 Found 3124 hits with Last Name = 'sun' and Initial = 'c'
Found 3124 hits with Last Name = 'sun' and Initial = 'c' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM50270877
((R)-4-(4-((2-(4-chlorophenyl)-5,5-dimethylcyclohex...)Show SMILES CC1(C)CCC(=C(CN2CCN(CC2)c2ccc(cc2)C(=O)NS(=O)(=O)c2ccc(N[C@H](CCN3CCOCC3)CSc3ccccc3)c(c2)S(=O)(=O)C(F)(F)F)C1)c1ccc(Cl)cc1 |r,t:5| Show InChI InChI=1S/C47H55ClF3N5O6S3/c1-46(2)20-18-42(34-8-12-37(48)13-9-34)36(31-46)32-55-22-24-56(25-23-55)39-14-10-35(11-15-39)45(57)53-65(60,61)41-16-17-43(44(30-41)64(58,59)47(49,50)51)52-38(19-21-54-26-28-62-29-27-54)33-63-40-6-4-3-5-7-40/h3-17,30,38,52H,18-29,31-33H2,1-2H3,(H,53,57)/t38-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| <0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of F-Bak binding to GST-tagged BCL-XL (unknown origin) measured after 1 hr by TR-FRET assay |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00162
BindingDB Entry DOI: 10.7270/Q2R78JW5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Apoptosis regulator Bcl-2
(Homo sapiens (Human)) | BDBM50270877

((R)-4-(4-((2-(4-chlorophenyl)-5,5-dimethylcyclohex...)Show SMILES CC1(C)CCC(=C(CN2CCN(CC2)c2ccc(cc2)C(=O)NS(=O)(=O)c2ccc(N[C@H](CCN3CCOCC3)CSc3ccccc3)c(c2)S(=O)(=O)C(F)(F)F)C1)c1ccc(Cl)cc1 |r,t:5| Show InChI InChI=1S/C47H55ClF3N5O6S3/c1-46(2)20-18-42(34-8-12-37(48)13-9-34)36(31-46)32-55-22-24-56(25-23-55)39-14-10-35(11-15-39)45(57)53-65(60,61)41-16-17-43(44(30-41)64(58,59)47(49,50)51)52-38(19-21-54-26-28-62-29-27-54)33-63-40-6-4-3-5-7-40/h3-17,30,38,52H,18-29,31-33H2,1-2H3,(H,53,57)/t38-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| <0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of F-Bak binding to GST-tagged BCL2 (unknown origin) measured after 1 hr by TR-FRET assay |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00162
BindingDB Entry DOI: 10.7270/Q2R78JW5 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50368427
(CHEMBL1790880 | CHEMBL3349491)Show SMILES [H][C@@](O)(CN1CCC[C@H]1C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](Cc1ccccc1)C(=O)OC)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)c1ccc2ccccc2n1 Show InChI InChI=1S/C45H55N7O8/c1-4-28(2)40(44(58)50-36(45(59)60-3)25-30-16-9-6-10-17-30)51-43(57)37-20-13-23-52(37)27-38(53)34(24-29-14-7-5-8-15-29)48-42(56)35(26-39(46)54)49-41(55)33-22-21-31-18-11-12-19-32(31)47-33/h5-12,14-19,21-22,28,34-38,40,53H,4,13,20,23-27H2,1-3H3,(H2,46,54)(H,48,56)(H,49,55)(H,50,58)(H,51,57)/t28-,34+,35+,36+,37+,38-,40+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Binding affinity to HIV protease |
J Med Chem 35: 3803-12 (1992)
BindingDB Entry DOI: 10.7270/Q2348M0D |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM139040
(US8883784, 1)Show SMILES CN(C)CC[C@H](CSc1ccccc1)Nc1ccc(cc1[N+]([O-])=O)S(=O)(=O)NC(=O)c1csc(n1)N1CCc2cccc(C(=O)Nc3nc4ccccc4s3)c2C1 Show InChI InChI=1S/C39H38N8O6S4/c1-45(2)19-18-26(23-54-27-10-4-3-5-11-27)40-31-16-15-28(21-34(31)47(50)51)57(52,53)44-37(49)33-24-55-39(42-33)46-20-17-25-9-8-12-29(30(25)22-46)36(48)43-38-41-32-13-6-7-14-35(32)56-38/h3-16,21,24,26,40H,17-20,22-23H2,1-2H3,(H,44,49)(H,41,43,48)/t26-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of F-Bak binding to GST-tagged BCL-XL (unknown origin) measured after 1 hr by TR-FRET assay |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00162
BindingDB Entry DOI: 10.7270/Q2R78JW5 |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50078161

(CHEMBL3417702)Show SMILES CCS(=O)(=O)N1CCN(CC1)c1ccc(OCc2c(c(C)nn2C)-c2cccc3c(CCCOc4cccc5ccccc45)c(C(O)=O)n(CCN4CCOCC4)c23)cc1 |(-21.22,-16.71,;-19.99,-16.7,;-19.23,-15.36,;-19.85,-14.3,;-20.46,-15.37,;-17.68,-15.35,;-16.91,-16.68,;-15.37,-16.67,;-14.6,-15.33,;-15.38,-14,;-16.92,-14.01,;-13.06,-15.32,;-12.28,-16.64,;-10.74,-16.63,;-9.98,-15.29,;-8.44,-15.28,;-7.69,-13.93,;-6.15,-13.92,;-5.26,-12.68,;-3.79,-13.11,;-2.81,-12.36,;-3.77,-14.65,;-5.22,-15.15,;-5.58,-16.33,;-5.74,-11.22,;-7.27,-10.9,;-7.75,-9.44,;-6.72,-8.27,;-5.21,-8.6,;-3.97,-7.71,;-3.98,-6.17,;-2.65,-5.4,;-2.66,-3.85,;-1.33,-3.08,;-1.33,-1.54,;-2.68,-.77,;-2.68,.77,;-1.33,1.54,;,.77,;1.33,1.54,;2.66,.77,;2.66,-.77,;1.33,-1.54,;,-.77,;-2.74,-8.57,;-1.29,-8.05,;-.35,-8.85,;-1.06,-6.84,;-3.2,-10.04,;-2.28,-11.27,;-.75,-11.09,;.17,-12.33,;1.7,-12.15,;2.62,-13.39,;2.01,-14.8,;.48,-14.98,;-.44,-13.74,;-4.73,-10.05,;-10.76,-13.96,;-12.3,-13.98,)| Show InChI InChI=1S/C46H54N6O7S/c1-4-60(55,56)51-24-22-50(23-25-51)35-17-19-36(20-18-35)59-32-41-43(33(2)47-48(41)3)40-14-8-13-38-39(15-9-29-58-42-16-7-11-34-10-5-6-12-37(34)42)45(46(53)54)52(44(38)40)26-21-49-27-30-57-31-28-49/h5-8,10-14,16-20H,4,9,15,21-32H2,1-3H3,(H,53,54) | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Displacement of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide) from MCL1 (unknown origin) after 1 hr by TR-FRET assay |
J Med Chem 58: 2180-94 (2015)
Article DOI: 10.1021/jm501258m
BindingDB Entry DOI: 10.7270/Q2HX1FC8 |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50078159
(CHEMBL3417700)Show SMILES CC(=O)N1CCN(CC1)c1ccc(OCc2c(c(C)nn2C)-c2cccc3c(CCCOc4cccc5ccccc45)c(C(O)=O)n(CCN4CCOCC4)c23)cc1 |(-19.84,-16.43,;-19.23,-15.36,;-19.85,-14.29,;-17.68,-15.35,;-16.91,-16.68,;-15.37,-16.67,;-14.6,-15.33,;-15.38,-14,;-16.92,-14.01,;-13.06,-15.32,;-12.28,-16.64,;-10.74,-16.63,;-9.98,-15.29,;-8.44,-15.28,;-7.69,-13.93,;-6.15,-13.92,;-5.26,-12.68,;-3.79,-13.11,;-2.81,-12.36,;-3.77,-14.65,;-5.22,-15.15,;-5.58,-16.33,;-5.74,-11.22,;-7.27,-10.9,;-7.75,-9.44,;-6.72,-8.27,;-5.21,-8.6,;-3.97,-7.71,;-3.98,-6.17,;-2.65,-5.4,;-2.66,-3.85,;-1.33,-3.08,;-1.33,-1.54,;-2.68,-.77,;-2.68,.77,;-1.33,1.54,;,.77,;1.33,1.54,;2.66,.77,;2.66,-.77,;1.33,-1.54,;,-.77,;-2.74,-8.57,;-1.29,-8.05,;-.35,-8.85,;-1.06,-6.84,;-3.2,-10.04,;-2.28,-11.27,;-.75,-11.09,;.17,-12.33,;1.7,-12.15,;2.62,-13.39,;2.01,-14.8,;.48,-14.98,;-.44,-13.74,;-4.73,-10.05,;-10.76,-13.96,;-12.3,-13.98,)| Show InChI InChI=1S/C46H52N6O6/c1-32-43(41(48(3)47-32)31-58-36-18-16-35(17-19-36)51-23-21-50(22-24-51)33(2)53)40-13-7-12-38-39(14-8-28-57-42-15-6-10-34-9-4-5-11-37(34)42)45(46(54)55)52(44(38)40)25-20-49-26-29-56-30-27-49/h4-7,9-13,15-19H,8,14,20-31H2,1-3H3,(H,54,55) | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Displacement of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide) from MCL1 (unknown origin) after 1 hr by TR-FRET assay |
J Med Chem 58: 2180-94 (2015)
Article DOI: 10.1021/jm501258m
BindingDB Entry DOI: 10.7270/Q2HX1FC8 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50368430

(CHEMBL1790874 | CHEMBL3349485)Show SMILES [H][C@@](O)(CN1CCC[C@H]1C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C(C)C)C(=O)OC)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(C)=O Show InChI InChI=1S/C42H68N8O11/c1-9-25(6)36(41(59)48-35(24(4)5)42(60)61-8)49-40(58)32-16-13-17-50(32)21-33(53)28(19-27-14-11-10-12-15-27)45-38(56)30(20-34(43)54)47-37(55)29(18-23(2)3)46-39(57)31(22-51)44-26(7)52/h10-12,14-15,23-25,28-33,35-36,51,53H,9,13,16-22H2,1-8H3,(H2,43,54)(H,44,52)(H,45,56)(H,46,57)(H,47,55)(H,48,59)(H,49,58)/t25-,28+,29+,30+,31+,32+,33-,35+,36+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Binding affinity to HIV protease |
J Med Chem 35: 3803-12 (1992)
BindingDB Entry DOI: 10.7270/Q2348M0D |
More data for this
Ligand-Target Pair | |
Polycomb protein EED
(Homo sapiens (Human)) | BDBM50235631
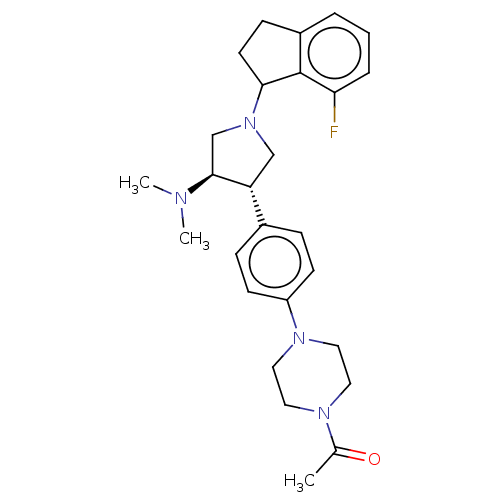
(CHEMBL4060827)Show SMILES CN(C)[C@H]1CN(C[C@@H]1c1ccc(cc1)N1CCN(CC1)C(C)=O)C1CCc2cccc(F)c12 |r| Show InChI InChI=1S/C27H35FN4O/c1-19(33)30-13-15-31(16-14-30)22-10-7-20(8-11-22)23-17-32(18-26(23)29(2)3)25-12-9-21-5-4-6-24(28)27(21)25/h4-8,10-11,23,25-26H,9,12-18H2,1-3H3/t23-,25?,26+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Inhibition of OG(488) labeled probe binding to GST-tagged EED (unknown origin) after 1 hr by LanthaScreen TR-FRET assay |
Bioorg Med Chem Lett 27: 1576-1583 (2017)
Article DOI: 10.1016/j.bmcl.2017.02.030
BindingDB Entry DOI: 10.7270/Q22F7QQG |
More data for this
Ligand-Target Pair | |
Polycomb protein EED
(Homo sapiens (Human)) | BDBM50235630
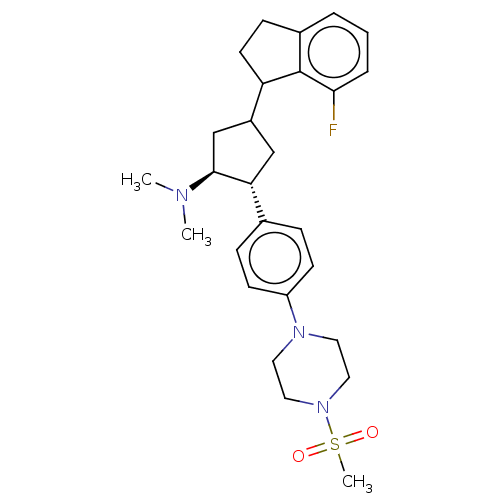
(CHEMBL4093096)Show SMILES CN(C)[C@H]1CC(C[C@@H]1c1ccc(cc1)N1CCN(CC1)S(C)(=O)=O)C1CCc2cccc(F)c12 |r| Show InChI InChI=1S/C27H36FN3O2S/c1-29(2)26-18-21(23-12-9-20-5-4-6-25(28)27(20)23)17-24(26)19-7-10-22(11-8-19)30-13-15-31(16-14-30)34(3,32)33/h4-8,10-11,21,23-24,26H,9,12-18H2,1-3H3/t21?,23?,24-,26+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Inhibition of OG(488) labeled probe binding to GST-tagged EED (unknown origin) after 1 hr by LanthaScreen TR-FRET assay |
Bioorg Med Chem Lett 27: 1576-1583 (2017)
Article DOI: 10.1016/j.bmcl.2017.02.030
BindingDB Entry DOI: 10.7270/Q22F7QQG |
More data for this
Ligand-Target Pair | |
Polycomb protein EED
(Homo sapiens (Human)) | BDBM50241662
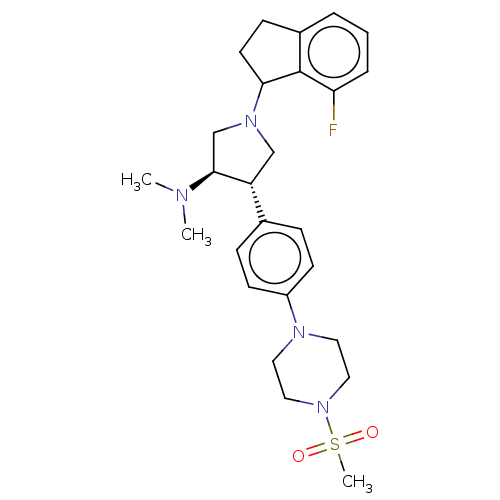
(CHEMBL4104741)Show SMILES CN(C)[C@H]1CN(C[C@@H]1c1ccc(cc1)N1CCN(CC1)S(C)(=O)=O)C1CCc2cccc(F)c12 |r| Show InChI InChI=1S/C26H35FN4O2S/c1-28(2)25-18-30(24-12-9-20-5-4-6-23(27)26(20)24)17-22(25)19-7-10-21(11-8-19)29-13-15-31(16-14-29)34(3,32)33/h4-8,10-11,22,24-25H,9,12-18H2,1-3H3/t22-,24?,25+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to GST tagged EED (unknown origin) after 1 hr by OG(488) probe based TR-FRET assay |
J Med Chem 60: 8321-8335 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00576
BindingDB Entry DOI: 10.7270/Q2TM7D8G |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18161

((1S,2S,7S,10R,11S,14S,15S)-14-hydroxy-2,15-dimethy...)Show SMILES [H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CC[C@@]2([H])CC(=O)CC[C@]12C |r| Show InChI InChI=1S/C19H30O2/c1-18-9-7-13(20)11-12(18)3-4-14-15-5-6-17(21)19(15,2)10-8-16(14)18/h12,14-17,21H,3-11H2,1-2H3/t12-,14-,15-,16-,17-,18-,19-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.300 | -53.8 | n/a | n/a | 2.80 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Polycomb protein EED
(Homo sapiens (Human)) | BDBM50241660
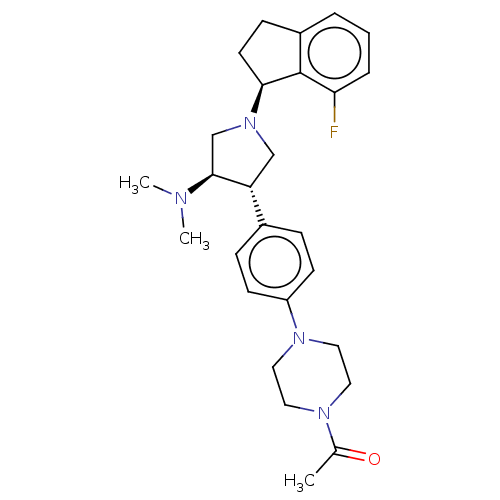
(CHEMBL4084398)Show SMILES CN(C)[C@H]1CN(C[C@@H]1c1ccc(cc1)N1CCN(CC1)C(C)=O)[C@H]1CCc2cccc(F)c12 |r| Show InChI InChI=1S/C27H35FN4O/c1-19(33)30-13-15-31(16-14-30)22-10-7-20(8-11-22)23-17-32(18-26(23)29(2)3)25-12-9-21-5-4-6-24(28)27(21)25/h4-8,10-11,23,25-26H,9,12-18H2,1-3H3/t23-,25+,26+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
The compound was tested for the inhibition of fibrinogen receptor |
J Med Chem 60: 8321-8335 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00576
BindingDB Entry DOI: 10.7270/Q2TM7D8G |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50078162
(CHEMBL3417703)Show SMILES CCCS(=O)(=O)N1CCN(CC1)c1ccc(OCc2c(c(C)nn2C)-c2cccc3c(CCCOc4cccc5ccccc45)c(C(O)=O)n(CCN4CCOCC4)c23)cc1 |(-22.14,-17.78,;-21.53,-16.71,;-19.99,-16.7,;-19.23,-15.36,;-19.85,-14.3,;-20.46,-15.37,;-17.68,-15.35,;-16.91,-16.68,;-15.37,-16.67,;-14.6,-15.33,;-15.38,-14,;-16.92,-14.01,;-13.06,-15.32,;-12.28,-16.64,;-10.74,-16.63,;-9.98,-15.29,;-8.44,-15.28,;-7.69,-13.93,;-6.15,-13.92,;-5.26,-12.68,;-3.79,-13.11,;-2.81,-12.36,;-3.77,-14.65,;-5.22,-15.15,;-5.58,-16.33,;-5.74,-11.22,;-7.27,-10.9,;-7.75,-9.44,;-6.72,-8.27,;-5.21,-8.6,;-3.97,-7.71,;-3.98,-6.17,;-2.65,-5.4,;-2.66,-3.85,;-1.33,-3.08,;-1.33,-1.54,;-2.68,-.77,;-2.68,.77,;-1.33,1.54,;,.77,;1.33,1.54,;2.66,.77,;2.66,-.77,;1.33,-1.54,;,-.77,;-2.74,-8.57,;-1.29,-8.05,;-.35,-8.85,;-1.06,-6.84,;-3.2,-10.04,;-2.28,-11.27,;-.75,-11.09,;.17,-12.33,;1.7,-12.15,;2.62,-13.39,;2.01,-14.8,;.48,-14.98,;-.44,-13.74,;-4.73,-10.05,;-10.76,-13.96,;-12.3,-13.98,)| Show InChI InChI=1S/C47H56N6O7S/c1-4-32-61(56,57)52-24-22-51(23-25-52)36-17-19-37(20-18-36)60-33-42-44(34(2)48-49(42)3)41-14-8-13-39-40(15-9-29-59-43-16-7-11-35-10-5-6-12-38(35)43)46(47(54)55)53(45(39)41)26-21-50-27-30-58-31-28-50/h5-8,10-14,16-20H,4,9,15,21-33H2,1-3H3,(H,54,55) | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.310 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Displacement of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide) from MCL1 (unknown origin) after 1 hr by TR-FRET assay |
J Med Chem 58: 2180-94 (2015)
Article DOI: 10.1021/jm501258m
BindingDB Entry DOI: 10.7270/Q2HX1FC8 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50368420
(CHEMBL1790890 | CHEMBL3349500)Show SMILES [H][C@@](O)(CN1CCCC1C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](Cc1ccccc1)C(=O)OC)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)c1ccc2ccccc2n1 Show InChI InChI=1S/C45H55N7O8/c1-4-28(2)40(44(58)50-36(45(59)60-3)25-30-16-9-6-10-17-30)51-43(57)37-20-13-23-52(37)27-38(53)34(24-29-14-7-5-8-15-29)48-42(56)35(26-39(46)54)49-41(55)33-22-21-31-18-11-12-19-32(31)47-33/h5-12,14-19,21-22,28,34-38,40,53H,4,13,20,23-27H2,1-3H3,(H2,46,54)(H,48,56)(H,49,55)(H,50,58)(H,51,57)/t28-,34+,35+,36+,37?,38-,40+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Binding affinity to HIV protease |
J Med Chem 35: 3803-12 (1992)
BindingDB Entry DOI: 10.7270/Q2348M0D |
More data for this
Ligand-Target Pair | |
Polycomb protein EED
(Homo sapiens (Human)) | BDBM223987
(A-395 (5) | rac-(3R,4S)-1-(7-fluoro-2,3-dihydro-1H...)Show SMILES CN(C)[C@H]1CN(C[C@@H]1c1ccc(cc1)N1CCN(CC1)S(C)(=O)=O)C1CCc2cccc(F)c12 |r,w:24.26| Show InChI InChI=1S/C26H35FN4O2S/c1-28(2)25-18-30(24-12-9-20-5-4-6-23(27)26(20)24)17-22(25)19-7-10-21(11-8-19)29-13-15-31(16-14-29)34(3,32)33/h4-8,10-11,22,24-25H,9,12-18H2,1-3H3/t22-,24?,25+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.400 | -53.6 | n/a | n/a | n/a | n/a | n/a | 7.5 | 25 |
AbbVie Inc.
| Assay Description
For the assay, compounds were dispensed in assay-ready plates using a three-fold serial dilution from 50 μM to ~850 pM using an Echo 550 Acousti... |
Nat Chem Biol 13: 389-395 (2017)
Article DOI: 10.1038/nchembio.2306
BindingDB Entry DOI: 10.7270/Q2NG4PGD |
More data for this
Ligand-Target Pair | |
Bcl-2-like protein 1
(Homo sapiens (Human)) | BDBM139041
(US8883784, 2)Show SMILES FC(F)(F)S(=O)(=O)c1cc(ccc1N[C@H](CCN1CCOCC1)CSc1ccccc1)S(=O)(=O)NC(=O)c1csc(n1)N1CCc2cccc(C(=O)Nc3nc4ccccc4s3)c2C1 Show InChI InChI=1S/C42H40F3N7O7S5/c43-42(44,45)63(55,56)37-23-30(13-14-34(37)46-28(16-17-51-19-21-59-22-20-51)25-60-29-8-2-1-3-9-29)64(57,58)50-39(54)35-26-61-41(48-35)52-18-15-27-7-6-10-31(32(27)24-52)38(53)49-40-47-33-11-4-5-12-36(33)62-40/h1-14,23,26,28,46H,15-22,24-25H2,(H,50,54)(H,47,49,53)/t28-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of F-Bak binding to GST-tagged BCL-XL (unknown origin) measured after 1 hr by TR-FRET assay |
Citation and Details
Article DOI: 10.1021/acsmedchemlett.1c00162
BindingDB Entry DOI: 10.7270/Q2R78JW5 |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50078160
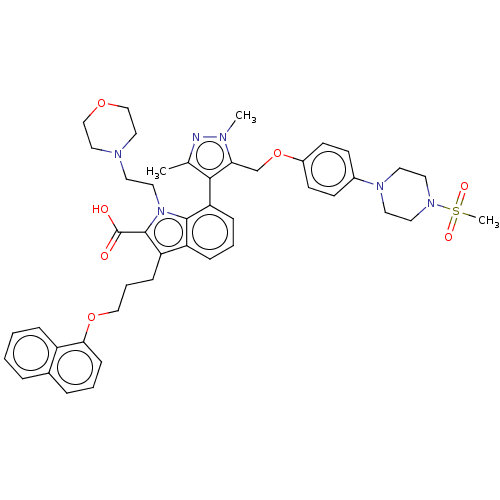
(CHEMBL3417701)Show SMILES Cc1nn(C)c(COc2ccc(cc2)N2CCN(CC2)S(C)(=O)=O)c1-c1cccc2c(CCCOc3cccc4ccccc34)c(C(O)=O)n(CCN3CCOCC3)c12 |(-2.81,-12.36,;-3.79,-13.11,;-3.77,-14.65,;-5.22,-15.15,;-5.58,-16.33,;-6.15,-13.92,;-7.69,-13.93,;-8.44,-15.28,;-9.98,-15.29,;-10.74,-16.63,;-12.28,-16.64,;-13.06,-15.32,;-12.3,-13.98,;-10.76,-13.96,;-14.6,-15.33,;-15.37,-16.67,;-16.91,-16.68,;-17.68,-15.35,;-16.92,-14.01,;-15.38,-14,;-19.23,-15.36,;-19.85,-14.3,;-19.83,-16.43,;-20.46,-15.37,;-5.26,-12.68,;-5.74,-11.22,;-7.27,-10.9,;-7.75,-9.44,;-6.72,-8.27,;-5.21,-8.6,;-3.97,-7.71,;-3.98,-6.17,;-2.65,-5.4,;-2.66,-3.85,;-1.33,-3.08,;-1.33,-1.54,;-2.68,-.77,;-2.68,.77,;-1.33,1.54,;,.77,;1.33,1.54,;2.66,.77,;2.66,-.77,;1.33,-1.54,;,-.77,;-2.74,-8.57,;-1.29,-8.05,;-.35,-8.85,;-1.06,-6.84,;-3.2,-10.04,;-2.28,-11.27,;-.75,-11.09,;.17,-12.33,;1.7,-12.15,;2.62,-13.39,;2.01,-14.8,;.48,-14.98,;-.44,-13.74,;-4.73,-10.05,)| Show InChI InChI=1S/C45H52N6O7S/c1-32-42(40(47(2)46-32)31-58-35-18-16-34(17-19-35)49-21-23-50(24-22-49)59(3,54)55)39-13-7-12-37-38(14-8-28-57-41-15-6-10-33-9-4-5-11-36(33)41)44(45(52)53)51(43(37)39)25-20-48-26-29-56-30-27-48/h4-7,9-13,15-19H,8,14,20-31H2,1-3H3,(H,52,53) | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Displacement of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide) from MCL1 (unknown origin) after 1 hr by TR-FRET assay |
J Med Chem 58: 2180-94 (2015)
Article DOI: 10.1021/jm501258m
BindingDB Entry DOI: 10.7270/Q2HX1FC8 |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50078163

(CHEMBL3417704)Show SMILES CN(C)S(=O)(=O)N1CCN(CC1)c1ccc(OCc2c(c(C)nn2C)-c2cccc3c(CCCOc4cccc5ccccc45)c(C(O)=O)n(CCN4CCOCC4)c23)cc1 |(-21.22,-16.71,;-19.99,-16.7,;-19.36,-17.76,;-19.23,-15.36,;-19.85,-14.3,;-20.46,-15.37,;-17.68,-15.35,;-16.91,-16.68,;-15.37,-16.67,;-14.6,-15.33,;-15.38,-14,;-16.92,-14.01,;-13.06,-15.32,;-12.28,-16.64,;-10.74,-16.63,;-9.98,-15.29,;-8.44,-15.28,;-7.69,-13.93,;-6.15,-13.92,;-5.26,-12.68,;-3.79,-13.11,;-2.81,-12.36,;-3.77,-14.65,;-5.22,-15.15,;-5.58,-16.33,;-5.74,-11.22,;-7.27,-10.9,;-7.75,-9.44,;-6.72,-8.27,;-5.21,-8.6,;-3.97,-7.71,;-3.98,-6.17,;-2.65,-5.4,;-2.66,-3.85,;-1.33,-3.08,;-1.33,-1.54,;-2.68,-.77,;-2.68,.77,;-1.33,1.54,;,.77,;1.33,1.54,;2.66,.77,;2.66,-.77,;1.33,-1.54,;,-.77,;-2.74,-8.57,;-1.29,-8.05,;-.35,-8.85,;-1.06,-6.84,;-3.2,-10.04,;-2.28,-11.27,;-.75,-11.09,;.17,-12.33,;1.7,-12.15,;2.62,-13.39,;2.01,-14.8,;.48,-14.98,;-.44,-13.74,;-4.73,-10.05,;-10.76,-13.96,;-12.3,-13.98,)| Show InChI InChI=1S/C46H55N7O7S/c1-33-43(41(49(4)47-33)32-60-36-19-17-35(18-20-36)51-22-24-52(25-23-51)61(56,57)48(2)3)40-14-8-13-38-39(15-9-29-59-42-16-7-11-34-10-5-6-12-37(34)42)45(46(54)55)53(44(38)40)26-21-50-27-30-58-31-28-50/h5-8,10-14,16-20H,9,15,21-32H2,1-4H3,(H,54,55) | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Displacement of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide) from MCL1 (unknown origin) after 1 hr by TR-FRET assay |
J Med Chem 58: 2180-94 (2015)
Article DOI: 10.1021/jm501258m
BindingDB Entry DOI: 10.7270/Q2HX1FC8 |
More data for this
Ligand-Target Pair | |
Polycomb protein EED
(Homo sapiens (Human)) | BDBM50235643
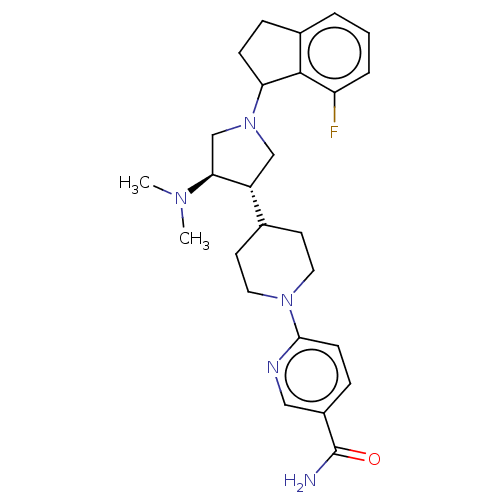
(CHEMBL4076017)Show SMILES CN(C)[C@H]1CN(C[C@@H]1C1CCN(CC1)c1ccc(cn1)C(N)=O)C1CCc2cccc(F)c12 |r| Show InChI InChI=1S/C26H34FN5O/c1-30(2)23-16-32(22-8-6-18-4-3-5-21(27)25(18)22)15-20(23)17-10-12-31(13-11-17)24-9-7-19(14-29-24)26(28)33/h3-5,7,9,14,17,20,22-23H,6,8,10-13,15-16H2,1-2H3,(H2,28,33)/t20-,22?,23+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
In vitro displacement of [3H]-LY 278584 from rat cerebral cortex 5-hydroxytryptamine 3 receptor |
Bioorg Med Chem Lett 27: 1576-1583 (2017)
Article DOI: 10.1016/j.bmcl.2017.02.030
BindingDB Entry DOI: 10.7270/Q22F7QQG |
More data for this
Ligand-Target Pair | |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50078164
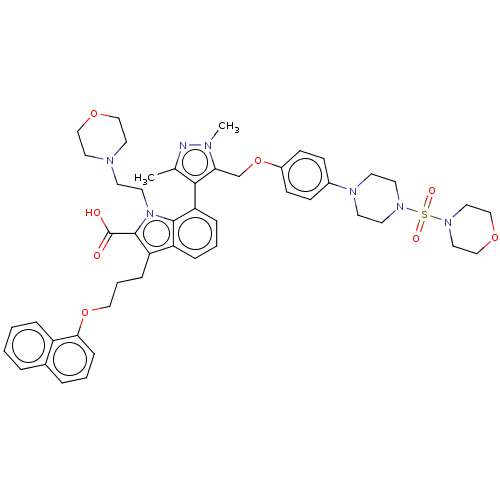
(CHEMBL3417705)Show SMILES Cc1nn(C)c(COc2ccc(cc2)N2CCN(CC2)S(=O)(=O)N2CCOCC2)c1-c1cccc2c(CCCOc3cccc4ccccc34)c(C(O)=O)n(CCN3CCOCC3)c12 |(-2.81,-12.36,;-3.79,-13.11,;-3.77,-14.65,;-5.22,-15.15,;-5.58,-16.33,;-6.15,-13.92,;-7.69,-13.93,;-8.44,-15.28,;-9.98,-15.29,;-10.74,-16.63,;-12.28,-16.64,;-13.06,-15.32,;-12.3,-13.98,;-10.76,-13.96,;-14.6,-15.33,;-15.37,-16.67,;-16.91,-16.68,;-17.68,-15.35,;-16.92,-14.01,;-15.38,-14,;-19.23,-15.36,;-19.85,-14.3,;-18.62,-14.29,;-19.99,-16.7,;-19.21,-18.03,;-19.97,-19.37,;-21.51,-19.38,;-22.29,-18.05,;-21.53,-16.71,;-5.26,-12.68,;-5.74,-11.22,;-7.27,-10.9,;-7.75,-9.44,;-6.72,-8.27,;-5.21,-8.6,;-3.97,-7.71,;-3.98,-6.17,;-2.65,-5.4,;-2.66,-3.85,;-1.33,-3.08,;-1.33,-1.54,;-2.68,-.77,;-2.68,.77,;-1.33,1.54,;,.77,;1.33,1.54,;2.66,.77,;2.66,-.77,;1.33,-1.54,;,-.77,;-2.74,-8.57,;-1.29,-8.05,;-.35,-8.85,;-1.06,-6.84,;-3.2,-10.04,;-2.28,-11.27,;-.75,-11.09,;.17,-12.33,;1.7,-12.15,;2.62,-13.39,;2.01,-14.8,;.48,-14.98,;-.44,-13.74,;-4.73,-10.05,)| Show InChI InChI=1S/C48H57N7O8S/c1-35-45(43(50(2)49-35)34-63-38-17-15-37(16-18-38)52-20-22-53(23-21-52)64(58,59)54-27-32-61-33-28-54)42-12-6-11-40-41(13-7-29-62-44-14-5-9-36-8-3-4-10-39(36)44)47(48(56)57)55(46(40)42)24-19-51-25-30-60-31-26-51/h3-6,8-12,14-18H,7,13,19-34H2,1-2H3,(H,56,57) | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Displacement of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide) from MCL1 (unknown origin) after 1 hr by TR-FRET assay |
J Med Chem 58: 2180-94 (2015)
Article DOI: 10.1021/jm501258m
BindingDB Entry DOI: 10.7270/Q2HX1FC8 |
More data for this
Ligand-Target Pair | |
Polycomb protein EED
(Homo sapiens (Human)) | BDBM223987

(A-395 (5) | rac-(3R,4S)-1-(7-fluoro-2,3-dihydro-1H...)Show SMILES CN(C)[C@H]1CN(C[C@@H]1c1ccc(cc1)N1CCN(CC1)S(C)(=O)=O)C1CCc2cccc(F)c12 |r,w:24.26| Show InChI InChI=1S/C26H35FN4O2S/c1-28(2)25-18-30(24-12-9-20-5-4-6-23(27)26(20)24)17-22(25)19-7-10-21(11-8-19)29-13-15-31(16-14-29)34(3,32)33/h4-8,10-11,22,24-25H,9,12-18H2,1-3H3/t22-,24?,25+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| 0.5 | -53.1 | n/a | n/a | n/a | n/a | n/a | 7.5 | 25 |
AbbVie Inc.
| Assay Description
For the assay, compounds were dispensed in assay-ready plates using a three-fold serial dilution from 50 μM to ~850 pM using an Echo 550 Acousti... |
Nat Chem Biol 13: 389-395 (2017)
Article DOI: 10.1038/nchembio.2306
BindingDB Entry DOI: 10.7270/Q2NG4PGD |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18188
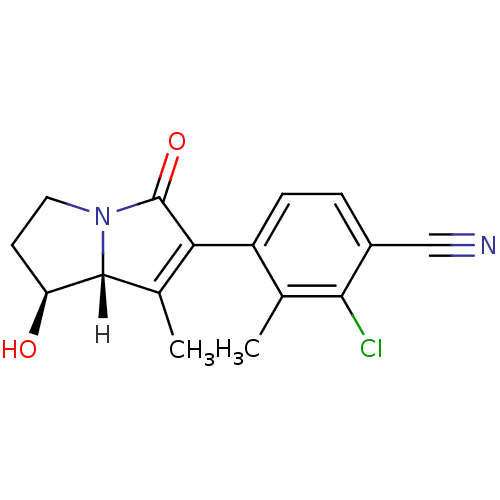
(4-[(1S,7aR)-1-hydroxy-7-methyl-5-oxo-2,3,5,7a-tetr...)Show SMILES [H][C@@]12[C@@H](O)CCN1C(=O)C(=C2C)c1ccc(C#N)c(Cl)c1C |r,c:10| Show InChI InChI=1S/C16H15ClN2O2/c1-8-11(4-3-10(7-18)14(8)17)13-9(2)15-12(20)5-6-19(15)16(13)21/h3-4,12,15,20H,5-6H2,1-2H3/t12-,15+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 0.5 | -52.6 | n/a | n/a | 2 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 50: 3015-3025 (2007)
Article DOI: 10.1021/jm070312d
BindingDB Entry DOI: 10.7270/Q24F1P1F |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50368409
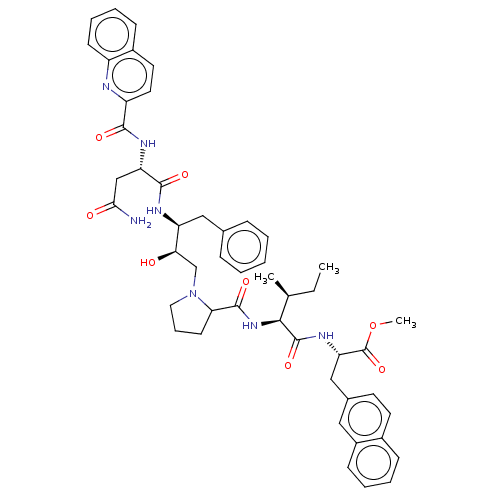
(CHEMBL1790877 | CHEMBL3349488)Show SMILES [H][C@@](O)(CN1CCCC1C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](Cc1ccc2ccccc2c1)C(=O)OC)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)c1ccc2ccccc2n1 Show InChI InChI=1S/C49H57N7O8/c1-4-30(2)44(48(62)54-40(49(63)64-3)27-32-20-21-33-15-8-9-17-35(33)25-32)55-47(61)41-19-12-24-56(41)29-42(57)38(26-31-13-6-5-7-14-31)52-46(60)39(28-43(50)58)53-45(59)37-23-22-34-16-10-11-18-36(34)51-37/h5-11,13-18,20-23,25,30,38-42,44,57H,4,12,19,24,26-29H2,1-3H3,(H2,50,58)(H,52,60)(H,53,59)(H,54,62)(H,55,61)/t30-,38+,39+,40+,41?,42-,44+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Binding affinity to HIV protease |
J Med Chem 35: 3803-12 (1992)
BindingDB Entry DOI: 10.7270/Q2348M0D |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50368429
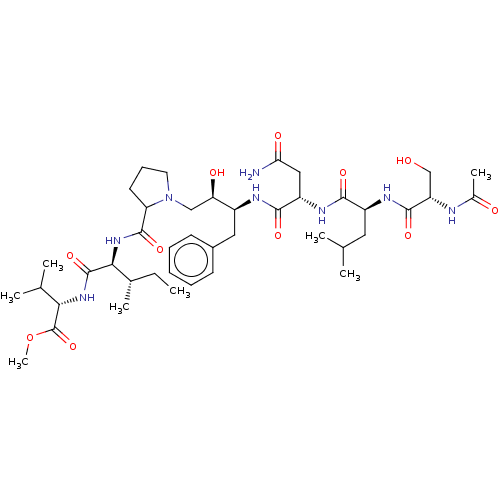
(CHEMBL1790875 | CHEMBL3349486)Show SMILES [H][C@@](O)(CN1CCCC1C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C(C)C)C(=O)OC)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(C)=O Show InChI InChI=1S/C42H68N8O11/c1-9-25(6)36(41(59)48-35(24(4)5)42(60)61-8)49-40(58)32-16-13-17-50(32)21-33(53)28(19-27-14-11-10-12-15-27)45-38(56)30(20-34(43)54)47-37(55)29(18-23(2)3)46-39(57)31(22-51)44-26(7)52/h10-12,14-15,23-25,28-33,35-36,51,53H,9,13,16-22H2,1-8H3,(H2,43,54)(H,44,52)(H,45,56)(H,46,57)(H,47,55)(H,48,59)(H,49,58)/t25-,28+,29+,30+,31+,32?,33-,35+,36+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Binding affinity to HIV protease |
J Med Chem 35: 3803-12 (1992)
BindingDB Entry DOI: 10.7270/Q2348M0D |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50010495

(Acetyl-Ser-Leu-Asn-Phe-[CH(OH)CH2N]Pro-Ile-Val-OMe...)Show SMILES CC[C@H](C)[C@H](NC(=O)[C@@H]1CCCN1CC(O)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(C)=O)C(=O)N[C@@H](C(C)C)C(=O)OC Show InChI InChI=1S/C42H68N8O11/c1-9-25(6)36(41(59)48-35(24(4)5)42(60)61-8)49-40(58)32-16-13-17-50(32)21-33(53)28(19-27-14-11-10-12-15-27)45-38(56)30(20-34(43)54)47-37(55)29(18-23(2)3)46-39(57)31(22-51)44-26(7)52/h10-12,14-15,23-25,28-33,35-36,51,53H,9,13,16-22H2,1-8H3,(H2,43,54)(H,44,52)(H,45,56)(H,46,57)(H,47,55)(H,48,59)(H,49,58)/t25-,28-,29-,30-,31-,32-,33?,35-,36-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
In vitro binding affinity of the compound against HIV protease was measured |
J Med Chem 34: 1222-5 (1991)
BindingDB Entry DOI: 10.7270/Q2057DWM |
More data for this
Ligand-Target Pair | |
Polycomb protein EED
(Homo sapiens (Human)) | BDBM50235658
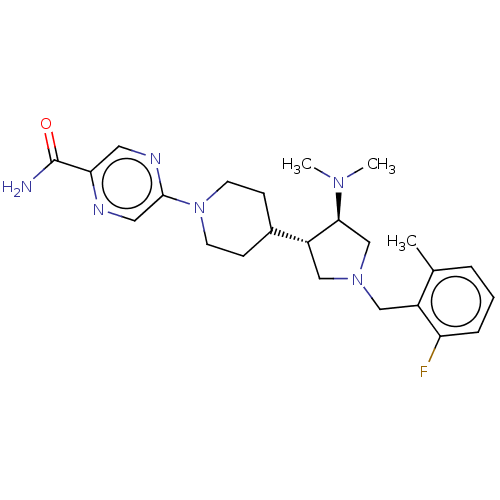
(CHEMBL4073166)Show SMILES CN(C)[C@H]1CN(Cc2c(C)cccc2F)C[C@@H]1C1CCN(CC1)c1cnc(cn1)C(N)=O |r| Show InChI InChI=1S/C24H33FN6O/c1-16-5-4-6-20(25)18(16)13-30-14-19(22(15-30)29(2)3)17-7-9-31(10-8-17)23-12-27-21(11-28-23)24(26)32/h4-6,11-12,17,19,22H,7-10,13-15H2,1-3H3,(H2,26,32)/t19-,22+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
In vitro displacement of [3H]-LY 278584 from rat cerebral cortex 5-hydroxytryptamine 3 receptor |
Bioorg Med Chem Lett 27: 1576-1583 (2017)
Article DOI: 10.1016/j.bmcl.2017.02.030
BindingDB Entry DOI: 10.7270/Q22F7QQG |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50368422
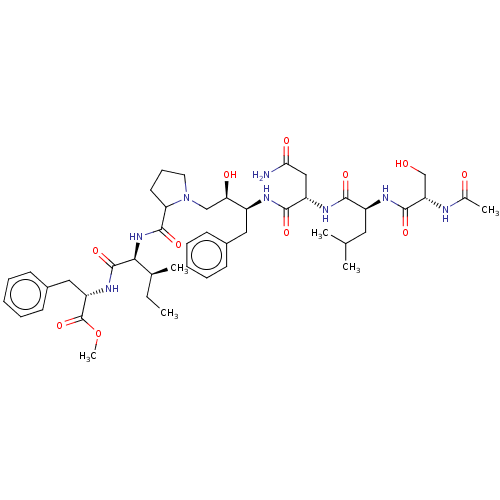
(CHEMBL1790871 | CHEMBL3349482)Show SMILES [H][C@@](O)(CN1CCCC1C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](Cc1ccccc1)C(=O)OC)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(C)=O Show InChI InChI=1S/C46H68N8O11/c1-7-28(4)40(45(63)52-35(46(64)65-6)23-31-17-12-9-13-18-31)53-44(62)37-19-14-20-54(37)25-38(57)32(22-30-15-10-8-11-16-30)49-42(60)34(24-39(47)58)51-41(59)33(21-27(2)3)50-43(61)36(26-55)48-29(5)56/h8-13,15-18,27-28,32-38,40,55,57H,7,14,19-26H2,1-6H3,(H2,47,58)(H,48,56)(H,49,60)(H,50,61)(H,51,59)(H,52,63)(H,53,62)/t28-,32+,33+,34+,35+,36+,37?,38-,40+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.670 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Binding affinity to HIV protease |
J Med Chem 35: 3803-12 (1992)
BindingDB Entry DOI: 10.7270/Q2348M0D |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18183
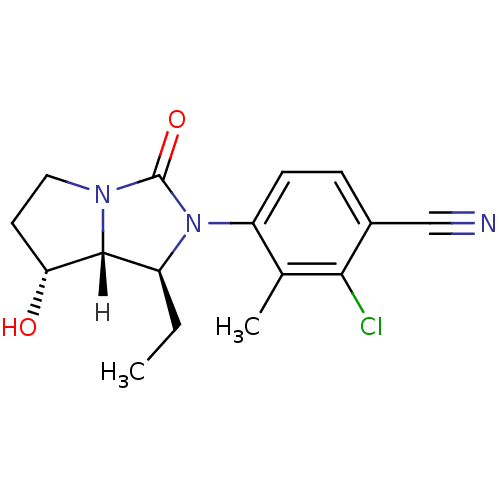
(4-[(1S,7R,7aR)-1-ethyl-7-hydroxy-3-oxo-hexahydro-1...)Show SMILES [H][C@@]12[C@H](O)CCN1C(=O)N([C@H]2CC)c1ccc(C#N)c(Cl)c1C |r| Show InChI InChI=1S/C16H18ClN3O2/c1-3-11-15-13(21)6-7-19(15)16(22)20(11)12-5-4-10(8-18)14(17)9(12)2/h4-5,11,13,15,21H,3,6-7H2,1-2H3/t11-,13+,15+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.700 | -51.7 | n/a | n/a | 2.60 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 50: 3015-3025 (2007)
Article DOI: 10.1021/jm070312d
BindingDB Entry DOI: 10.7270/Q24F1P1F |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205111
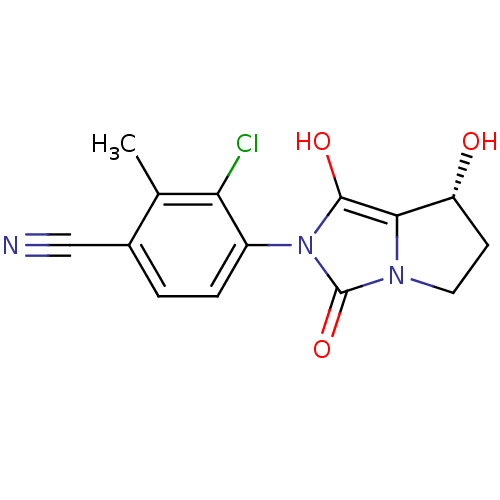
(3-chloro-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydr...)Show SMILES Cc1c(Cl)c(ccc1C#N)-n1c(O)c2[C@H](O)CCn2c1=O |wU:14.15,(22.97,-37.01,;22.21,-35.67,;20.67,-35.65,;19.89,-36.98,;19.91,-34.31,;20.69,-32.99,;22.22,-32.99,;22.99,-34.34,;24.53,-34.34,;26.08,-34.34,;18.37,-34.31,;17.48,-33.06,;17.96,-31.6,;16.01,-33.53,;14.55,-33.04,;14.08,-31.57,;13.63,-34.28,;14.54,-35.53,;16,-35.06,;17.46,-35.55,;17.93,-37.02,)| Show InChI InChI=1S/C14H12ClN3O3/c1-7-8(6-16)2-3-9(11(7)15)18-13(20)12-10(19)4-5-17(12)14(18)21/h2-3,10,19-20H,4-5H2,1H3/t10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Polycomb protein EED
(Homo sapiens (Human)) | BDBM50235631

(CHEMBL4060827)Show SMILES CN(C)[C@H]1CN(C[C@@H]1c1ccc(cc1)N1CCN(CC1)C(C)=O)C1CCc2cccc(F)c12 |r| Show InChI InChI=1S/C27H35FN4O/c1-19(33)30-13-15-31(16-14-30)22-10-7-20(8-11-22)23-17-32(18-26(23)29(2)3)25-12-9-21-5-4-6-24(28)27(21)25/h4-8,10-11,23,25-26H,9,12-18H2,1-3H3/t23-,25?,26+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Inhibition of ADP-induced platelet aggregation in human platelet-rich plasma |
J Med Chem 60: 8321-8335 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00576
BindingDB Entry DOI: 10.7270/Q2TM7D8G |
More data for this
Ligand-Target Pair | |
Polycomb protein EED
(Homo sapiens (Human)) | BDBM50235644
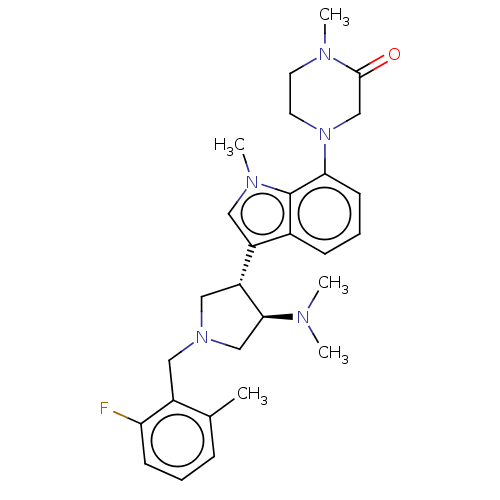
(CHEMBL4065766)Show SMILES CN(C)[C@H]1CN(Cc2c(C)cccc2F)C[C@@H]1c1cn(C)c2c(cccc12)N1CCN(C)C(=O)C1 |r| Show InChI InChI=1S/C28H36FN5O/c1-19-8-6-10-24(29)21(19)15-33-16-23(26(17-33)30(2)3)22-14-32(5)28-20(22)9-7-11-25(28)34-13-12-31(4)27(35)18-34/h6-11,14,23,26H,12-13,15-18H2,1-5H3/t23-,26+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
In vitro displacement of [3H]-LY 278584 from rat cerebral cortex 5-hydroxytryptamine 3 receptor |
Bioorg Med Chem Lett 27: 1576-1583 (2017)
Article DOI: 10.1016/j.bmcl.2017.02.030
BindingDB Entry DOI: 10.7270/Q22F7QQG |
More data for this
Ligand-Target Pair | |
Cathepsin D
(Homo sapiens (Human)) | BDBM50333945
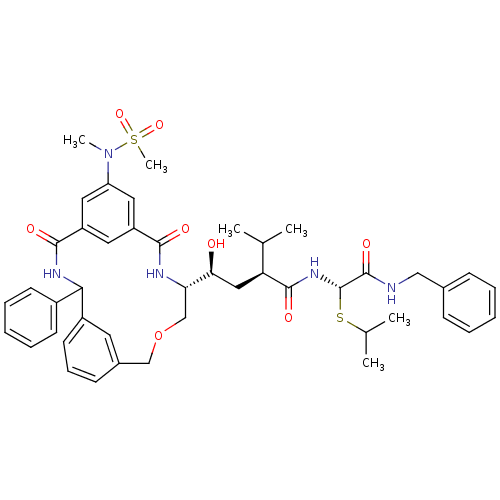
((S)-N-((S)-Benzylcarbamoyl-isopropylsulfanyl-methy...)Show SMILES CC(C)S[C@H](NC(=O)[C@@H](C[C@H](O)[C@@H]1COCc2cccc(c2)C(NC(=O)c2cc(cc(c2)C(=O)N1)N(C)S(C)(=O)=O)c1ccccc1)C(C)C)C(=O)NCc1ccccc1 |r| Show InChI InChI=1S/C45H55N5O8S2/c1-28(2)37(43(54)49-45(59-29(3)4)44(55)46-25-30-14-9-7-10-15-30)24-39(51)38-27-58-26-31-16-13-19-33(20-31)40(32-17-11-8-12-18-32)48-42(53)35-21-34(41(52)47-38)22-36(23-35)50(5)60(6,56)57/h7-23,28-29,37-40,45,51H,24-27H2,1-6H3,(H,46,55)(H,47,52)(H,48,53)(H,49,54)/t37-,38-,39-,40?,45-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Medivir AB
Curated by ChEMBL
| Assay Description
Inhibition of cathepsin D |
Bioorg Med Chem Lett 21: 358-62 (2010)
Article DOI: 10.1016/j.bmcl.2010.10.140
BindingDB Entry DOI: 10.7270/Q2HM58RJ |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50368423

(CHEMBL1790881 | CHEMBL3349492)Show SMILES [H][C@@](O)(CN1CCCC1C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](Cc1ccccc1)C(=O)NC)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)c1ccc2ccccc2n1 Show InChI InChI=1S/C45H56N8O7/c1-4-28(2)40(45(60)51-35(41(56)47-3)25-30-16-9-6-10-17-30)52-44(59)37-20-13-23-53(37)27-38(54)34(24-29-14-7-5-8-15-29)49-43(58)36(26-39(46)55)50-42(57)33-22-21-31-18-11-12-19-32(31)48-33/h5-12,14-19,21-22,28,34-38,40,54H,4,13,20,23-27H2,1-3H3,(H2,46,55)(H,47,56)(H,49,58)(H,50,57)(H,51,60)(H,52,59)/t28-,34+,35+,36+,37?,38-,40+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Binding affinity to HIV protease |
J Med Chem 35: 3803-12 (1992)
BindingDB Entry DOI: 10.7270/Q2348M0D |
More data for this
Ligand-Target Pair | |
Bromodomain-containing protein 4
(Homo sapiens (Human)) | BDBM50265666
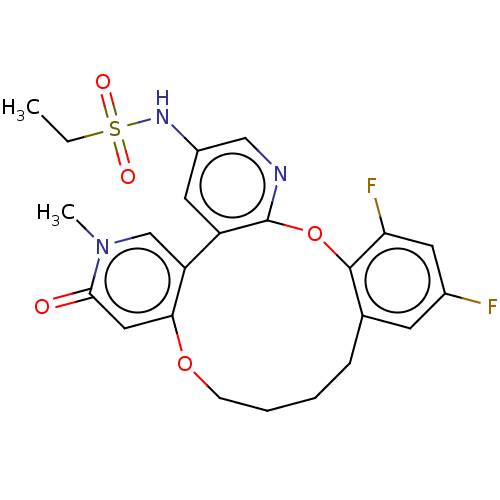
(CHEMBL4068431)Show SMILES CCS(=O)(=O)Nc1cnc2Oc3c(F)cc(F)cc3CCCCOc3cc(=O)n(C)cc3-c2c1 Show InChI InChI=1S/C23H23F2N3O5S/c1-3-34(30,31)27-16-10-17-18-13-28(2)21(29)11-20(18)32-7-5-4-6-14-8-15(24)9-19(25)22(14)33-23(17)26-12-16/h8-13,27H,3-7H2,1-2H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human N-terminal His6-tagged BRD4 BD1-BD2 (57 to 550 residues) expressed in EScherichia coli BL21(DE3) after 1 hr by Alexa-647-conjugat... |
J Med Chem 60: 3828-3850 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00017
BindingDB Entry DOI: 10.7270/Q2BR8VPN |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18177
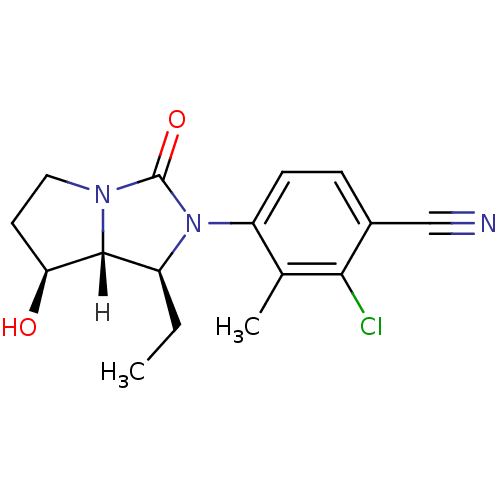
(4-[(1S,7S,7aR)-1-ethyl-7-hydroxy-3-oxo-hexahydro-1...)Show SMILES [H][C@@]12[C@@H](O)CCN1C(=O)N([C@H]2CC)c1ccc(C#N)c(Cl)c1C |r| Show InChI InChI=1S/C16H18ClN3O2/c1-3-11-15-13(21)6-7-19(15)16(22)20(11)12-5-4-10(8-18)14(17)9(12)2/h4-5,11,13,15,21H,3,6-7H2,1-2H3/t11-,13-,15+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.900 | -51.1 | n/a | n/a | 1.80 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 50: 3015-3025 (2007)
Article DOI: 10.1021/jm070312d
BindingDB Entry DOI: 10.7270/Q24F1P1F |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18178
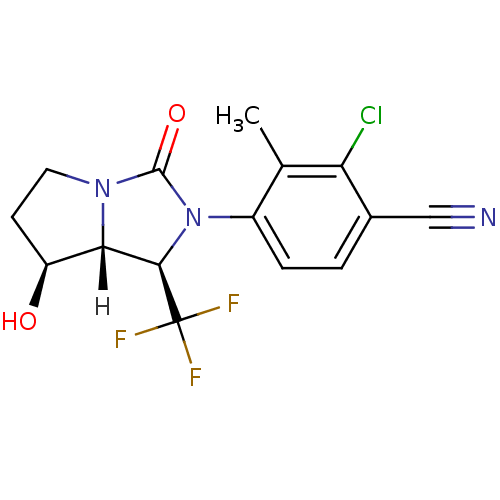
(4-[(1R,7S,7aR)-7-hydroxy-3-oxo-1-(trifluoromethyl)...)Show SMILES [H][C@@]12[C@@H](O)CCN1C(=O)N([C@H]2C(F)(F)F)c1ccc(C#N)c(Cl)c1C |r| Show InChI InChI=1S/C15H13ClF3N3O2/c1-7-9(3-2-8(6-20)11(7)16)22-13(15(17,18)19)12-10(23)4-5-21(12)14(22)24/h2-3,10,12-13,23H,4-5H2,1H3/t10-,12-,13+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.900 | -51.1 | n/a | n/a | 2.5 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 50: 3015-3025 (2007)
Article DOI: 10.1021/jm070312d
BindingDB Entry DOI: 10.7270/Q24F1P1F |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 8
(Homo sapiens (Human)) | BDBM15956
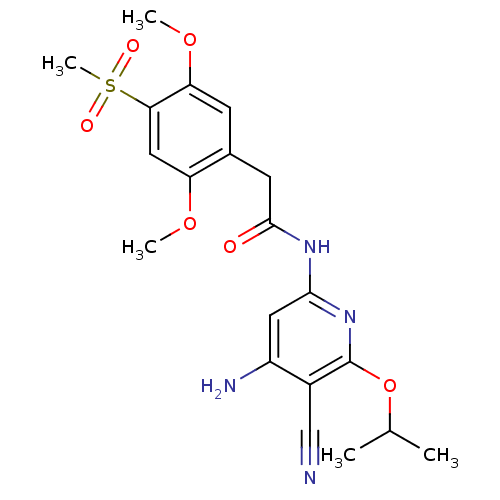
(Aminopyridine-Based Inhibitor 18b | N-(4-Amino-5-c...)Show SMILES COc1cc(c(OC)cc1CC(=O)Nc1cc(N)c(C#N)c(OC(C)C)n1)S(C)(=O)=O Show InChI InChI=1S/C20H24N4O6S/c1-11(2)30-20-13(10-21)14(22)8-18(24-20)23-19(25)7-12-6-16(29-4)17(31(5,26)27)9-15(12)28-3/h6,8-9,11H,7H2,1-5H3,(H3,22,23,24,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
| Assay Description
Ser/Thr-kinase selectivity assays were performed using a radioactive FlashPlate-based assay platform. Substrate incorporated radioactivity was counte... |
J Med Chem 49: 3563-80 (2006)
Article DOI: 10.1021/jm060199b
BindingDB Entry DOI: 10.7270/Q2P26WDX |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18162
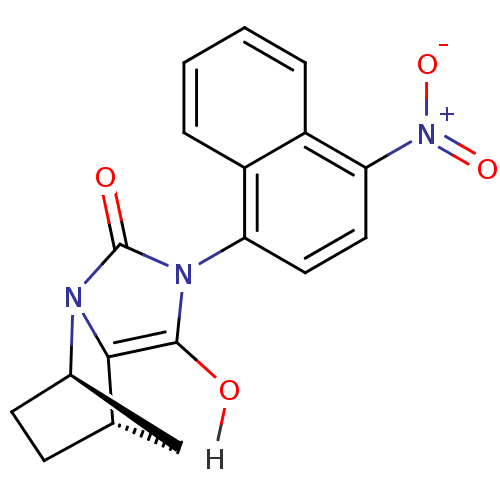
((1R,6R,7S)-4-(4-nitronaphthalen-1-yl)-2,4-diazatri...)Show SMILES Oc1c2[C@H]3CC[C@H](C3)n2c(=O)n1-c1ccc([N+]([O-])=O)c2ccccc12 |r,wU:6.6,3.7,(12.2,-7.28,;12.6,-8.77,;11.56,-9.96,;10.02,-9.96,;9.25,-11.29,;10.02,-12.62,;11.56,-12.62,;10.02,-11.73,;12.33,-11.29,;13.87,-10.92,;15.03,-11.93,;14,-9.4,;15.49,-9,;16.07,-7.57,;17.59,-7.36,;18.54,-8.57,;20.06,-8.36,;20.83,-9.69,;20.64,-6.93,;17.96,-10,;18.91,-11.21,;18.34,-12.64,;16.81,-12.86,;15.86,-11.64,;16.44,-10.22,)| Show InChI InChI=1S/C18H15N3O4/c22-17-16-10-5-6-11(9-10)19(16)18(23)20(17)14-7-8-15(21(24)25)13-4-2-1-3-12(13)14/h1-4,7-8,10-11,22H,5-6,9H2/t10-,11+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | -50.9 | n/a | n/a | 385 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Company
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 49: 7596-9 (2006)
Article DOI: 10.1021/jm061101w
BindingDB Entry DOI: 10.7270/Q2862DQ9 |
More data for this
Ligand-Target Pair | |
Polycomb protein EED
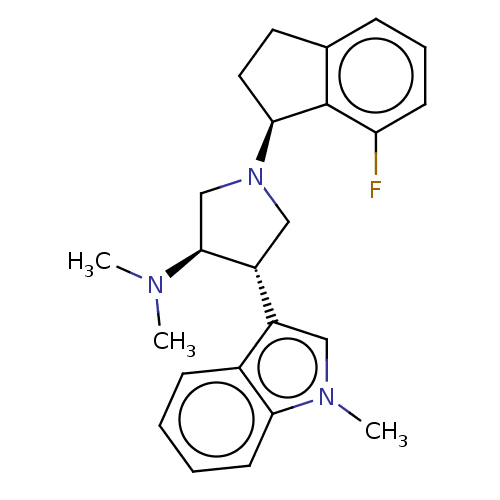
(Homo sapiens (Human)) | BDBM223986
((3R,4S)-1-[(1S)-7-fluoroindan-1-yl]-N,N-dimethyl-4...)Show SMILES CN(C)[C@H]1CN(C[C@@H]1c1cn(C)c2ccccc12)[C@H]1CCc2cccc(F)c12 |r| Show InChI InChI=1S/C24H28FN3/c1-26(2)23-15-28(22-12-11-16-7-6-9-20(25)24(16)22)14-19(23)18-13-27(3)21-10-5-4-8-17(18)21/h4-10,13,19,22-23H,11-12,14-15H2,1-3H3/t19-,22+,23+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Inhibition of ADP-induced platelet aggregation in human platelet-rich plasma |
J Med Chem 60: 8321-8335 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00576
BindingDB Entry DOI: 10.7270/Q2TM7D8G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Bromodomain-containing protein 2
(Homo sapiens (Human)) | BDBM220447

(US10633379, Compound X | US9296741, 36)Show SMILES CCS(=O)(=O)Nc1ccc(Oc2ccc(F)cc2F)c(c1)-c1cn(C)c(=O)c2[nH]ccc12 Show InChI InChI=1S/C22H19F2N3O4S/c1-3-32(29,30)26-14-5-7-19(31-20-6-4-13(23)10-18(20)24)16(11-14)17-12-27(2)22(28)21-15(17)8-9-25-21/h4-12,25-26H,3H2,1-2H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Binding affinity to BRD2 BD1 to BD2 (G73 to A560 residues) (unknown origin) |
J Med Chem 60: 8369-8384 (2017)
Article DOI: 10.1021/acs.jmedchem.7b00746
BindingDB Entry DOI: 10.7270/Q2251MB8 |
More data for this
Ligand-Target Pair | |
Polycomb protein EED
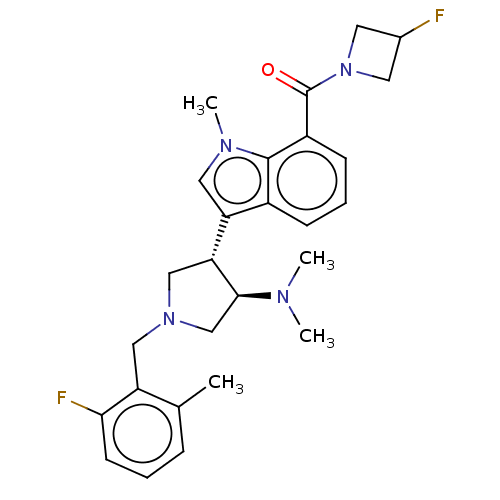
(Homo sapiens (Human)) | BDBM50235632
(CHEMBL4077363)Show SMILES CN(C)[C@H]1CN(Cc2c(C)cccc2F)C[C@@H]1c1cn(C)c2c(cccc12)C(=O)N1CC(F)C1 |r| Show InChI InChI=1S/C27H32F2N4O/c1-17-7-5-10-24(29)21(17)14-32-15-23(25(16-32)30(2)3)22-13-31(4)26-19(22)8-6-9-20(26)27(34)33-11-18(28)12-33/h5-10,13,18,23,25H,11-12,14-16H2,1-4H3/t23-,25+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
In vitro displacement of [3H]-LY 278584 from rat cerebral cortex 5-hydroxytryptamine 3 receptor |
Bioorg Med Chem Lett 27: 1576-1583 (2017)
Article DOI: 10.1016/j.bmcl.2017.02.030
BindingDB Entry DOI: 10.7270/Q22F7QQG |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50230041

(CHEMBL3349496)Show SMILES [H][C@@](O)(CN1CCC[C@H]1C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](Cc1ccccc1)C(=O)OC)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C43H56N6O9/c1-4-28(2)38(41(54)46-34(42(55)57-3)24-30-17-10-6-11-18-30)48-40(53)35-21-14-22-49(35)26-36(50)32(23-29-15-8-5-9-16-29)45-39(52)33(25-37(44)51)47-43(56)58-27-31-19-12-7-13-20-31/h5-13,15-20,28,32-36,38,50H,4,14,21-27H2,1-3H3,(H2,44,51)(H,45,52)(H,46,54)(H,47,56)(H,48,53)/t28-,32-,33-,34-,35-,36+,38-/m0/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Binding affinity to HIV protease |
J Med Chem 35: 3803-12 (1992)
BindingDB Entry DOI: 10.7270/Q2348M0D |
More data for this
Ligand-Target Pair | |
Polycomb protein EED
(Homo sapiens (Human)) | BDBM223986

((3R,4S)-1-[(1S)-7-fluoroindan-1-yl]-N,N-dimethyl-4...)Show SMILES CN(C)[C@H]1CN(C[C@@H]1c1cn(C)c2ccccc12)[C@H]1CCc2cccc(F)c12 |r| Show InChI InChI=1S/C24H28FN3/c1-26(2)23-15-28(22-12-11-16-7-6-9-20(25)24(16)22)14-19(23)18-13-27(3)21-10-5-4-8-17(18)21/h4-10,13,19,22-23H,11-12,14-15H2,1-3H3/t19-,22+,23+/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 1 | -51.4 | n/a | n/a | n/a | n/a | n/a | 7.5 | 25 |
AbbVie Inc.
| Assay Description
For the assay, compounds were dispensed in assay-ready plates using a three-fold serial dilution from 50 μM to ~850 pM using an Echo 550 Acousti... |
Nat Chem Biol 13: 389-395 (2017)
Article DOI: 10.1038/nchembio.2306
BindingDB Entry DOI: 10.7270/Q2NG4PGD |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Androgen receptor
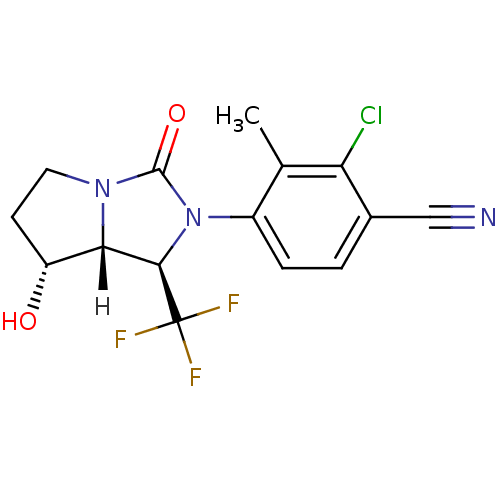
(Homo sapiens (Human)) | BDBM18184
(4-[(1R,7R,7aR)-7-hydroxy-3-oxo-1-(trifluoromethyl)...)Show SMILES [H][C@@]12[C@H](O)CCN1C(=O)N([C@H]2C(F)(F)F)c1ccc(C#N)c(Cl)c1C |r| Show InChI InChI=1S/C15H13ClF3N3O2/c1-7-9(3-2-8(6-20)11(7)16)22-13(15(17,18)19)12-10(23)4-5-21(12)14(22)24/h2-3,10,12-13,23H,4-5H2,1H3/t10-,12+,13-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1 | -50.9 | n/a | n/a | 2.90 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 50: 3015-3025 (2007)
Article DOI: 10.1021/jm070312d
BindingDB Entry DOI: 10.7270/Q24F1P1F |
More data for this
Ligand-Target Pair | |
Caspase-3
(Homo sapiens (Human)) | BDBM10323

((S)-1-Benzyl-5-{1-[2-(phenoxymethyl)pyrrolidinyl]s...)Show SMILES O=C1N(Cc2ccccc2)c2ccc(cc2C1=O)S(=O)(=O)N1CCC[C@H]1COc1ccccc1 |r| Show InChI InChI=1S/C26H24N2O5S/c29-25-23-16-22(13-14-24(23)27(26(25)30)17-19-8-3-1-4-9-19)34(31,32)28-15-7-10-20(28)18-33-21-11-5-2-6-12-21/h1-6,8-9,11-14,16,20H,7,10,15,17-18H2/t20-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.20 | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline
| Assay Description
The substrate peptides terminating in AMC/AFC are processed by caspases with or without inhibitors, and the accumulation of AMC/AFC was assessed with... |
J Med Chem 44: 2015-26 (2001)
Article DOI: 10.1021/jm0100537
BindingDB Entry DOI: 10.7270/Q2ZW1J42 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205116
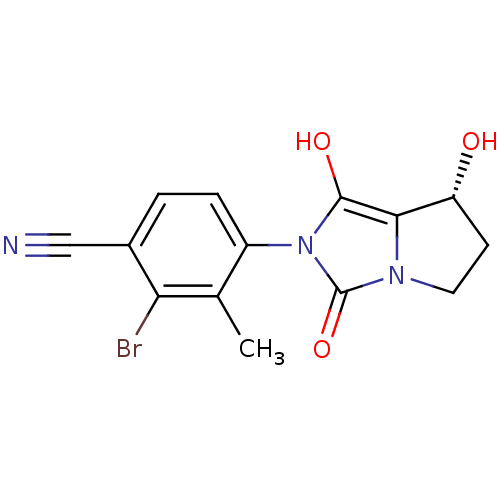
(2-bromo-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydro...)Show SMILES Cc1c(Br)c(ccc1-n1c(O)c2[C@H](O)CCn2c1=O)C#N |wU:12.13,(21.54,-10.86,;22.32,-9.54,;23.86,-9.55,;24.62,-10.89,;24.64,-8.22,;23.87,-6.88,;22.33,-6.87,;21.56,-8.2,;20.02,-8.19,;19.13,-6.94,;19.61,-5.48,;17.66,-7.41,;16.2,-6.92,;15.73,-5.45,;15.28,-8.17,;16.19,-9.42,;17.65,-8.95,;19.11,-9.43,;19.58,-10.9,;26.18,-8.22,;27.72,-8.23,)| Show InChI InChI=1S/C14H12BrN3O3/c1-7-9(3-2-8(6-16)11(7)15)18-13(20)12-10(19)4-5-17(12)14(18)21/h2-3,10,19-20H,4-5H2,1H3/t10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50205114
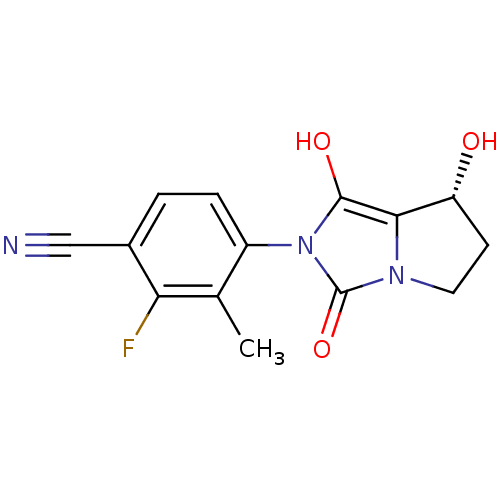
(2-fluoro-4-((7R,7aS)-7-hydroxy-1,3-dioxo-tetrahydr...)Show SMILES Cc1c(F)c(ccc1-n1c(O)c2[C@H](O)CCn2c1=O)C#N |wU:12.13,(21.37,-1.41,;22.15,-.08,;23.69,-.09,;24.45,-1.43,;24.47,1.24,;23.7,2.58,;22.16,2.59,;21.39,1.26,;19.85,1.27,;18.96,2.52,;19.44,3.98,;17.49,2.05,;16.03,2.54,;15.56,4.01,;15.11,1.29,;16.02,.04,;17.48,.51,;18.94,.03,;19.41,-1.44,;26.01,1.24,;27.55,1.23,)| Show InChI InChI=1S/C14H12FN3O3/c1-7-9(3-2-8(6-16)11(7)15)18-13(20)12-10(19)4-5-17(12)14(18)21/h2-3,10,19-20H,4-5H2,1H3/t10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmaceutical Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity to human AR |
Bioorg Med Chem Lett 17: 1860-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.01.076
BindingDB Entry DOI: 10.7270/Q2S46RN1 |
More data for this
Ligand-Target Pair | |
Gag-Pol polyprotein [489-587]
(Human immunodeficiency virus type 1) | BDBM50368425
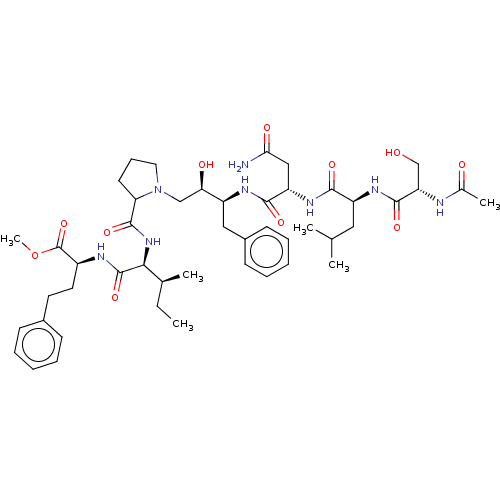
(CHEMBL1790885 | CHEMBL3349495)Show SMILES [H][C@@](O)(CN1CCCC1C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCc1ccccc1)C(=O)OC)[C@H](Cc1ccccc1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(C)=O Show InChI InChI=1S/C47H70N8O11/c1-7-29(4)41(46(64)50-33(47(65)66-6)21-20-31-15-10-8-11-16-31)54-45(63)38-19-14-22-55(38)26-39(58)34(24-32-17-12-9-13-18-32)51-43(61)36(25-40(48)59)53-42(60)35(23-28(2)3)52-44(62)37(27-56)49-30(5)57/h8-13,15-18,28-29,33-39,41,56,58H,7,14,19-27H2,1-6H3,(H2,48,59)(H,49,57)(H,50,64)(H,51,61)(H,52,62)(H,53,60)(H,54,63)/t29-,33+,34+,35+,36+,37+,38?,39-,41+/m1/s1 | PDB
MMDB
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Wisconsin-Madison
Curated by ChEMBL
| Assay Description
Binding affinity to HIV protease |
J Med Chem 35: 3803-12 (1992)
BindingDB Entry DOI: 10.7270/Q2348M0D |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM18173
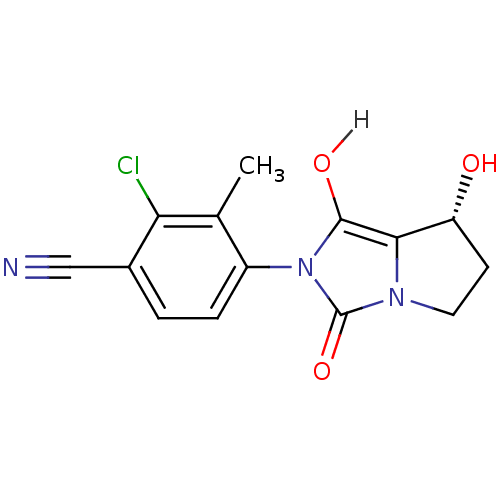
(4-[(7R,7aS)-7-hydroxy-1,3-dioxo-hexahydro-1H-pyrro...)Show SMILES Cc1c(Cl)c(ccc1-n1c(O)c2[C@H](O)CCn2c1=O)C#N |r,wU:12.13,(.01,.36,;.81,1.67,;2.35,1.63,;3.09,.28,;3.16,2.94,;2.42,4.3,;.88,4.34,;.08,3.02,;-1.46,3.02,;-1.94,4.49,;-1.03,5.73,;-3.48,4.49,;-4.72,5.39,;-4.72,6.93,;-5.97,4.49,;-5.5,3.02,;-3.95,3.02,;-2.71,2.12,;-2.71,.58,;4.7,2.9,;6.24,2.9,)| Show InChI InChI=1S/C14H12ClN3O3/c1-7-9(3-2-8(6-16)11(7)15)18-13(20)12-10(19)4-5-17(12)14(18)21/h2-3,10,19-20H,4-5H2,1H3/t10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 1.40 | -50.0 | n/a | n/a | 0.700 | n/a | n/a | 7.4 | 22 |
Bristol-Myers Squibb Pharmaceutical Research Institute
| Assay Description
Receptor Binding Assay (Ki)-Binding determined through direct displacement of ligand with [3H]-DHT in the MDA-453 cell line. Transactivation Assay (E... |
J Med Chem 50: 3015-3025 (2007)
Article DOI: 10.1021/jm070312d
BindingDB Entry DOI: 10.7270/Q24F1P1F |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Induced myeloid leukemia cell differentiation protein Mcl-1
(Homo sapiens (Human)) | BDBM50078155
(CHEMBL3417697)Show SMILES Cc1nn(C)c(C)c1-c1cccc2c(CCCOc3cccc4ccccc34)c(C(O)=O)n(CCN3CCOCC3)c12 |(-7.41,-13.93,;-6.18,-13.93,;-5.27,-15.18,;-3.8,-14.7,;-2.8,-15.42,;-3.8,-13.16,;-2.81,-12.42,;-5.26,-12.7,;-5.75,-11.24,;-7.28,-10.92,;-7.77,-9.46,;-6.73,-8.28,;-5.22,-8.61,;-3.98,-7.73,;-3.99,-6.18,;-2.65,-5.41,;-2.66,-3.86,;-1.33,-3.08,;-1.33,-1.54,;-2.69,-.77,;-2.69,.77,;-1.33,1.54,;,.77,;1.33,1.54,;2.67,.77,;2.67,-.77,;1.33,-1.54,;,-.77,;-2.74,-8.58,;-1.29,-8.07,;-.35,-8.87,;-1.06,-6.85,;-3.2,-10.06,;-2.28,-11.29,;-.75,-11.11,;.17,-12.35,;1.71,-12.18,;2.63,-13.42,;2.01,-14.83,;.48,-15.01,;-.44,-13.77,;-4.74,-10.07,)| Show InChI InChI=1S/C34H38N4O4/c1-23-31(24(2)36(3)35-23)29-13-7-12-27-28(14-8-20-42-30-15-6-10-25-9-4-5-11-26(25)30)33(34(39)40)38(32(27)29)17-16-37-18-21-41-22-19-37/h4-7,9-13,15H,8,14,16-22H2,1-3H3,(H,39,40) | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
AbbVie Inc.
Curated by ChEMBL
| Assay Description
Displacement of F-Bak (GQVGRQLAIIGDK(6-FAM)INR-amide) from MCL1 (unknown origin) after 1 hr by TR-FRET assay |
J Med Chem 58: 2180-94 (2015)
Article DOI: 10.1021/jm501258m
BindingDB Entry DOI: 10.7270/Q2HX1FC8 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data