccc(F)c(F)c1)[C@H]2O)C(O)=O |r| | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of [3H]-LY459477 from recombinant human mGlu3 receptor expressed in hamster AV12 cell membranes co-expressing rat EAAT1 incubated for 90... |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2611419 |
More data for this
Ligand-Target Pair | |
Metabotropic glutamate receptor 3
(Homo sapiens (Human)) | BDBM50204261
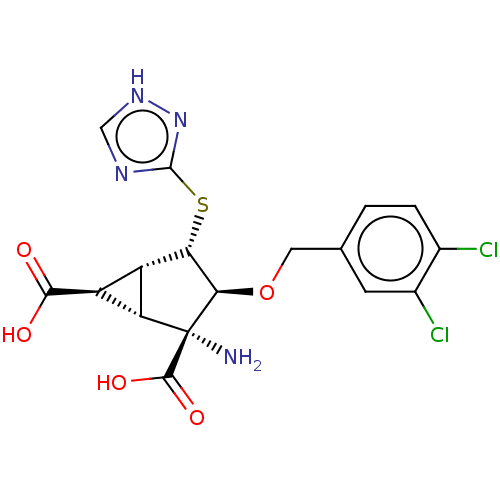
(CHEMBL3969063)Show SMILES [H][C@]12[C@H](C(O)=O)[C@@]1([H])[C@@](N)([C@H](OCc1ccc(Cl)c(Cl)c1)[C@H]2Sc1nc[nH]n1)C(O)=O |r| Show InChI InChI=1S/C17H16Cl2N4O5S/c18-7-2-1-6(3-8(7)19)4-28-13-12(29-16-21-5-22-23-16)9-10(14(24)25)11(9)17(13,20)15(26)27/h1-3,5,9-13H,4,20H2,(H,24,25)(H,26,27)(H,21,22,23)/t9-,10-,11-,12-,13+,17+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Lilly Research Laboratories
Curated by ChEMBL
| Assay Description
Displacement of [3H]-LY459477 from human mGlu3 receptor expressed in hamster AV12 cell membranes co-expressing human EAAT1 after 90 mins by liquid sc... |
Bioorg Med Chem Lett 26: 5663-5668 (2016)
Article DOI: 10.1016/j.bmcl.2016.10.067
BindingDB Entry DOI: 10.7270/Q2JS9SDT |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 1
(Homo sapiens (Human)) | BDBM50067737
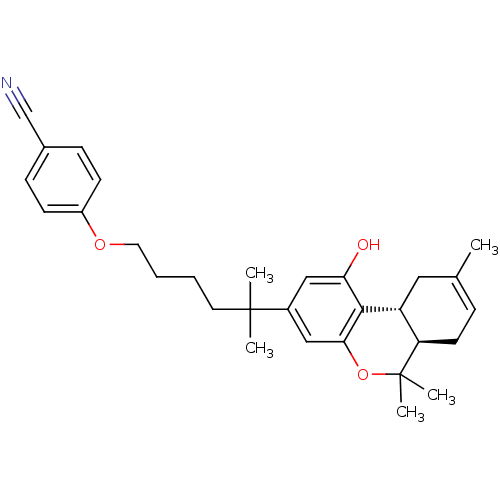
(4-(5-((6aR,10aR)-1-hydroxy-6,6,9-trimethyl-6a,7,10...)Show SMILES CC1=CC[C@@H]2[C@@H](C1)c1c(O)cc(cc1OC2(C)C)C(C)(C)CCCCOc1ccc(cc1)C#N |t:1| Show InChI InChI=1S/C30H37NO3/c1-20-8-13-25-24(16-20)28-26(32)17-22(18-27(28)34-30(25,4)5)29(2,3)14-6-7-15-33-23-11-9-21(19-31)10-12-23/h8-12,17-18,24-25,32H,6-7,13-16H2,1-5H3/t24-,25-/m1/s1 | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Organix, Inc.
Curated by ChEMBL
| Assay Description
Evaluated for its binding affinity towards Cannabinoid receptor 1 (CB1) |
J Med Chem 41: 4400-7 (1998)
Article DOI: 10.1021/jm9803875
BindingDB Entry DOI: 10.7270/Q2M909CZ |
More data for this
Ligand-Target Pair | |
Mu-type opioid receptor
(CALF) | BDBM50095150

((R)-1-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionyl]...)Show SMILES N[C@@H](Cc1ccc(O)cc1)C(=O)N1CCC[C@@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1ccccc1)C(N)=O Show InChI InChI=1S/C34H38N6O5/c35-26(17-22-12-14-24(41)15-13-22)34(45)40-16-6-11-30(40)33(44)39-29(19-23-20-37-27-10-5-4-9-25(23)27)32(43)38-28(31(36)42)18-21-7-2-1-3-8-21/h1-5,7-10,12-15,20,26,28-30,37,41H,6,11,16-19,35H2,(H2,36,42)(H,38,43)(H,39,44)/t26-,28-,29-,30+/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Memorial Sloan-Kettering Cancer Center
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 286: 1007-13 (1998)
BindingDB Entry DOI: 10.7270/Q2QC022N |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data