 Found 900 hits with Last Name = 'hamilton' and Initial = 's'
Found 900 hits with Last Name = 'hamilton' and Initial = 's' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM23334

(3-(pyridin-3-yl)propyl (2S)-1-(3,3-dimethyl-2-oxop...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCC[C@H]1C(=O)OCCCc1cccnc1 |r| Show InChI InChI=1S/C20H28N2O4/c1-4-20(2,3)17(23)18(24)22-12-6-10-16(22)19(25)26-13-7-9-15-8-5-11-21-14-15/h5,8,11,14,16H,4,6-7,9-10,12-13H2,1-3H3/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| 0.0100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Ability to inhibit peptidyl-prolyl isomerase (PPIase, or rotamase) activity of FK506 binding protein 12 |
Bioorg Med Chem Lett 12: 1429-33 (2002)
BindingDB Entry DOI: 10.7270/Q23J3C9S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM36609
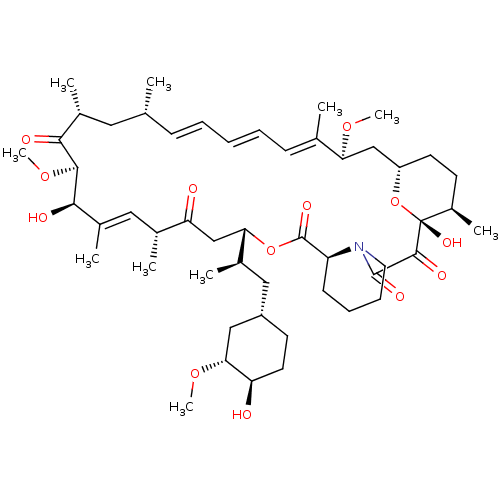
(Rapamycin C-7, analog 4 | SIROLIMUS | US11603377, ...)Show SMILES CO[C@@H]1C[C@H](C[C@@H](C)[C@@H]2CC(=O)[C@H](C)\C=C(C)\[C@@H](O)[C@@H](OC)C(=O)[C@H](C)C[C@H](C)\C=C\C=C\C=C(C)\[C@H](C[C@@H]3CC[C@@H](C)[C@@](O)(O3)C(=O)C(=O)N3CCCC[C@H]3C(=O)O2)OC)CC[C@H]1O |c:14,33,t:29,31| Show InChI InChI=1S/C51H79NO13/c1-30-16-12-11-13-17-31(2)42(61-8)28-38-21-19-36(7)51(60,65-38)48(57)49(58)52-23-15-14-18-39(52)50(59)64-43(33(4)26-37-20-22-40(53)44(27-37)62-9)29-41(54)32(3)25-35(6)46(56)47(63-10)45(55)34(5)24-30/h11-13,16-17,25,30,32-34,36-40,42-44,46-47,53,56,60H,14-15,18-24,26-29H2,1-10H3/b13-11+,16-12+,31-17+,35-25+/t30-,32-,33-,34-,36-,37+,38+,39+,40-,42+,43+,44-,46-,47+,51-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50030448

(8-DEETHYL-8-[BUT-3-ENYL]-ASCOMYCIN | CHEMBL269732 ...)Show SMILES CO[C@@H]1C[C@@H](CC[C@H]1O)\C=C(/C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@]2(O)O[C@@H]([C@H](C[C@H]2C)OC)[C@H](C[C@@H](C)C\C(C)=C\[C@@H](CC=C)C(=O)C[C@H](O)[C@H]1C)OC |r,t:45| Show InChI InChI=1S/C44H69NO12/c1-10-13-31-19-25(2)18-26(3)20-37(54-8)40-38(55-9)22-28(5)44(52,57-40)41(49)42(50)45-17-12-11-14-32(45)43(51)56-39(29(6)34(47)24-35(31)48)27(4)21-30-15-16-33(46)36(23-30)53-7/h10,19,21,26,28-34,36-40,46-47,52H,1,11-18,20,22-24H2,2-9H3/b25-19+,27-21+/t26-,28+,29+,30-,31+,32-,33+,34-,36+,37-,38-,39+,40+,44+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
PDB
Article
PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068608
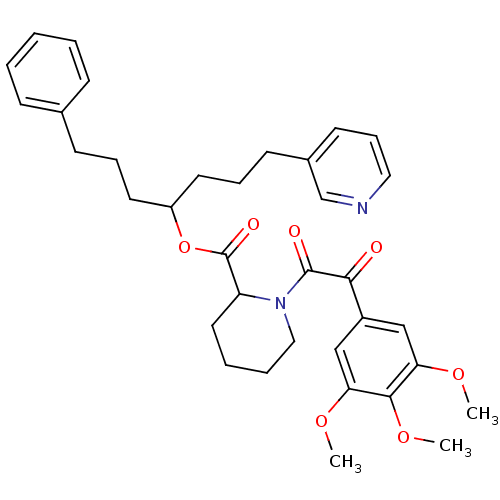
(1-[2-Oxo-2-(3,4,5-trimethoxy-phenyl)-acetyl]-piper...)Show SMILES COc1cc(cc(OC)c1OC)C(=O)C(=O)N1CCCCC1C(=O)OC(CCCc1ccccc1)CCCc1cccnc1 Show InChI InChI=1S/C35H42N2O7/c1-41-30-22-27(23-31(42-2)33(30)43-3)32(38)34(39)37-21-8-7-19-29(37)35(40)44-28(17-9-14-25-12-5-4-6-13-25)18-10-15-26-16-11-20-36-24-26/h4-6,11-13,16,20,22-24,28-29H,7-10,14-15,17-19,21H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50408684
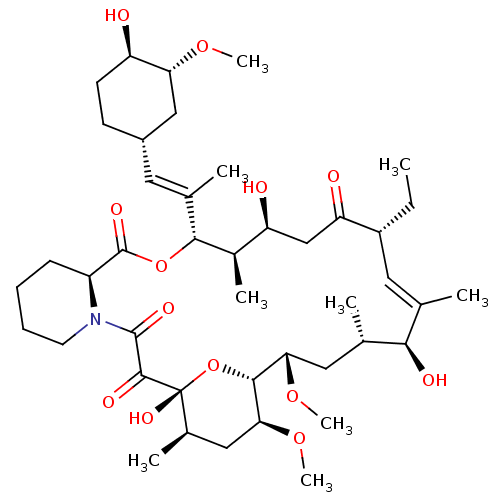
(CHEMBL2052020 | L-685818)Show SMILES CC[C@@H]1\C=C(C)/[C@@H](O)[C@@H](C)C[C@H](OC)[C@H]2O[C@](O)([C@H](C)C[C@@H]2OC)C(=O)C(=O)N2CCCC[C@H]2C(=O)O[C@@H]([C@H](C)[C@@H](O)CC1=O)C(\C)=C\[C@@H]1CC[C@@H](O)[C@@H](C1)OC |t:3| Show InChI InChI=1S/C43H69NO13/c1-10-29-18-23(2)37(48)24(3)19-35(54-8)39-36(55-9)20-26(5)43(52,57-39)40(49)41(50)44-16-12-11-13-30(44)42(51)56-38(27(6)32(46)22-33(29)47)25(4)17-28-14-15-31(45)34(21-28)53-7/h17-18,24,26-32,34-39,45-46,48,52H,10-16,19-22H2,1-9H3/b23-18-,25-17+/t24-,26+,27+,28-,29+,30-,31+,32-,34+,35-,36-,37+,38+,39+,43+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidylprolyl isomerase
(Gallus gallus) | BDBM50068597
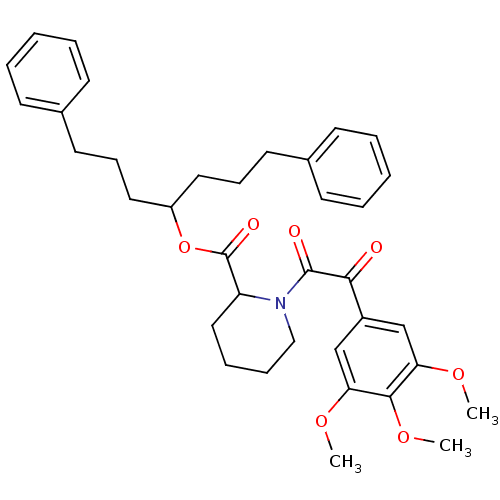
(1-[2-Oxo-2-(3,4,5-trimethoxy-phenyl)-acetyl]-piper...)Show SMILES COc1cc(cc(OC)c1OC)C(=O)C(=O)N1CCCCC1C(=O)OC(CCCc1ccccc1)CCCc1ccccc1 Show InChI InChI=1S/C36H43NO7/c1-41-31-24-28(25-32(42-2)34(31)43-3)33(38)35(39)37-23-11-10-22-30(37)36(40)44-29(20-12-18-26-14-6-4-7-15-26)21-13-19-27-16-8-5-9-17-27/h4-9,14-17,24-25,29-30H,10-13,18-23H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of binding to FKBP12 receptor |
Bioorg Med Chem Lett 7: 1785-1790 (1997)
Article DOI: 10.1016/S0960-894X(97)00304-1
BindingDB Entry DOI: 10.7270/Q2PZ58TF |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068556
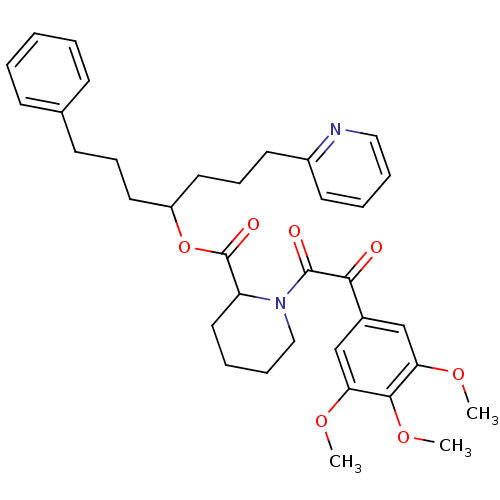
(1-[2-Oxo-2-(3,4,5-trimethoxy-phenyl)-acetyl]-piper...)Show SMILES COc1cc(cc(OC)c1OC)C(=O)C(=O)N1CCCCC1C(=O)OC(CCCc1ccccc1)CCCc1ccccn1 Show InChI InChI=1S/C35H42N2O7/c1-41-30-23-26(24-31(42-2)33(30)43-3)32(38)34(39)37-22-10-8-20-29(37)35(40)44-28(18-11-15-25-13-5-4-6-14-25)19-12-17-27-16-7-9-21-36-27/h4-7,9,13-14,16,21,23-24,28-29H,8,10-12,15,17-20,22H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50113103
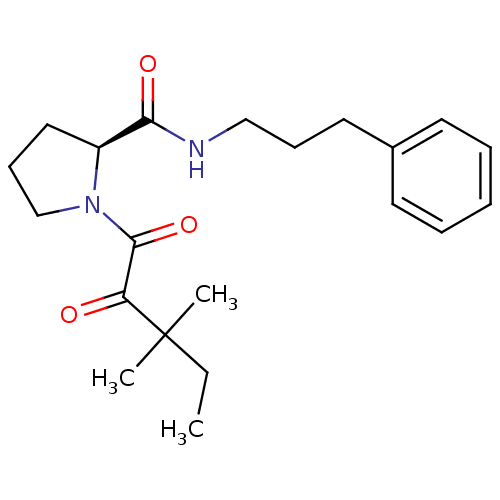
(1-(3,3-Dimethyl-2-oxo-pentanoyl)-pyrrolidine-2-car...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCC[C@H]1C(=O)NCCCc1ccccc1 Show InChI InChI=1S/C21H30N2O3/c1-4-21(2,3)18(24)20(26)23-15-9-13-17(23)19(25)22-14-8-12-16-10-6-5-7-11-16/h5-7,10-11,17H,4,8-9,12-15H2,1-3H3,(H,22,25)/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Ability to inhibit peptidyl-prolyl isomerase (PPIase, or rotamase) activity of FK506 binding protein 12 |
Bioorg Med Chem Lett 12: 1429-33 (2002)
BindingDB Entry DOI: 10.7270/Q23J3C9S |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068597

(1-[2-Oxo-2-(3,4,5-trimethoxy-phenyl)-acetyl]-piper...)Show SMILES COc1cc(cc(OC)c1OC)C(=O)C(=O)N1CCCCC1C(=O)OC(CCCc1ccccc1)CCCc1ccccc1 Show InChI InChI=1S/C36H43NO7/c1-41-31-24-28(25-32(42-2)34(31)43-3)33(38)35(39)37-23-11-10-22-30(37)36(40)44-29(20-12-18-26-14-6-4-7-15-26)21-13-19-27-16-8-5-9-17-27/h4-9,14-17,24-25,29-30H,10-13,18-23H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068564
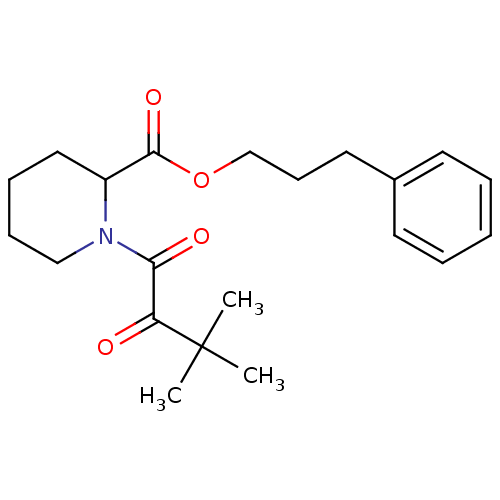
(1-(3,3-Dimethyl-2-oxo-butyryl)-piperidine-2-carbox...)Show SMILES CC(C)(C)C(=O)C(=O)N1CCCCC1C(=O)OCCCc1ccccc1 Show InChI InChI=1S/C21H29NO4/c1-21(2,3)18(23)19(24)22-14-8-7-13-17(22)20(25)26-15-9-12-16-10-5-4-6-11-16/h4-6,10-11,17H,7-9,12-15H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidylprolyl isomerase
(Gallus gallus) | BDBM50068564

(1-(3,3-Dimethyl-2-oxo-butyryl)-piperidine-2-carbox...)Show SMILES CC(C)(C)C(=O)C(=O)N1CCCCC1C(=O)OCCCc1ccccc1 Show InChI InChI=1S/C21H29NO4/c1-21(2,3)18(23)19(24)22-14-8-7-13-17(22)20(25)26-15-9-12-16-10-5-4-6-11-16/h4-6,10-11,17H,7-9,12-15H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of binding to FKBP12 receptor |
Bioorg Med Chem Lett 7: 1785-1790 (1997)
Article DOI: 10.1016/S0960-894X(97)00304-1
BindingDB Entry DOI: 10.7270/Q2PZ58TF |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068590

(1-(3,3-Dimethyl-2-oxo-butyryl)-pyrrolidine-2-carbo...)Show InChI InChI=1S/C19H26N2O4/c1-19(2,3)16(22)17(23)21-11-5-9-15(21)18(24)25-12-6-8-14-7-4-10-20-13-14/h4,7,10,13,15H,5-6,8-9,11-12H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068573
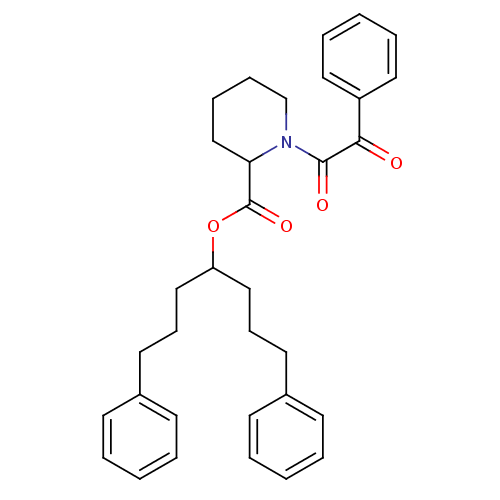
(1-(2-Oxo-2-phenyl-acetyl)-piperidine-2-carboxylic ...)Show SMILES O=C(OC(CCCc1ccccc1)CCCc1ccccc1)C1CCCCN1C(=O)C(=O)c1ccccc1 Show InChI InChI=1S/C33H37NO4/c35-31(28-20-8-3-9-21-28)32(36)34-25-11-10-24-30(34)33(37)38-29(22-12-18-26-14-4-1-5-15-26)23-13-19-27-16-6-2-7-17-27/h1-9,14-17,20-21,29-30H,10-13,18-19,22-25H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidylprolyl isomerase
(Gallus gallus) | BDBM50068573

(1-(2-Oxo-2-phenyl-acetyl)-piperidine-2-carboxylic ...)Show SMILES O=C(OC(CCCc1ccccc1)CCCc1ccccc1)C1CCCCN1C(=O)C(=O)c1ccccc1 Show InChI InChI=1S/C33H37NO4/c35-31(28-20-8-3-9-21-28)32(36)34-25-11-10-24-30(34)33(37)38-29(22-12-18-26-14-4-1-5-15-26)23-13-19-27-16-6-2-7-17-27/h1-9,14-17,20-21,29-30H,10-13,18-19,22-25H2 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of binding to FKBP12 receptor |
Bioorg Med Chem Lett 7: 1785-1790 (1997)
Article DOI: 10.1016/S0960-894X(97)00304-1
BindingDB Entry DOI: 10.7270/Q2PZ58TF |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068578
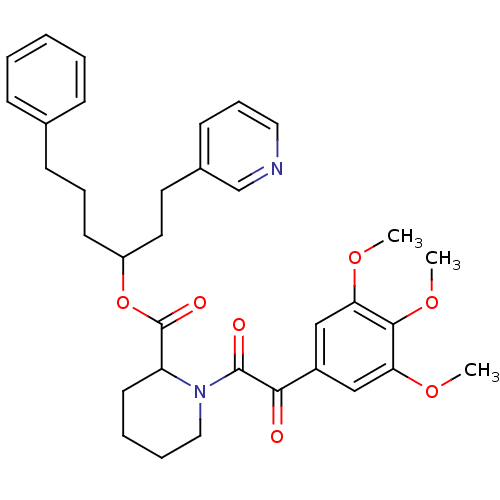
(1-[2-Oxo-2-(3,4,5-trimethoxy-phenyl)-acetyl]-piper...)Show SMILES COc1cc(cc(OC)c1OC)C(=O)C(=O)N1CCCCC1C(=O)OC(CCCc1ccccc1)CCc1cccnc1 Show InChI InChI=1S/C34H40N2O7/c1-40-29-21-26(22-30(41-2)32(29)42-3)31(37)33(38)36-20-8-7-16-28(36)34(39)43-27(18-17-25-14-10-19-35-23-25)15-9-13-24-11-5-4-6-12-24/h4-6,10-12,14,19,21-23,27-28H,7-9,13,15-18,20H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidylprolyl isomerase
(Gallus gallus) | BDBM50068582
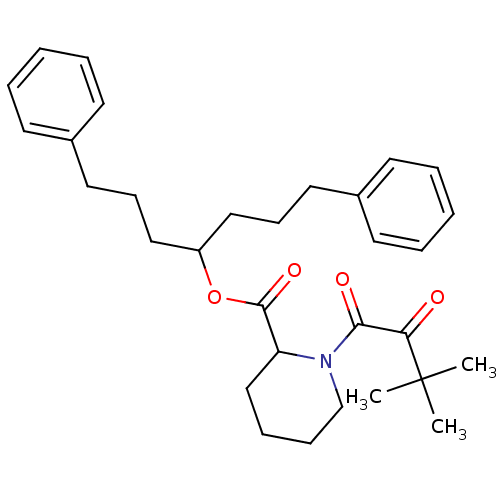
(1-(3,3-Dimethyl-2-oxo-butyryl)-piperidine-2-carbox...)Show SMILES CC(C)(C)C(=O)C(=O)N1CCCCC1C(=O)OC(CCCc1ccccc1)CCCc1ccccc1 Show InChI InChI=1S/C31H41NO4/c1-31(2,3)28(33)29(34)32-23-11-10-22-27(32)30(35)36-26(20-12-18-24-14-6-4-7-15-24)21-13-19-25-16-8-5-9-17-25/h4-9,14-17,26-27H,10-13,18-23H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of binding to FKBP12 receptor |
Bioorg Med Chem Lett 7: 1785-1790 (1997)
Article DOI: 10.1016/S0960-894X(97)00304-1
BindingDB Entry DOI: 10.7270/Q2PZ58TF |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068582

(1-(3,3-Dimethyl-2-oxo-butyryl)-piperidine-2-carbox...)Show SMILES CC(C)(C)C(=O)C(=O)N1CCCCC1C(=O)OC(CCCc1ccccc1)CCCc1ccccc1 Show InChI InChI=1S/C31H41NO4/c1-31(2,3)28(33)29(34)32-23-11-10-22-27(32)30(35)36-26(20-12-18-24-14-6-4-7-15-24)21-13-19-25-16-8-5-9-17-25/h4-9,14-17,26-27H,10-13,18-23H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50116632
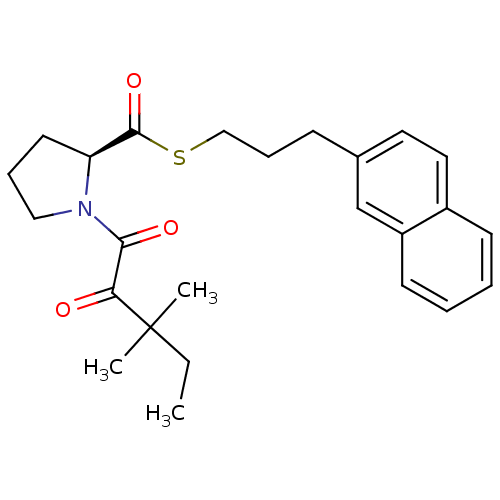
(1-(3,3-Dimethyl-2-oxo-pentanoyl)-pyrrolidine-2-car...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCC[C@H]1C(=O)SCCCc1ccc2ccccc2c1 Show InChI InChI=1S/C25H31NO3S/c1-4-25(2,3)22(27)23(28)26-15-7-12-21(26)24(29)30-16-8-9-18-13-14-19-10-5-6-11-20(19)17-18/h5-6,10-11,13-14,17,21H,4,7-9,12,15-16H2,1-3H3/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 6 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against FK506 binding protein 12 |
J Med Chem 45: 3549-57 (2002)
BindingDB Entry DOI: 10.7270/Q2416WC5 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50116633

(1-(3,3-Dimethyl-2-oxo-pentanoyl)-pyrrolidine-2-car...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCC[C@H]1C(=O)SCCC(c1ccccc1)c1ccccc1 Show InChI InChI=1S/C27H33NO3S/c1-4-27(2,3)24(29)25(30)28-18-11-16-23(28)26(31)32-19-17-22(20-12-7-5-8-13-20)21-14-9-6-10-15-21/h5-10,12-15,22-23H,4,11,16-19H2,1-3H3/t23-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 7 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against FK506 binding protein 12 |
J Med Chem 45: 3549-57 (2002)
BindingDB Entry DOI: 10.7270/Q2416WC5 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068547
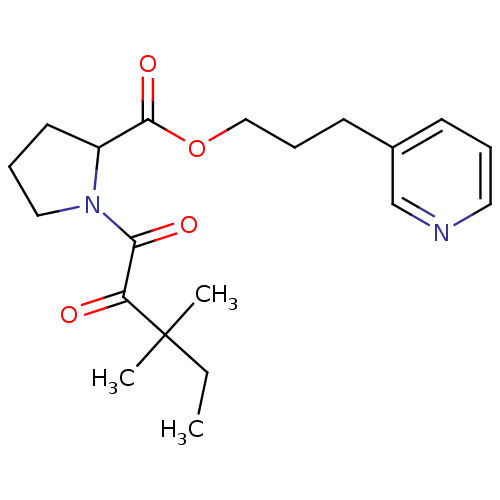
(1-(3,3-Dimethyl-2-oxo-pentanoyl)-pyrrolidine-2-car...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCCC1C(=O)OCCCc1cccnc1 Show InChI InChI=1S/C20H28N2O4/c1-4-20(2,3)17(23)18(24)22-12-6-10-16(22)19(25)26-13-7-9-15-8-5-11-21-14-15/h5,8,11,14,16H,4,6-7,9-10,12-13H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 7.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Bos taurus (bovine)) | BDBM23334

(3-(pyridin-3-yl)propyl (2S)-1-(3,3-dimethyl-2-oxop...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCC[C@H]1C(=O)OCCCc1cccnc1 |r| Show InChI InChI=1S/C20H28N2O4/c1-4-20(2,3)17(23)18(24)22-12-6-10-16(22)19(25)26-13-7-9-15-8-5-11-21-14-15/h5,8,11,14,16H,4,6-7,9-10,12-13H2,1-3H3/t16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 7.5 | -42.5 | n/a | n/a | n/a | n/a | n/a | 8.0 | 0 |
Guilford Pharmaceuticals, Inc.
| Assay Description
PPIase(Rotamase) activity of FKBP12 was assayed using the peptide N-succinyl Ala-Leu-Pro-Phe p-nitroanilide as substrate. It is based on the observat... |
J Med Chem 45: 3558-68 (2002)
Article DOI: 10.1021/jm0200456
BindingDB Entry DOI: 10.7270/Q2GH9G7G |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM23334

(3-(pyridin-3-yl)propyl (2S)-1-(3,3-dimethyl-2-oxop...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCC[C@H]1C(=O)OCCCc1cccnc1 |r| Show InChI InChI=1S/C20H28N2O4/c1-4-20(2,3)17(23)18(24)22-12-6-10-16(22)19(25)26-13-7-9-15-8-5-11-21-14-15/h5,8,11,14,16H,4,6-7,9-10,12-13H2,1-3H3/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| 7.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against FK506 binding protein 12 |
J Med Chem 45: 3549-57 (2002)
BindingDB Entry DOI: 10.7270/Q2416WC5 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Peptidylprolyl isomerase
(Gallus gallus) | BDBM50068610

(1-(3,3-Dimethyl-2-oxo-pentanoyl)-piperidine-2-carb...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCCCC1C(=O)OCCC(c1ccccc1)c1ccccc1 Show InChI InChI=1S/C28H35NO4/c1-4-28(2,3)25(30)26(31)29-19-12-11-17-24(29)27(32)33-20-18-23(21-13-7-5-8-14-21)22-15-9-6-10-16-22/h5-10,13-16,23-24H,4,11-12,17-20H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 8.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of binding to FKBP12 receptor |
Bioorg Med Chem Lett 7: 1785-1790 (1997)
Article DOI: 10.1016/S0960-894X(97)00304-1
BindingDB Entry DOI: 10.7270/Q2PZ58TF |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068610

(1-(3,3-Dimethyl-2-oxo-pentanoyl)-piperidine-2-carb...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCCCC1C(=O)OCCC(c1ccccc1)c1ccccc1 Show InChI InChI=1S/C28H35NO4/c1-4-28(2,3)25(30)26(31)29-19-12-11-17-24(29)27(32)33-20-18-23(21-13-7-5-8-14-21)22-15-9-6-10-16-22/h5-10,13-16,23-24H,4,11-12,17-20H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 8.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50116636

(1-(3,3-Dimethyl-2-oxo-pentanoyl)-pyrrolidine-2-car...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCC[C@H]1C(=O)SCCCc1cccnc1 Show InChI InChI=1S/C20H28N2O3S/c1-4-20(2,3)17(23)18(24)22-12-6-10-16(22)19(25)26-13-7-9-15-8-5-11-21-14-15/h5,8,11,14,16H,4,6-7,9-10,12-13H2,1-3H3/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 8.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against FK506 binding protein 12 |
J Med Chem 45: 3549-57 (2002)
BindingDB Entry DOI: 10.7270/Q2416WC5 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50116630

(1-(3,3-Dimethyl-2-oxo-pentanoyl)-pyrrolidine-2-car...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCC[C@H]1C(=O)SCCCc1ccc(OC)cc1 Show InChI InChI=1S/C22H31NO4S/c1-5-22(2,3)19(24)20(25)23-14-6-9-18(23)21(26)28-15-7-8-16-10-12-17(27-4)13-11-16/h10-13,18H,5-9,14-15H2,1-4H3/t18-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 8.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against FK506 binding protein 12 |
J Med Chem 45: 3549-57 (2002)
BindingDB Entry DOI: 10.7270/Q2416WC5 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068576
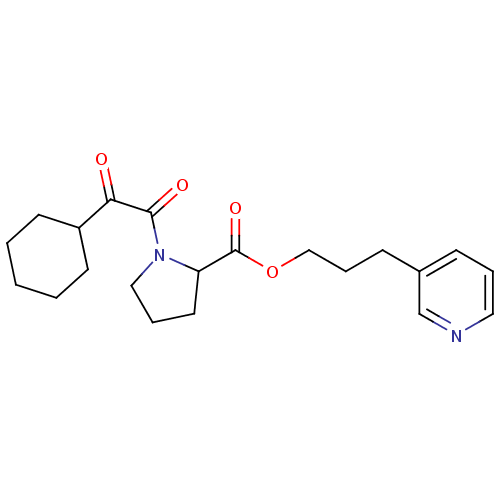
(1-(2-Cyclohexyl-2-oxo-acetyl)-pyrrolidine-2-carbox...)Show InChI InChI=1S/C21H28N2O4/c24-19(17-9-2-1-3-10-17)20(25)23-13-5-11-18(23)21(26)27-14-6-8-16-7-4-12-22-15-16/h4,7,12,15,17-18H,1-3,5-6,8-11,13-14H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase A
(Homo sapiens (Human)) | BDBM50150950

(30-Ethyl-33-(1-hydroxy-2-methyl-hex-4-enyl)-6,9,18...)Show SMILES CC[C@@H]1NC(=O)[C@H](C(O)[C@H](C)C\C=C\C)N(C)C(=O)[C@H](C(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](CC(C)C)N(C)C(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](NC(=O)[C@@H](CC(C)C)N(C)C(=O)CN(C)C1=O)C(C)C Show InChI InChI=1S/C62H111N11O12/c1-25-27-28-40(15)52(75)51-56(79)65-43(26-2)58(81)67(18)33-48(74)68(19)44(29-34(3)4)55(78)66-49(38(11)12)61(84)69(20)45(30-35(5)6)54(77)63-41(16)53(76)64-42(17)57(80)70(21)46(31-36(7)8)59(82)71(22)47(32-37(9)10)60(83)72(23)50(39(13)14)62(85)73(51)24/h25,27,34-47,49-52,75H,26,28-33H2,1-24H3,(H,63,77)(H,64,76)(H,65,79)(H,66,78)/b27-25+/t40-,41+,42-,43+,44-,45+,46-,47+,49-,50+,51+,52?/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of cyclophilin A rotamase |
Bioorg Med Chem Lett 14: 4549-51 (2004)
Article DOI: 10.1016/j.bmcl.2004.06.028
BindingDB Entry DOI: 10.7270/Q2WH2QQQ |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50116631
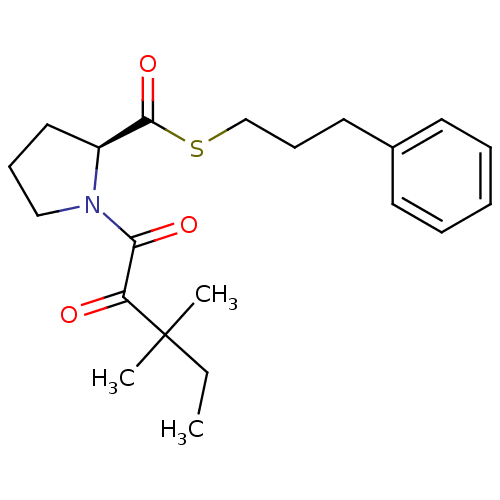
(1-(3,3-Dimethyl-2-oxo-pentanoyl)-pyrrolidine-2-car...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCC[C@H]1C(=O)SCCCc1ccccc1 Show InChI InChI=1S/C21H29NO3S/c1-4-21(2,3)18(23)19(24)22-14-8-13-17(22)20(25)26-15-9-12-16-10-6-5-7-11-16/h5-7,10-11,17H,4,8-9,12-15H2,1-3H3/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against FK506 binding protein 12 |
J Med Chem 45: 3549-57 (2002)
BindingDB Entry DOI: 10.7270/Q2416WC5 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068586
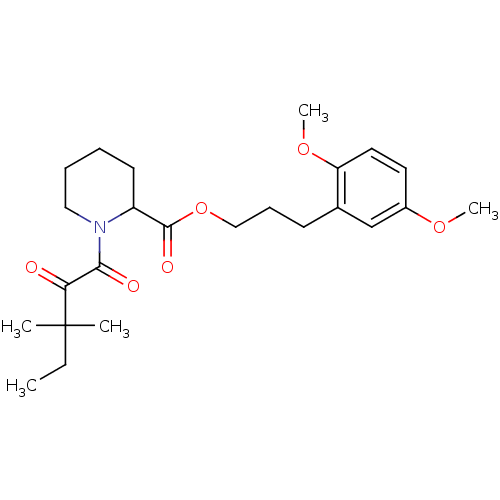
(1-(3,3-Dimethyl-2-oxo-pentanoyl)-piperidine-2-carb...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCCCC1C(=O)OCCCc1cc(OC)ccc1OC Show InChI InChI=1S/C24H35NO6/c1-6-24(2,3)21(26)22(27)25-14-8-7-11-19(25)23(28)31-15-9-10-17-16-18(29-4)12-13-20(17)30-5/h12-13,16,19H,6-11,14-15H2,1-5H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50113058

(1-(Adamantan-1-ylcarbamoyl)-piperidine-2-carboxyli...)Show SMILES COc1ccc(CCCOC(=O)C2CCCCN2C(=O)NC23CC4CC(CC(C4)C2)C3)c(OC)c1 |TLB:24:25:29:23.22.28,THB:24:23:29:25.30.26,26:25:22:27.29.28,26:27:22:25.30.24| Show InChI InChI=1S/C28H40N2O5/c1-33-23-9-8-22(25(15-23)34-2)6-5-11-35-26(31)24-7-3-4-10-30(24)27(32)29-28-16-19-12-20(17-28)14-21(13-19)18-28/h8-9,15,19-21,24H,3-7,10-14,16-18H2,1-2H3,(H,29,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of peptidyl-prolyl isomerase (PPIase, or rotamase) activity of FK506 binding protein 12 |
Bioorg Med Chem Lett 12: 1421-8 (2002)
BindingDB Entry DOI: 10.7270/Q279441H |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068601
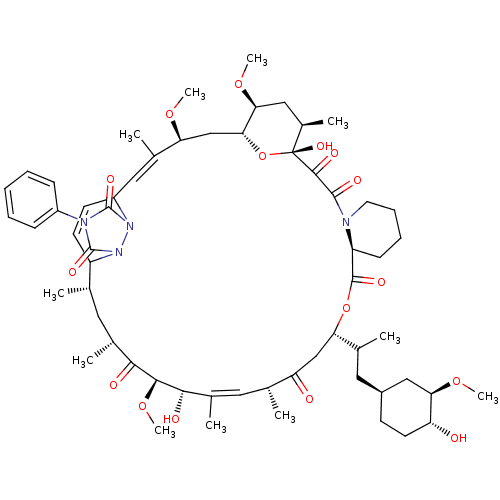
(CHEMBL265972 | WAY-124466)Show SMILES CO[C@@H]1C[C@H](CC(C)[C@@H]2CC(=O)[C@H](C)\C=C(C)\[C@H](O)[C@@H](OC)C(=O)[C@H](C)C[C@H](C)C3C=CC(\C=C(C)\[C@H](C[C@H]4O[C@](O)([C@H](C)C[C@@H]4OC)C(=O)C(=O)N4CCCC[C@H]4C(=O)O2)OC)n2n3c(=O)n(-c3ccccc3)c2=O)CC[C@H]1O |c:14,30,33| Show InChI InChI=1S/C60H86N4O16/c1-33-25-37(5)52(67)54(78-11)53(68)38(6)26-34(2)46(66)31-48(35(3)27-40-20-23-45(65)49(30-40)76-9)79-57(71)44-19-15-16-24-61(44)56(70)55(69)60(74)39(7)29-50(77-10)51(80-60)32-47(75-8)36(4)28-42-21-22-43(33)64-59(73)62(58(72)63(42)64)41-17-13-12-14-18-41/h12-14,17-18,21-22,26,28,33-35,37,39-40,42-45,47-51,53-54,65,68,74H,15-16,19-20,23-25,27,29-32H2,1-11H3/b36-28+,38-26+/t33-,34+,35?,37+,39+,40-,42?,43?,44-,45+,47-,48-,49+,50-,51+,53-,54-,60+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 12 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50116635

(1-(3,3-Dimethyl-2-oxo-pentanoyl)-pyrrolidine-2-car...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCC[C@H]1C(=O)SCCc1ccccc1 Show InChI InChI=1S/C20H27NO3S/c1-4-20(2,3)17(22)18(23)21-13-8-11-16(21)19(24)25-14-12-15-9-6-5-7-10-15/h5-7,9-10,16H,4,8,11-14H2,1-3H3/t16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 12.2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against FK506 binding protein 12 |
J Med Chem 45: 3549-57 (2002)
BindingDB Entry DOI: 10.7270/Q2416WC5 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068545

(1-[2-Oxo-2-(3,4,5-trimethoxy-phenyl)-acetyl]-piper...)Show SMILES COc1ccc(CCCCOC(=O)C2CCCCN2C(=O)C(=O)c2cc(OC)c(OC)c(OC)c2)cc1 Show InChI InChI=1S/C28H35NO8/c1-33-21-13-11-19(12-14-21)9-6-8-16-37-28(32)22-10-5-7-15-29(22)27(31)25(30)20-17-23(34-2)26(36-4)24(18-20)35-3/h11-14,17-18,22H,5-10,15-16H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 13 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Lactoylglutathione lyase
(Homo sapiens (Human)) | BDBM50092826
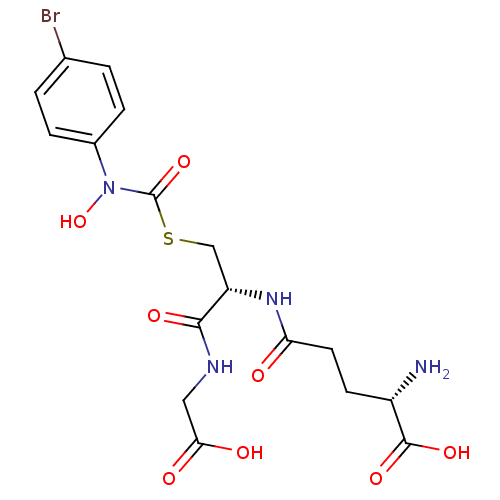
((2S)-2-amino-5-{[(1R)-1-[({[(4-bromophenyl)(hydrox...)Show SMILES N[C@@H](CCC(=O)N[C@@H](CSC(=O)N(O)c1ccc(Br)cc1)C(=O)NCC(O)=O)C(O)=O Show InChI InChI=1S/C17H21BrN4O8S/c18-9-1-3-10(4-2-9)22(30)17(29)31-8-12(15(26)20-7-14(24)25)21-13(23)6-5-11(19)16(27)28/h1-4,11-12,30H,5-8,19H2,(H,20,26)(H,21,23)(H,24,25)(H,27,28)/t11-,12-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
MMDB
PC cid
PC sid
UniChem
Similars
| PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Maryland
Curated by ChEMBL
| Assay Description
Binding affinity for Glyoxalase I |
J Med Chem 43: 3981-6 (2000)
BindingDB Entry DOI: 10.7270/Q2BC3XS4 |
More data for this
Ligand-Target Pair | |
Lactoylglutathione lyase
(Homo sapiens (Human)) | BDBM50039111
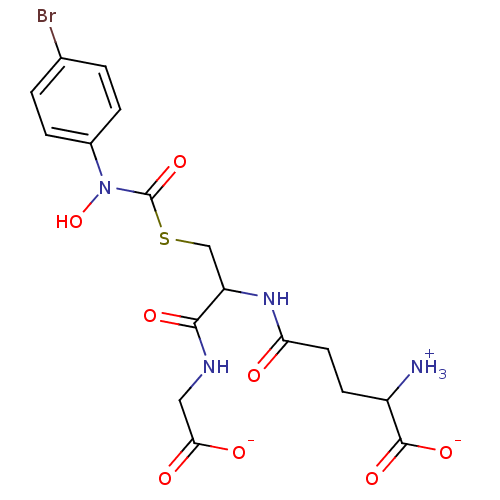
(S-(N-Hydroxy-N-(4-bromophenyl)carbamoyl)glutathion...)Show SMILES [NH3+]C(CCC(=O)NC(CSC(=O)N(O)c1ccc(Br)cc1)C(=O)NCC([O-])=O)C([O-])=O Show InChI InChI=1S/C17H21BrN4O8S/c18-9-1-3-10(4-2-9)22(30)17(29)31-8-12(15(26)20-7-14(24)25)21-13(23)6-5-11(19)16(27)28/h1-4,11-12,30H,5-8,19H2,(H,20,26)(H,21,23)(H,24,25)(H,27,28)/p-1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Maryland
Curated by ChEMBL
| Assay Description
Tested for inhibitory activity against human erythrocyte glyoxalase I |
J Med Chem 37: 2161-6 (1994)
BindingDB Entry DOI: 10.7270/Q2TT4RKK |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068553
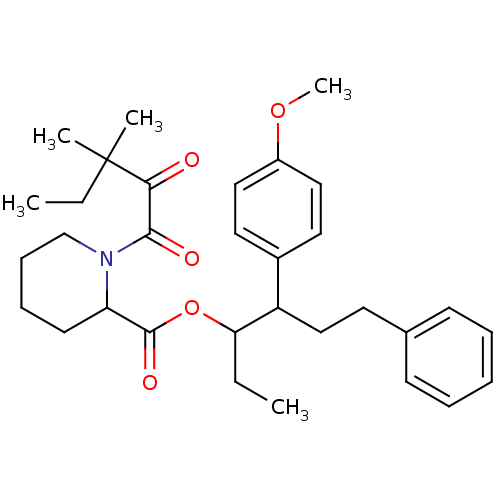
(1-(3,3-Dimethyl-2-oxo-pentanoyl)-piperidine-2-carb...)Show SMILES CCC(OC(=O)C1CCCCN1C(=O)C(=O)C(C)(C)CC)C(CCc1ccccc1)c1ccc(OC)cc1 Show InChI InChI=1S/C32H43NO5/c1-6-28(26(21-16-23-13-9-8-10-14-23)24-17-19-25(37-5)20-18-24)38-31(36)27-15-11-12-22-33(27)30(35)29(34)32(3,4)7-2/h8-10,13-14,17-20,26-28H,6-7,11-12,15-16,21-22H2,1-5H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidylprolyl isomerase
(Gallus gallus) | BDBM50289698

(1-(3,3-Dimethyl-2-oxo-pentanoyl)-piperidine-2-carb...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCCCC1C(=O)OC(CCCc1ccccc1)CCc1ccc(OC)cc1 Show InChI InChI=1S/C32H43NO5/c1-5-32(2,3)29(34)30(35)33-23-10-9-16-28(33)31(36)38-27(15-11-14-24-12-7-6-8-13-24)22-19-25-17-20-26(37-4)21-18-25/h6-8,12-13,17-18,20-21,27-28H,5,9-11,14-16,19,22-23H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of binding to FKBP12 receptor |
Bioorg Med Chem Lett 7: 1785-1790 (1997)
Article DOI: 10.1016/S0960-894X(97)00304-1
BindingDB Entry DOI: 10.7270/Q2PZ58TF |
More data for this
Ligand-Target Pair | |
Lactoylglutathione lyase
(Homo sapiens (Human)) | BDBM50092827
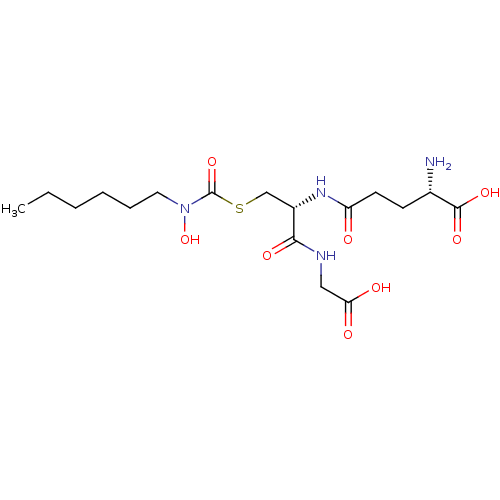
(CHEMBL127840 | S-(N-hexyl-N-hydroxycarbamoyl)gluta...)Show SMILES CCCCCCN(O)C(=O)SC[C@H](NC(=O)CC[C@H](N)C(O)=O)C(=O)NCC(O)=O Show InChI InChI=1S/C17H30N4O8S/c1-2-3-4-5-8-21(29)17(28)30-10-12(15(25)19-9-14(23)24)20-13(22)7-6-11(18)16(26)27/h11-12,29H,2-10,18H2,1H3,(H,19,25)(H,20,22)(H,23,24)(H,26,27)/t11-,12-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Maryland
Curated by ChEMBL
| Assay Description
Binding affinity for Glyoxalase I |
J Med Chem 43: 3981-6 (2000)
BindingDB Entry DOI: 10.7270/Q2BC3XS4 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068577

(1-(3,3-Dimethyl-2-oxo-pentanoyl)-piperidine-2-carb...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCCCC1C(=O)O[C@H](CCc1ccccc1)c1ccccc1 Show InChI InChI=1S/C28H35NO4/c1-4-28(2,3)25(30)26(31)29-20-12-11-17-23(29)27(32)33-24(22-15-9-6-10-16-22)19-18-21-13-7-5-8-14-21/h5-10,13-16,23-24H,4,11-12,17-20H2,1-3H3/t23?,24-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068605
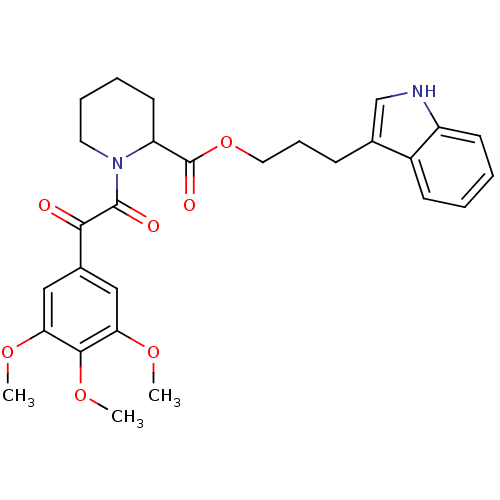
(1-[2-Oxo-2-(3,4,5-trimethoxy-phenyl)-acetyl]-piper...)Show SMILES COc1cc(cc(OC)c1OC)C(=O)C(=O)N1CCCCC1C(=O)OCCCc1c[nH]c2ccccc12 Show InChI InChI=1S/C28H32N2O7/c1-34-23-15-19(16-24(35-2)26(23)36-3)25(31)27(32)30-13-7-6-12-22(30)28(33)37-14-8-9-18-17-29-21-11-5-4-10-20(18)21/h4-5,10-11,15-17,22,29H,6-9,12-14H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 17 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase A
(Homo sapiens (Human)) | BDBM50150948

(12-Ethyl-3-(1-hydroxy-ethyl)-15-(1-hydroxy-2-methy...)Show SMILES CC[C@@H]1NC(=O)[C@H](C(O)[C@H](C)C\C=C\C)N(C)C(=O)[C@H](C(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](CC(C)C)N(C)C(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@](C)(NC(=O)[C@@H](CC(C)C)N(C)C(=O)CN(C)C1=O)C(C)O Show InChI InChI=1S/C62H111N11O13/c1-25-27-28-39(13)51(76)50-55(80)65-43(26-2)57(82)67(18)33-48(75)68(19)45(30-35(5)6)54(79)66-62(17,42(16)74)61(86)71(22)44(29-34(3)4)53(78)63-40(14)52(77)64-41(15)56(81)69(20)46(31-36(7)8)58(83)70(21)47(32-37(9)10)59(84)72(23)49(38(11)12)60(85)73(50)24/h25,27,34-47,49-51,74,76H,26,28-33H2,1-24H3,(H,63,78)(H,64,77)(H,65,80)(H,66,79)/b27-25+/t39-,40+,41-,42?,43+,44+,45-,46-,47+,49+,50+,51?,62-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of cyclophilin A rotamase |
Bioorg Med Chem Lett 14: 4549-51 (2004)
Article DOI: 10.1016/j.bmcl.2004.06.028
BindingDB Entry DOI: 10.7270/Q2WH2QQQ |
More data for this
Ligand-Target Pair | |
Lactoylglutathione lyase
(Homo sapiens (Human)) | BDBM50092831
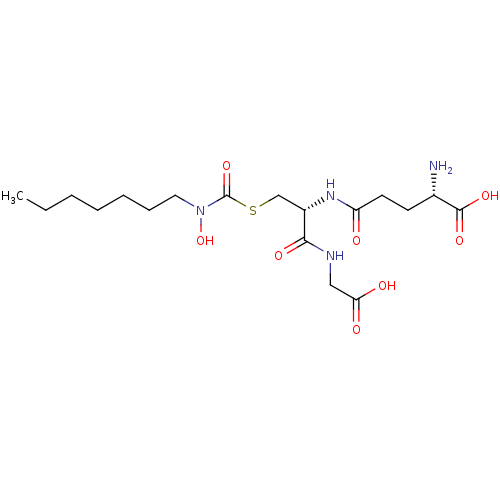
(CHEMBL128836 | S-(N-heptyl-N-hydroxycarbamoyl)glut...)Show SMILES CCCCCCCN(O)C(=O)SC[C@H](NC(=O)CC[C@H](N)C(O)=O)C(=O)NCC(O)=O Show InChI InChI=1S/C18H32N4O8S/c1-2-3-4-5-6-9-22(30)18(29)31-11-13(16(26)20-10-15(24)25)21-14(23)8-7-12(19)17(27)28/h12-13,30H,2-11,19H2,1H3,(H,20,26)(H,21,23)(H,24,25)(H,27,28)/t12-,13-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Maryland
Curated by ChEMBL
| Assay Description
Binding affinity for Glyoxalase I |
J Med Chem 43: 3981-6 (2000)
BindingDB Entry DOI: 10.7270/Q2BC3XS4 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068568
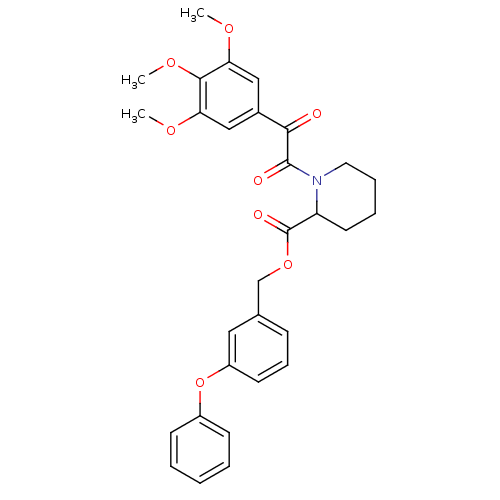
(1-[2-Oxo-2-(3,4,5-trimethoxy-phenyl)-acetyl]-piper...)Show SMILES COc1cc(cc(OC)c1OC)C(=O)C(=O)N1CCCCC1C(=O)OCc1cccc(Oc2ccccc2)c1 Show InChI InChI=1S/C30H31NO8/c1-35-25-17-21(18-26(36-2)28(25)37-3)27(32)29(33)31-15-8-7-14-24(31)30(34)38-19-20-10-9-13-23(16-20)39-22-11-5-4-6-12-22/h4-6,9-13,16-18,24H,7-8,14-15,19H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase A
(Homo sapiens (Human)) | BDBM50022815
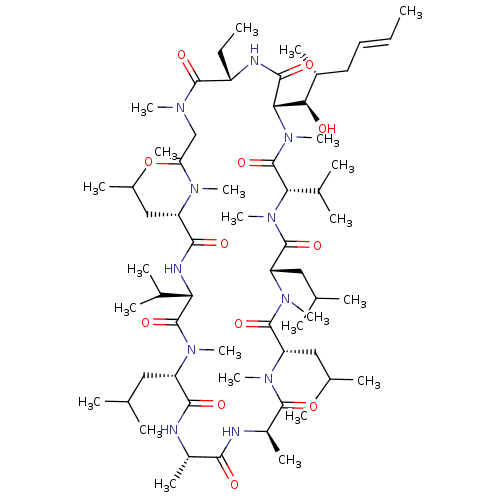
((3S,6S,9S,12R,15S,18S,21S,24S,30S,33S)-30-ethyl-33...)Show SMILES CC[C@@H]1NC(=O)[C@H]([C@H](O)[C@H](C)C\C=C\C)N(C)C(=O)[C@H](C(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](NC(=O)[C@H](CC(C)C)N(C)C(=O)CN(C)C1=O)C(C)C |r| Show InChI InChI=1S/C62H111N11O12/c1-25-27-28-40(15)52(75)51-56(79)65-43(26-2)58(81)67(18)33-48(74)68(19)44(29-34(3)4)55(78)66-49(38(11)12)61(84)69(20)45(30-35(5)6)54(77)63-41(16)53(76)64-42(17)57(80)70(21)46(31-36(7)8)59(82)71(22)47(32-37(9)10)60(83)72(23)50(39(13)14)62(85)73(51)24/h25,27,34-47,49-52,75H,26,28-33H2,1-24H3,(H,63,77)(H,64,76)(H,65,79)(H,66,78)/b27-25+/t40-,41+,42-,43+,44+,45+,46+,47+,49+,50+,51+,52-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of cyclophilin A rotamase |
Bioorg Med Chem Lett 14: 4549-51 (2004)
Article DOI: 10.1016/j.bmcl.2004.06.028
BindingDB Entry DOI: 10.7270/Q2WH2QQQ |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068594
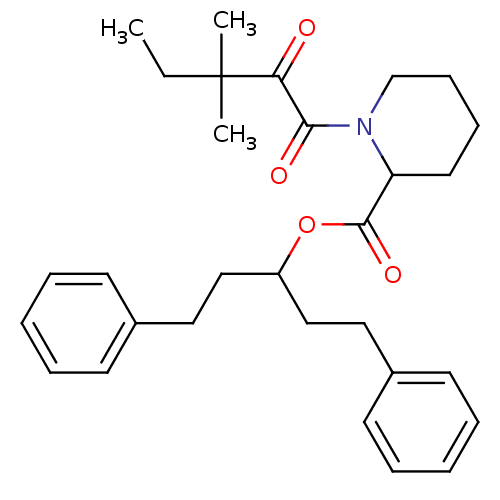
(1-(3,3-Dimethyl-2-oxo-pentanoyl)-piperidine-2-carb...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCCCC1C(=O)OC(CCc1ccccc1)CCc1ccccc1 Show InChI InChI=1S/C30H39NO4/c1-4-30(2,3)27(32)28(33)31-22-12-11-17-26(31)29(34)35-25(20-18-23-13-7-5-8-14-23)21-19-24-15-9-6-10-16-24/h5-10,13-16,25-26H,4,11-12,17-22H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50022815

((3S,6S,9S,12R,15S,18S,21S,24S,30S,33S)-30-ethyl-33...)Show SMILES CC[C@@H]1NC(=O)[C@H]([C@H](O)[C@H](C)C\C=C\C)N(C)C(=O)[C@H](C(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](NC(=O)[C@H](CC(C)C)N(C)C(=O)CN(C)C1=O)C(C)C |r| Show InChI InChI=1S/C62H111N11O12/c1-25-27-28-40(15)52(75)51-56(79)65-43(26-2)58(81)67(18)33-48(74)68(19)44(29-34(3)4)55(78)66-49(38(11)12)61(84)69(20)45(30-35(5)6)54(77)63-41(16)53(76)64-42(17)57(80)70(21)46(31-36(7)8)59(82)71(22)47(32-37(9)10)60(83)72(23)50(39(13)14)62(85)73(51)24/h25,27,34-47,49-52,75H,26,28-33H2,1-24H3,(H,63,77)(H,64,76)(H,65,79)(H,66,78)/b27-25+/t40-,41+,42-,43+,44+,45+,46+,47+,49+,50+,51+,52-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |
Peptidylprolyl isomerase
(Gallus gallus) | BDBM50068594

(1-(3,3-Dimethyl-2-oxo-pentanoyl)-piperidine-2-carb...)Show SMILES CCC(C)(C)C(=O)C(=O)N1CCCCC1C(=O)OC(CCc1ccccc1)CCc1ccccc1 Show InChI InChI=1S/C30H39NO4/c1-4-30(2,3)27(32)28(33)31-22-12-11-17-26(31)29(34)35-25(20-18-23-13-7-5-8-14-23)21-19-24-15-9-6-10-16-24/h5-10,13-16,25-26H,4,11-12,17-22H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/TrEMBL
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of binding to FKBP12 receptor |
Bioorg Med Chem Lett 7: 1785-1790 (1997)
Article DOI: 10.1016/S0960-894X(97)00304-1
BindingDB Entry DOI: 10.7270/Q2PZ58TF |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50113095
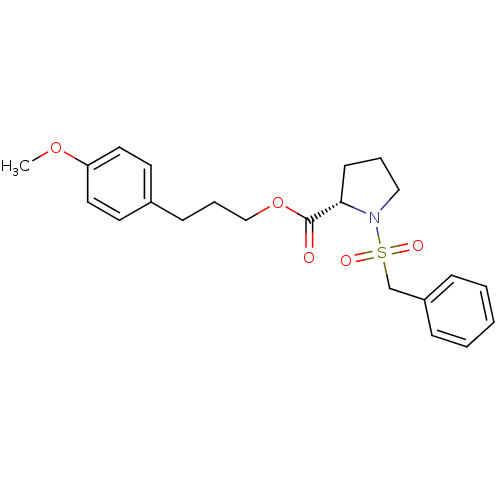
((S)-1-Phenylmethanesulfonyl-pyrrolidine-2-carboxyl...)Show SMILES COc1ccc(CCCOC(=O)[C@@H]2CCCN2S(=O)(=O)Cc2ccccc2)cc1 Show InChI InChI=1S/C22H27NO5S/c1-27-20-13-11-18(12-14-20)9-6-16-28-22(24)21-10-5-15-23(21)29(25,26)17-19-7-3-2-4-8-19/h2-4,7-8,11-14,21H,5-6,9-10,15-17H2,1H3/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of peptidyl-prolyl isomerase (PPIase, or rotamase) activity of FK506 binding protein 12 |
Bioorg Med Chem Lett 12: 1421-8 (2002)
BindingDB Entry DOI: 10.7270/Q279441H |
More data for this
Ligand-Target Pair | |
Peptidyl-prolyl cis-trans isomerase FKBP1A
(Homo sapiens (Human)) | BDBM50068591

(1-(3,3-Dimethyl-2-oxo-butyryl)-piperidine-2-carbox...)Show SMILES COc1ccc(CCCCOC(=O)C2CCCCN2C(=O)C(=O)C(C)(C)C)cc1 Show InChI InChI=1S/C23H33NO5/c1-23(2,3)20(25)21(26)24-15-7-5-10-19(24)22(27)29-16-8-6-9-17-11-13-18(28-4)14-12-17/h11-14,19H,5-10,15-16H2,1-4H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Guilford Pharmaceuticals, Inc.
Curated by ChEMBL
| Assay Description
Compound was tested for its ability to inhibit FK506 binding protein 12 rotamase activity |
J Med Chem 41: 5119-43 (1999)
Article DOI: 10.1021/jm980307x
BindingDB Entry DOI: 10.7270/Q22B8ZP8 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data