 Found 1581 hits with Last Name = 'dale' and Initial = 'j'
Found 1581 hits with Last Name = 'dale' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Cathepsin K
(Homo sapiens (Human)) | BDBM50408519
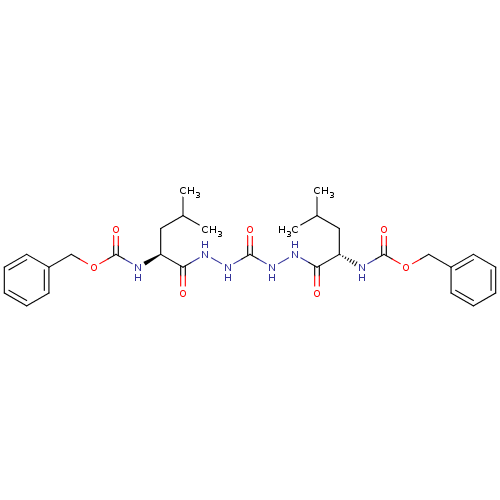
(CHEMBL115357)Show SMILES CC(C)C[C@H](NC(=O)OCc1ccccc1)C(=O)NNC(=O)NNC(=O)[C@H](CC(C)C)NC(=O)OCc1ccccc1 |r| Show InChI InChI=1S/C29H40N6O7/c1-19(2)15-23(30-28(39)41-17-21-11-7-5-8-12-21)25(36)32-34-27(38)35-33-26(37)24(16-20(3)4)31-29(40)42-18-22-13-9-6-10-14-22/h5-14,19-20,23-24H,15-18H2,1-4H3,(H,30,39)(H,31,40)(H,32,36)(H,33,37)(H2,34,35,38)/t23-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Relative binding affinity was measured for Cathepsin K |
J Med Chem 41: 3923-7 (1998)
Article DOI: 10.1021/jm980474x
BindingDB Entry DOI: 10.7270/Q2Q81F87 |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50408522
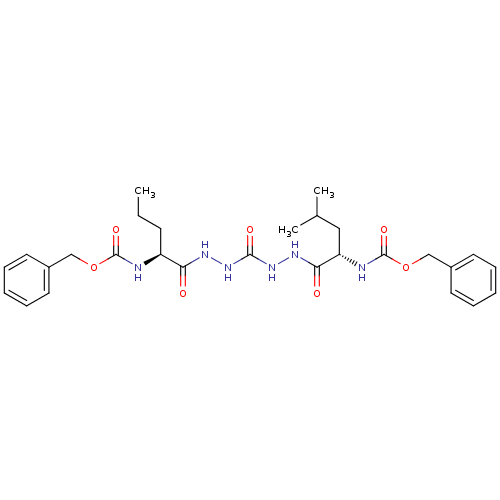
(CHEMBL126820)Show SMILES CCC[C@H](NC(=O)OCc1ccccc1)C(=O)NNC(=O)NNC(=O)[C@H](CC(C)C)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C28H38N6O7/c1-4-11-22(29-27(38)40-17-20-12-7-5-8-13-20)24(35)31-33-26(37)34-32-25(36)23(16-19(2)3)30-28(39)41-18-21-14-9-6-10-15-21/h5-10,12-15,19,22-23H,4,11,16-18H2,1-3H3,(H,29,38)(H,30,39)(H,31,35)(H,32,36)(H2,33,34,37)/t22-,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Relative binding affinity was measured for Cathepsin K |
J Med Chem 41: 3923-7 (1998)
Article DOI: 10.1021/jm980474x
BindingDB Entry DOI: 10.7270/Q2Q81F87 |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50408520
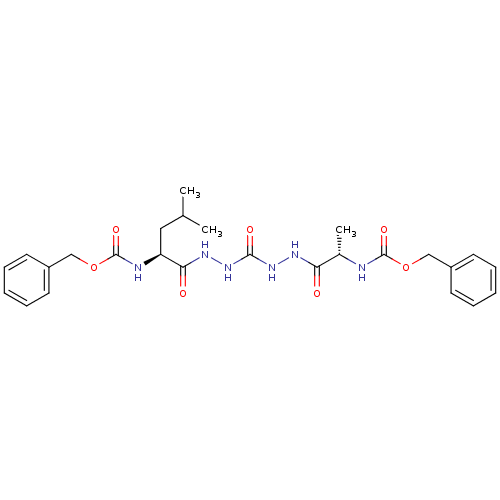
(CHEMBL126352)Show SMILES CC(C)C[C@H](NC(=O)OCc1ccccc1)C(=O)NNC(=O)NNC(=O)[C@H](C)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C26H34N6O7/c1-17(2)14-21(28-26(37)39-16-20-12-8-5-9-13-20)23(34)30-32-24(35)31-29-22(33)18(3)27-25(36)38-15-19-10-6-4-7-11-19/h4-13,17-18,21H,14-16H2,1-3H3,(H,27,36)(H,28,37)(H,29,33)(H,30,34)(H2,31,32,35)/t18-,21-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Relative binding affinity was measured for Cathepsin K |
J Med Chem 41: 3923-7 (1998)
Article DOI: 10.1021/jm980474x
BindingDB Entry DOI: 10.7270/Q2Q81F87 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394058
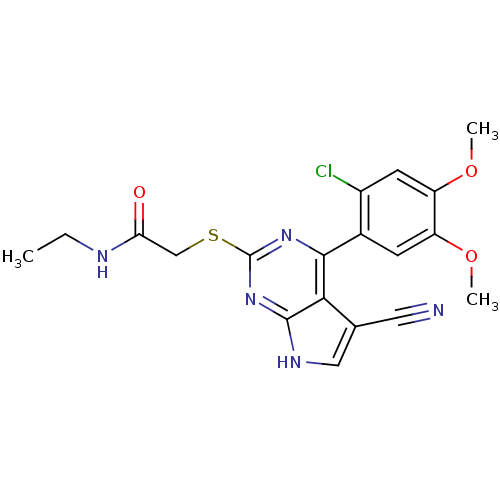
(CHEMBL2158570)Show SMILES CCNC(=O)CSc1nc(-c2cc(OC)c(OC)cc2Cl)c2c(c[nH]c2n1)C#N Show InChI InChI=1S/C19H18ClN5O3S/c1-4-22-15(26)9-29-19-24-17(16-10(7-21)8-23-18(16)25-19)11-5-13(27-2)14(28-3)6-12(11)20/h5-6,8H,4,9H2,1-3H3,(H,22,26)(H,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394056
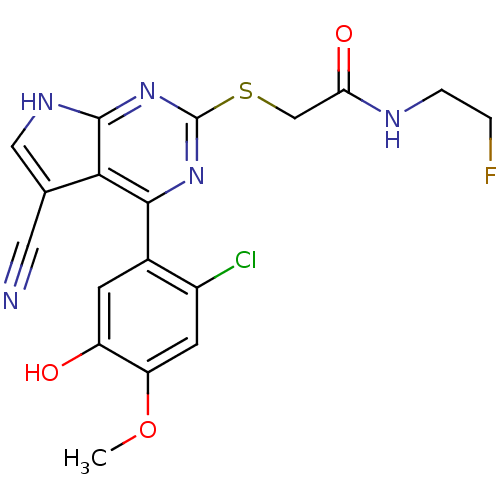
(CHEMBL2158577)Show SMILES COc1cc(Cl)c(cc1O)-c1nc(SCC(=O)NCCF)nc2[nH]cc(C#N)c12 Show InChI InChI=1S/C18H15ClFN5O3S/c1-28-13-5-11(19)10(4-12(13)26)16-15-9(6-21)7-23-17(15)25-18(24-16)29-8-14(27)22-3-2-20/h4-5,7,26H,2-3,8H2,1H3,(H,22,27)(H,23,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394065
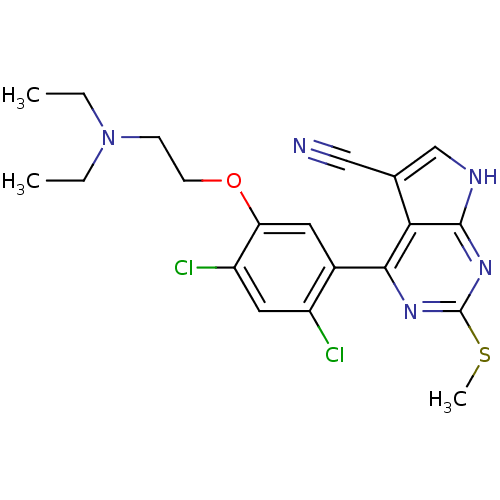
(CHEMBL2158626)Show SMILES CCN(CC)CCOc1cc(c(Cl)cc1Cl)-c1nc(SC)nc2[nH]cc(C#N)c12 Show InChI InChI=1S/C20H21Cl2N5OS/c1-4-27(5-2)6-7-28-16-8-13(14(21)9-15(16)22)18-17-12(10-23)11-24-19(17)26-20(25-18)29-3/h8-9,11H,4-7H2,1-3H3,(H,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394059
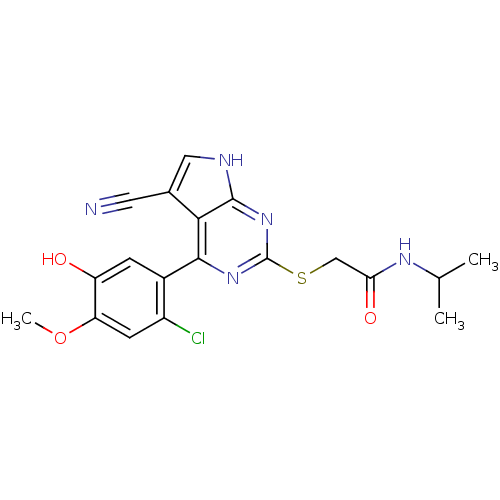
(CHEMBL2158569)Show SMILES COc1cc(Cl)c(cc1O)-c1nc(SCC(=O)NC(C)C)nc2[nH]cc(C#N)c12 Show InChI InChI=1S/C19H18ClN5O3S/c1-9(2)23-15(27)8-29-19-24-17(16-10(6-21)7-22-18(16)25-19)11-4-13(26)14(28-3)5-12(11)20/h4-5,7,9,26H,8H2,1-3H3,(H,23,27)(H,22,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394064
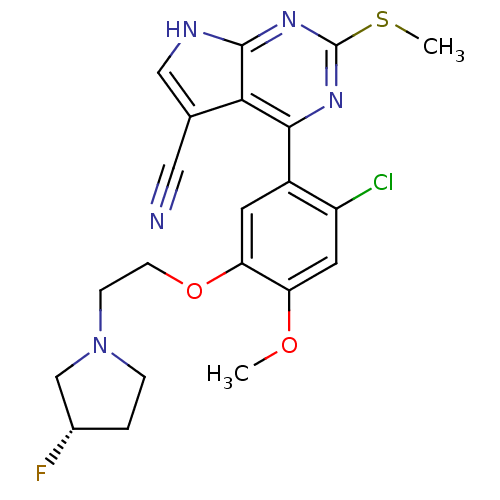
(CHEMBL2158627)Show SMILES COc1cc(Cl)c(cc1OCCN1CC[C@H](F)C1)-c1nc(SC)nc2[nH]cc(C#N)c12 |r| Show InChI InChI=1S/C21H21ClFN5O2S/c1-29-16-8-15(22)14(7-17(16)30-6-5-28-4-3-13(23)11-28)19-18-12(9-24)10-25-20(18)27-21(26-19)31-2/h7-8,10,13H,3-6,11H2,1-2H3,(H,25,26,27)/t13-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50408521
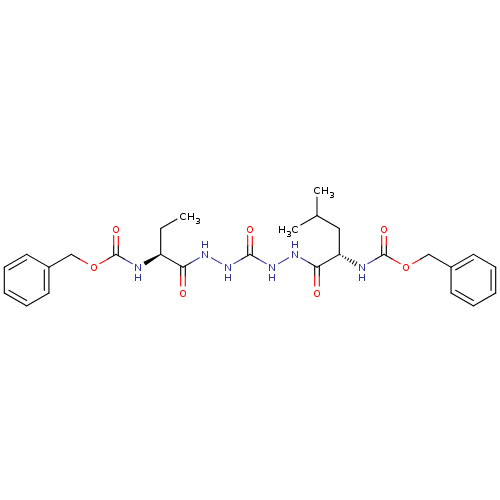
(CHEMBL129773)Show SMILES CC[C@H](NC(=O)OCc1ccccc1)C(=O)NNC(=O)NNC(=O)[C@H](CC(C)C)NC(=O)OCc1ccccc1 Show InChI InChI=1S/C27H36N6O7/c1-4-21(28-26(37)39-16-19-11-7-5-8-12-19)23(34)30-32-25(36)33-31-24(35)22(15-18(2)3)29-27(38)40-17-20-13-9-6-10-14-20/h5-14,18,21-22H,4,15-17H2,1-3H3,(H,28,37)(H,29,38)(H,30,34)(H,31,35)(H2,32,33,36)/t21-,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Relative binding affinity was measured for Cathepsin K |
J Med Chem 41: 3923-7 (1998)
Article DOI: 10.1021/jm980474x
BindingDB Entry DOI: 10.7270/Q2Q81F87 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394066
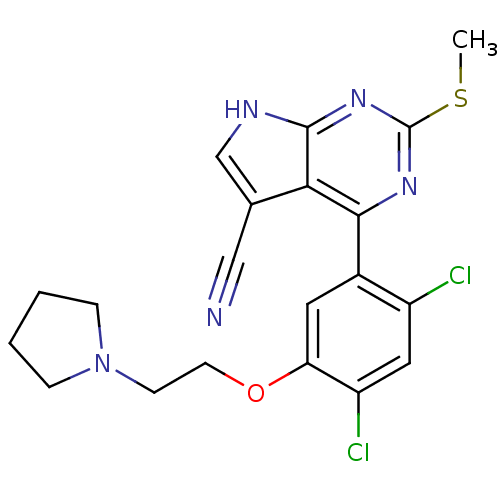
(CHEMBL2158625)Show SMILES CSc1nc(-c2cc(OCCN3CCCC3)c(Cl)cc2Cl)c2c(c[nH]c2n1)C#N Show InChI InChI=1S/C20H19Cl2N5OS/c1-29-20-25-18(17-12(10-23)11-24-19(17)26-20)13-8-16(15(22)9-14(13)21)28-7-6-27-4-2-3-5-27/h8-9,11H,2-7H2,1H3,(H,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394057
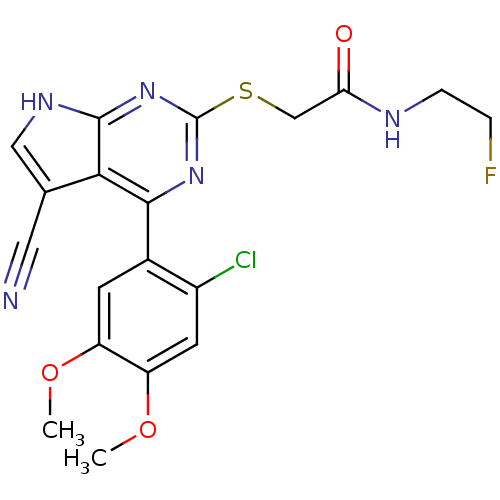
(CHEMBL2158576)Show SMILES COc1cc(Cl)c(cc1OC)-c1nc(SCC(=O)NCCF)nc2[nH]cc(C#N)c12 Show InChI InChI=1S/C19H17ClFN5O3S/c1-28-13-5-11(12(20)6-14(13)29-2)17-16-10(7-22)8-24-18(16)26-19(25-17)30-9-15(27)23-4-3-21/h5-6,8H,3-4,9H2,1-2H3,(H,23,27)(H,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394062
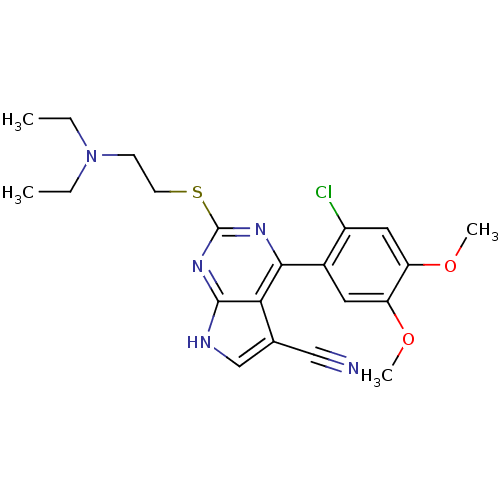
(CHEMBL2158630)Show SMILES CCN(CC)CCSc1nc(-c2cc(OC)c(OC)cc2Cl)c2c(c[nH]c2n1)C#N Show InChI InChI=1S/C21H24ClN5O2S/c1-5-27(6-2)7-8-30-21-25-19(18-13(11-23)12-24-20(18)26-21)14-9-16(28-3)17(29-4)10-15(14)22/h9-10,12H,5-8H2,1-4H3,(H,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394055
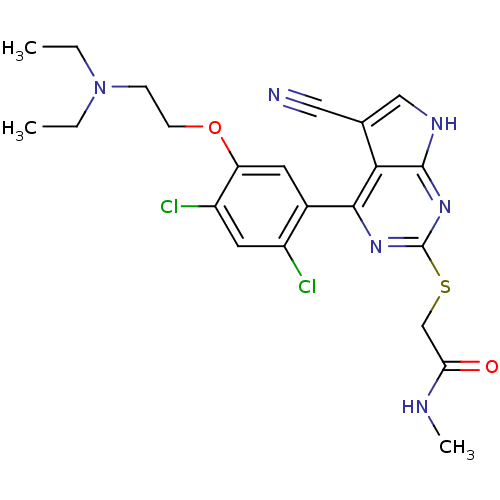
(CHEMBL2158563)Show SMILES CCN(CC)CCOc1cc(c(Cl)cc1Cl)-c1nc(SCC(=O)NC)nc2[nH]cc(C#N)c12 Show InChI InChI=1S/C22H24Cl2N6O2S/c1-4-30(5-2)6-7-32-17-8-14(15(23)9-16(17)24)20-19-13(10-25)11-27-21(19)29-22(28-20)33-12-18(31)26-3/h8-9,11H,4-7,12H2,1-3H3,(H,26,31)(H,27,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50408515
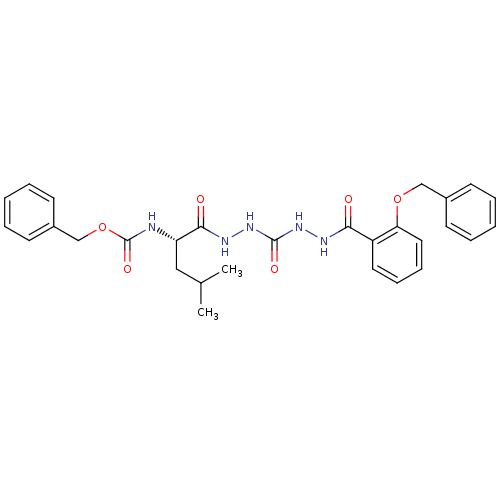
(CHEMBL338770)Show SMILES CC(C)C[C@H](NC(=O)OCc1ccccc1)C(=O)NNC(=O)NNC(=O)c1ccccc1OCc1ccccc1 Show InChI InChI=1S/C29H33N5O6/c1-20(2)17-24(30-29(38)40-19-22-13-7-4-8-14-22)27(36)32-34-28(37)33-31-26(35)23-15-9-10-16-25(23)39-18-21-11-5-3-6-12-21/h3-16,20,24H,17-19H2,1-2H3,(H,30,38)(H,31,35)(H,32,36)(H2,33,34,37)/t24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Relative binding affinity was measured for Cathepsin K |
J Med Chem 41: 3923-7 (1998)
Article DOI: 10.1021/jm980474x
BindingDB Entry DOI: 10.7270/Q2Q81F87 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394063
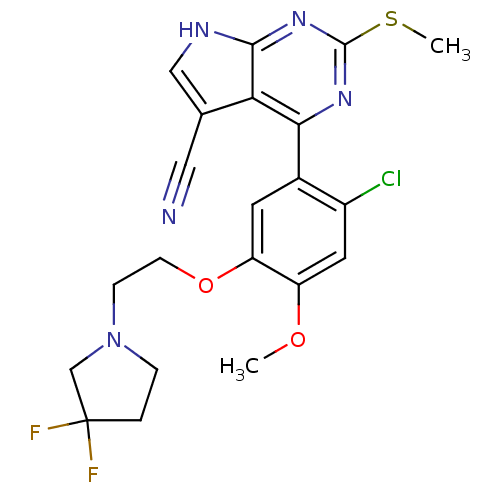
(CHEMBL2158628)Show SMILES COc1cc(Cl)c(cc1OCCN1CCC(F)(F)C1)-c1nc(SC)nc2[nH]cc(C#N)c12 Show InChI InChI=1S/C21H20ClF2N5O2S/c1-30-15-8-14(22)13(7-16(15)31-6-5-29-4-3-21(23,24)11-29)18-17-12(9-25)10-26-19(17)28-20(27-18)32-2/h7-8,10H,3-6,11H2,1-2H3,(H,26,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50408517
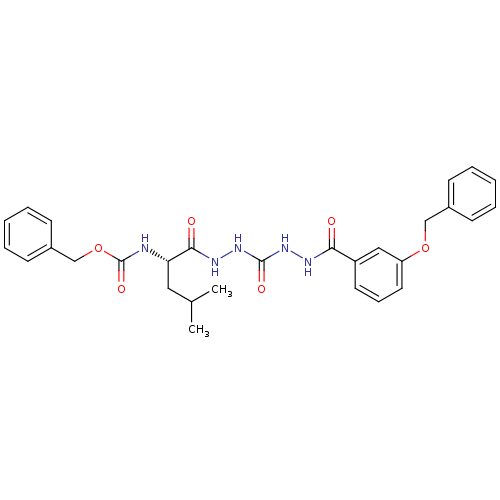
(CHEMBL340191)Show SMILES CC(C)C[C@H](NC(=O)OCc1ccccc1)C(=O)NNC(=O)NNC(=O)c1cccc(OCc2ccccc2)c1 |r| Show InChI InChI=1S/C29H33N5O6/c1-20(2)16-25(30-29(38)40-19-22-12-7-4-8-13-22)27(36)32-34-28(37)33-31-26(35)23-14-9-15-24(17-23)39-18-21-10-5-3-6-11-21/h3-15,17,20,25H,16,18-19H2,1-2H3,(H,30,38)(H,31,35)(H,32,36)(H2,33,34,37)/t25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Relative binding affinity was measured for Cathepsin K |
J Med Chem 41: 3923-7 (1998)
Article DOI: 10.1021/jm980474x
BindingDB Entry DOI: 10.7270/Q2Q81F87 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394061
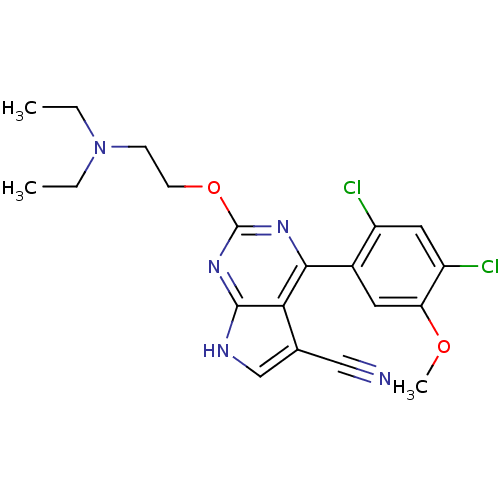
(CHEMBL2158008)Show SMILES CCN(CC)CCOc1nc(-c2cc(OC)c(Cl)cc2Cl)c2c(c[nH]c2n1)C#N Show InChI InChI=1S/C20H21Cl2N5O2/c1-4-27(5-2)6-7-29-20-25-18(17-12(10-23)11-24-19(17)26-20)13-8-16(28-3)15(22)9-14(13)21/h8-9,11H,4-7H2,1-3H3,(H,24,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 62 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50345738
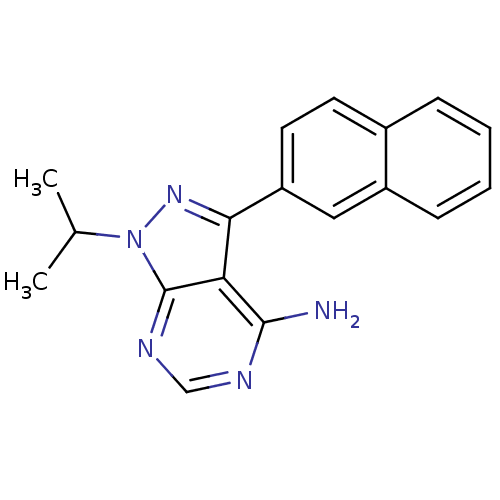
(1-isopropyl-3-(naphthalen-2-yl)-1H-pyrazolo[3,4-d]...)Show InChI InChI=1S/C18H17N5/c1-11(2)23-18-15(17(19)20-10-21-18)16(22-23)14-8-7-12-5-3-4-6-13(12)9-14/h3-11H,1-2H3,(H2,19,20,21) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human SRC by radiometric assay |
J Med Chem 55: 2803-10 (2012)
Article DOI: 10.1021/jm201725v
BindingDB Entry DOI: 10.7270/Q2ST7QVK |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50345738

(1-isopropyl-3-(naphthalen-2-yl)-1H-pyrazolo[3,4-d]...)Show InChI InChI=1S/C18H17N5/c1-11(2)23-18-15(17(19)20-10-21-18)16(22-23)14-8-7-12-5-3-4-6-13(12)9-14/h3-11H,1-2H3,(H2,19,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 69 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human ABL by radiometric assay |
J Med Chem 55: 2803-10 (2012)
Article DOI: 10.1021/jm201725v
BindingDB Entry DOI: 10.7270/Q2ST7QVK |
More data for this
Ligand-Target Pair | |
Kynureninase
(Homo sapiens (Human)) | BDBM50069730
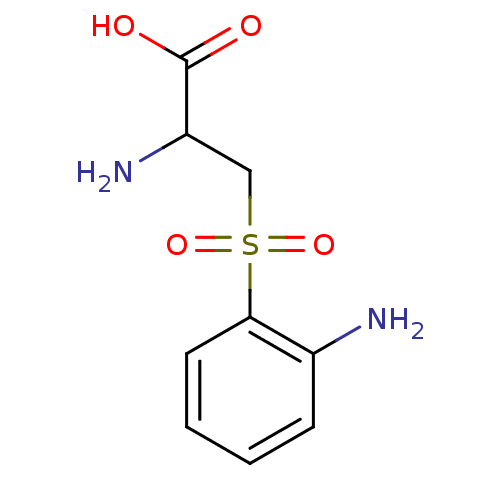
(2-Amino-3-(2-amino-benzenesulfonyl)-propionic acid...)Show InChI InChI=1S/C9H12N2O4S/c10-6-3-1-2-4-8(6)16(14,15)5-7(11)9(12)13/h1-4,7H,5,10-11H2,(H,12,13) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Compound was tested for the binding affinity against bacterial kynureninase |
Bioorg Med Chem Lett 8: 133-8 (1999)
BindingDB Entry DOI: 10.7270/Q2639NXW |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50408518
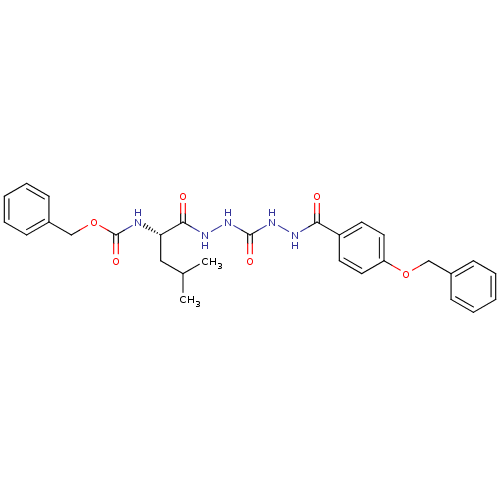
(CHEMBL338150)Show SMILES CC(C)C[C@H](NC(=O)OCc1ccccc1)C(=O)NNC(=O)NNC(=O)c1ccc(OCc2ccccc2)cc1 Show InChI InChI=1S/C29H33N5O6/c1-20(2)17-25(30-29(38)40-19-22-11-7-4-8-12-22)27(36)32-34-28(37)33-31-26(35)23-13-15-24(16-14-23)39-18-21-9-5-3-6-10-21/h3-16,20,25H,17-19H2,1-2H3,(H,30,38)(H,31,35)(H,32,36)(H2,33,34,37)/t25-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Relative binding affinity was measured for Cathepsin K |
J Med Chem 41: 3923-7 (1998)
Article DOI: 10.1021/jm980474x
BindingDB Entry DOI: 10.7270/Q2Q81F87 |
More data for this
Ligand-Target Pair | |
Heat shock 70 kDa protein 1A
(Homo sapiens (Human)) | BDBM82124
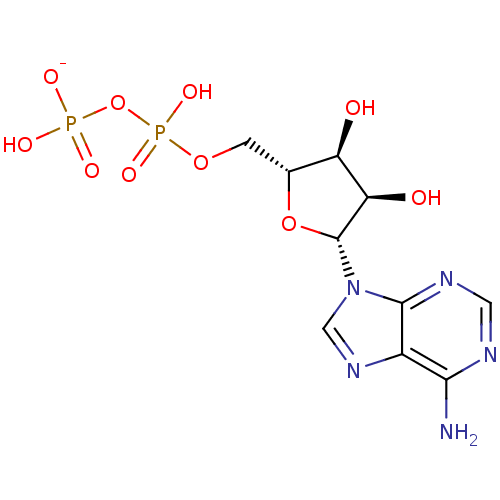
(adenosine-derived inhibitor (Grp78), 1)Show SMILES Nc1ncnc2n(cnc12)[C@@H]1O[C@H](COP(O)(=O)OP(O)([O-])=O)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C10H15N5O10P2/c11-8-5-9(13-2-12-8)15(3-14-5)10-7(17)6(16)4(24-10)1-23-27(21,22)25-26(18,19)20/h2-4,6-7,10,16-17H,1H2,(H,21,22)(H2,11,12,13)(H2,18,19,20)/p-1/t4-,6-,7-,10-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 110 | -39.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd.
| Assay Description
The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... |
J Med Chem 54: 4034-41 (2011)
Article DOI: 10.1021/jm101625x
BindingDB Entry DOI: 10.7270/Q2R49P83 |
More data for this
Ligand-Target Pair | |
Heat shock 70 kDa protein 1A
(Homo sapiens (Human)) | BDBM32378
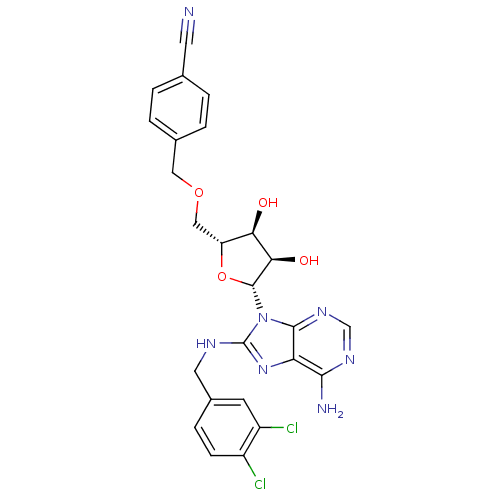
(adenosine-derived inhibitor (Grp78), 13 | adenosin...)Show SMILES Nc1ncnc2n([C@@H]3O[C@H](COCc4ccc(cc4)C#N)[C@@H](O)[C@H]3O)c(NCc3ccc(Cl)c(Cl)c3)nc12 |r| Show InChI InChI=1S/C25H23Cl2N7O4/c26-16-6-5-15(7-17(16)27)9-30-25-33-19-22(29)31-12-32-23(19)34(25)24-21(36)20(35)18(38-24)11-37-10-14-3-1-13(8-28)2-4-14/h1-7,12,18,20-21,24,35-36H,9-11H2,(H,30,33)(H2,29,31,32)/t18-,20-,21-,24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 120 | -39.5 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd.
| Assay Description
The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... |
J Med Chem 54: 4034-41 (2011)
Article DOI: 10.1021/jm101625x
BindingDB Entry DOI: 10.7270/Q2R49P83 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein phosphatase non-receptor type 1
(Rattus norvegicus (rat)) | BDBM13614

(2-(carboxymethoxy)-5-[(2S)-2-(pentylcarbamoyl)-2-[...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(OCC(O)=O)c(c1)C(O)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)Cn1[nH]ncc1=S |r| Show InChI InChI=1S/C30H36N6O8S/c1-2-3-7-12-31-28(40)22(15-20-10-11-24(44-18-27(38)39)21(13-20)30(42)43)34-29(41)23(14-19-8-5-4-6-9-19)33-25(37)17-36-26(45)16-32-35-36/h4-6,8-11,13,16,22-23,35H,2-3,7,12,14-15,17-18H2,1H3,(H,31,40)(H,33,37)(H,34,41)(H,38,39)(H,42,43)/t22-,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 120 | -39.1 | n/a | n/a | n/a | n/a | n/a | 7.2 | 22 |
Pharmacia Corporation
| Assay Description
Hydrolysis of substrate p-nitrophenyl phosphate was monitored on a 96-well microplate reader. Concentrations of PTP1B inhibitors that resulted in 50%... |
Biochemistry 40: 5642-54 (2001)
Article DOI: 10.1021/bi002865v
BindingDB Entry DOI: 10.7270/Q2D50K6S |
More data for this
Ligand-Target Pair | |
Cathepsin K
(Homo sapiens (Human)) | BDBM50408516
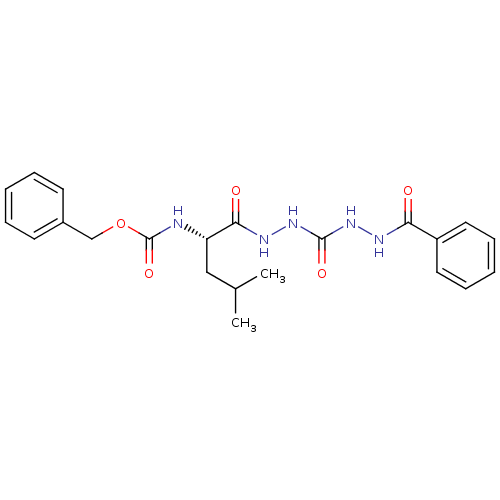
(CHEMBL126835)Show SMILES CC(C)C[C@H](NC(=O)OCc1ccccc1)C(=O)NNC(=O)NNC(=O)c1ccccc1 Show InChI InChI=1S/C22H27N5O5/c1-15(2)13-18(23-22(31)32-14-16-9-5-3-6-10-16)20(29)25-27-21(30)26-24-19(28)17-11-7-4-8-12-17/h3-12,15,18H,13-14H2,1-2H3,(H,23,31)(H,24,28)(H,25,29)(H2,26,27,30)/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
SmithKline Beecham Pharmaceuticals
Curated by ChEMBL
| Assay Description
Relative binding affinity was measured for Cathepsin K |
J Med Chem 41: 3923-7 (1998)
Article DOI: 10.1021/jm980474x
BindingDB Entry DOI: 10.7270/Q2Q81F87 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394060
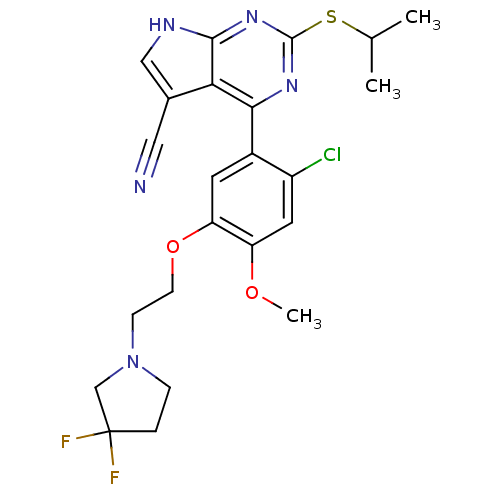
(CHEMBL2158565)Show SMILES COc1cc(Cl)c(cc1OCCN1CCC(F)(F)C1)-c1nc(SC(C)C)nc2[nH]cc(C#N)c12 Show InChI InChI=1S/C23H24ClF2N5O2S/c1-13(2)34-22-29-20(19-14(10-27)11-28-21(19)30-22)15-8-18(17(32-3)9-16(15)24)33-7-6-31-5-4-23(25,26)12-31/h8-9,11,13H,4-7,12H2,1-3H3,(H,28,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 172 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394079
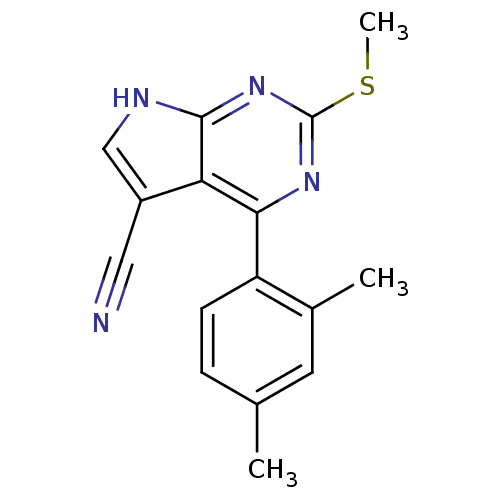
(CHEMBL2158581)Show InChI InChI=1S/C16H14N4S/c1-9-4-5-12(10(2)6-9)14-13-11(7-17)8-18-15(13)20-16(19-14)21-3/h4-6,8H,1-3H3,(H,18,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 195 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50345740

(3-(6-ethoxynaphthalen-2-yl)-1-isopropyl-1H-pyrazol...)Show SMILES CCOc1ccc2cc(ccc2c1)-c1nn(C(C)C)c2ncnc(N)c12 Show InChI InChI=1S/C20H21N5O/c1-4-26-16-8-7-13-9-15(6-5-14(13)10-16)18-17-19(21)22-11-23-20(17)25(24-18)12(2)3/h5-12H,4H2,1-3H3,(H2,21,22,23) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human SRC by radiometric assay |
J Med Chem 55: 2803-10 (2012)
Article DOI: 10.1021/jm201725v
BindingDB Entry DOI: 10.7270/Q2ST7QVK |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50345742

(1-isopropyl-3-(quinolin-3-yl)-1H-pyrazolo[3,4-d]py...)Show InChI InChI=1S/C17H16N6/c1-10(2)23-17-14(16(18)20-9-21-17)15(22-23)12-7-11-5-3-4-6-13(11)19-8-12/h3-10H,1-2H3,(H2,18,20,21) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human SRC by radiometric assay |
J Med Chem 55: 2803-10 (2012)
Article DOI: 10.1021/jm201725v
BindingDB Entry DOI: 10.7270/Q2ST7QVK |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394077
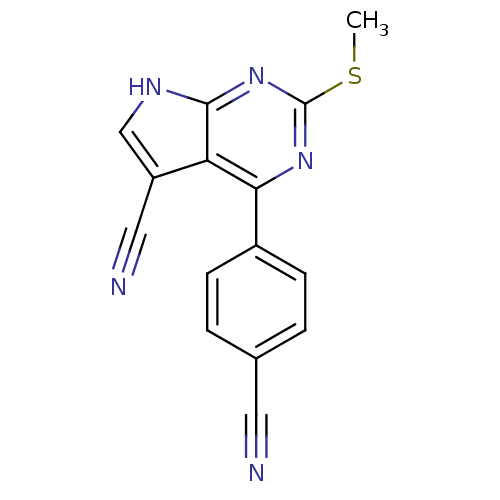
(CHEMBL2158583)Show InChI InChI=1S/C15H9N5S/c1-21-15-19-13(10-4-2-9(6-16)3-5-10)12-11(7-17)8-18-14(12)20-15/h2-5,8H,1H3,(H,18,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 204 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM13611
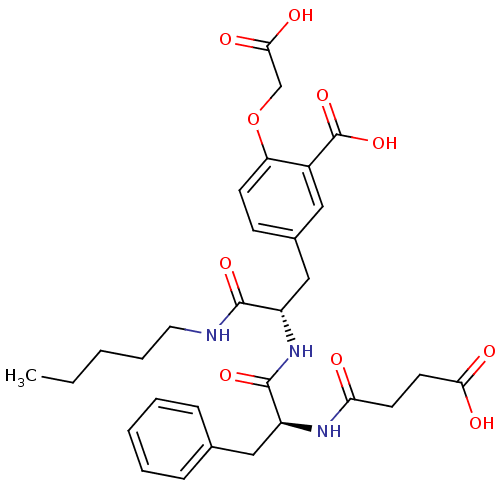
(2-(carboxymethoxy)-5-[(2S)-2-[(2S)-2-(3-formamidop...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(OCC(O)=O)c(c1)C(O)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)CCC(O)=O |r| Show InChI InChI=1S/C30H37N3O10/c1-2-3-7-14-31-28(39)22(17-20-10-11-24(43-18-27(37)38)21(15-20)30(41)42)33-29(40)23(16-19-8-5-4-6-9-19)32-25(34)12-13-26(35)36/h4-6,8-11,15,22-23H,2-3,7,12-14,16-18H2,1H3,(H,31,39)(H,32,34)(H,33,40)(H,35,36)(H,37,38)(H,41,42)/t22-,23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
PubMed
| 220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacia Corporation
Curated by ChEMBL
| Assay Description
Inhibitory activity against human protein tyrosine phosphatase 1B (PTP1B) |
Bioorg Med Chem Lett 13: 971-5 (2003)
BindingDB Entry DOI: 10.7270/Q24M93WC |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein phosphatase non-receptor type 1
(Rattus norvegicus (rat)) | BDBM13611

(2-(carboxymethoxy)-5-[(2S)-2-[(2S)-2-(3-formamidop...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(OCC(O)=O)c(c1)C(O)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)CCC(O)=O |r| Show InChI InChI=1S/C30H37N3O10/c1-2-3-7-14-31-28(39)22(17-20-10-11-24(43-18-27(37)38)21(15-20)30(41)42)33-29(40)23(16-19-8-5-4-6-9-19)32-25(34)12-13-26(35)36/h4-6,8-11,15,22-23H,2-3,7,12-14,16-18H2,1H3,(H,31,39)(H,32,34)(H,33,40)(H,35,36)(H,37,38)(H,41,42)/t22-,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 250 | -37.3 | n/a | n/a | n/a | n/a | n/a | 7.2 | 22 |
Pharmacia Corporation
| Assay Description
Hydrolysis of substrate p-nitrophenyl phosphate was monitored on a 96-well microplate reader. Concentrations of PTP1B inhibitors that resulted in 50%... |
Biochemistry 40: 5642-54 (2001)
Article DOI: 10.1021/bi002865v
BindingDB Entry DOI: 10.7270/Q2D50K6S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50383382

(CHEMBL2030556 | CHEMBL2069948 | US10172858, Table ...)Show SMILES CC(C)n1nc(-c2cc3c(Cl)cccc3[nH]2)c2c(N)ncnc12 Show InChI InChI=1S/C16H15ClN6/c1-8(2)23-16-13(15(18)19-7-20-16)14(22-23)12-6-9-10(17)4-3-5-11(9)21-12/h3-8,21H,1-2H3,(H2,18,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human ABL by radiometric assay |
J Med Chem 55: 2803-10 (2012)
Article DOI: 10.1021/jm201725v
BindingDB Entry DOI: 10.7270/Q2ST7QVK |
More data for this
Ligand-Target Pair | |
Heat shock 70 kDa protein 1A
(Homo sapiens (Human)) | BDBM32381
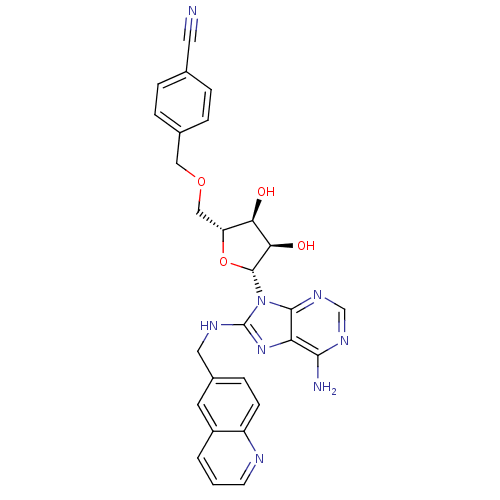
(adenosine-derived inhibitor (Grp78), 14 | adenosin...)Show SMILES Nc1ncnc2n([C@@H]3O[C@H](COCc4ccc(cc4)C#N)[C@@H](O)[C@H]3O)c(NCc3ccc4ncccc4c3)nc12 |r| Show InChI InChI=1S/C28H26N8O4/c29-11-16-3-5-17(6-4-16)13-39-14-21-23(37)24(38)27(40-21)36-26-22(25(30)33-15-34-26)35-28(36)32-12-18-7-8-20-19(10-18)2-1-9-31-20/h1-10,15,21,23-24,27,37-38H,12-14H2,(H,32,35)(H2,30,33,34)/t21-,23-,24-,27-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| MMDB
PC cid
PC sid
PDB
UniChem
Similars
| MMDB
PDB
Article
PubMed
| 310 | -37.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd.
| Assay Description
The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... |
J Med Chem 54: 4034-41 (2011)
Article DOI: 10.1021/jm101625x
BindingDB Entry DOI: 10.7270/Q2R49P83 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394078
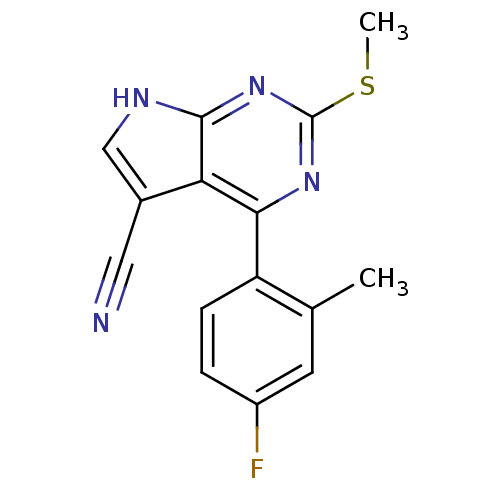
(CHEMBL2158582)Show InChI InChI=1S/C15H11FN4S/c1-8-5-10(16)3-4-11(8)13-12-9(6-17)7-18-14(12)20-15(19-13)21-2/h3-5,7H,1-2H3,(H,18,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 358 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Heat shock 70 kDa protein 1A
(Homo sapiens (Human)) | BDBM82127
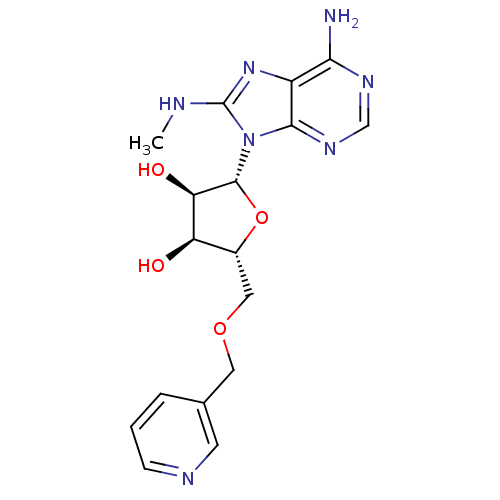
((2R,3R,4S,5R)-2-(6-Amino-8-methylaminopurin-9-yl)-...)Show SMILES CNc1nc2c(N)ncnc2n1[C@@H]1O[C@H](COCc2cccnc2)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C17H21N7O4/c1-19-17-23-11-14(18)21-8-22-15(11)24(17)16-13(26)12(25)10(28-16)7-27-6-9-3-2-4-20-5-9/h2-5,8,10,12-13,16,25-26H,6-7H2,1H3,(H,19,23)(H2,18,21,22)/t10-,12-,13-,16-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 450 | -36.2 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd.
| Assay Description
The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... |
J Med Chem 54: 4034-41 (2011)
Article DOI: 10.1021/jm101625x
BindingDB Entry DOI: 10.7270/Q2R49P83 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50124517
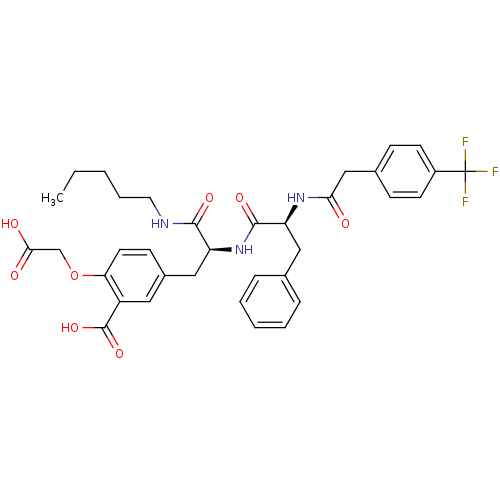
(2-Carboxymethoxy-5-(2-pentylcarbamoyl-2-{(S)-(S)-3...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(OCC(O)=O)c(c1)C(O)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)Cc1ccc(cc1)C(F)(F)F Show InChI InChI=1S/C35H38F3N3O8/c1-2-3-7-16-39-32(45)27(19-24-12-15-29(49-21-31(43)44)26(17-24)34(47)48)41-33(46)28(18-22-8-5-4-6-9-22)40-30(42)20-23-10-13-25(14-11-23)35(36,37)38/h4-6,8-15,17,27-28H,2-3,7,16,18-21H2,1H3,(H,39,45)(H,40,42)(H,41,46)(H,43,44)(H,47,48)/t27-,28-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacia Corporation
Curated by ChEMBL
| Assay Description
Inhibitory activity against human protein tyrosine phosphatase 1B (PTP1B) |
Bioorg Med Chem Lett 13: 971-5 (2003)
BindingDB Entry DOI: 10.7270/Q24M93WC |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394069
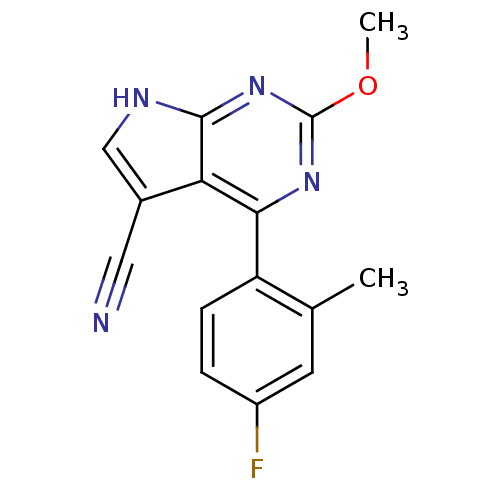
(CHEMBL2158622)Show InChI InChI=1S/C15H11FN4O/c1-8-5-10(16)3-4-11(8)13-12-9(6-17)7-18-14(12)20-15(19-13)21-2/h3-5,7H,1-2H3,(H,18,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 628 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50345735
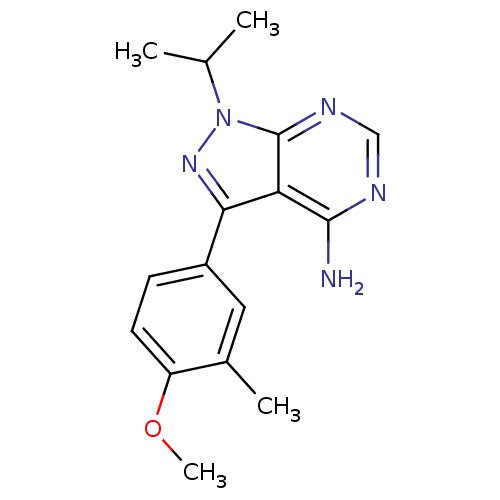
(1-isopropyl-3-(4-methoxy-3-methylphenyl)-1H-pyrazo...)Show InChI InChI=1S/C16H19N5O/c1-9(2)21-16-13(15(17)18-8-19-16)14(20-21)11-5-6-12(22-4)10(3)7-11/h5-9H,1-4H3,(H2,17,18,19) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human SRC by radiometric assay |
J Med Chem 55: 2803-10 (2012)
Article DOI: 10.1021/jm201725v
BindingDB Entry DOI: 10.7270/Q2ST7QVK |
More data for this
Ligand-Target Pair | |
Heat shock protein HSP 90-beta
(Homo sapiens (Human)) | BDBM50394085
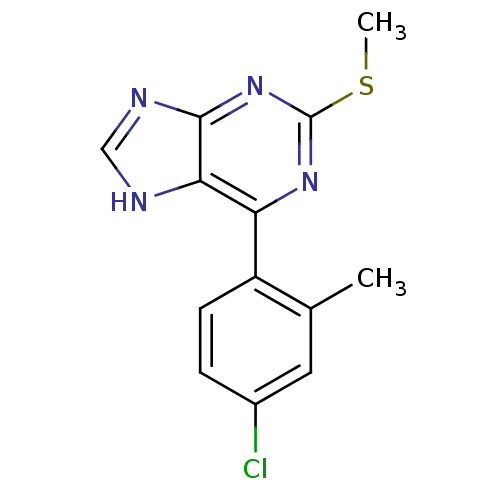
(CHEMBL2158635)Show InChI InChI=1S/C13H11ClN4S/c1-7-5-8(14)3-4-9(7)10-11-12(16-6-15-11)18-13(17-10)19-2/h3-6H,1-2H3,(H,15,16,17,18) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 661 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd.
Curated by ChEMBL
| Assay Description
Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay |
Bioorg Med Chem 20: 6770-89 (2012)
Article DOI: 10.1016/j.bmc.2012.08.050
BindingDB Entry DOI: 10.7270/Q2KS6SN4 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50124511

(2-Carboxymethoxy-5-(2-{(S)-(S)-2-[2-(4-methoxy-phe...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(OCC(O)=O)c(c1)C(O)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)Cc1ccc(OC)cc1 Show InChI InChI=1S/C35H41N3O9/c1-3-4-8-17-36-33(42)28(20-25-13-16-30(47-22-32(40)41)27(18-25)35(44)45)38-34(43)29(19-23-9-6-5-7-10-23)37-31(39)21-24-11-14-26(46-2)15-12-24/h5-7,9-16,18,28-29H,3-4,8,17,19-22H2,1-2H3,(H,36,42)(H,37,39)(H,38,43)(H,40,41)(H,44,45)/t28-,29-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 670 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacia Corporation
Curated by ChEMBL
| Assay Description
Inhibitory activity against human protein tyrosine phosphatase 1B (PTP1B) |
Bioorg Med Chem Lett 13: 971-5 (2003)
BindingDB Entry DOI: 10.7270/Q24M93WC |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50124514
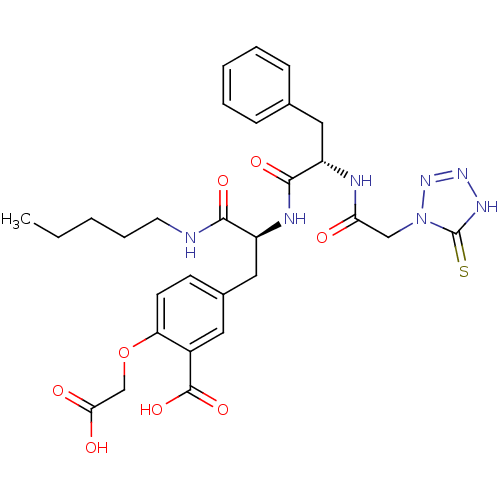
(2-Carboxymethoxy-5-[2-{(S)-2-[2-(5-mercapto-tetraz...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(OCC(O)=O)c(c1)C(O)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)Cn1nn[nH]c1=S Show InChI InChI=1S/C29H35N7O8S/c1-2-3-7-12-30-26(40)21(15-19-10-11-23(44-17-25(38)39)20(13-19)28(42)43)32-27(41)22(14-18-8-5-4-6-9-18)31-24(37)16-36-29(45)33-34-35-36/h4-6,8-11,13,21-22H,2-3,7,12,14-17H2,1H3,(H,30,40)(H,31,37)(H,32,41)(H,38,39)(H,42,43)(H,33,35,45)/t21-,22-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacia Corporation
Curated by ChEMBL
| Assay Description
Inhibitory activity against human protein tyrosine phosphatase 1B (PTP1B) |
Bioorg Med Chem Lett 13: 971-5 (2003)
BindingDB Entry DOI: 10.7270/Q24M93WC |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50345739
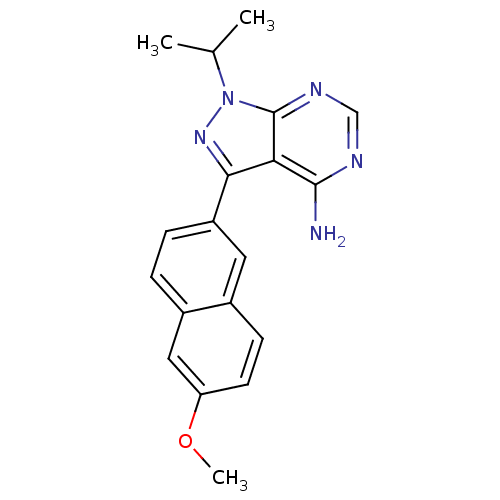
(1-isopropyl-3-(6-methoxynaphthalen-2-yl)-1H-pyrazo...)Show InChI InChI=1S/C19H19N5O/c1-11(2)24-19-16(18(20)21-10-22-19)17(23-24)14-5-4-13-9-15(25-3)7-6-12(13)8-14/h4-11H,1-3H3,(H2,20,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 750 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human ABL by radiometric assay |
J Med Chem 55: 2803-10 (2012)
Article DOI: 10.1021/jm201725v
BindingDB Entry DOI: 10.7270/Q2ST7QVK |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50345739

(1-isopropyl-3-(6-methoxynaphthalen-2-yl)-1H-pyrazo...)Show InChI InChI=1S/C19H19N5O/c1-11(2)24-19-16(18(20)21-10-22-19)17(23-24)14-5-4-13-9-15(25-3)7-6-12(13)8-14/h4-11H,1-3H3,(H2,20,21,22) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 770 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human SRC by radiometric assay |
J Med Chem 55: 2803-10 (2012)
Article DOI: 10.1021/jm201725v
BindingDB Entry DOI: 10.7270/Q2ST7QVK |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Homo sapiens (Human)) | BDBM50124513

(2-Carboxymethoxy-5-[(S)-2-pentylcarbamoyl-2-((S)-2...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(OCC(O)=O)c(c1)C(O)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)Oc1ccccc1 Show InChI InChI=1S/C33H37N3O9/c1-2-3-10-17-34-30(39)26(20-23-15-16-28(44-21-29(37)38)25(18-23)32(41)42)35-31(40)27(19-22-11-6-4-7-12-22)36-33(43)45-24-13-8-5-9-14-24/h4-9,11-16,18,26-27H,2-3,10,17,19-21H2,1H3,(H,34,39)(H,35,40)(H,36,43)(H,37,38)(H,41,42)/t26-,27-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacia Corporation
Curated by ChEMBL
| Assay Description
Inhibitory activity against human protein tyrosine phosphatase 1B (PTP1B) |
Bioorg Med Chem Lett 13: 971-5 (2003)
BindingDB Entry DOI: 10.7270/Q24M93WC |
More data for this
Ligand-Target Pair | |
Heat shock 70 kDa protein 1A
(Homo sapiens (Human)) | BDBM32377
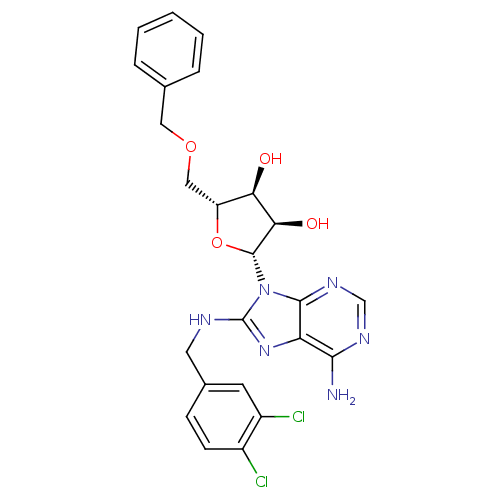
(adenosine-derived inhibitor (Grp78), 12 | adenosin...)Show SMILES Nc1ncnc2n([C@@H]3O[C@H](COCc4ccccc4)[C@@H](O)[C@H]3O)c(NCc3ccc(Cl)c(Cl)c3)nc12 |r| Show InChI InChI=1S/C24H24Cl2N6O4/c25-15-7-6-14(8-16(15)26)9-28-24-31-18-21(27)29-12-30-22(18)32(24)23-20(34)19(33)17(36-23)11-35-10-13-4-2-1-3-5-13/h1-8,12,17,19-20,23,33-34H,9-11H2,(H,28,31)(H2,27,29,30)/t17-,19-,20-,23-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.05E+3 | -34.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd.
| Assay Description
The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... |
J Med Chem 54: 4034-41 (2011)
Article DOI: 10.1021/jm101625x
BindingDB Entry DOI: 10.7270/Q2R49P83 |
More data for this
Ligand-Target Pair | |
Heat shock 70 kDa protein 1A
(Homo sapiens (Human)) | BDBM82125
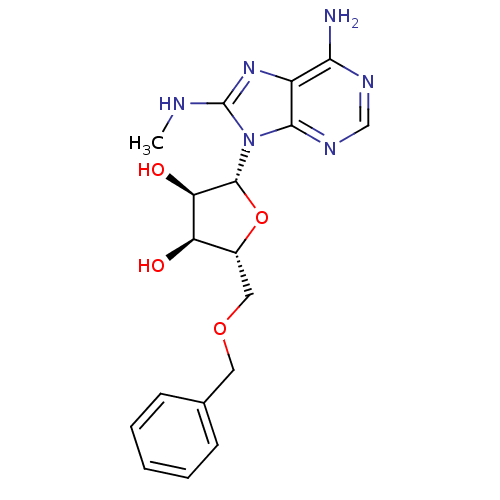
((2R,3R,4S,5R)-2-(6-Amino-8-methylaminopurin-9-yl)-...)Show SMILES CNc1nc2c(N)ncnc2n1[C@@H]1O[C@H](COCc2ccccc2)[C@@H](O)[C@H]1O |r| Show InChI InChI=1S/C18H22N6O4/c1-20-18-23-12-15(19)21-9-22-16(12)24(18)17-14(26)13(25)11(28-17)8-27-7-10-5-3-2-4-6-10/h2-6,9,11,13-14,17,25-26H,7-8H2,1H3,(H,20,23)(H2,19,21,22)/t11-,13-,14-,17-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10E+3 | -34.0 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd.
| Assay Description
The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... |
J Med Chem 54: 4034-41 (2011)
Article DOI: 10.1021/jm101625x
BindingDB Entry DOI: 10.7270/Q2R49P83 |
More data for this
Ligand-Target Pair | |
Tyrosine-protein phosphatase non-receptor type 1
(Rattus norvegicus (rat)) | BDBM13609

(2-{4-[(2S)-2-[(2S)-2-(3-formamidopropanoic acid)-3...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(OC(C(O)=O)C(O)=O)cc1)NC(=O)[C@H](Cc1ccccc1)NC(=O)CCC(O)=O |r| Show InChI InChI=1S/C30H37N3O10/c1-2-3-7-16-31-27(37)22(18-20-10-12-21(13-11-20)43-26(29(39)40)30(41)42)33-28(38)23(17-19-8-5-4-6-9-19)32-24(34)14-15-25(35)36/h4-6,8-13,22-23,26H,2-3,7,14-18H2,1H3,(H,31,37)(H,32,34)(H,33,38)(H,35,36)(H,39,40)(H,41,42)/t22-,23-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.20E+3 | -33.5 | n/a | n/a | n/a | n/a | n/a | 7.2 | 22 |
Pharmacia Corporation
| Assay Description
Hydrolysis of substrate p-nitrophenyl phosphate was monitored on a 96-well microplate reader. Concentrations of PTP1B inhibitors that resulted in 50%... |
Biochemistry 40: 5642-54 (2001)
Article DOI: 10.1021/bi002865v
BindingDB Entry DOI: 10.7270/Q2D50K6S |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50383382

(CHEMBL2030556 | CHEMBL2069948 | US10172858, Table ...)Show SMILES CC(C)n1nc(-c2cc3c(Cl)cccc3[nH]2)c2c(N)ncnc12 Show InChI InChI=1S/C16H15ClN6/c1-8(2)23-16-13(15(18)19-7-20-16)14(22-23)12-6-9-10(17)4-3-5-11(9)21-12/h3-8,21H,1-2H3,(H2,18,19,20) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.24E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Washington
Curated by ChEMBL
| Assay Description
Inhibition of human SRC by radiometric assay |
J Med Chem 55: 2803-10 (2012)
Article DOI: 10.1021/jm201725v
BindingDB Entry DOI: 10.7270/Q2ST7QVK |
More data for this
Ligand-Target Pair | |
Heat shock 70 kDa protein 1A
(Homo sapiens (Human)) | BDBM32375
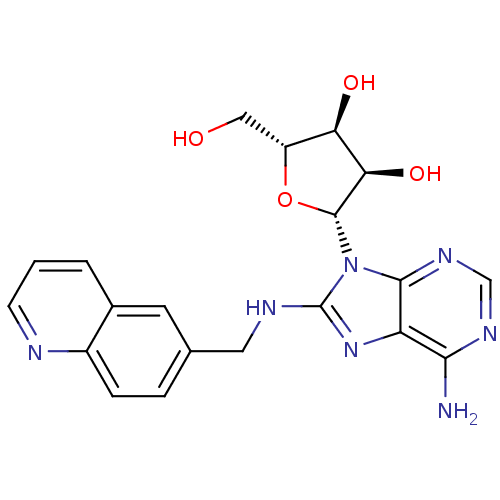
(adenosine-derived inhibitor (Grp78), 9 | adenosine...)Show SMILES Nc1ncnc2n([C@@H]3O[C@H](CO)[C@@H](O)[C@H]3O)c(NCc3ccc4ncccc4c3)nc12 |r| Show InChI InChI=1S/C20H21N7O4/c21-17-14-18(25-9-24-17)27(19-16(30)15(29)13(8-28)31-19)20(26-14)23-7-10-3-4-12-11(6-10)2-1-5-22-12/h1-6,9,13,15-16,19,28-30H,7-8H2,(H,23,26)(H2,21,24,25)/t13-,15-,16-,19-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 1.29E+3 | -33.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
Vernalis (R&D) Ltd.
| Assay Description
The assay was based on the fluorescence polarization of the unbound small binding partner will be low, and its binding to a larger binding partner wi... |
J Med Chem 54: 4034-41 (2011)
Article DOI: 10.1021/jm101625x
BindingDB Entry DOI: 10.7270/Q2R49P83 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data