

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31173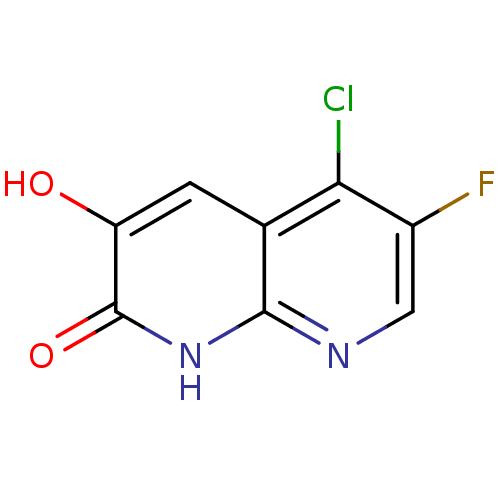 (naphthyridinone analog., 27) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31149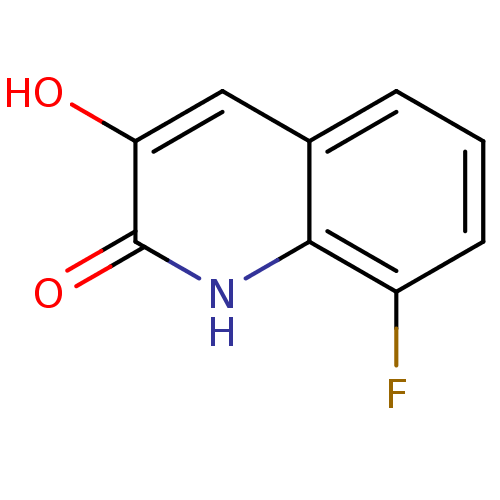 (3-hydroxyquinolin-2(1H)-one, 3) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31148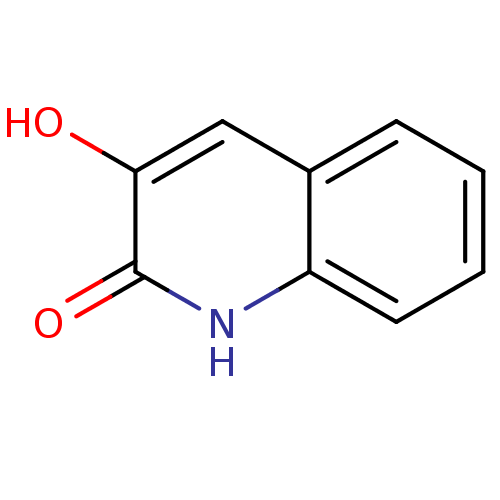 (3-hydroxyquinolin-2(1H)-one, 2 | US9701638, 1) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| D-amino-acid oxidase (Rattus norvegicus (rat)) | BDBM31173 (naphthyridinone analog., 27) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31156 (3-hydroxyquinolin-2(1H)-one, 10) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31172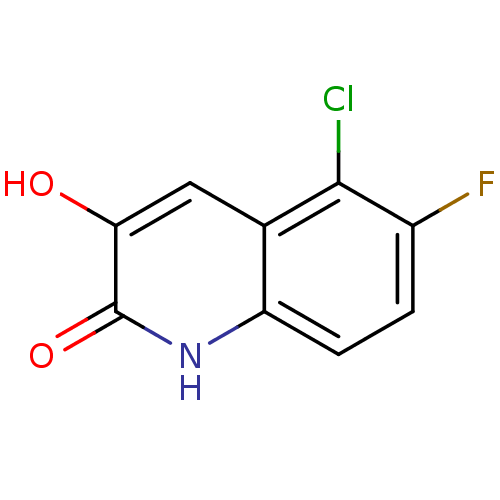 (3-hydroxyquinolin-2(1H)-one, 26) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31152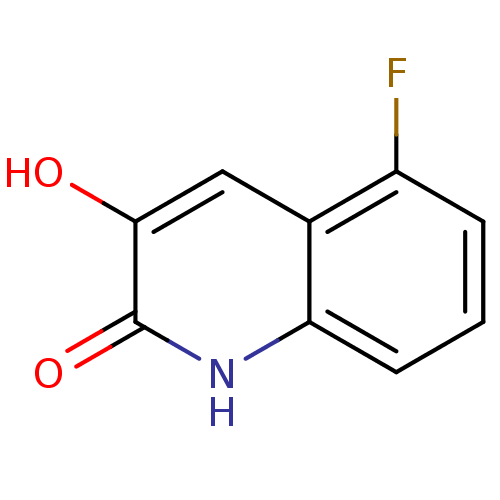 (3-hydroxyquinolin-2(1H)-one, 6) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31164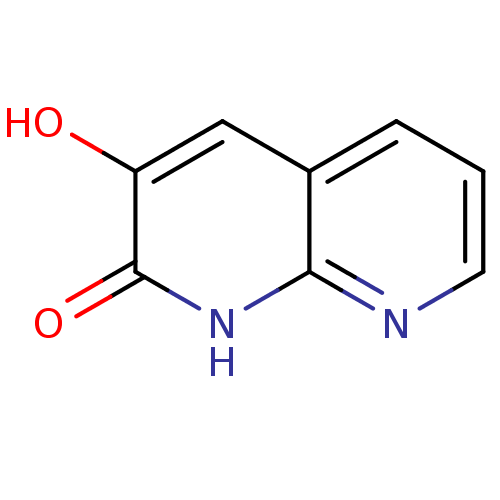 (naphthyridinone analog.,18) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31161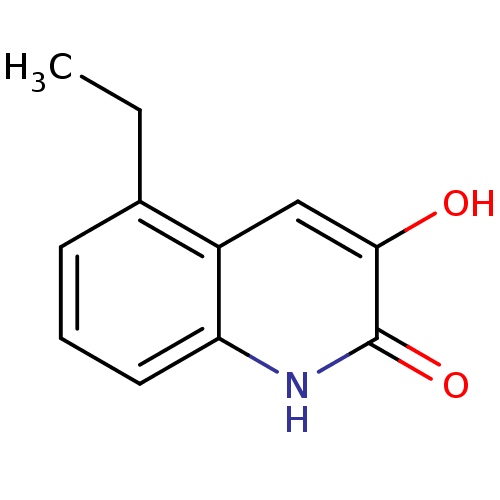 (3-hydroxyquinolin-2(1H)-one, 15) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31151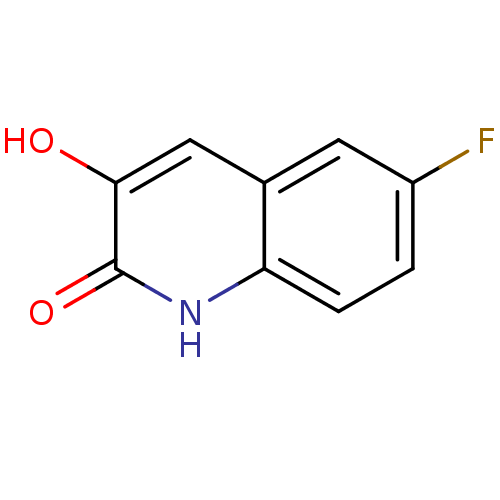 (3-hydroxyquinolin-2(1H)-one, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 9 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31147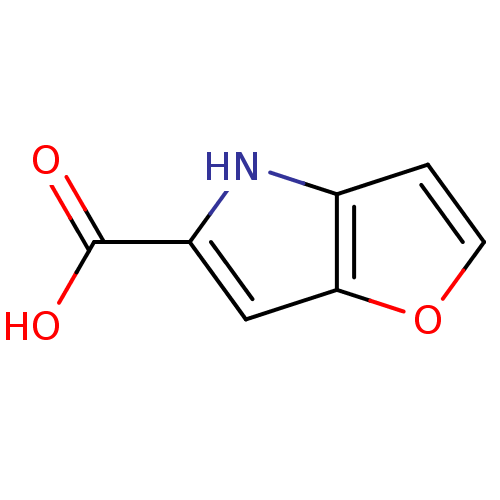 (4H-furo[3,2-b]pyrrole-5-carboxylic acid | 5-carbox...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents | MMDB PDB Article PubMed | n/a | n/a | 9 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31150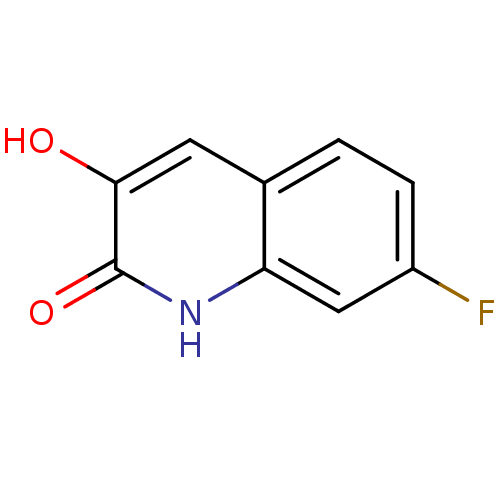 (3-hydroxyquinolin-2(1H)-one, 4) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 10 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| Ketohexokinase (Homo sapiens (Human)) | BDBM50380246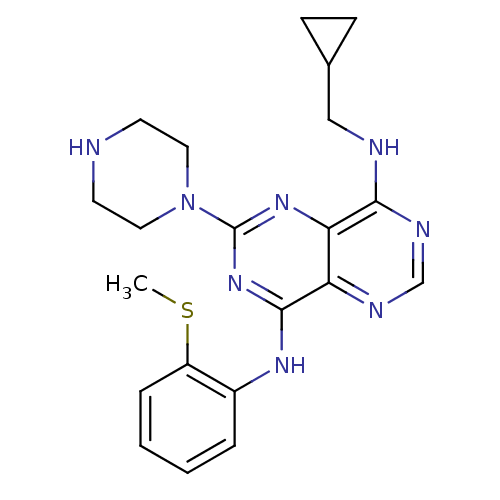 (CHEMBL2017214) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of KHK (unknown origin) using D-fructose as substrate after 60 mins in presence of ATP by LC-MS analysis | J Med Chem 60: 7835-7849 (2017) Article DOI: 10.1021/acs.jmedchem.7b00947 BindingDB Entry DOI: 10.7270/Q2H997CM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31160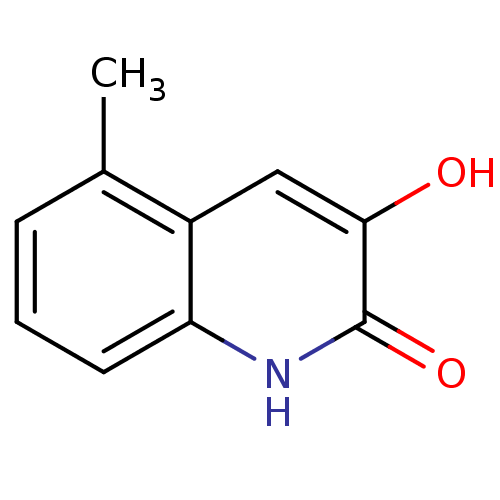 (3-hydroxyquinolin-2(1H)-one, 14) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 16 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31165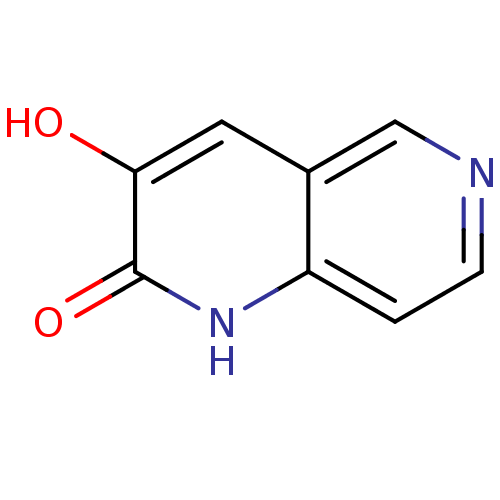 (naphthyridinone analog.,19) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 32 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31153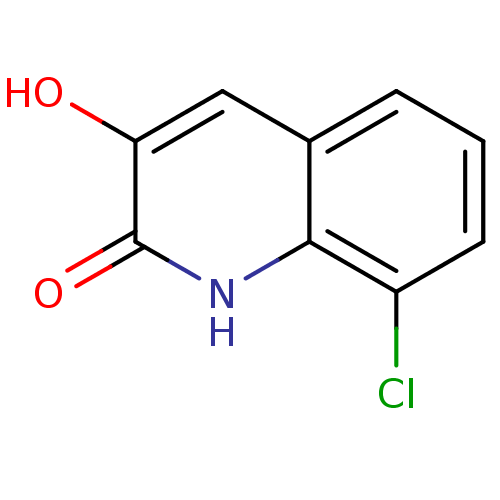 (3-hydroxyquinolin-2(1H)-one, 7) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 33 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31157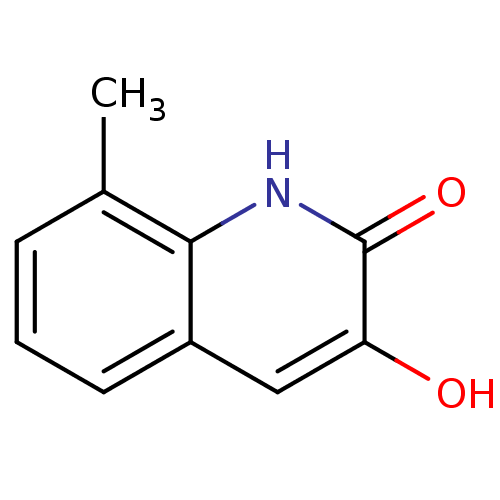 (3-hydroxyquinolin-2(1H)-one, 11) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 38 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| D-amino-acid oxidase (Rattus norvegicus (rat)) | BDBM31156 (3-hydroxyquinolin-2(1H)-one, 10) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D-amino-acid oxidase (Rattus norvegicus (rat)) | BDBM31172 (3-hydroxyquinolin-2(1H)-one, 26) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50398610 (CHEMBL2177470) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of BACE1-mediated sAPPbeta production in human H4 cells expressing APP 695 after 18 hrs by ELISA | J Med Chem 55: 9224-39 (2012) Article DOI: 10.1021/jm3009426 BindingDB Entry DOI: 10.7270/Q22F7PK8 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31154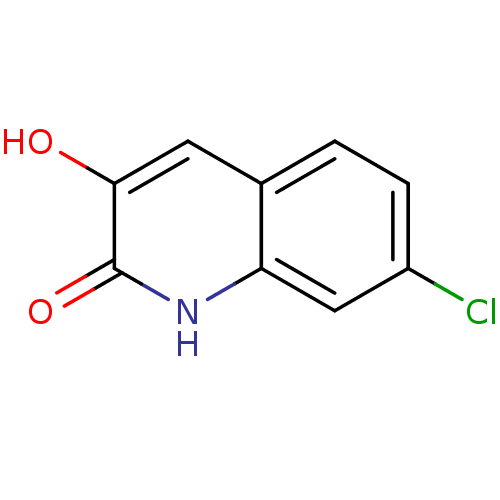 (3-hydroxyquinolin-2(1H)-one, 8) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50398610 (CHEMBL2177470) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of human recombinant BACE1 using K(biotin)RGLTTRPGSGLTNIKTEEISEVNLDAEFRHDSGA as substrate after 1 hr by ELISA | J Med Chem 55: 9224-39 (2012) Article DOI: 10.1021/jm3009426 BindingDB Entry DOI: 10.7270/Q22F7PK8 | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31167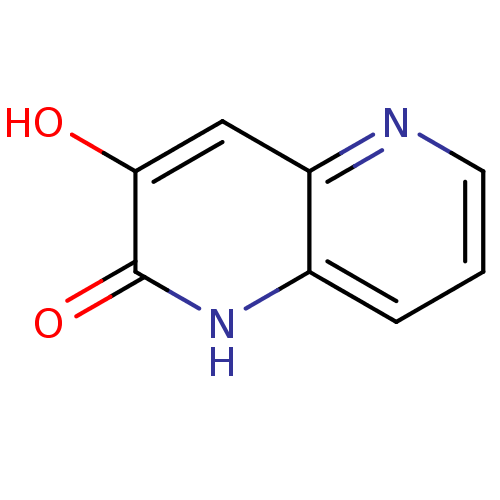 (naphthyridinone analog.,21) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 128 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31155 (3-hydroxyquinolin-2(1H)-one, 9) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 155 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| D-amino-acid oxidase (Rattus norvegicus (rat)) | BDBM31149 (3-hydroxyquinolin-2(1H)-one, 3) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 180 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D-amino-acid oxidase (Rattus norvegicus (rat)) | BDBM31152 (3-hydroxyquinolin-2(1H)-one, 6) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 196 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D-amino-acid oxidase (Homo sapiens (Human)) | BDBM31158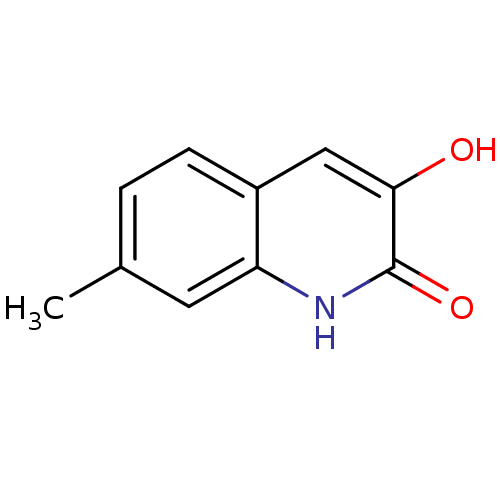 (3-hydroxyquinolin-2(1H)-one, 12) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 197 | n/a | n/a | n/a | n/a | 8.5 | 22 |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (docked) | ||||||||||||
| D-amino-acid oxidase (Rattus norvegicus (rat)) | BDBM31164 (naphthyridinone analog.,18) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase PC cid PC sid UniChem | Article PubMed | n/a | n/a | 200 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D-amino-acid oxidase (Rattus norvegicus (rat)) | BDBM31148 (3-hydroxyquinolin-2(1H)-one, 2 | US9701638, 1) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 215 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50398613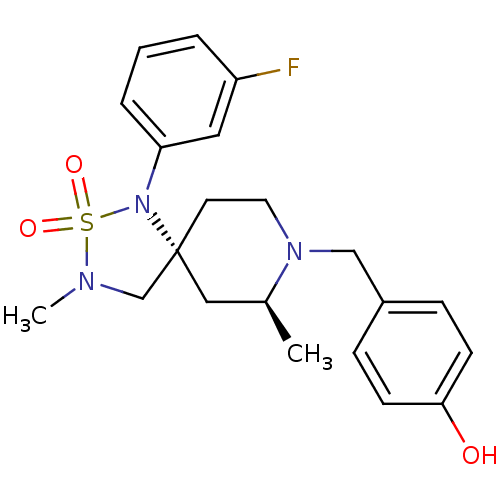 (CHEMBL2177484) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 240 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of BACE1-mediated sAPPbeta production in human H4 cells expressing APP 695 after 18 hrs by ELISA | J Med Chem 55: 9224-39 (2012) Article DOI: 10.1021/jm3009426 BindingDB Entry DOI: 10.7270/Q22F7PK8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50398613 (CHEMBL2177484) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 270 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of human recombinant BACE1 using K(biotin)RGLTTRPGSGLTNIKTEEISEVNLDAEFRHDSGA as substrate after 1 hr by ELISA | J Med Chem 55: 9224-39 (2012) Article DOI: 10.1021/jm3009426 BindingDB Entry DOI: 10.7270/Q22F7PK8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50398614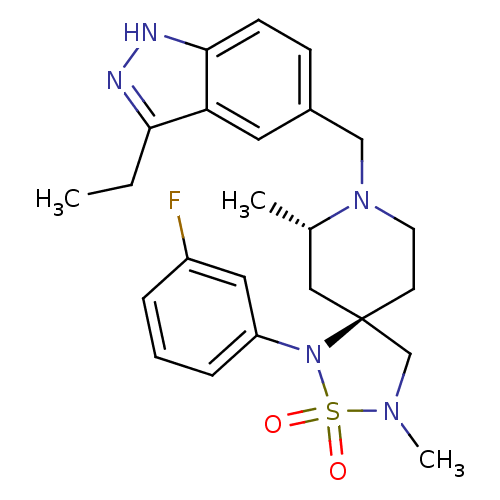 (CHEMBL2177483) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of BACE1-mediated sAPPbeta production in human H4 cells expressing APP 695 after 18 hrs by ELISA | J Med Chem 55: 9224-39 (2012) Article DOI: 10.1021/jm3009426 BindingDB Entry DOI: 10.7270/Q22F7PK8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| D-amino-acid oxidase (Rattus norvegicus (rat)) | BDBM31151 (3-hydroxyquinolin-2(1H)-one, 5) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 308 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ketohexokinase (Rattus norvegicus) | BDBM50241178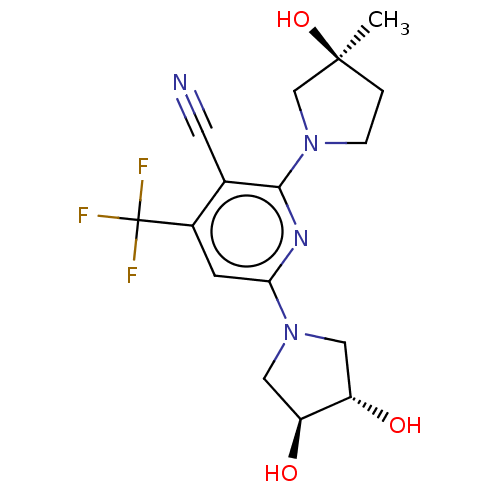 (CHEMBL4070442) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of recombinant rat N-terminal His-tagged KHK expressed in Escherichia coli BL21(DE3) using fructose as substrate incubated for 30 mins fol... | J Med Chem 60: 7835-7849 (2017) Article DOI: 10.1021/acs.jmedchem.7b00947 BindingDB Entry DOI: 10.7270/Q2H997CM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Ketohexokinase (Rattus norvegicus) | BDBM50241178 (CHEMBL4070442) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 340 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of recombinant rat N-terminal His-tagged KHK expressed in Escherichia coli BL21(DE3) using fructose as substrate incubated for 30 mins fol... | J Med Chem 60: 7835-7849 (2017) Article DOI: 10.1021/acs.jmedchem.7b00947 BindingDB Entry DOI: 10.7270/Q2H997CM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50398612 (CHEMBL2177468) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 350 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of BACE1-mediated sAPPbeta production in human H4 cells expressing APP 695 after 18 hrs by ELISA | J Med Chem 55: 9224-39 (2012) Article DOI: 10.1021/jm3009426 BindingDB Entry DOI: 10.7270/Q22F7PK8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ketohexokinase (Homo sapiens (Human)) | BDBM50241178 (CHEMBL4070442) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 389 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal His-tagged KHK-A expressed in Escherichia coli BL21(DE3) using fructose as substrate incubated for 30 mins... | J Med Chem 60: 7835-7849 (2017) Article DOI: 10.1021/acs.jmedchem.7b00947 BindingDB Entry DOI: 10.7270/Q2H997CM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Ketohexokinase (Homo sapiens (Human)) | BDBM50241178 (CHEMBL4070442) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 390 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal His-tagged KHK-A expressed in Escherichia coli BL21(DE3) using fructose as substrate incubated for 30 mins... | J Med Chem 60: 7835-7849 (2017) Article DOI: 10.1021/acs.jmedchem.7b00947 BindingDB Entry DOI: 10.7270/Q2H997CM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| 5'-AMP-activated protein kinase subunit beta-1 (Homo sapiens (Human)) | BDBM237920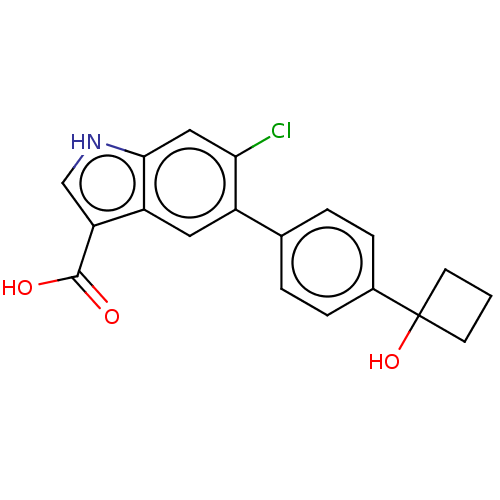 (US9394285, 1) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid PDB UniChem | PDB Article PubMed | n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Activation of recombinant AMPKbeta1 in cryopreserved human hepatocytes assessed as reduction in 14C-2-acetic acid incorporation after 1 hr by scintil... | J Med Chem 61: 7273-7288 (2018) Article DOI: 10.1021/acs.jmedchem.8b00807 BindingDB Entry DOI: 10.7270/Q2Q242WT | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| 5'-AMP-activated protein kinase subunit beta-1 (Homo sapiens (Human)) | BDBM238079 (US9394285, 173) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase MCE PC cid PC sid PDB UniChem | PDB Article PubMed | n/a | n/a | 420 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Activation of recombinant AMPKbeta1 in cryopreserved human hepatocytes assessed as reduction in 14C-2-acetic acid incorporation after 1 hr by scintil... | J Med Chem 61: 7273-7288 (2018) Article DOI: 10.1021/acs.jmedchem.8b00807 BindingDB Entry DOI: 10.7270/Q2Q242WT | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| D-amino-acid oxidase (Rattus norvegicus (rat)) | BDBM31147 (4H-furo[3,2-b]pyrrole-5-carboxylic acid | 5-carbox...) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents | Article PubMed | n/a | n/a | 424 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer | Assay Description Inhibitory effect of compounds was determined in a cell free fluorescence assay. The H2O2 generated from the degradation of D-serine was linked to ox... | J Med Chem 52: 3576-85 (2009) Article DOI: 10.1021/jm900128w BindingDB Entry DOI: 10.7270/Q2CZ35HM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ketohexokinase (Homo sapiens (Human)) | BDBM50241178 (CHEMBL4070442) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 450 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal His-tagged KHK-C expressed in Escherichia coli BL21(DE3) using fructose as substrate incubated for 30 mins... | J Med Chem 60: 7835-7849 (2017) Article DOI: 10.1021/acs.jmedchem.7b00947 BindingDB Entry DOI: 10.7270/Q2H997CM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Ketohexokinase (Homo sapiens (Human)) | BDBM50241178 (CHEMBL4070442) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Similars | PDB Article PubMed | n/a | n/a | 450 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal His-tagged KHK-C expressed in Escherichia coli BL21(DE3) using fructose as substrate incubated for 30 mins... | J Med Chem 60: 7835-7849 (2017) Article DOI: 10.1021/acs.jmedchem.7b00947 BindingDB Entry DOI: 10.7270/Q2H997CM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Ketohexokinase (Homo sapiens (Human)) | BDBM50349755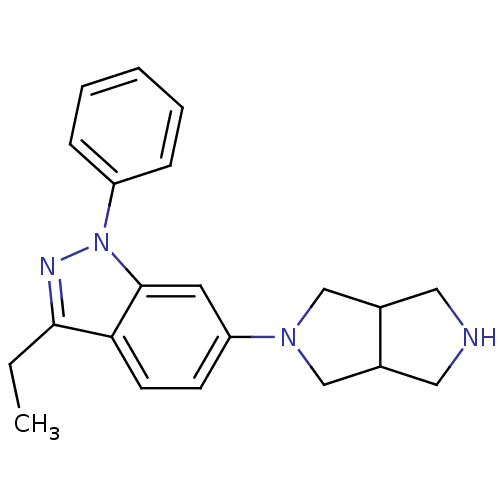 (CHEMBL1809182 | US8822447, 50 | US9771375, Example...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 590 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of KHK (unknown origin) using D-fructose as substrate after 60 mins in presence of ATP by LC-MS analysis | J Med Chem 60: 7835-7849 (2017) Article DOI: 10.1021/acs.jmedchem.7b00947 BindingDB Entry DOI: 10.7270/Q2H997CM | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Ketohexokinase (Rattus norvegicus) | BDBM50241195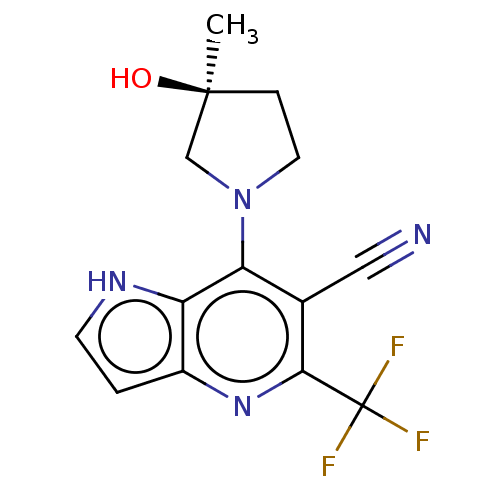 (CHEMBL4063606) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 640 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of recombinant rat N-terminal His-tagged KHK expressed in Escherichia coli BL21(DE3) using fructose as substrate incubated for 30 mins fol... | J Med Chem 60: 7835-7849 (2017) Article DOI: 10.1021/acs.jmedchem.7b00947 BindingDB Entry DOI: 10.7270/Q2H997CM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ketohexokinase (Rattus norvegicus) | BDBM50241195 (CHEMBL4063606) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 646 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of recombinant rat N-terminal His-tagged KHK expressed in Escherichia coli BL21(DE3) using fructose as substrate incubated for 30 mins fol... | J Med Chem 60: 7835-7849 (2017) Article DOI: 10.1021/acs.jmedchem.7b00947 BindingDB Entry DOI: 10.7270/Q2H997CM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ketohexokinase (Homo sapiens (Human)) | BDBM50241195 (CHEMBL4063606) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 670 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal His-tagged KHK expressed in Escherichia coli BL21(DE3) using fructose as substrate incubated for 30 mins f... | J Med Chem 60: 7835-7849 (2017) Article DOI: 10.1021/acs.jmedchem.7b00947 BindingDB Entry DOI: 10.7270/Q2H997CM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Ketohexokinase (Homo sapiens (Human)) | BDBM50241195 (CHEMBL4063606) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 676 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of recombinant human N-terminal His-tagged KHK expressed in Escherichia coli BL21(DE3) using fructose as substrate incubated for 30 mins f... | J Med Chem 60: 7835-7849 (2017) Article DOI: 10.1021/acs.jmedchem.7b00947 BindingDB Entry DOI: 10.7270/Q2H997CM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50398624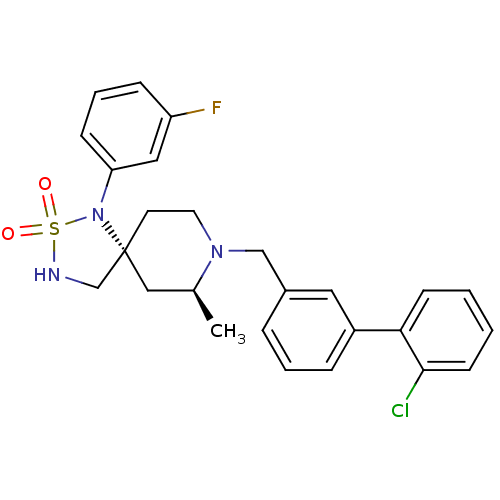 (CHEMBL2177472) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 710 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of BACE1-mediated sAPPbeta production in human H4 cells expressing APP 695 after 18 hrs by ELISA | J Med Chem 55: 9224-39 (2012) Article DOI: 10.1021/jm3009426 BindingDB Entry DOI: 10.7270/Q22F7PK8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50398615 (CHEMBL2177482) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 770 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Inhibition of BACE1-mediated sAPPbeta production in human H4 cells expressing APP 695 after 18 hrs by ELISA | J Med Chem 55: 9224-39 (2012) Article DOI: 10.1021/jm3009426 BindingDB Entry DOI: 10.7270/Q22F7PK8 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 252 total ) | Next | Last >> |