 Found 3746 hits with Last Name = 'chakka' and Initial = 'n'
Found 3746 hits with Last Name = 'chakka' and Initial = 'n' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379458
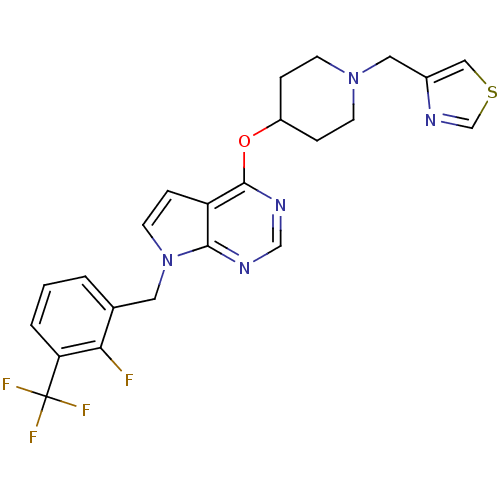
(CHEMBL2012273)Show SMILES Fc1c(Cn2ccc3c(OC4CCN(Cc5cscn5)CC4)ncnc23)cccc1C(F)(F)F Show InChI InChI=1S/C23H21F4N5OS/c24-20-15(2-1-3-19(20)23(25,26)27)10-32-9-6-18-21(32)28-13-29-22(18)33-17-4-7-31(8-5-17)11-16-12-34-14-30-16/h1-3,6,9,12-14,17H,4-5,7-8,10-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379439
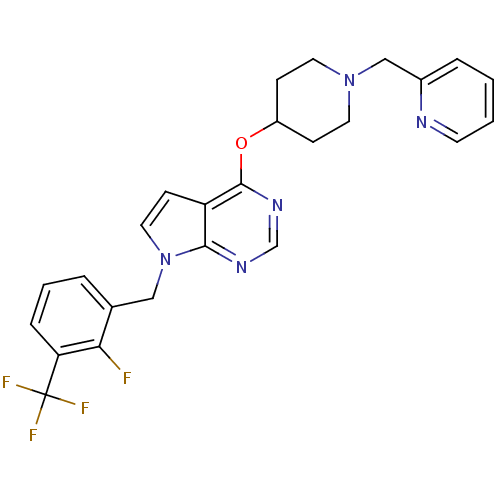
(CHEMBL2012301)Show SMILES Fc1c(Cn2ccc3c(OC4CCN(Cc5ccccn5)CC4)ncnc23)cccc1C(F)(F)F Show InChI InChI=1S/C25H23F4N5O/c26-22-17(4-3-6-21(22)25(27,28)29)14-34-13-9-20-23(34)31-16-32-24(20)35-19-7-11-33(12-8-19)15-18-5-1-2-10-30-18/h1-6,9-10,13,16,19H,7-8,11-12,14-15H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379455
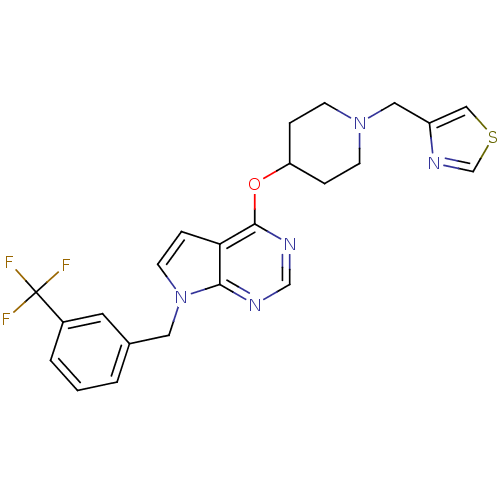
(CHEMBL2012269)Show SMILES FC(F)(F)c1cccc(Cn2ccc3c(OC4CCN(Cc5cscn5)CC4)ncnc23)c1 Show InChI InChI=1S/C23H22F3N5OS/c24-23(25,26)17-3-1-2-16(10-17)11-31-9-6-20-21(31)27-14-28-22(20)32-19-4-7-30(8-5-19)12-18-13-33-15-29-18/h1-3,6,9-10,13-15,19H,4-5,7-8,11-12H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379438

(CHEMBL2012300)Show SMILES Fc1c(Cl)cccc1Cn1ccc2c(OC3CCN(Cc4ccccn4)CC3)ncnc12 Show InChI InChI=1S/C24H23ClFN5O/c25-21-6-3-4-17(22(21)26)14-31-13-9-20-23(31)28-16-29-24(20)32-19-7-11-30(12-8-19)15-18-5-1-2-10-27-18/h1-6,9-10,13,16,19H,7-8,11-12,14-15H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 680 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379467

(CHEMBL2012293)Show SMILES Cc1cccnc1CN1CCC(CC1)Oc1ncnc2n(Cc3ccccc3)ccc12 Show InChI InChI=1S/C25H27N5O/c1-19-6-5-12-26-23(19)17-29-13-9-21(10-14-29)31-25-22-11-15-30(24(22)27-18-28-25)16-20-7-3-2-4-8-20/h2-8,11-12,15,18,21H,9-10,13-14,16-17H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379452
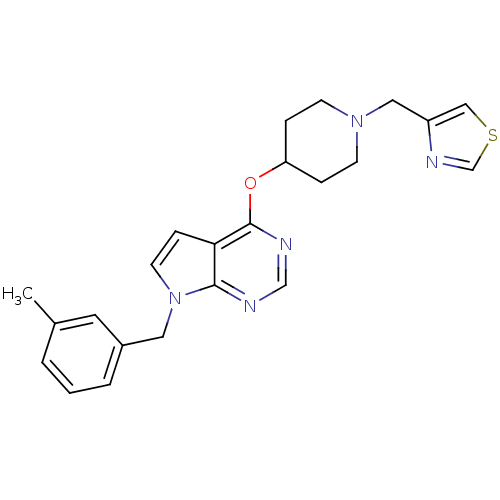
(CHEMBL2012266)Show SMILES Cc1cccc(Cn2ccc3c(OC4CCN(Cc5cscn5)CC4)ncnc23)c1 Show InChI InChI=1S/C23H25N5OS/c1-17-3-2-4-18(11-17)12-28-10-7-21-22(28)24-15-25-23(21)29-20-5-8-27(9-6-20)13-19-14-30-16-26-19/h2-4,7,10-11,14-16,20H,5-6,8-9,12-13H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 930 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379457
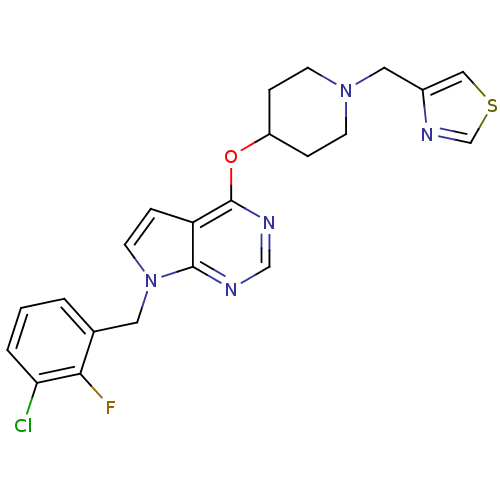
(CHEMBL2012272)Show SMILES Fc1c(Cl)cccc1Cn1ccc2c(OC3CCN(Cc4cscn4)CC3)ncnc12 Show InChI InChI=1S/C22H21ClFN5OS/c23-19-3-1-2-15(20(19)24)10-29-9-6-18-21(29)25-13-26-22(18)30-17-4-7-28(8-5-17)11-16-12-31-14-27-16/h1-3,6,9,12-14,17H,4-5,7-8,10-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 950 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379465
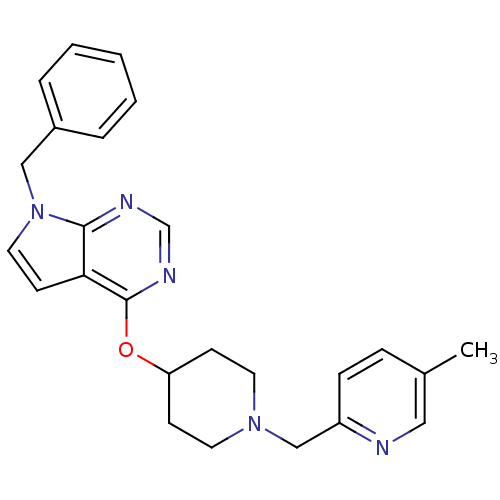
(CHEMBL2012291)Show SMILES Cc1ccc(CN2CCC(CC2)Oc2ncnc3n(Cc4ccccc4)ccc23)nc1 Show InChI InChI=1S/C25H27N5O/c1-19-7-8-21(26-15-19)17-29-12-9-22(10-13-29)31-25-23-11-14-30(24(23)27-18-28-25)16-20-5-3-2-4-6-20/h2-8,11,14-15,18,22H,9-10,12-13,16-17H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 980 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379444

(CHEMBL2012246)Show SMILES FC(F)(F)c1nc(OC2CCN(Cc3cscn3)CC2)c2ccn(Cc3ccccc3)c2n1 Show InChI InChI=1S/C23H22F3N5OS/c24-23(25,26)22-28-20-19(8-11-31(20)12-16-4-2-1-3-5-16)21(29-22)32-18-6-9-30(10-7-18)13-17-14-33-15-27-17/h1-5,8,11,14-15,18H,6-7,9-10,12-13H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 990 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379437

(CHEMBL2012299)Show SMILES Fc1cccc(Cn2ccc3c(OC4CCN(Cc5ccccn5)CC4)ncnc23)c1F Show InChI InChI=1S/C24H23F2N5O/c25-21-6-3-4-17(22(21)26)14-31-13-9-20-23(31)28-16-29-24(20)32-19-7-11-30(12-8-19)15-18-5-1-2-10-27-18/h1-6,9-10,13,16,19H,7-8,11-12,14-15H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379471
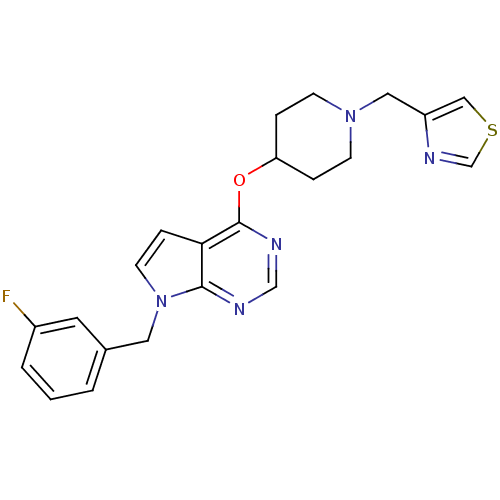
(CHEMBL2012260)Show SMILES Fc1cccc(Cn2ccc3c(OC4CCN(Cc5cscn5)CC4)ncnc23)c1 Show InChI InChI=1S/C22H22FN5OS/c23-17-3-1-2-16(10-17)11-28-9-6-20-21(28)24-14-25-22(20)29-19-4-7-27(8-5-19)12-18-13-30-15-26-18/h1-3,6,9-10,13-15,19H,4-5,7-8,11-12H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379469
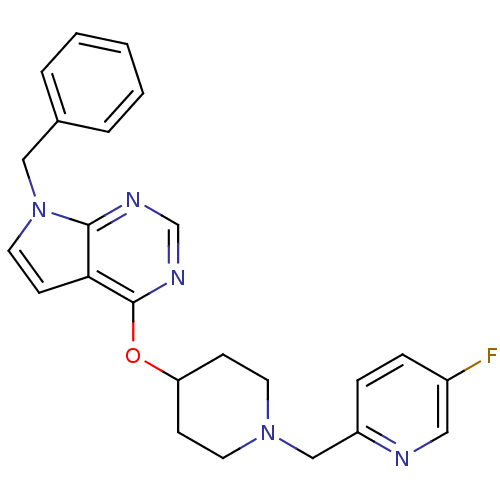
(CHEMBL2012295)Show SMILES Fc1ccc(CN2CCC(CC2)Oc2ncnc3n(Cc4ccccc4)ccc23)nc1 Show InChI InChI=1S/C24H24FN5O/c25-19-6-7-20(26-14-19)16-29-11-8-21(9-12-29)31-24-22-10-13-30(23(22)27-17-28-24)15-18-4-2-1-3-5-18/h1-7,10,13-14,17,21H,8-9,11-12,15-16H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379470
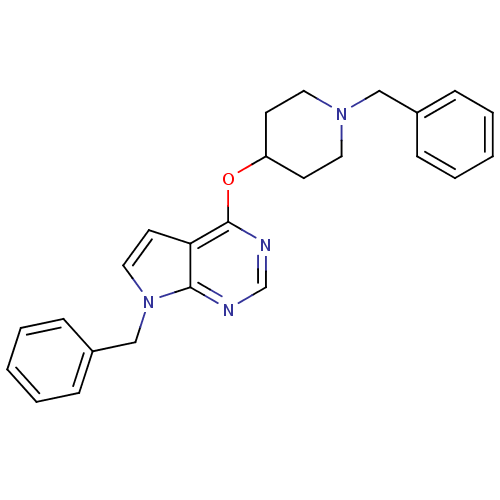
(CHEMBL2012277)Show SMILES C(N1CCC(CC1)Oc1ncnc2n(Cc3ccccc3)ccc12)c1ccccc1 Show InChI InChI=1S/C25H26N4O/c1-3-7-20(8-4-1)17-28-14-11-22(12-15-28)30-25-23-13-16-29(24(23)26-19-27-25)18-21-9-5-2-6-10-21/h1-10,13,16,19,22H,11-12,14-15,17-18H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379450

(CHEMBL2012263)Show SMILES Clc1cccc(Cn2ccc3c(OC4CCN(Cc5cscn5)CC4)ncnc23)c1 Show InChI InChI=1S/C22H22ClN5OS/c23-17-3-1-2-16(10-17)11-28-9-6-20-21(28)24-14-25-22(20)29-19-4-7-27(8-5-19)12-18-13-30-15-26-18/h1-3,6,9-10,13-15,19H,4-5,7-8,11-12H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379440

(CHEMBL2012179)Show SMILES Cc1cn(Cc2ccccc2)c2ncnc(OC3CCN(Cc4cscn4)CC3)c12 Show InChI InChI=1S/C23H25N5OS/c1-17-11-28(12-18-5-3-2-4-6-18)22-21(17)23(25-15-24-22)29-20-7-9-27(10-8-20)13-19-14-30-16-26-19/h2-6,11,14-16,20H,7-10,12-13H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379448
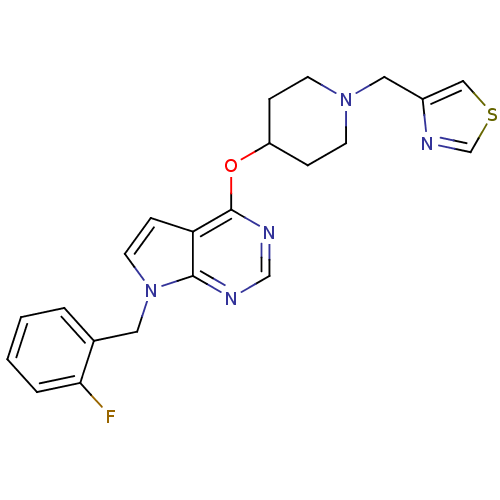
(CHEMBL2012259)Show SMILES Fc1ccccc1Cn1ccc2c(OC3CCN(Cc4cscn4)CC3)ncnc12 Show InChI InChI=1S/C22H22FN5OS/c23-20-4-2-1-3-16(20)11-28-10-7-19-21(28)24-14-25-22(19)29-18-5-8-27(9-6-18)12-17-13-30-15-26-17/h1-4,7,10,13-15,18H,5-6,8-9,11-12H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379442

(CHEMBL2012181)Show SMILES Clc1cn(Cc2ccccc2)c2ncnc(OC3CCN(Cc4cscn4)CC3)c12 Show InChI InChI=1S/C22H22ClN5OS/c23-19-12-28(10-16-4-2-1-3-5-16)21-20(19)22(25-14-24-21)29-18-6-8-27(9-7-18)11-17-13-30-15-26-17/h1-5,12-15,18H,6-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379436

(CHEMBL2012298)Show SMILES Fc1ccccc1Cn1ccc2c(OC3CCN(Cc4ccccn4)CC3)ncnc12 Show InChI InChI=1S/C24H24FN5O/c25-22-7-2-1-5-18(22)15-30-14-10-21-23(30)27-17-28-24(21)31-20-8-12-29(13-9-20)16-19-6-3-4-11-26-19/h1-7,10-11,14,17,20H,8-9,12-13,15-16H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM154061
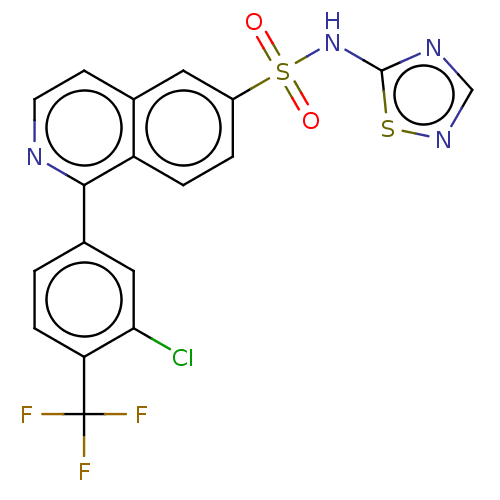
(US9012443, 57)Show SMILES FC(F)(F)c1ccc(cc1Cl)-c1nccc2cc(ccc12)S(=O)(=O)Nc1ncns1 Show InChI InChI=1S/C18H10ClF3N4O2S2/c19-15-8-11(1-4-14(15)18(20,21)22)16-13-3-2-12(7-10(13)5-6-23-16)30(27,28)26-17-24-9-25-29-17/h1-9H,(H,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in HEK293 cells after 45 mins by scintillation counting analysis |
J Med Chem 59: 7818-39 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00425
BindingDB Entry DOI: 10.7270/Q25Q50KX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379466
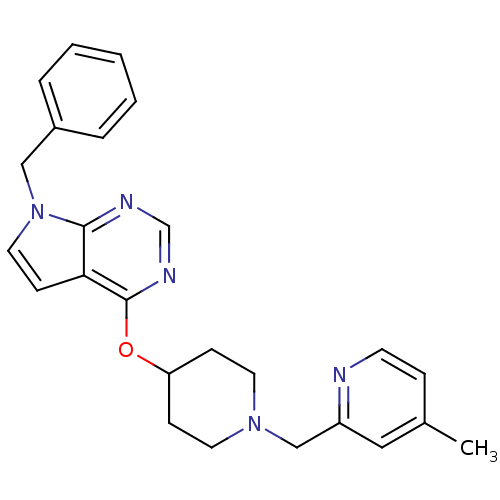
(CHEMBL2012292)Show SMILES Cc1ccnc(CN2CCC(CC2)Oc2ncnc3n(Cc4ccccc4)ccc23)c1 Show InChI InChI=1S/C25H27N5O/c1-19-7-11-26-21(15-19)17-29-12-8-22(9-13-29)31-25-23-10-14-30(24(23)27-18-28-25)16-20-5-3-2-4-6-20/h2-7,10-11,14-15,18,22H,8-9,12-13,16-17H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379459

(CHEMBL2012274)Show SMILES Fc1cccc(F)c1Cn1ccc2c(OC3CCN(Cc4cscn4)CC3)ncnc12 Show InChI InChI=1S/C22H21F2N5OS/c23-19-2-1-3-20(24)18(19)11-29-9-6-17-21(29)25-13-26-22(17)30-16-4-7-28(8-5-16)10-15-12-31-14-27-15/h1-3,6,9,12-14,16H,4-5,7-8,10-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379456

(CHEMBL2012271)Show SMILES Fc1cccc(Cn2ccc3c(OC4CCN(Cc5cscn5)CC4)ncnc23)c1F Show InChI InChI=1S/C22H21F2N5OS/c23-19-3-1-2-15(20(19)24)10-29-9-6-18-21(29)25-13-26-22(18)30-17-4-7-28(8-5-17)11-16-12-31-14-27-16/h1-3,6,9,12-14,17H,4-5,7-8,10-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379464

(CHEMBL2012290)Show SMILES Cc1cccc(CN2CCC(CC2)Oc2ncnc3n(Cc4ccccc4)ccc23)n1 Show InChI InChI=1S/C25H27N5O/c1-19-6-5-9-21(28-19)17-29-13-10-22(11-14-29)31-25-23-12-15-30(24(23)26-18-27-25)16-20-7-3-2-4-8-20/h2-9,12,15,18,22H,10-11,13-14,16-17H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379454

(CHEMBL2012268)Show SMILES FC(F)(F)c1ccccc1Cn1ccc2c(OC3CCN(Cc4cscn4)CC3)ncnc12 Show InChI InChI=1S/C23H22F3N5OS/c24-23(25,26)20-4-2-1-3-16(20)11-31-10-7-19-21(31)27-14-28-22(19)32-18-5-8-30(9-6-18)12-17-13-33-15-29-17/h1-4,7,10,13-15,18H,5-6,8-9,11-12H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.46E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379431
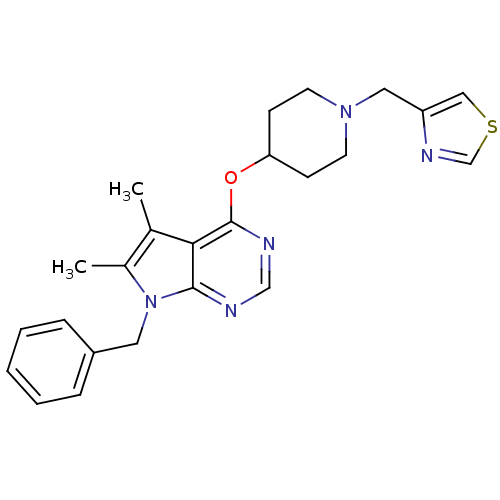
(CHEMBL2012178)Show SMILES Cc1c(C)c2c(OC3CCN(Cc4cscn4)CC3)ncnc2n1Cc1ccccc1 Show InChI InChI=1S/C24H27N5OS/c1-17-18(2)29(12-19-6-4-3-5-7-19)23-22(17)24(26-15-25-23)30-21-8-10-28(11-9-21)13-20-14-31-16-27-20/h3-7,14-16,21H,8-13H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379441

(CHEMBL2012180)Show SMILES Fc1cn(Cc2ccccc2)c2ncnc(OC3CCN(Cc4cscn4)CC3)c12 Show InChI InChI=1S/C22H22FN5OS/c23-19-12-28(10-16-4-2-1-3-5-16)21-20(19)22(25-14-24-21)29-18-6-8-27(9-7-18)11-17-13-30-15-26-17/h1-5,12-15,18H,6-11H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379453
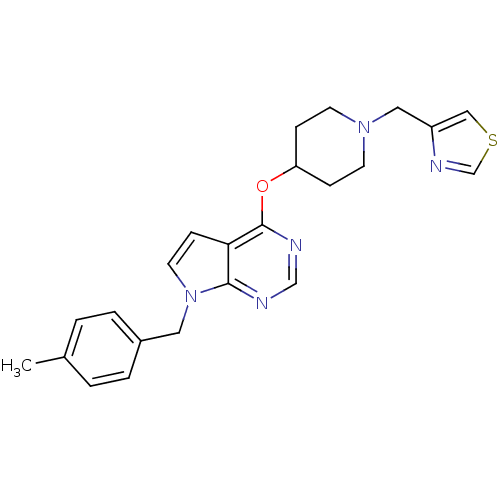
(CHEMBL2012267)Show SMILES Cc1ccc(Cn2ccc3c(OC4CCN(Cc5cscn5)CC4)ncnc23)cc1 Show InChI InChI=1S/C23H25N5OS/c1-17-2-4-18(5-3-17)12-28-11-8-21-22(28)24-15-25-23(21)29-20-6-9-27(10-7-20)13-19-14-30-16-26-19/h2-5,8,11,14-16,20H,6-7,9-10,12-13H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.75E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379468

(CHEMBL2012294)Show SMILES Fc1cccc(CN2CCC(CC2)Oc2ncnc3n(Cc4ccccc4)ccc23)n1 Show InChI InChI=1S/C24H24FN5O/c25-22-8-4-7-19(28-22)16-29-12-9-20(10-13-29)31-24-21-11-14-30(23(21)26-17-27-24)15-18-5-2-1-3-6-18/h1-8,11,14,17,20H,9-10,12-13,15-16H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50379451
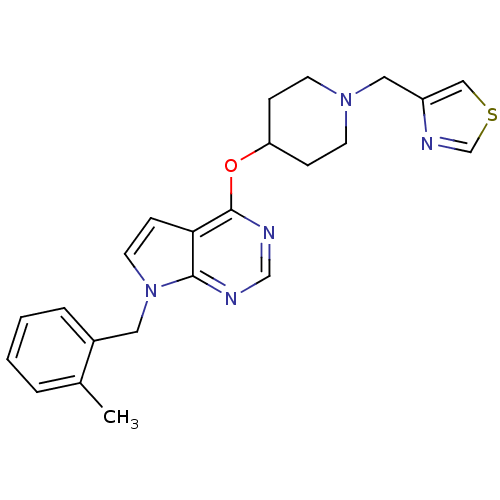
(CHEMBL2012265)Show SMILES Cc1ccccc1Cn1ccc2c(OC3CCN(Cc4cscn4)CC3)ncnc12 Show InChI InChI=1S/C23H25N5OS/c1-17-4-2-3-5-18(17)12-28-11-8-21-22(28)24-15-25-23(21)29-20-6-9-27(10-7-20)13-19-14-30-16-26-19/h2-5,8,11,14-16,20H,6-7,9-10,12-13H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Displacement of dofetilide from human ERG |
Bioorg Med Chem Lett 22: 2052-62 (2012)
Article DOI: 10.1016/j.bmcl.2012.01.015
BindingDB Entry DOI: 10.7270/Q2HM59GC |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50533552
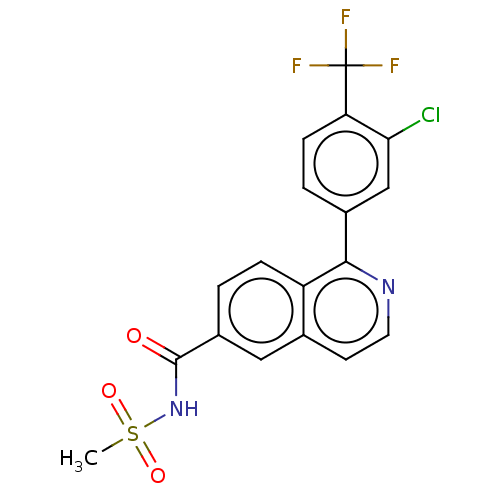
(CHEMBL4450471)Show SMILES CS(=O)(=O)NC(=O)c1ccc2c(nccc2c1)-c1ccc(c(Cl)c1)C(F)(F)F Show InChI InChI=1S/C18H12ClF3N2O3S/c1-28(26,27)24-17(25)12-2-4-13-10(8-12)6-7-23-16(13)11-3-5-14(15(19)9-11)18(20,21)22/h2-9H,1H3,(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in HEK293 cells after 45 mins by scintillation counting analysis |
J Med Chem 59: 7818-39 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00425
BindingDB Entry DOI: 10.7270/Q25Q50KX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50533547

(CHEMBL4537339)Show SMILES CS(=O)(=O)NC(=O)c1cc(F)c(Oc2ccc(Cl)c(Cl)c2)cc1F Show InChI InChI=1S/C14H9Cl2F2NO4S/c1-24(21,22)19-14(20)8-5-12(18)13(6-11(8)17)23-7-2-3-9(15)10(16)4-7/h2-6H,1H3,(H,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in HEK293 cells after 45 mins by scintillation counting analysis |
J Med Chem 59: 7818-39 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00425
BindingDB Entry DOI: 10.7270/Q25Q50KX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50533541
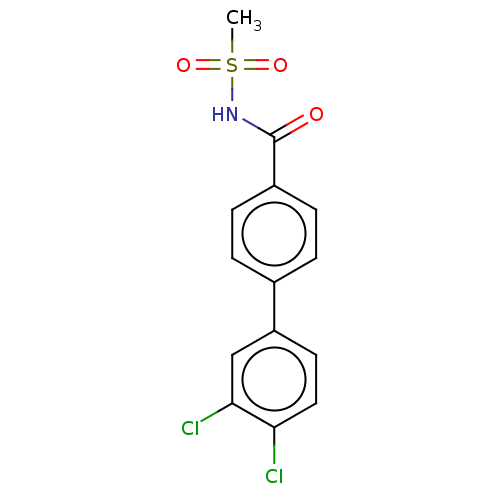
(CHEMBL4559824)Show SMILES CS(=O)(=O)NC(=O)c1ccc(cc1)-c1ccc(Cl)c(Cl)c1 Show InChI InChI=1S/C14H11Cl2NO3S/c1-21(19,20)17-14(18)10-4-2-9(3-5-10)11-6-7-12(15)13(16)8-11/h2-8H,1H3,(H,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in HEK293 cells after 45 mins by scintillation counting analysis |
J Med Chem 59: 7818-39 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00425
BindingDB Entry DOI: 10.7270/Q25Q50KX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50533553

(CHEMBL4445237)Show SMILES CS(=O)(=O)NC(=O)c1cc(F)c(cc1F)-c1ccc(Cl)c(Cl)c1 Show InChI InChI=1S/C14H9Cl2F2NO3S/c1-23(21,22)19-14(20)9-6-12(17)8(5-13(9)18)7-2-3-10(15)11(16)4-7/h2-6H,1H3,(H,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in HEK293 cells after 45 mins by scintillation counting analysis |
J Med Chem 59: 7818-39 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00425
BindingDB Entry DOI: 10.7270/Q25Q50KX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM329203
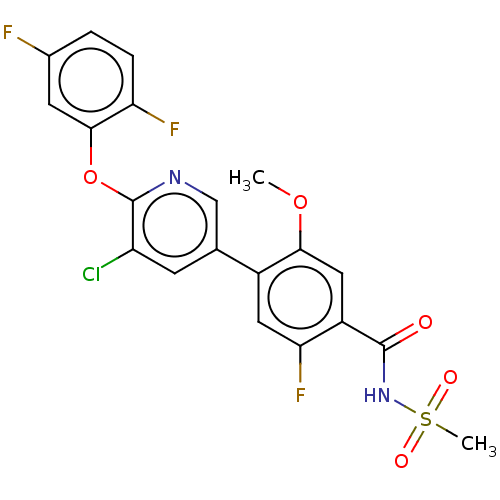
(4-(5-Chloro-6-((1-Methylcyclopropyl)Methoxy)Pyridi...)Show SMILES COc1cc(C(=O)NS(C)(=O)=O)c(F)cc1-c1cnc(Oc2cc(F)ccc2F)c(Cl)c1 Show InChI InChI=1S/C20H14ClF3N2O5S/c1-30-17-8-13(19(27)26-32(2,28)29)16(24)7-12(17)10-5-14(21)20(25-9-10)31-18-6-11(22)3-4-15(18)23/h3-9H,1-2H3,(H,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in HEK293 cells after 45 mins by scintillation counting analysis |
J Med Chem 59: 7818-39 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00425
BindingDB Entry DOI: 10.7270/Q25Q50KX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50533549
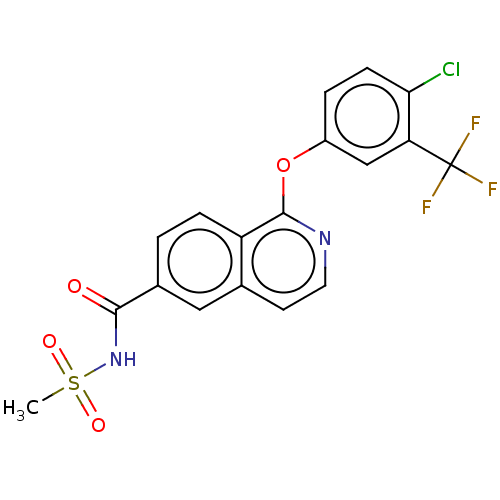
(CHEMBL4579742)Show SMILES CS(=O)(=O)NC(=O)c1ccc2c(Oc3ccc(Cl)c(c3)C(F)(F)F)nccc2c1 Show InChI InChI=1S/C18H12ClF3N2O4S/c1-29(26,27)24-16(25)11-2-4-13-10(8-11)6-7-23-17(13)28-12-3-5-15(19)14(9-12)18(20,21)22/h2-9H,1H3,(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in HEK293 cells after 45 mins by scintillation counting analysis |
J Med Chem 59: 7818-39 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00425
BindingDB Entry DOI: 10.7270/Q25Q50KX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50533554

(CHEMBL4470763)Show SMILES CC(C)COc1ncc(cc1Cl)-c1cc(F)c(cc1F)C(=O)NS(C)(=O)=O Show InChI InChI=1S/C17H17ClF2N2O4S/c1-9(2)8-26-17-13(18)4-10(7-21-17)11-5-15(20)12(6-14(11)19)16(23)22-27(3,24)25/h4-7,9H,8H2,1-3H3,(H,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in HEK293 cells after 45 mins by scintillation counting analysis |
J Med Chem 59: 7818-39 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00425
BindingDB Entry DOI: 10.7270/Q25Q50KX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50269118

(CHEMBL4100413)Show SMILES FC(F)Oc1cccnc1-c1cccc(c1)C(=O)Nc1cccc(c1-c1cccnc1)C(F)(F)F Show InChI InChI=1S/C25H16F5N3O2/c26-24(27)35-20-10-4-12-32-22(20)15-5-1-6-16(13-15)23(34)33-19-9-2-8-18(25(28,29)30)21(19)17-7-3-11-31-14-17/h1-14,24H,(H,33,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human ERG |
Bioorg Med Chem Lett 27: 3817-3824 (2017)
Article DOI: 10.1016/j.bmcl.2017.06.054
BindingDB Entry DOI: 10.7270/Q2RB773S |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50533546

(CHEMBL4462738)Show SMILES CC(C)COc1ncc(Oc2nccc3cc(ccc23)C(=O)NS(C)(=O)=O)cc1Cl Show InChI InChI=1S/C20H20ClN3O5S/c1-12(2)11-28-20-17(21)9-15(10-23-20)29-19-16-5-4-14(8-13(16)6-7-22-19)18(25)24-30(3,26)27/h4-10,12H,11H2,1-3H3,(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in HEK293 cells after 45 mins by scintillation counting analysis |
J Med Chem 59: 7818-39 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00425
BindingDB Entry DOI: 10.7270/Q25Q50KX |
More data for this
Ligand-Target Pair | |
Potassium voltage-gated channel subfamily H member 2
(Homo sapiens (Human)) | BDBM50272533
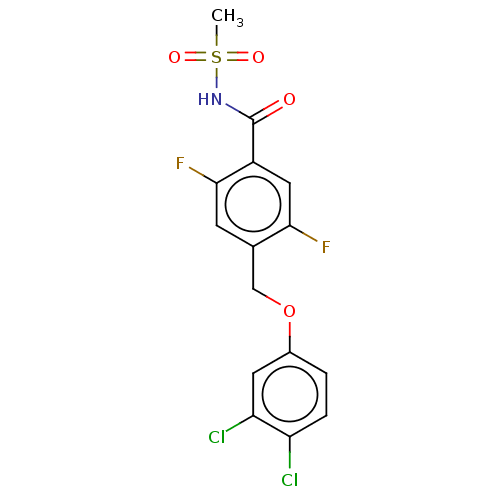
(CHEMBL4129030)Show SMILES CS(=O)(=O)NC(=O)c1cc(F)c(COc2ccc(Cl)c(Cl)c2)cc1F Show InChI InChI=1S/C15H11Cl2F2NO4S/c1-25(22,23)20-15(21)10-6-13(18)8(4-14(10)19)7-24-9-2-3-11(16)12(17)5-9/h2-6H,7H2,1H3,(H,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Displacement of [3H]dofetilide from human ERG expressed in HEK293 cells after 45 mins by scintillation counting analysis |
J Med Chem 59: 7818-39 (2016)
Article DOI: 10.1021/acs.jmedchem.6b00425
BindingDB Entry DOI: 10.7270/Q25Q50KX |
More data for this
Ligand-Target Pair | |
Poly [ADP-ribose] polymerase tankyrase-1 [1091-1325]
(Homo sapiens (Human)) | BDBM259592

(US9505749, 104)Show SMILES O=C(CCSc1nc2ccccc2c(=O)[nH]1)N[C@H]1CC[C@@H](CC1)n1c2ccc(cc2[nH]c1=O)C#N |r,wU:17.18,wD:20.25,(-2.42,-1.15,;-2.42,-2.69,;-3.75,-3.47,;-5.09,-2.69,;-5.09,-1.15,;-6.42,-.38,;-7.75,-1.15,;-9.09,-.38,;-10.42,-1.15,;-11.75,-.38,;-11.75,1.15,;-10.42,1.93,;-9.09,1.15,;-7.75,1.93,;-7.75,3.47,;-6.42,1.15,;-1.08,-3.47,;.25,-2.69,;.25,-1.15,;1.58,-.38,;2.92,-1.15,;2.92,-2.69,;1.58,-3.47,;4.25,-.38,;5.72,-.86,;6.34,-2.27,;7.87,-2.43,;8.78,-1.18,;8.15,.22,;6.62,.38,;5.72,1.63,;4.25,1.15,;2.92,1.93,;10.27,-1.58,;11.75,-1.98,)| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0450 | n/a | n/a | n/a | n/a | n/a | n/a |
AMGEN INC.
US Patent
| Assay Description
The tankyrase 1 biochemical activity of the compounds was assayed in the following assay buffer (50 mM MOPS pH7.5, 100 mM NaCl, 2.5 mM MgCl2, 0.01% T... |
US Patent US9505749 (2016)
BindingDB Entry DOI: 10.7270/Q2FN154F |
More data for this
Ligand-Target Pair | |
Poly [ADP-ribose] polymerase tankyrase-1 [1091-1325]
(Homo sapiens (Human)) | BDBM259576

(US9505749, 87)Show SMILES Clc1ccc2n([C@H]3CC[C@@H](CC3)NC(=O)CSc3nc4ccccc4c(=O)[nH]3)c(=O)[nH]c2c1 |r,wU:6.5,wD:9.12,(-.35,-6.38,;.74,-5.29,;.26,-3.82,;1.29,-2.68,;2.8,-3,;4.04,-2.09,;4.04,-.55,;2.71,.22,;2.71,1.76,;4.04,2.53,;5.38,1.76,;5.38,.22,;4.04,4.07,;2.71,4.84,;2.71,6.38,;1.38,4.07,;.04,4.84,;-1.29,4.07,;-2.62,4.84,;-3.96,4.07,;-5.29,4.84,;-6.62,4.07,;-6.62,2.53,;-5.29,1.76,;-3.96,2.53,;-2.62,1.76,;-2.62,.22,;-1.29,2.53,;5.29,-3,;6.62,-2.23,;4.81,-4.46,;3.27,-4.46,;2.24,-5.61,)| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0597 | n/a | n/a | n/a | n/a | n/a | n/a |
AMGEN INC.
US Patent
| Assay Description
The tankyrase 1 biochemical activity of the compounds was assayed in the following assay buffer (50 mM MOPS pH7.5, 100 mM NaCl, 2.5 mM MgCl2, 0.01% T... |
US Patent US9505749 (2016)
BindingDB Entry DOI: 10.7270/Q2FN154F |
More data for this
Ligand-Target Pair | |
Poly [ADP-ribose] polymerase tankyrase-1 [1091-1325]
(Homo sapiens (Human)) | BDBM259607
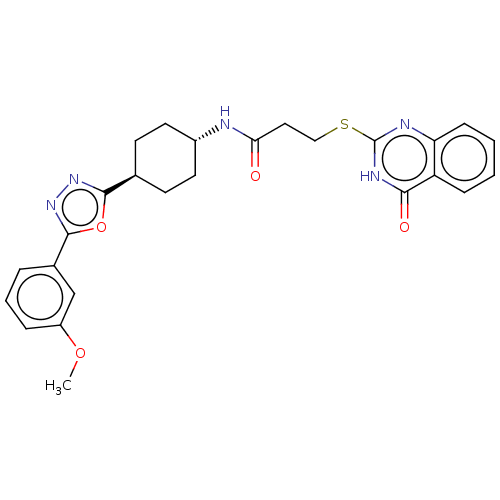
(US9505749, 120)Show SMILES COc1cccc(c1)-c1nnc(o1)[C@H]1CC[C@@H](CC1)NC(=O)CCSc1nc2ccccc2c(=O)[nH]1 |r,wU:16.21,wD:13.14,(11.82,-3.63,;10.33,-3.23,;9.93,-1.74,;11.02,-.65,;10.62,.84,;9.13,1.24,;8.04,.15,;8.44,-1.34,;6.56,.55,;5.65,1.79,;4.19,1.32,;4.19,-.22,;5.65,-.7,;2.85,-.99,;1.52,-.22,;.19,-.99,;.19,-2.53,;1.52,-3.3,;2.85,-2.53,;-1.15,-3.3,;-2.48,-2.53,;-2.48,-.99,;-3.81,-3.3,;-5.15,-2.53,;-5.15,-.99,;-6.48,-.22,;-7.82,-.99,;-9.15,-.22,;-10.48,-.99,;-11.82,-.22,;-11.82,1.32,;-10.48,2.09,;-9.15,1.32,;-7.82,2.09,;-7.82,3.63,;-6.48,1.32,)| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0647 | n/a | n/a | n/a | n/a | n/a | n/a |
AMGEN INC.
US Patent
| Assay Description
The tankyrase 1 biochemical activity of the compounds was assayed in the following assay buffer (50 mM MOPS pH7.5, 100 mM NaCl, 2.5 mM MgCl2, 0.01% T... |
US Patent US9505749 (2016)
BindingDB Entry DOI: 10.7270/Q2FN154F |
More data for this
Ligand-Target Pair | |
Poly [ADP-ribose] polymerase tankyrase-1 [1091-1325]
(Homo sapiens (Human)) | BDBM259608

(US9505749, 121)Show SMILES COc1ccc(cc1)-c1nnc(o1)[C@H]1CC[C@@H](CC1)NC(=O)CCSc1nc2ccccc2c(=O)[nH]1 |r,wU:16.21,wD:13.14,(12.71,-.12,;11.62,-1.21,;10.13,-.81,;9.04,-1.9,;7.55,-1.5,;7.15,-.01,;8.24,1.08,;9.73,.68,;5.67,.38,;4.76,1.63,;3.3,1.15,;3.3,-.38,;4.76,-.86,;1.96,-1.15,;.63,-.38,;-.7,-1.15,;-.7,-2.69,;.63,-3.47,;1.96,-2.69,;-2.04,-3.47,;-3.37,-2.69,;-3.37,-1.15,;-4.7,-3.47,;-6.04,-2.69,;-6.04,-1.15,;-7.37,-.38,;-8.71,-1.15,;-10.04,-.38,;-11.37,-1.15,;-12.71,-.38,;-12.71,1.15,;-11.37,1.93,;-10.04,1.15,;-8.71,1.93,;-8.71,3.47,;-7.37,1.15,)| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0724 | n/a | n/a | n/a | n/a | n/a | n/a |
AMGEN INC.
US Patent
| Assay Description
The tankyrase 1 biochemical activity of the compounds was assayed in the following assay buffer (50 mM MOPS pH7.5, 100 mM NaCl, 2.5 mM MgCl2, 0.01% T... |
US Patent US9505749 (2016)
BindingDB Entry DOI: 10.7270/Q2FN154F |
More data for this
Ligand-Target Pair | |
Poly [ADP-ribose] polymerase tankyrase-2 [946-1162]
(Homo sapiens (Human)) | BDBM259594

(US9505749, 107)Show SMILES O=C(CCSc1nc2ccccc2c(=O)[nH]1)N[C@H]1CC[C@@H](CC1)Oc1ccccn1 |r,wU:17.18,wD:20.25,(2.67,5,;1.33,4.23,;,5,;-1.33,4.23,;-2.67,5,;-4,4.23,;-4,2.69,;-5.33,1.93,;-5.33,.38,;-6.67,-.38,;-8,.38,;-8,1.93,;-6.67,2.69,;-6.67,4.23,;-8,5,;-5.33,5,;1.33,2.69,;2.67,1.93,;4,2.69,;5.33,1.93,;5.33,.38,;4,-.38,;2.67,.38,;6.67,-.38,;6.67,-1.93,;8,-2.69,;8,-4.23,;6.67,-5,;5.33,-4.23,;5.33,-2.69,)| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0882 | n/a | n/a | n/a | n/a | n/a | n/a |
AMGEN INC.
US Patent
| Assay Description
The tankyrase 1 biochemical activity of the compounds was assayed in the following assay buffer (50 mM MOPS pH7.5, 100 mM NaCl, 2.5 mM MgCl2, 0.01% T... |
US Patent US9505749 (2016)
BindingDB Entry DOI: 10.7270/Q2FN154F |
More data for this
Ligand-Target Pair | |
Poly [ADP-ribose] polymerase tankyrase-1 [1091-1325]
(Homo sapiens (Human)) | BDBM259529

(US9505749, 40)Show SMILES O=C(CCSc1nc2ccccc2c(=O)[nH]1)N[C@H]1C[C@@H](C1)c1nnc(o1)-c1ccccc1 |r,wU:19.23,wD:17.18,(-1.01,-.23,;-1.41,-1.72,;-2.9,-2.11,;-3.98,-1.03,;-5.47,-1.42,;-6.56,-.34,;-8.05,-.73,;-9.14,.36,;-10.63,-.04,;-11.71,1.05,;-11.32,2.53,;-9.83,2.93,;-8.74,1.84,;-7.25,2.24,;-6.85,3.73,;-6.16,1.15,;-.32,-2.8,;1.17,-2.41,;2.5,-3.18,;3.27,-1.84,;1.94,-1.07,;4.76,-1.44,;5.24,.02,;6.78,.02,;7.25,-1.44,;6.01,-2.35,;8.74,-1.84,;9.83,-.75,;11.32,-1.15,;11.71,-2.64,;10.63,-3.73,;9.14,-3.33,)| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0891 | n/a | n/a | n/a | n/a | n/a | n/a |
AMGEN INC.
US Patent
| Assay Description
The tankyrase 1 biochemical activity of the compounds was assayed in the following assay buffer (50 mM MOPS pH7.5, 100 mM NaCl, 2.5 mM MgCl2, 0.01% T... |
US Patent US9505749 (2016)
BindingDB Entry DOI: 10.7270/Q2FN154F |
More data for this
Ligand-Target Pair | |
Poly [ADP-ribose] polymerase tankyrase-1 [1091-1325]
(Homo sapiens (Human)) | BDBM50446537
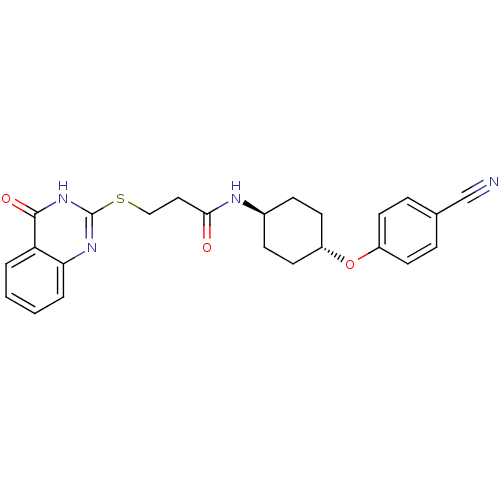
(CHEMBL3110100 | US9505749, 106)Show SMILES O=C(CCSc1nc2ccccc2c(=O)[nH]1)N[C@H]1CC[C@@H](CC1)Oc1ccc(cc1)C#N |r,wU:20.25,wD:17.18,(59.2,-17.39,;59.2,-18.93,;57.86,-19.7,;56.53,-18.93,;55.19,-19.69,;53.86,-18.92,;52.51,-19.69,;51.18,-18.92,;49.85,-19.68,;48.51,-18.91,;48.51,-17.37,;49.84,-16.6,;51.18,-17.36,;52.52,-16.58,;52.52,-15.04,;53.86,-17.36,;60.53,-19.71,;61.86,-18.92,;63.2,-19.69,;64.53,-18.91,;64.52,-17.37,;63.17,-16.61,;61.85,-17.39,;65.85,-16.59,;67.19,-17.35,;67.2,-18.89,;68.54,-19.64,;69.87,-18.86,;69.85,-17.31,;68.51,-16.56,;71.21,-19.62,;72.55,-20.37,)| Show InChI InChI=1S/C24H24N4O3S/c25-15-16-5-9-18(10-6-16)31-19-11-7-17(8-12-19)26-22(29)13-14-32-24-27-21-4-2-1-3-20(21)23(30)28-24/h1-6,9-10,17,19H,7-8,11-14H2,(H,26,29)(H,27,28,30)/t17-,19- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| PDB
US Patent
| n/a | n/a | 0.0973 | n/a | n/a | n/a | n/a | n/a | n/a |
AMGEN INC.
US Patent
| Assay Description
The tankyrase 1 biochemical activity of the compounds was assayed in the following assay buffer (50 mM MOPS pH7.5, 100 mM NaCl, 2.5 mM MgCl2, 0.01% T... |
US Patent US9505749 (2016)
BindingDB Entry DOI: 10.7270/Q2FN154F |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Poly [ADP-ribose] polymerase tankyrase-1 [1091-1325]
(Homo sapiens (Human)) | BDBM259584

(US9505749, 95)Show SMILES O=C(CCCc1nc2ccccc2c(=O)[nH]1)N[C@H]1CC[C@@H](CC1)c1ncc(o1)-c1ccccc1 |r,wU:17.18,wD:20.25,(-2.08,.92,;-2.08,-.62,;-3.42,-1.39,;-3.42,-2.93,;-4.75,-3.7,;-6.08,-2.93,;-7.42,-3.7,;-8.75,-2.93,;-10.08,-3.7,;-11.42,-2.93,;-11.42,-1.39,;-10.08,-.62,;-8.75,-1.39,;-7.42,-.62,;-7.42,.92,;-6.08,-1.39,;-.75,-1.39,;.58,-.62,;.58,.92,;1.92,1.69,;3.25,.92,;3.25,-.62,;1.92,-1.39,;4.59,1.69,;4.59,3.23,;6.05,3.7,;6.96,2.46,;6.05,1.21,;8.44,2.06,;8.84,.57,;10.33,.17,;11.42,1.26,;11.02,2.75,;9.53,3.15,)| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0979 | n/a | n/a | n/a | n/a | n/a | n/a |
AMGEN INC.
US Patent
| Assay Description
The tankyrase 1 biochemical activity of the compounds was assayed in the following assay buffer (50 mM MOPS pH7.5, 100 mM NaCl, 2.5 mM MgCl2, 0.01% T... |
US Patent US9505749 (2016)
BindingDB Entry DOI: 10.7270/Q2FN154F |
More data for this
Ligand-Target Pair | |
Poly [ADP-ribose] polymerase tankyrase-2 [946-1162]
(Homo sapiens (Human)) | BDBM259593
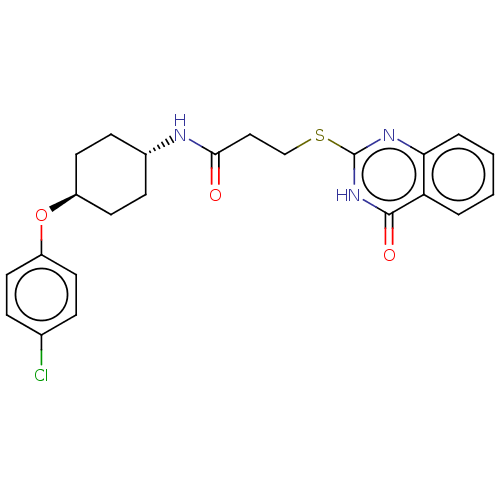
(US9505749, 105)Show SMILES Clc1ccc(O[C@H]2CC[C@@H](CC2)NC(=O)CCSc2nc3ccccc3c(=O)[nH]2)cc1 |r,wU:9.12,wD:6.5,(6.67,-5.78,;6.67,-4.23,;5.33,-3.47,;5.33,-1.93,;6.67,-1.15,;6.67,.38,;5.33,1.15,;5.33,2.69,;4,3.47,;2.67,2.69,;2.67,1.15,;4,.38,;1.33,3.47,;1.33,5,;2.67,5.78,;,5.78,;-1.33,5,;-2.67,5.78,;-4,5,;-4,3.47,;-5.33,2.69,;-5.33,1.15,;-6.67,.38,;-8,1.15,;-8,2.69,;-6.67,3.47,;-6.67,5,;-8,5.78,;-5.33,5.78,;8,-1.93,;8,-3.47,)| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.0982 | n/a | n/a | n/a | n/a | n/a | n/a |
AMGEN INC.
US Patent
| Assay Description
The tankyrase 1 biochemical activity of the compounds was assayed in the following assay buffer (50 mM MOPS pH7.5, 100 mM NaCl, 2.5 mM MgCl2, 0.01% T... |
US Patent US9505749 (2016)
BindingDB Entry DOI: 10.7270/Q2FN154F |
More data for this
Ligand-Target Pair | |
Poly [ADP-ribose] polymerase tankyrase-1 [1091-1325]
(Homo sapiens (Human)) | BDBM259605

(US9505749, 118)Show SMILES Clc1cccc(c1)-c1nnc(o1)[C@H]1CC[C@@H](CC1)NC(=O)CCSc1nc2ccccc2c(=O)[nH]1 |r,wU:15.20,wD:12.13,(10.73,-3.39,;10.33,-1.9,;11.42,-.81,;11.02,.68,;9.53,1.08,;8.44,-.01,;8.84,-1.5,;6.96,.38,;6.05,1.63,;4.59,1.15,;4.59,-.38,;6.05,-.86,;3.25,-1.15,;1.92,-.38,;.58,-1.15,;.58,-2.69,;1.92,-3.47,;3.25,-2.69,;-.75,-3.47,;-2.08,-2.69,;-2.08,-1.15,;-3.42,-3.47,;-4.75,-2.69,;-4.75,-1.15,;-6.08,-.38,;-7.42,-1.15,;-8.75,-.38,;-10.08,-1.15,;-11.42,-.38,;-11.42,1.15,;-10.08,1.93,;-8.75,1.15,;-7.42,1.93,;-7.42,3.47,;-6.08,1.15,)| | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 0.107 | n/a | n/a | n/a | n/a | n/a | n/a |
AMGEN INC.
US Patent
| Assay Description
The tankyrase 1 biochemical activity of the compounds was assayed in the following assay buffer (50 mM MOPS pH7.5, 100 mM NaCl, 2.5 mM MgCl2, 0.01% T... |
US Patent US9505749 (2016)
BindingDB Entry DOI: 10.7270/Q2FN154F |
More data for this
Ligand-Target Pair | |
Poly [ADP-ribose] polymerase tankyrase-1 [1091-1325]
(Homo sapiens (Human)) | BDBM50446538
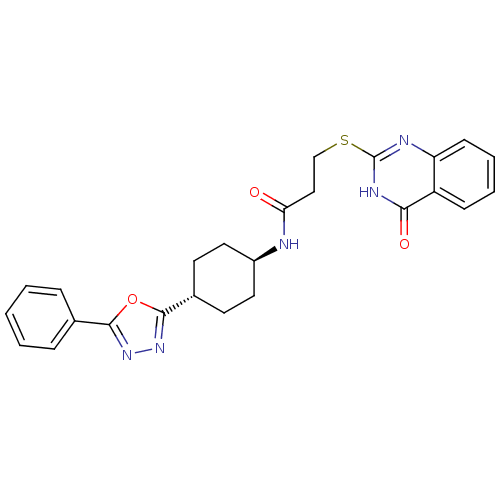
(CHEMBL3110117 | US9505749, 97)Show SMILES O=C(CCSc1nc2ccccc2c(=O)[nH]1)N[C@H]1CC[C@@H](CC1)c1nnc(o1)-c1ccccc1 |r,wU:20.25,wD:17.18,(59.36,-49.64,;59.35,-51.18,;58.02,-51.95,;56.68,-51.17,;55.35,-51.94,;54.02,-51.16,;52.67,-51.94,;51.33,-51.16,;50,-51.93,;48.67,-51.16,;48.67,-49.61,;50,-48.84,;51.33,-49.61,;52.67,-48.83,;52.67,-47.29,;54.02,-49.61,;60.68,-51.95,;62.02,-51.17,;63.35,-51.94,;64.68,-51.16,;64.67,-49.62,;63.33,-48.86,;62,-49.64,;66,-48.83,;67.42,-49.44,;68.44,-48.29,;67.65,-46.96,;66.15,-47.29,;68.4,-45.62,;69.94,-45.6,;70.69,-44.26,;69.9,-42.93,;68.35,-42.96,;67.61,-44.31,)| Show InChI InChI=1S/C25H25N5O3S/c31-21(14-15-34-25-27-20-9-5-4-8-19(20)22(32)28-25)26-18-12-10-17(11-13-18)24-30-29-23(33-24)16-6-2-1-3-7-16/h1-9,17-18H,10-15H2,(H,26,31)(H,27,28,32)/t17-,18- | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| PDB
US Patent
| n/a | n/a | 0.109 | n/a | n/a | n/a | n/a | n/a | n/a |
AMGEN INC.
US Patent
| Assay Description
The tankyrase 1 biochemical activity of the compounds was assayed in the following assay buffer (50 mM MOPS pH7.5, 100 mM NaCl, 2.5 mM MgCl2, 0.01% T... |
US Patent US9505749 (2016)
BindingDB Entry DOI: 10.7270/Q2FN154F |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data