 Found 357 hits with Last Name = 'burton' and Initial = 'eg'
Found 357 hits with Last Name = 'burton' and Initial = 'eg' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029613
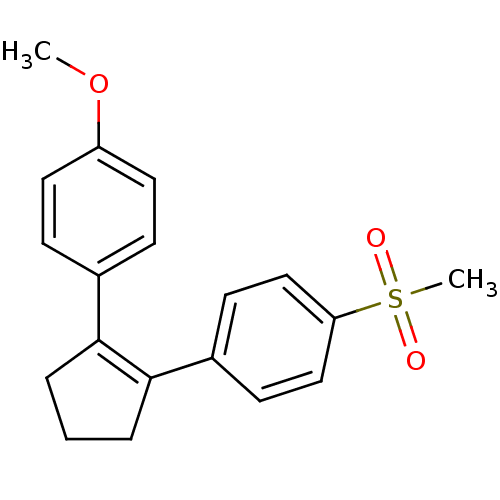
(1-Methoxy-4-(2-(4-(methanesulfonyl)phenyl)cyclopen...)Show SMILES COc1ccc(cc1)C1=C(CCC1)c1ccc(cc1)S(C)(=O)=O |t:9| Show InChI InChI=1S/C19H20O3S/c1-22-16-10-6-14(7-11-16)18-4-3-5-19(18)15-8-12-17(13-9-15)23(2,20)21/h6-13H,3-5H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 38: 4570-8 (1995)
BindingDB Entry DOI: 10.7270/Q23N22D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049042

(4-[6-(4-Trifluoromethyl-phenyl)-spiro[2.4]hept-5-e...)Show SMILES NS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(cc1)C(F)(F)F |t:11| Show InChI InChI=1S/C20H18F3NO2S/c21-20(22,23)15-5-1-13(2-6-15)17-11-19(9-10-19)12-18(17)14-3-7-16(8-4-14)27(24,25)26/h1-8H,9-12H2,(H2,24,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049014

(4-[6-(4-Chloro-phenyl)-spiro[2.4]hept-5-en-5-yl]-b...)Show SMILES NS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(Cl)cc1 |t:11| Show InChI InChI=1S/C19H18ClNO2S/c20-15-5-1-13(2-6-15)17-11-19(9-10-19)12-18(17)14-3-7-16(8-4-14)24(21,22)23/h1-8H,9-12H2,(H2,21,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049011
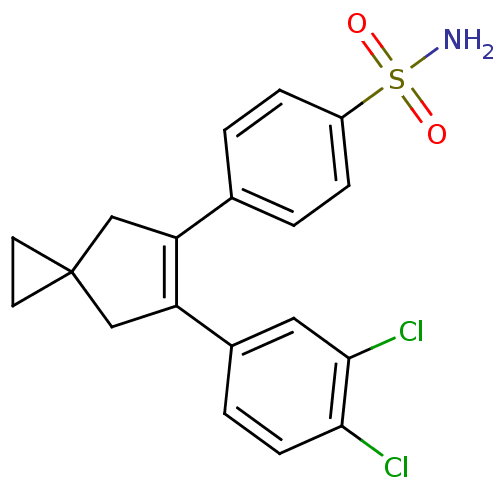
(4-[6-(3,4-Dichloro-phenyl)-spiro[2.4]hept-5-en-5-y...)Show SMILES NS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(Cl)c(Cl)c1 |t:11| Show InChI InChI=1S/C19H17Cl2NO2S/c20-17-6-3-13(9-18(17)21)16-11-19(7-8-19)10-15(16)12-1-4-14(5-2-12)25(22,23)24/h1-6,9H,7-8,10-11H2,(H2,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049038
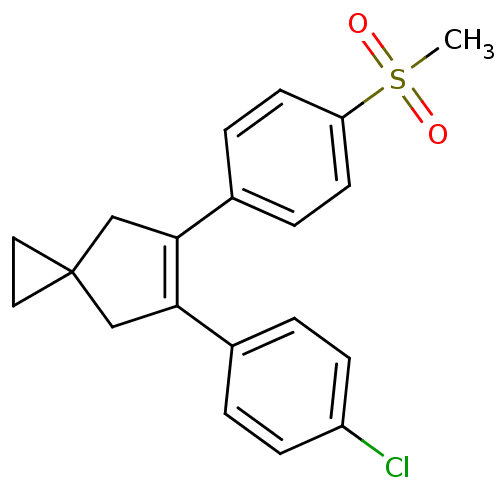
(5-(4-Chloro-phenyl)-6-(4-methanesulfonyl-phenyl)-s...)Show SMILES CS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(Cl)cc1 |t:11| Show InChI InChI=1S/C20H19ClO2S/c1-24(22,23)17-8-4-15(5-9-17)19-13-20(10-11-20)12-18(19)14-2-6-16(21)7-3-14/h2-9H,10-13H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049025
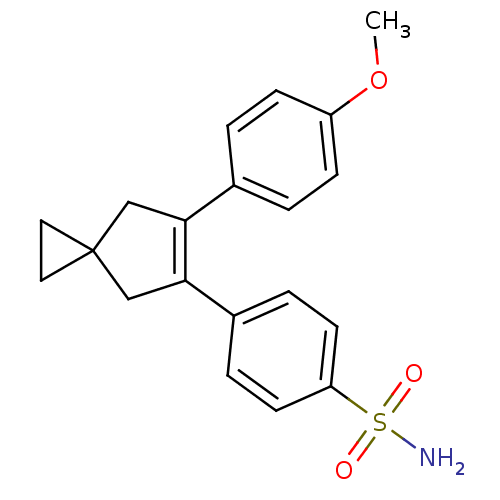
(4-[6-(4-Methoxy-phenyl)-spiro[2.4]hept-5-en-5-yl]-...)Show SMILES COc1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(cc1)S(N)(=O)=O |t:9| Show InChI InChI=1S/C20H21NO3S/c1-24-16-6-2-14(3-7-16)18-12-20(10-11-20)13-19(18)15-4-8-17(9-5-15)25(21,22)23/h2-9H,10-13H2,1H3,(H2,21,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049030
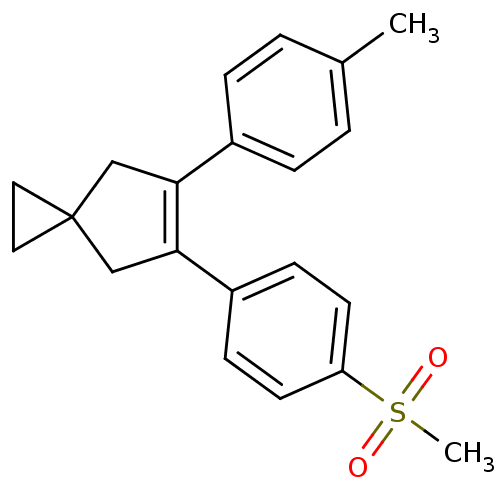
(5-(4-Methanesulfonyl-phenyl)-6-p-tolyl-spiro[2.4]h...)Show SMILES Cc1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(cc1)S(C)(=O)=O |t:8| Show InChI InChI=1S/C21H22O2S/c1-15-3-5-16(6-4-15)19-13-21(11-12-21)14-20(19)17-7-9-18(10-8-17)24(2,22)23/h3-10H,11-14H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049033
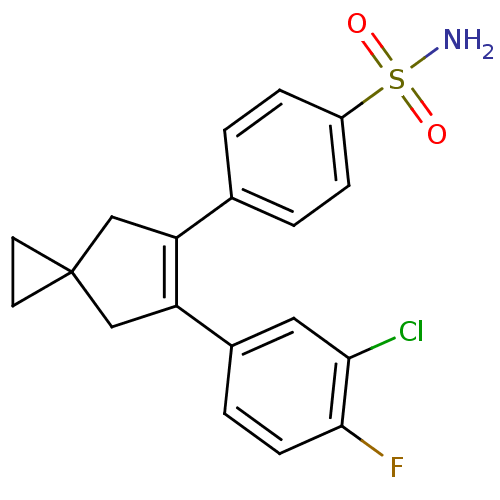
(4-[6-(3-Chloro-4-fluoro-phenyl)-spiro[2.4]hept-5-e...)Show SMILES NS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(F)c(Cl)c1 |t:11| Show InChI InChI=1S/C19H17ClFNO2S/c20-17-9-13(3-6-18(17)21)16-11-19(7-8-19)10-15(16)12-1-4-14(5-2-12)25(22,23)24/h1-6,9H,7-8,10-11H2,(H2,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057581
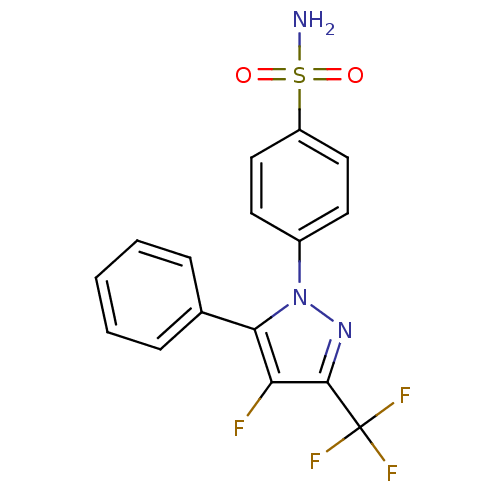
(4-(4-Fluoro-5-phenyl-3-trifluoromethyl-pyrazol-1-y...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(c(F)c1-c1ccccc1)C(F)(F)F Show InChI InChI=1S/C16H11F4N3O2S/c17-13-14(10-4-2-1-3-5-10)23(22-15(13)16(18,19)20)11-6-8-12(9-7-11)26(21,24)25/h1-9H,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029596
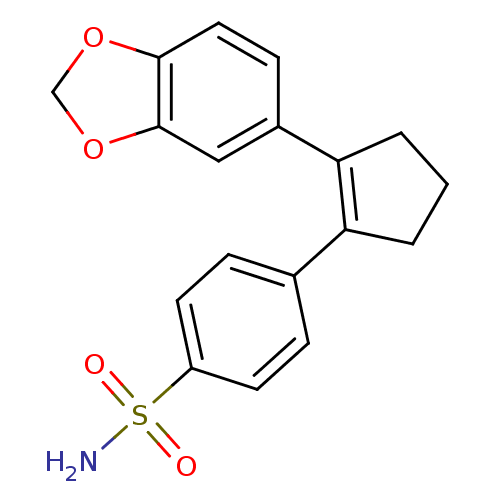
(4-(2-Benzo[1,3]dioxol-5-yl-cyclopent-1-enyl)-benze...)Show SMILES NS(=O)(=O)c1ccc(cc1)C1=C(CCC1)c1ccc2OCOc2c1 |t:11| Show InChI InChI=1S/C18H17NO4S/c19-24(20,21)14-7-4-12(5-8-14)15-2-1-3-16(15)13-6-9-17-18(10-13)23-11-22-17/h4-10H,1-3,11H2,(H2,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 38: 4570-8 (1995)
BindingDB Entry DOI: 10.7270/Q23N22D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029618

(4-(2-(4-methoxyphenyl)cyclopent-1-enyl)benzenesulf...)Show SMILES COc1ccc(cc1)C1=C(CCC1)c1ccc(cc1)S(N)(=O)=O |t:9| Show InChI InChI=1S/C18H19NO3S/c1-22-15-9-5-13(6-10-15)17-3-2-4-18(17)14-7-11-16(12-8-14)23(19,20)21/h5-12H,2-4H2,1H3,(H2,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 38: 4570-8 (1995)
BindingDB Entry DOI: 10.7270/Q23N22D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029595
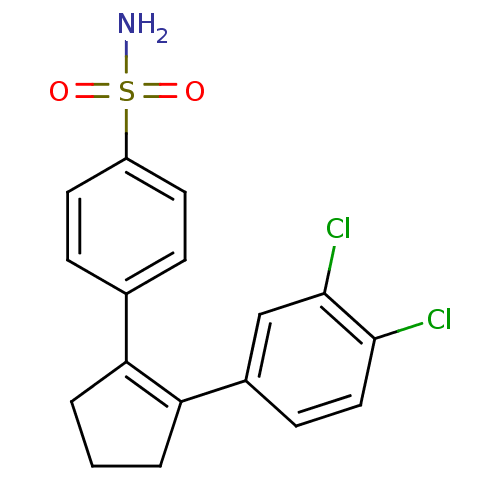
(4-[2-(3,4-Dichloro-phenyl)-cyclopent-1-enyl]-benze...)Show SMILES NS(=O)(=O)c1ccc(cc1)C1=C(CCC1)c1ccc(Cl)c(Cl)c1 |t:11| Show InChI InChI=1S/C17H15Cl2NO2S/c18-16-9-6-12(10-17(16)19)15-3-1-2-14(15)11-4-7-13(8-5-11)23(20,21)22/h4-10H,1-3H2,(H2,20,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 38: 4570-8 (1995)
BindingDB Entry DOI: 10.7270/Q23N22D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029624
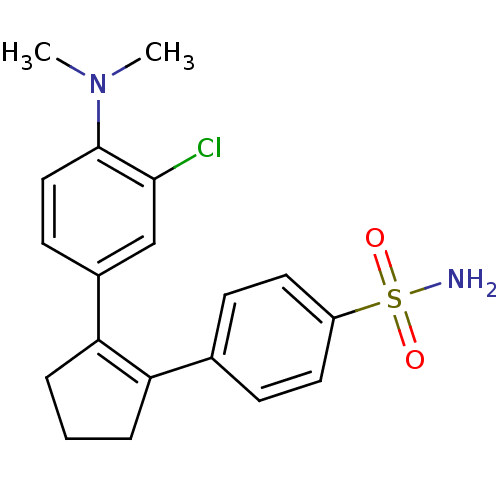
(4-[2-(3-Chloro-4-dimethylamino-phenyl)-cyclopent-1...)Show SMILES CN(C)c1ccc(cc1Cl)C1=C(CCC1)c1ccc(cc1)S(N)(=O)=O |t:11| Show InChI InChI=1S/C19H21ClN2O2S/c1-22(2)19-11-8-14(12-18(19)20)17-5-3-4-16(17)13-6-9-15(10-7-13)25(21,23)24/h6-12H,3-5H2,1-2H3,(H2,21,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 38: 4570-8 (1995)
BindingDB Entry DOI: 10.7270/Q23N22D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049037
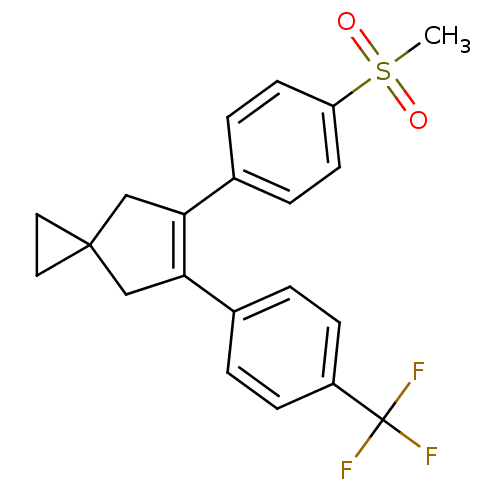
(5-(4-Methanesulfonyl-phenyl)-6-(4-trifluoromethyl-...)Show SMILES CS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(cc1)C(F)(F)F |t:11| Show InChI InChI=1S/C21H19F3O2S/c1-27(25,26)17-8-4-15(5-9-17)19-13-20(10-11-20)12-18(19)14-2-6-16(7-3-14)21(22,23)24/h2-9H,10-13H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049040
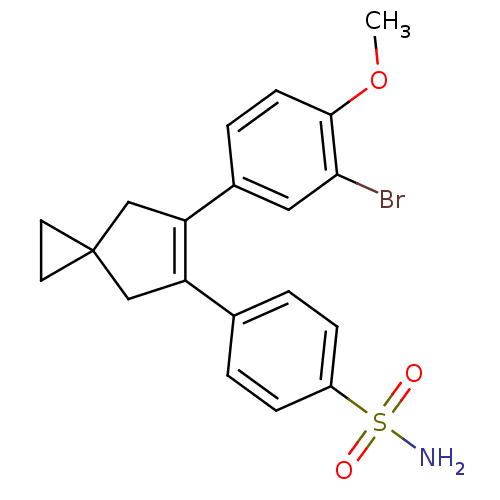
(4-[6-(3-Bromo-4-methoxy-phenyl)-spiro[2.4]hept-5-e...)Show SMILES COc1ccc(cc1Br)C1=C(CC2(CC2)C1)c1ccc(cc1)S(N)(=O)=O |t:10| Show InChI InChI=1S/C20H20BrNO3S/c1-25-19-7-4-14(10-18(19)21)17-12-20(8-9-20)11-16(17)13-2-5-15(6-3-13)26(22,23)24/h2-7,10H,8-9,11-12H2,1H3,(H2,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049029

(4-[6-(3-Chloro-4-methoxy-phenyl)-spiro[2.4]hept-5-...)Show SMILES COc1ccc(cc1Cl)C1=C(CC2(CC2)C1)c1ccc(cc1)S(N)(=O)=O |t:10| Show InChI InChI=1S/C20H20ClNO3S/c1-25-19-7-4-14(10-18(19)21)17-12-20(8-9-20)11-16(17)13-2-5-15(6-3-13)26(22,23)24/h2-7,10H,8-9,11-12H2,1H3,(H2,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049023
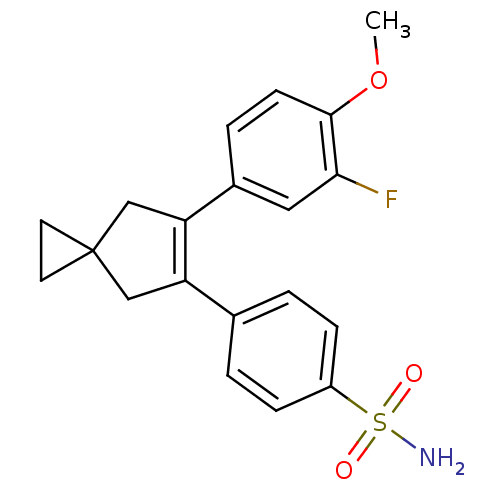
(4-[6-(3-Fluoro-4-methoxy-phenyl)-spiro[2.4]hept-5-...)Show SMILES COc1ccc(cc1F)C1=C(CC2(CC2)C1)c1ccc(cc1)S(N)(=O)=O |t:10| Show InChI InChI=1S/C20H20FNO3S/c1-25-19-7-4-14(10-18(19)21)17-12-20(8-9-20)11-16(17)13-2-5-15(6-3-13)26(22,23)24/h2-7,10H,8-9,11-12H2,1H3,(H2,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049028
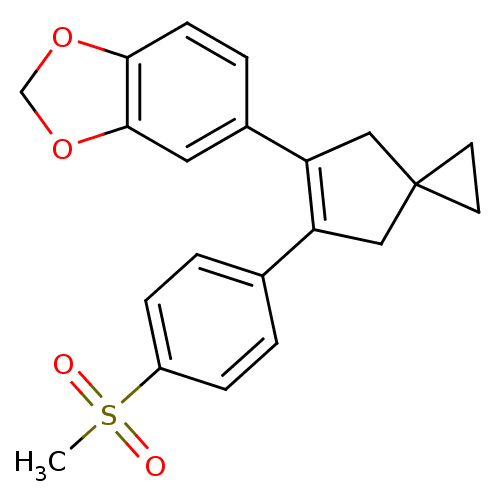
(5-[6-(4-Methanesulfonyl-phenyl)-spiro[2.4]hept-5-e...)Show SMILES CS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc2OCOc2c1 |t:11| Show InChI InChI=1S/C21H20O4S/c1-26(22,23)16-5-2-14(3-6-16)17-11-21(8-9-21)12-18(17)15-4-7-19-20(10-15)25-13-24-19/h2-7,10H,8-9,11-13H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029614
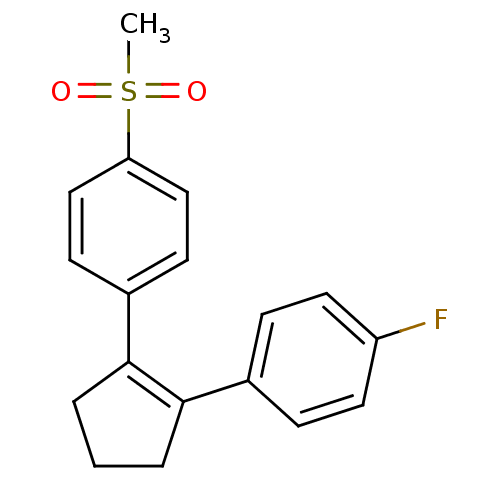
((SC-57666)1-[2-(4-fluorophenyl)-1-cyclopentenyl]-4...)Show SMILES CS(=O)(=O)c1ccc(cc1)C1=C(CCC1)c1ccc(F)cc1 |t:11| Show InChI InChI=1S/C18H17FO2S/c1-22(20,21)16-11-7-14(8-12-16)18-4-2-3-17(18)13-5-9-15(19)10-6-13/h5-12H,2-4H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049026

(5-(4-Fluoro-phenyl)-6-(4-methanesulfonyl-phenyl)-s...)Show SMILES CS(=O)(=O)c1ccc(cc1)C1=CC2(CC2)C=C1c1ccc(F)cc1 |c:17,t:11| Show InChI InChI=1S/C20H17FO2S/c1-24(22,23)17-8-4-15(5-9-17)19-13-20(10-11-20)12-18(19)14-2-6-16(21)7-3-14/h2-9,12-13H,10-11H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| n/a | n/a | 2.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029625

(1-methyl-4-[2-(4-methylsulfonylphenyl)-1-cyclopent...)Show SMILES Cc1ccc(cc1)C1=C(CCC1)c1ccc(cc1)S(C)(=O)=O |t:8| Show InChI InChI=1S/C19H20O2S/c1-14-6-8-15(9-7-14)18-4-3-5-19(18)16-10-12-17(13-11-16)22(2,20)21/h6-13H,3-5H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 38: 4570-8 (1995)
BindingDB Entry DOI: 10.7270/Q23N22D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049018

(5-(3,4-Dichloro-phenyl)-6-(4-methanesulfonyl-pheny...)Show SMILES CS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(Cl)c(Cl)c1 |t:11| Show InChI InChI=1S/C20H18Cl2O2S/c1-25(23,24)15-5-2-13(3-6-15)16-11-20(8-9-20)12-17(16)14-4-7-18(21)19(22)10-14/h2-7,10H,8-9,11-12H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029615

(4-[2-(4-Chloro-phenyl)-cyclopent-1-enyl]-benzenesu...)Show SMILES NS(=O)(=O)c1ccc(cc1)C1=C(CCC1)c1ccc(Cl)cc1 |t:11| Show InChI InChI=1S/C17H16ClNO2S/c18-14-8-4-12(5-9-14)16-2-1-3-17(16)13-6-10-15(11-7-13)22(19,20)21/h4-11H,1-3H2,(H2,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 38: 4570-8 (1995)
BindingDB Entry DOI: 10.7270/Q23N22D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029623
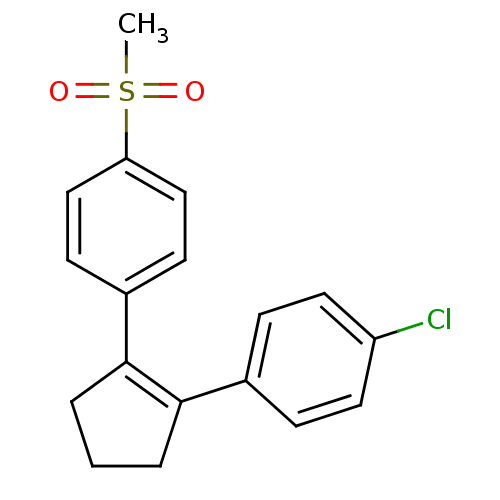
(1-chloro-4-[2-(4-methylsulfonylphenyl)-1-cyclopent...)Show SMILES CS(=O)(=O)c1ccc(cc1)C1=C(CCC1)c1ccc(Cl)cc1 |t:11| Show InChI InChI=1S/C18H17ClO2S/c1-22(20,21)16-11-7-14(8-12-16)18-4-2-3-17(18)13-5-9-15(19)10-6-13/h5-12H,2-4H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 38: 4570-8 (1995)
BindingDB Entry DOI: 10.7270/Q23N22D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049024

(4-[6-(4-Fluoro-phenyl)-spiro[2.4]hept-5-en-5-yl]-b...)Show SMILES NS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(F)cc1 |t:11| Show InChI InChI=1S/C19H18FNO2S/c20-15-5-1-13(2-6-15)17-11-19(9-10-19)12-18(17)14-3-7-16(8-4-14)24(21,22)23/h1-8H,9-12H2,(H2,21,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049010

(4-[6-(3,4-Difluoro-phenyl)-spiro[2.4]hept-5-en-5-y...)Show SMILES NS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(F)c(F)c1 |t:11| Show InChI InChI=1S/C19H17F2NO2S/c20-17-6-3-13(9-18(17)21)16-11-19(7-8-19)10-15(16)12-1-4-14(5-2-12)25(22,23)24/h1-6,9H,7-8,10-11H2,(H2,22,23,24) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049013
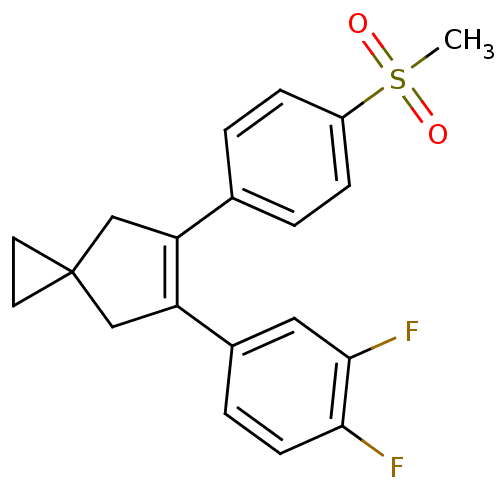
(5-(3,4-Difluoro-phenyl)-6-(4-methanesulfonyl-pheny...)Show SMILES CS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(F)c(F)c1 |t:11| Show InChI InChI=1S/C20H18F2O2S/c1-25(23,24)15-5-2-13(3-6-15)16-11-20(8-9-20)12-17(16)14-4-7-18(21)19(22)10-14/h2-7,10H,8-9,11-12H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057618
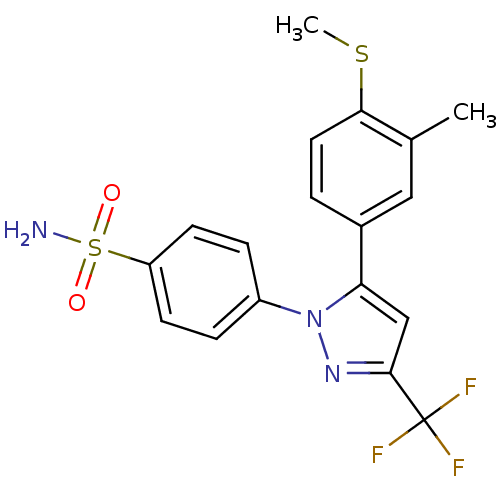
(4-[5-(3-Methyl-4-methylsulfanyl-phenyl)-3-trifluor...)Show SMILES CSc1ccc(cc1C)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C18H16F3N3O2S2/c1-11-9-12(3-8-16(11)27-2)15-10-17(18(19,20)21)23-24(15)13-4-6-14(7-5-13)28(22,25)26/h3-10H,1-2H3,(H2,22,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049027
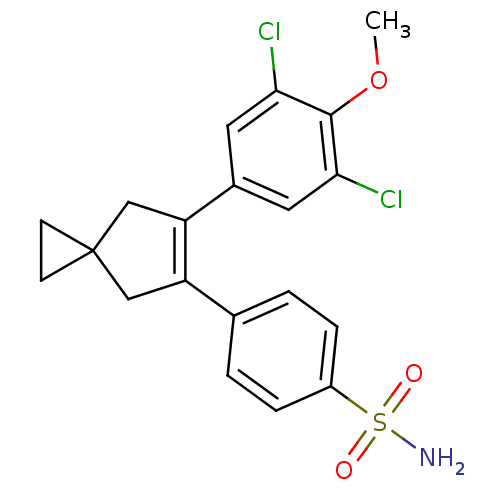
(4-[6-(3,5-Dichloro-4-methoxy-phenyl)-spiro[2.4]hep...)Show SMILES COc1c(Cl)cc(cc1Cl)C1=C(CC2(CC2)C1)c1ccc(cc1)S(N)(=O)=O |t:11| Show InChI InChI=1S/C20H19Cl2NO3S/c1-26-19-17(21)8-13(9-18(19)22)16-11-20(6-7-20)10-15(16)12-2-4-14(5-3-12)27(23,24)25/h2-5,8-9H,6-7,10-11H2,1H3,(H2,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049022

(6-(4-Fluoro-phenyl)-7-(4-methanesulfonyl-phenyl)-s...)Show SMILES CS(=O)(=O)c1ccc(cc1)C1=C(CC2(CCC2)C1)c1ccc(F)cc1 |t:11| Show InChI InChI=1S/C21H21FO2S/c1-25(23,24)18-9-5-16(6-10-18)20-14-21(11-2-12-21)13-19(20)15-3-7-17(22)8-4-15/h3-10H,2,11-14H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057551
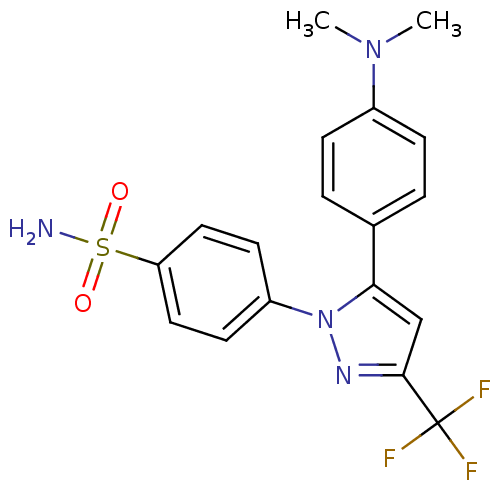
(4-[5-(4-Dimethylamino-phenyl)-3-trifluoromethyl-py...)Show SMILES CN(C)c1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C18H17F3N4O2S/c1-24(2)13-5-3-12(4-6-13)16-11-17(18(19,20)21)23-25(16)14-7-9-15(10-8-14)28(22,26)27/h3-11H,1-2H3,(H2,22,26,27) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 4.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029601
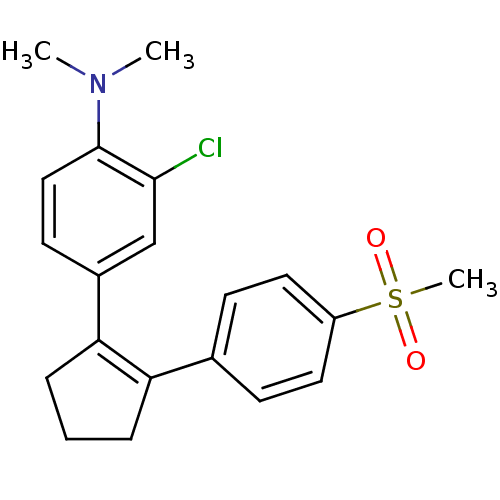
(CHEMBL109623 | N-(2-chloro-4-{2-[4-(methylsulfonyl...)Show SMILES CN(C)c1ccc(cc1Cl)C1=C(CCC1)c1ccc(cc1)S(C)(=O)=O |t:11| Show InChI InChI=1S/C20H22ClNO2S/c1-22(2)20-12-9-15(13-19(20)21)18-6-4-5-17(18)14-7-10-16(11-8-14)25(3,23)24/h7-13H,4-6H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 38: 4570-8 (1995)
BindingDB Entry DOI: 10.7270/Q23N22D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50057554
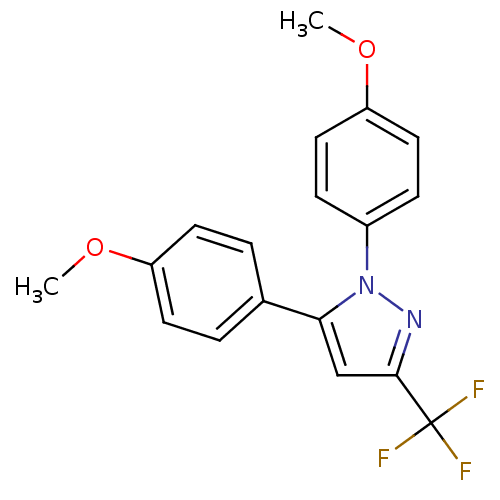
(1,5-Bis-(4-methoxy-phenyl)-3-trifluoromethyl-1H-py...)Show SMILES COc1ccc(cc1)-c1cc(nn1-c1ccc(OC)cc1)C(F)(F)F Show InChI InChI=1S/C18H15F3N2O2/c1-24-14-7-3-12(4-8-14)16-11-17(18(19,20)21)22-23(16)13-5-9-15(25-2)10-6-13/h3-11H,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block recombinant human prostaglandin G/H synthase 1 (COX-1) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049034
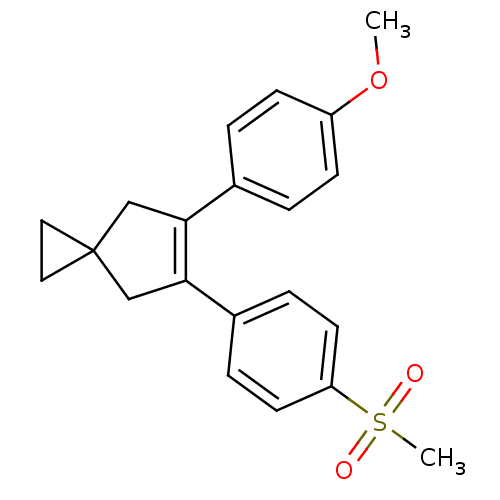
(5-(4-Methanesulfonyl-phenyl)-6-(4-methoxy-phenyl)-...)Show SMILES COc1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(cc1)S(C)(=O)=O |t:9| Show InChI InChI=1S/C21H22O3S/c1-24-17-7-3-15(4-8-17)19-13-21(11-12-21)14-20(19)16-5-9-18(10-6-16)25(2,22)23/h3-10H,11-14H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM50049025

(4-[6-(4-Methoxy-phenyl)-spiro[2.4]hept-5-en-5-yl]-...)Show SMILES COc1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(cc1)S(N)(=O)=O |t:9| Show InChI InChI=1S/C20H21NO3S/c1-24-16-6-2-14(3-7-16)18-12-20(10-11-20)13-19(18)15-4-8-17(9-5-15)25(21,22)23/h2-9H,10-13H2,1H3,(H2,21,22,23) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against prostaglandin G/H synthase 1 (COX-1). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057606
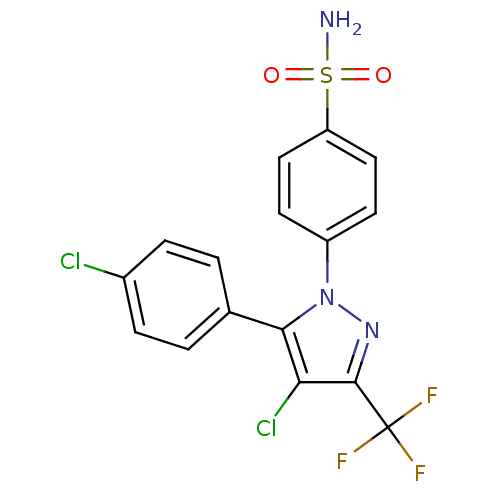
(4-[4-Chloro-5-(4-chloro-phenyl)-3-trifluoromethyl-...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(c(Cl)c1-c1ccc(Cl)cc1)C(F)(F)F Show InChI InChI=1S/C16H10Cl2F3N3O2S/c17-10-3-1-9(2-4-10)14-13(18)15(16(19,20)21)23-24(14)11-5-7-12(8-6-11)27(22,25)26/h1-8H,(H2,22,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057575
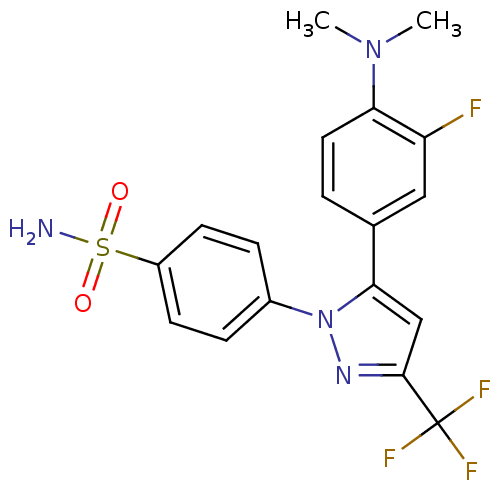
(4-[5-(4-Dimethylamino-3-fluoro-phenyl)-3-trifluoro...)Show SMILES CN(C)c1ccc(cc1F)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C18H16F4N4O2S/c1-25(2)15-8-3-11(9-14(15)19)16-10-17(18(20,21)22)24-26(16)12-4-6-13(7-5-12)29(23,27)28/h3-10H,1-2H3,(H2,23,27,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049031
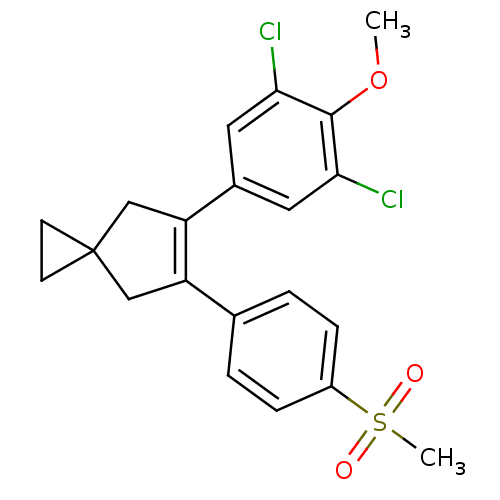
(5-(3,5-Dichloro-4-methoxy-phenyl)-6-(4-methanesulf...)Show SMILES COc1c(Cl)cc(cc1Cl)C1=C(CC2(CC2)C1)c1ccc(cc1)S(C)(=O)=O |t:11| Show InChI InChI=1S/C21H20Cl2O3S/c1-26-20-18(22)9-14(10-19(20)23)17-12-21(7-8-21)11-16(17)13-3-5-15(6-4-13)27(2,24)25/h3-6,9-10H,7-8,11-12H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057609
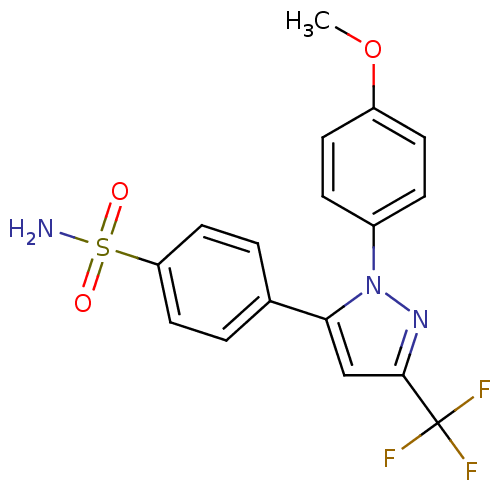
(4-[2-(4-Methoxy-phenyl)-5-trifluoromethyl-2H-pyraz...)Show SMILES COc1ccc(cc1)-n1nc(cc1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O3S/c1-26-13-6-4-12(5-7-13)23-15(10-16(22-23)17(18,19)20)11-2-8-14(9-3-11)27(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029626

(4-[2-(4-Fluoro-phenyl)-cyclopent-1-enyl]-benzenesu...)Show SMILES NS(=O)(=O)c1ccc(cc1)C1=C(CCC1)c1ccc(F)cc1 |t:11| Show InChI InChI=1S/C17H16FNO2S/c18-14-8-4-12(5-9-14)16-2-1-3-17(16)13-6-10-15(11-7-13)22(19,20)21/h4-11H,1-3H2,(H2,19,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 38: 4570-8 (1995)
BindingDB Entry DOI: 10.7270/Q23N22D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049021

(5-(3-Chloro-4-fluoro-phenyl)-6-(4-methanesulfonyl-...)Show SMILES CS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(F)c(Cl)c1 |t:11| Show InChI InChI=1S/C20H18ClFO2S/c1-25(23,24)15-5-2-13(3-6-15)16-11-20(8-9-20)12-17(16)14-4-7-19(22)18(21)10-14/h2-7,10H,8-9,11-12H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 1
(Homo sapiens (Human)) | BDBM13065

(5-(4-chlorophenyl)-1-(4-methoxyphenyl)-3-(trifluor...)Show SMILES COc1ccc(cc1)-n1nc(cc1-c1ccc(Cl)cc1)C(F)(F)F Show InChI InChI=1S/C17H12ClF3N2O/c1-24-14-8-6-13(7-9-14)23-15(10-16(22-23)17(19,20)21)11-2-4-12(18)5-3-11/h2-10H,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block recombinant human prostaglandin G/H synthase 1 (COX-1) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50049041
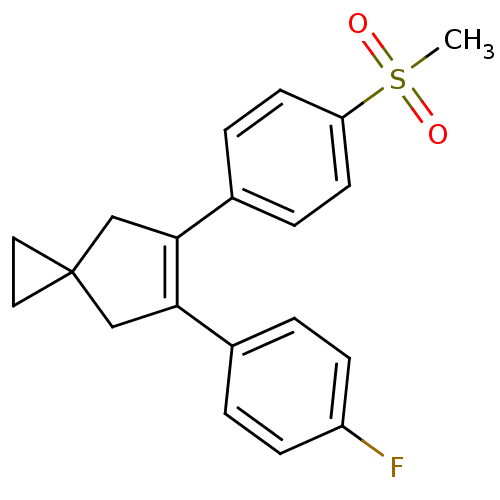
(5-(4-Fluoro-phenyl)-6-(4-methanesulfonyl-phenyl)-s...)Show SMILES CS(=O)(=O)c1ccc(cc1)C1=C(CC2(CC2)C1)c1ccc(F)cc1 |t:11| Show InChI InChI=1S/C20H19FO2S/c1-24(22,23)17-8-4-15(5-9-17)19-13-20(10-11-20)12-18(19)14-2-6-16(21)7-3-14/h2-9H,10-13H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 39: 253-66 (1996)
Article DOI: 10.1021/jm950664x
BindingDB Entry DOI: 10.7270/Q2XG9Q6M |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057589
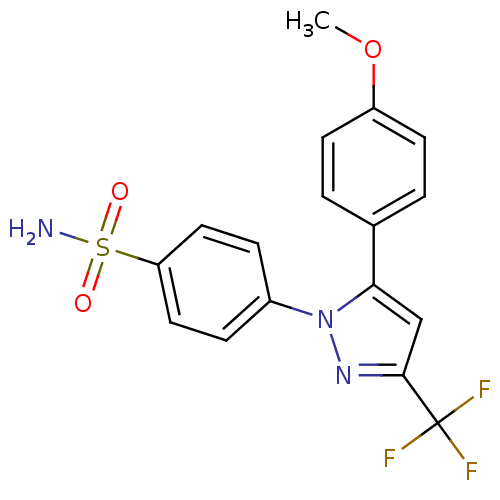
(4-[5-(4-Methoxy-phenyl)-3-trifluoromethyl-pyrazol-...)Show SMILES COc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O3S/c1-26-13-6-2-11(3-7-13)15-10-16(17(18,19)20)22-23(15)12-4-8-14(9-5-12)27(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057591
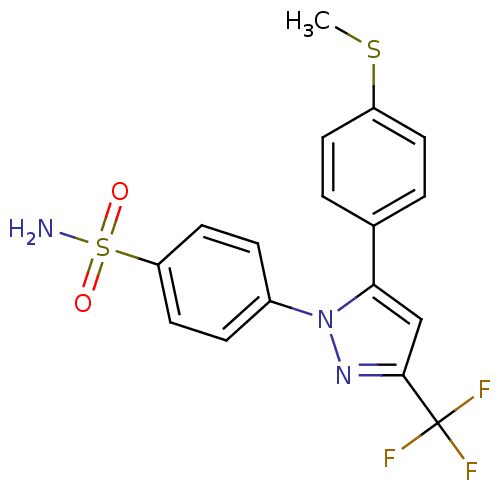
(4-[5-(4-Methylsulfanyl-phenyl)-3-trifluoromethyl-p...)Show SMILES CSc1ccc(cc1)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C17H14F3N3O2S2/c1-26-13-6-2-11(3-7-13)15-10-16(17(18,19)20)22-23(15)12-4-8-14(9-5-12)27(21,24)25/h2-10H,1H3,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029622

(4-[2-(3-Chloro-4-methoxy-phenyl)-cyclopent-1-enyl]...)Show SMILES COc1ccc(cc1Cl)C1=C(CCC1)c1ccc(cc1)S(N)(=O)=O |t:10| Show InChI InChI=1S/C18H18ClNO3S/c1-23-18-10-7-13(11-17(18)19)16-4-2-3-15(16)12-5-8-14(9-6-12)24(20,21)22/h5-11H,2-4H2,1H3,(H2,20,21,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 38: 4570-8 (1995)
BindingDB Entry DOI: 10.7270/Q23N22D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50029607
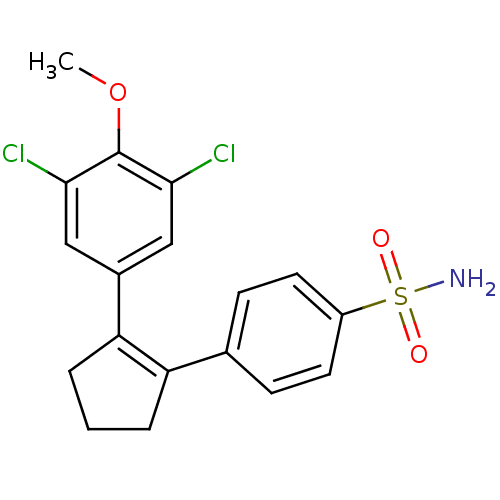
(4-[2-(3,5-Dichloro-4-methoxy-phenyl)-cyclopent-1-e...)Show SMILES COc1c(Cl)cc(cc1Cl)C1=C(CCC1)c1ccc(cc1)S(N)(=O)=O |t:11| Show InChI InChI=1S/C18H17Cl2NO3S/c1-24-18-16(19)9-12(10-17(18)20)15-4-2-3-14(15)11-5-7-13(8-6-11)25(21,22)23/h5-10H,2-4H2,1H3,(H2,21,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibition of prostaglandin G/H synthase 2 (COX-2). |
J Med Chem 38: 4570-8 (1995)
BindingDB Entry DOI: 10.7270/Q23N22D2 |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057620
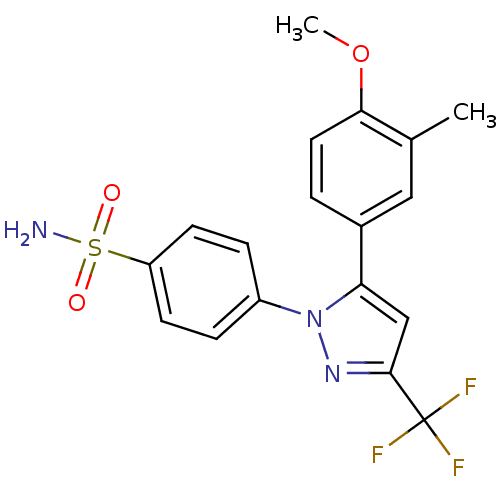
(4-[5-(4-Methoxy-3-methyl-phenyl)-3-trifluoromethyl...)Show SMILES COc1ccc(cc1C)-c1cc(nn1-c1ccc(cc1)S(N)(=O)=O)C(F)(F)F Show InChI InChI=1S/C18H16F3N3O3S/c1-11-9-12(3-8-16(11)27-2)15-10-17(18(19,20)21)23-24(15)13-4-6-14(7-5-13)28(22,25)26/h3-10H,1-2H3,(H2,22,25,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 9.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057527
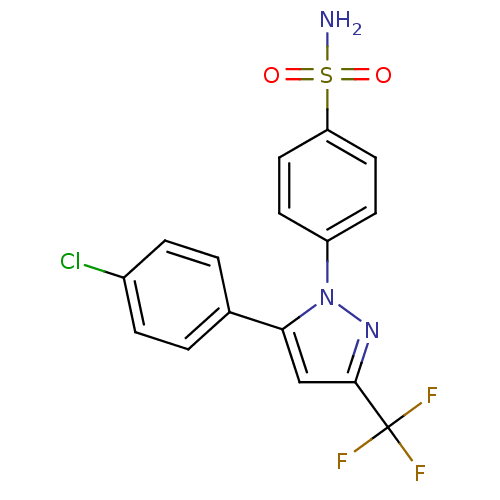
(4-(5-(4-chlorophenyl)-3-(trifluoromethyl)-1H-pyraz...)Show SMILES NS(=O)(=O)c1ccc(cc1)-n1nc(cc1-c1ccc(Cl)cc1)C(F)(F)F Show InChI InChI=1S/C16H11ClF3N3O2S/c17-11-3-1-10(2-4-11)14-9-15(16(18,19)20)22-23(14)12-5-7-13(8-6-12)26(21,24)25/h1-9H,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |
Prostaglandin G/H synthase 2
(Homo sapiens (Human)) | BDBM50057602
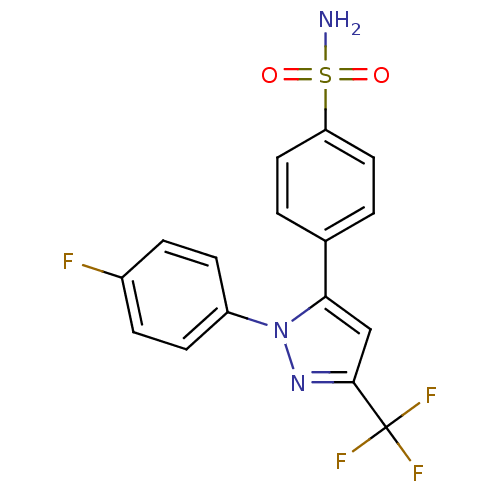
(4-[2-(4-Fluoro-phenyl)-5-trifluoromethyl-2H-pyrazo...)Show SMILES NS(=O)(=O)c1ccc(cc1)-c1cc(nn1-c1ccc(F)cc1)C(F)(F)F Show InChI InChI=1S/C16H11F4N3O2S/c17-11-3-5-12(6-4-11)23-14(9-15(22-23)16(18,19)20)10-1-7-13(8-2-10)26(21,24)25/h1-9H,(H2,21,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 10 | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
In vitro inhibitory concentration required to block human recombinant prostaglandin G/H synthase 2 (COX-2) |
J Med Chem 40: 1347-65 (1997)
Article DOI: 10.1021/jm960803q
BindingDB Entry DOI: 10.7270/Q2Z89BHB |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data