 Found 589 hits with Last Name = 'mano' and Initial = 'y'
Found 589 hits with Last Name = 'mano' and Initial = 'y' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM50292348
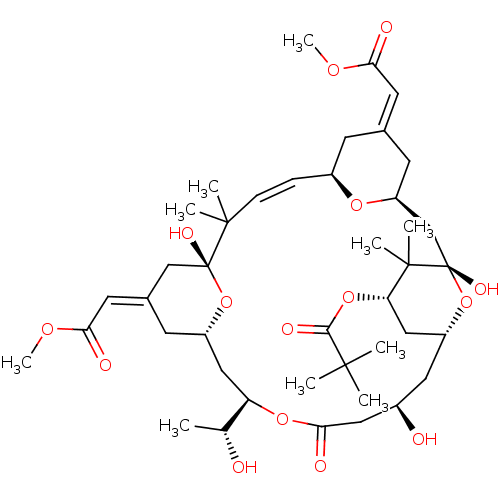
(CHEMBL501262 | bryostatin 10)Show SMILES COC(=O)\C=C1\C[C@H]2C[C@]3(O)O[C@@H](C[C@H](OC(=O)C(C)(C)C)C3(C)C)C[C@@H](O)CC(=O)O[C@H](C[C@@H]3C\C(C[C@@](O)(O3)C(C)(C)\C=C\[C@@H](C1)O2)=C/C(=O)OC)[C@@H](C)O |r,t:46| Show InChI InChI=1S/C42H64O15/c1-24(43)32-20-29-15-26(17-35(46)52-10)22-41(49,56-29)39(5,6)12-11-28-13-25(16-34(45)51-9)14-31(53-28)23-42(50)40(7,8)33(55-37(48)38(2,3)4)21-30(57-42)18-27(44)19-36(47)54-32/h11-12,16-17,24,27-33,43-44,49-50H,13-15,18-23H2,1-10H3/b12-11+,25-16+,26-17+/t24-,27-,28+,29+,30-,31+,32-,33+,41-,42+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Arizona State University
Curated by ChEMBL
| Assay Description
Displacement of [3H]PDBu form PKCalpha |
J Nat Prod 59: 286-9 (1997)
Article DOI: 10.1021/np960100b
BindingDB Entry DOI: 10.7270/Q2FQ9WNK |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM50292349

(CHEMBL505767 | bryostatin 18)Show SMILES COC(=O)\C=C1\C[C@H]2C[C@]3(O)O[C@@H](C[C@H](OC(=O)C(C)(C)C)C3(C)C)C[C@@H](O)CC(=O)O[C@H](C[C@@H]3C\C(C[C@@](O)(O3)C(C)(C)\C=C\[C@@H](C1)O2)=C\C(=O)OC)[C@@H](C)O |r,t:46| Show InChI InChI=1S/C42H64O15/c1-24(43)32-20-29-15-26(17-35(46)52-10)22-41(49,56-29)39(5,6)12-11-28-13-25(16-34(45)51-9)14-31(53-28)23-42(50)40(7,8)33(55-37(48)38(2,3)4)21-30(57-42)18-27(44)19-36(47)54-32/h11-12,16-17,24,27-33,43-44,49-50H,13-15,18-23H2,1-10H3/b12-11+,25-16+,26-17-/t24-,27-,28+,29+,30-,31+,32-,33+,41-,42+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Arizona State University
Curated by ChEMBL
| Assay Description
Displacement of [3H]PDBu form PKCalpha |
J Nat Prod 59: 286-9 (1997)
Article DOI: 10.1021/np960100b
BindingDB Entry DOI: 10.7270/Q2FQ9WNK |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240130
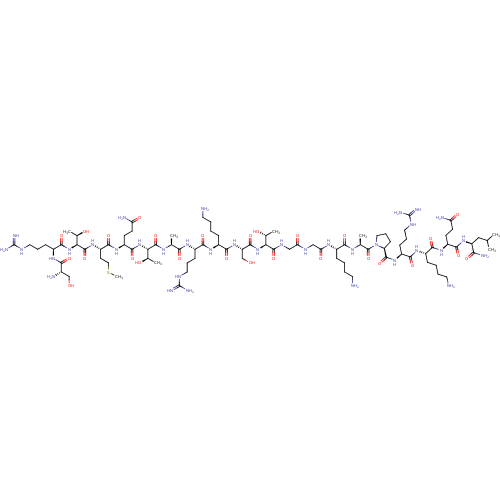
(CHEMBL4060961)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](N)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C90H165N35O27S/c1-44(2)39-60(70(97)135)120-78(143)57(26-28-63(95)131)116-74(139)52(20-10-13-32-92)114-76(141)55(23-16-35-105-89(100)101)118-83(148)62-25-18-37-125(62)87(152)46(4)110-73(138)51(19-9-12-31-91)111-66(134)41-107-65(133)40-108-84(149)67(47(5)128)122-82(147)61(43-127)121-77(142)53(21-11-14-33-93)115-75(140)54(22-15-34-104-88(98)99)112-71(136)45(3)109-85(150)68(48(6)129)123-81(146)58(27-29-64(96)132)117-79(144)59(30-38-153-8)119-86(151)69(49(7)130)124-80(145)56(24-17-36-106-90(102)103)113-72(137)50(94)42-126/h44-62,67-69,126-130H,9-43,91-94H2,1-8H3,(H2,95,131)(H2,96,132)(H2,97,135)(H,107,133)(H,108,149)(H,109,150)(H,110,138)(H,111,134)(H,112,136)(H,113,137)(H,114,141)(H,115,140)(H,116,139)(H,117,144)(H,118,148)(H,119,151)(H,120,143)(H,121,142)(H,122,147)(H,123,146)(H,124,145)(H4,98,99,104)(H4,100,101,105)(H4,102,103,106)/t45-,46-,47+,48+,49+,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,67-,68-,69-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240124
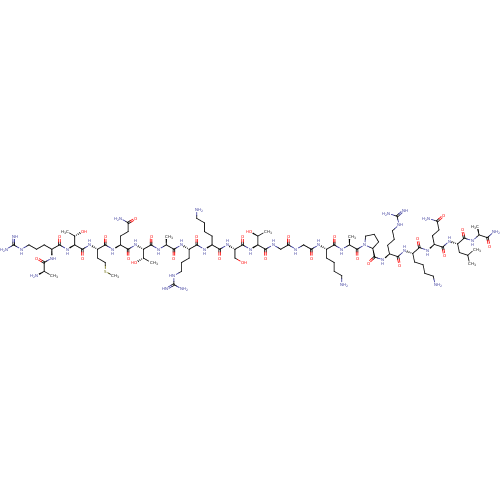
(CHEMBL4090728)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](C)N)[C@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(N)=O |r| Show InChI InChI=1S/C93H170N36O27S/c1-45(2)41-62(84(150)112-47(4)72(100)138)124-80(146)59(28-30-65(98)134)120-76(142)54(22-12-15-34-95)118-78(144)57(25-18-37-108-92(103)104)122-86(152)64-27-20-39-129(64)90(156)49(6)114-75(141)53(21-11-14-33-94)115-68(137)43-110-67(136)42-111-87(153)69(50(7)131)126-85(151)63(44-130)125-79(145)55(23-13-16-35-96)119-77(143)56(24-17-36-107-91(101)102)117-74(140)48(5)113-88(154)70(51(8)132)127-83(149)60(29-31-66(99)135)121-81(147)61(32-40-157-10)123-89(155)71(52(9)133)128-82(148)58(116-73(139)46(3)97)26-19-38-109-93(105)106/h45-64,69-71,130-133H,11-44,94-97H2,1-10H3,(H2,98,134)(H2,99,135)(H2,100,138)(H,110,136)(H,111,153)(H,112,150)(H,113,154)(H,114,141)(H,115,137)(H,116,139)(H,117,140)(H,118,144)(H,119,143)(H,120,142)(H,121,147)(H,122,152)(H,123,155)(H,124,146)(H,125,145)(H,126,151)(H,127,149)(H,128,148)(H4,101,102,107)(H4,103,104,108)(H4,105,106,109)/t46-,47+,48+,49+,50+,51+,52+,53+,54+,55+,56+,57+,58+,59+,60+,61+,62+,63+,64+,69+,70+,71+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 assessed as reduction in H2O2 production using pLys4Met H3 peptide as substrate by peroxidase coupled UV-visible spectrophot... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240122

(CHEMBL4103690)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](N)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCNC(=O)[C@@H](N)CO)C(N)=O |r| Show InChI InChI=1S/C99H182N38O30S/c1-49(2)43-67(90(161)123-57(77(107)148)21-12-16-37-114-79(150)55(103)46-138)132-86(157)64(29-31-70(105)144)128-82(153)59(23-10-14-35-101)126-84(155)62(26-18-39-116-98(110)111)130-92(163)69-28-20-41-137(69)96(167)51(4)121-81(152)58(22-9-13-34-100)122-73(147)45-118-72(146)44-119-93(164)74(52(5)141)134-91(162)68(48-140)133-85(156)60(24-11-15-36-102)127-83(154)61(25-17-38-115-97(108)109)124-78(149)50(3)120-94(165)75(53(6)142)135-89(160)65(30-32-71(106)145)129-87(158)66(33-42-168-8)131-95(166)76(54(7)143)136-88(159)63(27-19-40-117-99(112)113)125-80(151)56(104)47-139/h49-69,74-76,138-143H,9-48,100-104H2,1-8H3,(H2,105,144)(H2,106,145)(H2,107,148)(H,114,150)(H,118,146)(H,119,164)(H,120,165)(H,121,152)(H,122,147)(H,123,161)(H,124,149)(H,125,151)(H,126,155)(H,127,154)(H,128,153)(H,129,158)(H,130,163)(H,131,166)(H,132,157)(H,133,156)(H,134,162)(H,135,160)(H,136,159)(H4,108,109,115)(H4,110,111,116)(H4,112,113,117)/t50-,51-,52+,53+,54+,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,69-,74-,75-,76-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240126

(CHEMBL4105288)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)N)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C90H165N35O26S/c1-44(2)40-60(70(97)134)120-78(142)57(27-29-63(95)130)116-74(138)52(21-11-14-33-92)114-76(140)55(24-17-36-105-89(100)101)118-83(147)62-26-19-38-125(62)87(151)47(5)110-73(137)51(20-10-13-32-91)111-66(133)42-107-65(132)41-108-84(148)67(48(6)127)122-82(146)61(43-126)121-77(141)53(22-12-15-34-93)115-75(139)54(23-16-35-104-88(98)99)113-72(136)46(4)109-85(149)68(49(7)128)123-81(145)58(28-30-64(96)131)117-79(143)59(31-39-152-9)119-86(150)69(50(8)129)124-80(144)56(112-71(135)45(3)94)25-18-37-106-90(102)103/h44-62,67-69,126-129H,10-43,91-94H2,1-9H3,(H2,95,130)(H2,96,131)(H2,97,134)(H,107,132)(H,108,148)(H,109,149)(H,110,137)(H,111,133)(H,112,135)(H,113,136)(H,114,140)(H,115,139)(H,116,138)(H,117,143)(H,118,147)(H,119,150)(H,120,142)(H,121,141)(H,122,146)(H,123,145)(H,124,144)(H4,98,99,104)(H4,100,101,105)(H4,102,103,106)/t45-,46-,47-,48+,49+,50+,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,67-,68-,69-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 98 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM50292351
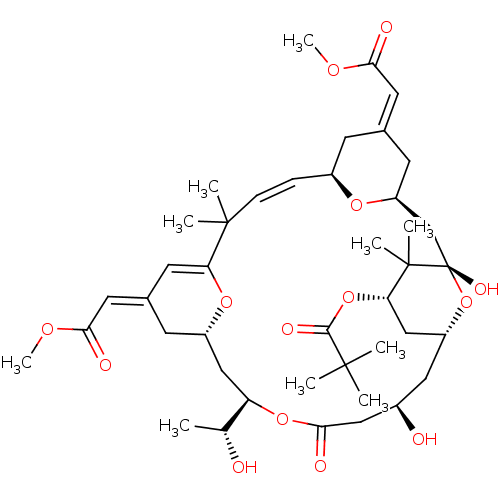
(CHEMBL509516 | bryostatin 16)Show SMILES COC(=O)\C=C1\C[C@H]2C[C@]3(O)O[C@@H](C[C@H](OC(=O)C(C)(C)C)C3(C)C)C[C@@H](O)CC(=O)O[C@H](C[C@@H]3C\C(=C/C(=O)OC)C=C(O3)C(C)(C)\C=C\[C@@H](C1)O2)[C@@H](C)O |r,c:43,t:50| Show InChI InChI=1S/C42H62O14/c1-24(43)32-21-29-14-26(18-36(46)51-10)16-33(53-29)40(5,6)12-11-28-13-25(17-35(45)50-9)15-31(52-28)23-42(49)41(7,8)34(55-38(48)39(2,3)4)22-30(56-42)19-27(44)20-37(47)54-32/h11-12,16-18,24,27-32,34,43-44,49H,13-15,19-23H2,1-10H3/b12-11+,25-17+,26-18+/t24-,27-,28+,29+,30-,31+,32-,34+,42+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 118 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Arizona State University
Curated by ChEMBL
| Assay Description
Displacement of [3H]PDBu form PKCalpha |
J Nat Prod 59: 286-9 (1997)
Article DOI: 10.1021/np960100b
BindingDB Entry DOI: 10.7270/Q2FQ9WNK |
More data for this
Ligand-Target Pair | |
REST corepressor 1 [4-485]
(Homo sapiens (Human)) | BDBM50158884

(CHEMBL3785550)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]1CCCN1)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(N)=O |r| Show InChI InChI=1S/C51H87N17O10/c1-29(2)26-36(46(75)67-40(30(3)4)48(77)62-34(19-12-24-60-51(56)57)43(72)63-35(16-8-9-21-52)49(78)68-25-13-20-39(68)41(53)70)64-45(74)37(27-31-14-6-5-7-15-31)65-47(76)38(28-69)66-44(73)33(18-11-23-59-50(54)55)61-42(71)32-17-10-22-58-32/h5-7,14-15,29-30,32-40,58,69H,8-13,16-28,52H2,1-4H3,(H2,53,70)(H,61,71)(H,62,77)(H,63,72)(H,64,74)(H,65,76)(H,66,73)(H,67,75)(H4,54,55,59)(H4,56,57,60)/t32-,33-,34-,35-,36-,37-,38-,39-,40-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human His-tagged LSD1 (171 to 836 residues)/GST-tagged CoREST (308 to 440 residues) complex using H3K4 peptide substrate by... |
Bioorg Med Chem 25: 1227-1234 (2017)
Article DOI: 10.1016/j.bmc.2016.12.033
BindingDB Entry DOI: 10.7270/Q2X92D9V |
More data for this
Ligand-Target Pair | |
Beta-lactamase
(Pseudomonas aeruginosa) | BDBM50336511

((4R,5S,6S)-3-(benzo[b]thiophen-2-ylthio)-6-((R)-1-...)Show SMILES C[C@@H](O)[C@@H]1[C@H]2[C@@H](C)C(Sc3cc4ccccc4s3)=C(N2C1=O)C(O)=O |r,c:19| Show InChI InChI=1S/C18H17NO4S2/c1-8-14-13(9(2)20)17(21)19(14)15(18(22)23)16(8)25-12-7-10-5-3-4-6-11(10)24-12/h3-9,13-14,20H,1-2H3,(H,22,23)/t8-,9-,13-,14-/m1/s1 | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Toho University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of Pseudomonas aeruginosa beta-lactamase IMP-1 assessed as hydrolysis of nitrocefin by UV spectrophotometric analysis |
Antimicrob Agents Chemother 54: 3625-9 (2010)
Article DOI: 10.1128/AAC.01397-09
BindingDB Entry DOI: 10.7270/Q29C6XQ2 |
More data for this
Ligand-Target Pair | |
Beta-lactamase
(Pseudomonas aeruginosa) | BDBM50121953
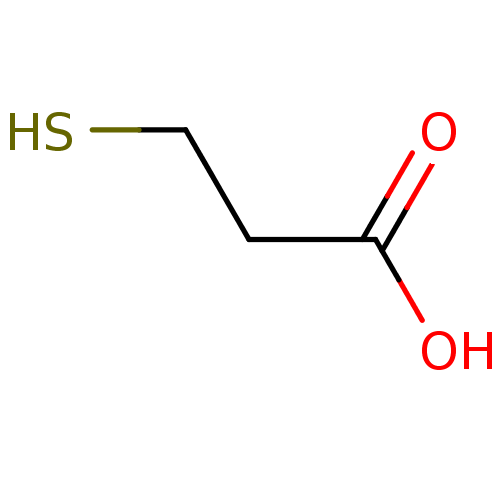
(2-mercaptoethanecarboxylic acid | 3-mercaptopropan...)Show InChI InChI=1S/C3H6O2S/c4-3(5)1-2-6/h6H,1-2H2,(H,4,5) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| Article
PubMed
| 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Toho University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of Pseudomonas aeruginosa beta-lactamase IMP-1 assessed as hydrolysis of nitrocefin by UV spectrophotometric analysis |
Antimicrob Agents Chemother 54: 3625-9 (2010)
Article DOI: 10.1128/AAC.01397-09
BindingDB Entry DOI: 10.7270/Q29C6XQ2 |
More data for this
Ligand-Target Pair | |
Beta-lactamase
(Pseudomonas aeruginosa) | BDBM50336510

(7,8-dihydroxy-1-methoxy-3-methyl-10-oxo-1,10-dihyd...)Show SMILES COC1OC(C)=Cc2oc3cc(O)c(O)c(C(O)=O)c3c(=O)c12 |c:5| Show InChI InChI=1S/C15H12O8/c1-5-3-7-10(15(21-2)22-5)13(18)9-8(23-7)4-6(16)12(17)11(9)14(19)20/h3-4,15-17H,1-2H3,(H,19,20) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Toho University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of Pseudomonas aeruginosa beta-lactamase IMP-1 assessed as hydrolysis of nitrocefin by UV spectrophotometric analysis |
Antimicrob Agents Chemother 54: 3625-9 (2010)
Article DOI: 10.1128/AAC.01397-09
BindingDB Entry DOI: 10.7270/Q29C6XQ2 |
More data for this
Ligand-Target Pair | |
Beta-lactamase
(Pseudomonas aeruginosa) | BDBM50336509

(CHEMBL116455 | Mercapto-acetic acid | Mercaptoacet...)Show InChI InChI=1S/C2H4O2S/c3-2(4)1-5/h5H,1H2,(H,3,4) | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| Purchase
CHEMBL
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
| PDB
Article
PubMed
| 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Toho University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of Pseudomonas aeruginosa beta-lactamase IMP-1 assessed as hydrolysis of nitrocefin by UV spectrophotometric analysis |
Antimicrob Agents Chemother 54: 3625-9 (2010)
Article DOI: 10.1128/AAC.01397-09
BindingDB Entry DOI: 10.7270/Q29C6XQ2 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM50292350

(CHEMBL452391 | bryostatin 17 | delta 19,20-bryosta...)Show SMILES COC(=O)\C=C1\C[C@H]2C[C@]3(O)O[C@@H](C[C@H](OC(=O)C(C)(C)C)C3(C)C)C[C@@H](O)CC(=O)O[C@H](C[C@@H]3C\C(=C\C(=O)OC)C=C(O3)C(C)(C)\C=C\[C@@H](C1)O2)[C@@H](C)O |r,c:43,t:50| Show InChI InChI=1S/C42H62O14/c1-24(43)32-21-29-14-26(18-36(46)51-10)16-33(53-29)40(5,6)12-11-28-13-25(17-35(45)50-9)15-31(52-28)23-42(49)41(7,8)34(55-38(48)39(2,3)4)22-30(56-42)19-27(44)20-37(47)54-32/h11-12,16-18,24,27-32,34,43-44,49H,13-15,19-23H2,1-10H3/b12-11+,25-17+,26-18-/t24-,27-,28+,29+,30-,31+,32-,34+,42+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 188 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Arizona State University
Curated by ChEMBL
| Assay Description
Displacement of [3H]PDBu form PKCalpha |
J Nat Prod 59: 286-9 (1997)
Article DOI: 10.1021/np960100b
BindingDB Entry DOI: 10.7270/Q2FQ9WNK |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240123
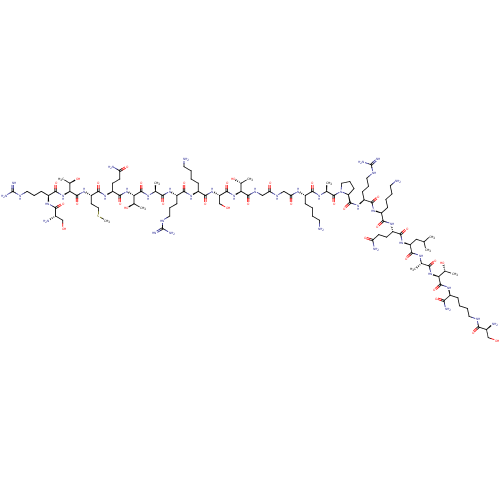
(CHEMBL4085763)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](N)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCCNC(=O)[C@@H](N)CO)C(N)=O |r| Show InChI InChI=1S/C106H194N40O33S/c1-51(2)45-71(96(172)127-53(4)84(160)142-80(57(8)152)101(177)131-61(82(114)158)23-14-18-39-121-85(161)59(110)48-147)140-92(168)68(31-33-74(112)154)136-88(164)63(25-12-16-37-108)134-90(166)66(28-20-41-123-105(117)118)138-98(174)73-30-22-43-146(73)103(179)54(5)129-87(163)62(24-11-15-36-107)130-77(157)47-125-76(156)46-126-99(175)78(55(6)150)143-97(173)72(50-149)141-91(167)64(26-13-17-38-109)135-89(165)65(27-19-40-122-104(115)116)132-83(159)52(3)128-100(176)79(56(7)151)144-95(171)69(32-34-75(113)155)137-93(169)70(35-44-180-10)139-102(178)81(58(9)153)145-94(170)67(29-21-42-124-106(119)120)133-86(162)60(111)49-148/h51-73,78-81,147-153H,11-50,107-111H2,1-10H3,(H2,112,154)(H2,113,155)(H2,114,158)(H,121,161)(H,125,156)(H,126,175)(H,127,172)(H,128,176)(H,129,163)(H,130,157)(H,131,177)(H,132,159)(H,133,162)(H,134,166)(H,135,165)(H,136,164)(H,137,169)(H,138,174)(H,139,178)(H,140,168)(H,141,167)(H,142,160)(H,143,173)(H,144,171)(H,145,170)(H4,115,116,122)(H4,117,118,123)(H4,119,120,124)/t52-,53-,54-,55+,56+,57+,58+,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,69-,70-,71-,72-,73-,78-,79-,80-,81-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240121
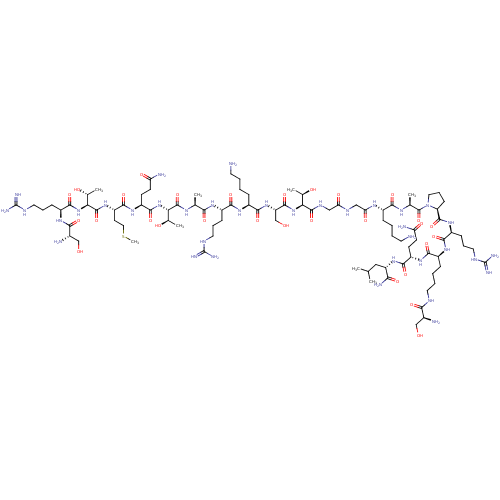
(CHEMBL4089148)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](N)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCNC(=O)[C@@H](N)CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C93H170N36O29S/c1-45(2)39-62(72(100)140)124-81(149)59(26-28-65(98)136)120-77(145)55(21-11-14-33-107-74(142)51(96)42-130)119-79(147)57(23-16-35-109-92(103)104)122-86(154)64-25-18-37-129(64)90(158)47(4)114-76(144)53(19-9-12-31-94)115-68(139)41-111-67(138)40-112-87(155)69(48(5)133)126-85(153)63(44-132)125-80(148)54(20-10-13-32-95)118-78(146)56(22-15-34-108-91(101)102)116-73(141)46(3)113-88(156)70(49(6)134)127-84(152)60(27-29-66(99)137)121-82(150)61(30-38-159-8)123-89(157)71(50(7)135)128-83(151)58(24-17-36-110-93(105)106)117-75(143)52(97)43-131/h45-64,69-71,130-135H,9-44,94-97H2,1-8H3,(H2,98,136)(H2,99,137)(H2,100,140)(H,107,142)(H,111,138)(H,112,155)(H,113,156)(H,114,144)(H,115,139)(H,116,141)(H,117,143)(H,118,146)(H,119,147)(H,120,145)(H,121,150)(H,122,154)(H,123,157)(H,124,149)(H,125,148)(H,126,153)(H,127,152)(H,128,151)(H4,101,102,108)(H4,103,104,109)(H4,105,106,110)/t46-,47-,48+,49+,50+,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,69-,70-,71-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Beta-lactamase
(Pseudomonas aeruginosa) | BDBM50336508
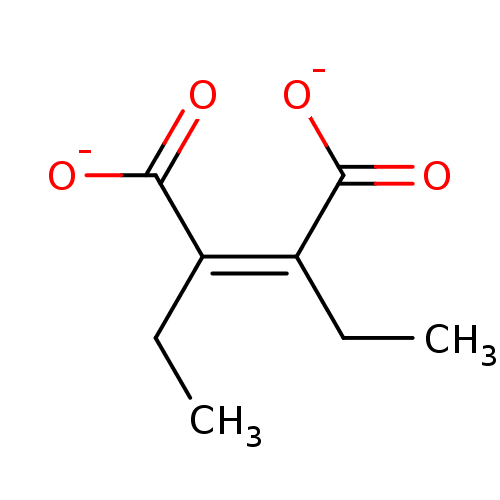
(CHEMBL1673299 | sodium 2,3-diethylmaleate)Show InChI InChI=1S/C8H12O4/c1-3-5(7(9)10)6(4-2)8(11)12/h3-4H2,1-2H3,(H,9,10)(H,11,12)/p-2/b6-5- | PDB
MMDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Toho University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of Pseudomonas aeruginosa beta-lactamase IMP-1 assessed as hydrolysis of nitrocefin by UV spectrophotometric analysis |
Antimicrob Agents Chemother 54: 3625-9 (2010)
Article DOI: 10.1128/AAC.01397-09
BindingDB Entry DOI: 10.7270/Q29C6XQ2 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240129
(CHEMBL4080345)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)N)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCNC(=O)CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C92H167N35O28S/c1-45(2)40-61(72(98)138)122-80(146)58(27-29-64(96)133)118-76(142)54(22-12-15-34-105-68(137)44-129)117-78(144)56(24-17-36-107-91(101)102)120-85(151)63-26-19-38-127(63)89(155)48(5)112-75(141)52(20-10-13-32-93)113-67(136)42-109-66(135)41-110-86(152)69(49(6)130)124-84(150)62(43-128)123-79(145)53(21-11-14-33-94)116-77(143)55(23-16-35-106-90(99)100)115-74(140)47(4)111-87(153)70(50(7)131)125-83(149)59(28-30-65(97)134)119-81(147)60(31-39-156-9)121-88(154)71(51(8)132)126-82(148)57(114-73(139)46(3)95)25-18-37-108-92(103)104/h45-63,69-71,128-132H,10-44,93-95H2,1-9H3,(H2,96,133)(H2,97,134)(H2,98,138)(H,105,137)(H,109,135)(H,110,152)(H,111,153)(H,112,141)(H,113,136)(H,114,139)(H,115,140)(H,116,143)(H,117,144)(H,118,142)(H,119,147)(H,120,151)(H,121,154)(H,122,146)(H,123,145)(H,124,150)(H,125,149)(H,126,148)(H4,99,100,106)(H4,101,102,107)(H4,103,104,108)/t46-,47-,48-,49+,50+,51+,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,69-,70-,71-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50210048

(CHEMBL3884919)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CO)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]2CCCN2C(=O)OCCSSCCOC(=O)NCCCC[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC1=O)C(C)C)C(=O)N1CCC[C@H]1C(N)=O |r| Show InChI InChI=1S/C57H93N17O14S2/c1-33(2)30-39-49(80)72-44(34(3)4)52(83)67-37(18-11-23-64-55(61)62)46(77)68-38(53(84)73-24-12-19-42(73)45(58)76)16-8-9-21-65-56(85)87-26-28-89-90-29-27-88-57(86)74-25-13-20-43(74)51(82)66-36(17-10-22-63-54(59)60)47(78)71-41(32-75)50(81)70-40(48(79)69-39)31-35-14-6-5-7-15-35/h5-7,14-15,33-34,36-44,75H,8-13,16-32H2,1-4H3,(H2,58,76)(H,65,85)(H,66,82)(H,67,83)(H,68,77)(H,69,79)(H,70,81)(H,71,78)(H,72,80)(H4,59,60,63)(H4,61,62,64)/t36-,37-,38-,39-,40-,41-,42-,43-,44-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... |
Bioorg Med Chem 25: 1227-1234 (2017)
Article DOI: 10.1016/j.bmc.2016.12.033
BindingDB Entry DOI: 10.7270/Q2X92D9V |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50158884

(CHEMBL3785550)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]1CCCN1)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(N)=O |r| Show InChI InChI=1S/C51H87N17O10/c1-29(2)26-36(46(75)67-40(30(3)4)48(77)62-34(19-12-24-60-51(56)57)43(72)63-35(16-8-9-21-52)49(78)68-25-13-20-39(68)41(53)70)64-45(74)37(27-31-14-6-5-7-15-31)65-47(76)38(28-69)66-44(73)33(18-11-23-59-50(54)55)61-42(71)32-17-10-22-58-32/h5-7,14-15,29-30,32-40,58,69H,8-13,16-28,52H2,1-4H3,(H2,53,70)(H,61,71)(H,62,77)(H,63,72)(H,64,74)(H,65,76)(H,66,73)(H,67,75)(H4,54,55,59)(H4,56,57,60)/t32-,33-,34-,35-,36-,37-,38-,39-,40-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... |
Bioorg Med Chem 25: 1227-1234 (2017)
Article DOI: 10.1016/j.bmc.2016.12.033
BindingDB Entry DOI: 10.7270/Q2X92D9V |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240131
(CHEMBL4083113)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](N)CCO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCNC(=O)[C@@H](N)CCO)C(N)=O |r| Show InChI InChI=1S/C101H186N38O30S/c1-51(2)47-69(92(163)125-59(79(109)150)21-12-16-39-116-81(152)57(105)33-44-140)134-88(159)66(29-31-72(107)146)130-84(155)61(23-10-14-37-103)128-86(157)64(26-18-41-118-100(112)113)132-94(165)71-28-20-43-139(71)98(169)53(4)123-83(154)60(22-9-13-36-102)124-75(149)49-120-74(148)48-121-95(166)76(54(5)143)136-93(164)70(50-142)135-87(158)62(24-11-15-38-104)129-85(156)63(25-17-40-117-99(110)111)126-80(151)52(3)122-96(167)77(55(6)144)137-91(162)67(30-32-73(108)147)131-89(160)68(35-46-170-8)133-97(168)78(56(7)145)138-90(161)65(27-19-42-119-101(114)115)127-82(153)58(106)34-45-141/h51-71,76-78,140-145H,9-50,102-106H2,1-8H3,(H2,107,146)(H2,108,147)(H2,109,150)(H,116,152)(H,120,148)(H,121,166)(H,122,167)(H,123,154)(H,124,149)(H,125,163)(H,126,151)(H,127,153)(H,128,157)(H,129,156)(H,130,155)(H,131,160)(H,132,165)(H,133,168)(H,134,159)(H,135,158)(H,136,164)(H,137,162)(H,138,161)(H4,110,111,117)(H4,112,113,118)(H4,114,115,119)/t52-,53-,54+,55+,56+,57-,58-,59-,60-,61-,62-,63-,64-,65-,66-,67-,68-,69-,70-,71-,76-,77-,78-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 6.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240118
(CHEMBL4096854)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)N)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCNC(=O)CO)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C92H167N35O28S/c1-45(2)40-61(72(98)138)122-80(146)58(27-29-64(96)133)118-76(142)53(20-10-13-32-93)116-78(144)56(24-17-36-107-91(101)102)120-85(151)63-26-19-38-127(63)89(155)48(5)112-75(141)52(22-12-15-34-105-68(137)44-129)113-67(136)42-109-66(135)41-110-86(152)69(49(6)130)124-84(150)62(43-128)123-79(145)54(21-11-14-33-94)117-77(143)55(23-16-35-106-90(99)100)115-74(140)47(4)111-87(153)70(50(7)131)125-83(149)59(28-30-65(97)134)119-81(147)60(31-39-156-9)121-88(154)71(51(8)132)126-82(148)57(114-73(139)46(3)95)25-18-37-108-92(103)104/h45-63,69-71,128-132H,10-44,93-95H2,1-9H3,(H2,96,133)(H2,97,134)(H2,98,138)(H,105,137)(H,109,135)(H,110,152)(H,111,153)(H,112,141)(H,113,136)(H,114,139)(H,115,140)(H,116,144)(H,117,143)(H,118,142)(H,119,147)(H,120,151)(H,121,154)(H,122,146)(H,123,145)(H,124,150)(H,125,149)(H,126,148)(H4,99,100,106)(H4,101,102,107)(H4,103,104,108)/t46-,47-,48-,49+,50+,51+,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,69-,70-,71-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
KEGG
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 9.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50210048

(CHEMBL3884919)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CO)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]2CCCN2C(=O)OCCSSCCOC(=O)NCCCC[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC1=O)C(C)C)C(=O)N1CCC[C@H]1C(N)=O |r| Show InChI InChI=1S/C57H93N17O14S2/c1-33(2)30-39-49(80)72-44(34(3)4)52(83)67-37(18-11-23-64-55(61)62)46(77)68-38(53(84)73-24-12-19-42(73)45(58)76)16-8-9-21-65-56(85)87-26-28-89-90-29-27-88-57(86)74-25-13-20-43(74)51(82)66-36(17-10-22-63-54(59)60)47(78)71-41(32-75)50(81)70-40(48(79)69-39)31-35-14-6-5-7-15-35/h5-7,14-15,33-34,36-44,75H,8-13,16-32H2,1-4H3,(H2,58,76)(H,65,85)(H,66,82)(H,67,83)(H,68,77)(H,69,79)(H,70,81)(H,71,78)(H,72,80)(H4,59,60,63)(H4,61,62,64)/t36-,37-,38-,39-,40-,41-,42-,43-,44-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... |
Bioorg Med Chem 25: 1227-1234 (2017)
Article DOI: 10.1016/j.bmc.2016.12.033
BindingDB Entry DOI: 10.7270/Q2X92D9V |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50210048

(CHEMBL3884919)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CO)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]2CCCN2C(=O)OCCSSCCOC(=O)NCCCC[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC1=O)C(C)C)C(=O)N1CCC[C@H]1C(N)=O |r| Show InChI InChI=1S/C57H93N17O14S2/c1-33(2)30-39-49(80)72-44(34(3)4)52(83)67-37(18-11-23-64-55(61)62)46(77)68-38(53(84)73-24-12-19-42(73)45(58)76)16-8-9-21-65-56(85)87-26-28-89-90-29-27-88-57(86)74-25-13-20-43(74)51(82)66-36(17-10-22-63-54(59)60)47(78)71-41(32-75)50(81)70-40(48(79)69-39)31-35-14-6-5-7-15-35/h5-7,14-15,33-34,36-44,75H,8-13,16-32H2,1-4H3,(H2,58,76)(H,65,85)(H,66,82)(H,67,83)(H,68,77)(H,69,79)(H,70,81)(H,71,78)(H,72,80)(H4,59,60,63)(H4,61,62,64)/t36-,37-,38-,39-,40-,41-,42-,43-,44-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 4.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... |
Bioorg Med Chem 25: 1227-1234 (2017)
Article DOI: 10.1016/j.bmc.2016.12.033
BindingDB Entry DOI: 10.7270/Q2X92D9V |
More data for this
Ligand-Target Pair | |
Metallo-beta-lactamase VIM-2
(Pseudomonas aeruginosa (g-Proteobacteria)) | BDBM50336508

(CHEMBL1673299 | sodium 2,3-diethylmaleate)Show InChI InChI=1S/C8H12O4/c1-3-5(7(9)10)6(4-2)8(11)12/h3-4H2,1-2H3,(H,9,10)(H,11,12)/p-2/b6-5- | PDB
UniProtKB/TrEMBL
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 1.20E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Toho University School of Medicine
Curated by ChEMBL
| Assay Description
Inhibition of Pseudomonas aeruginosa beta-lactamase VIM-2 assessed as hydrolysis of nitrocefin by UV spectrophotometric analysis |
Antimicrob Agents Chemother 54: 3625-9 (2010)
Article DOI: 10.1128/AAC.01397-09
BindingDB Entry DOI: 10.7270/Q29C6XQ2 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240125

(CHEMBL4081282)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C89H162N34O27S/c1-44(2)39-59(70(95)134)118-77(141)56(26-28-62(93)129)114-73(137)52(20-10-13-32-91)112-75(139)55(24-17-36-104-89(100)101)116-82(146)61-25-18-37-123(61)86(150)46(4)108-72(136)50(19-9-12-31-90)109-65(132)41-105-64(131)40-106-83(147)67(47(5)126)120-81(145)60(42-124)119-76(140)53(21-11-14-33-92)113-74(138)54(23-16-35-103-88(98)99)111-71(135)45(3)107-84(148)68(48(6)127)121-80(144)57(27-29-63(94)130)115-78(142)58(30-38-151-8)117-85(149)69(49(7)128)122-79(143)51(110-66(133)43-125)22-15-34-102-87(96)97/h44-61,67-69,124-128H,9-43,90-92H2,1-8H3,(H2,93,129)(H2,94,130)(H2,95,134)(H,105,131)(H,106,147)(H,107,148)(H,108,136)(H,109,132)(H,110,133)(H,111,135)(H,112,139)(H,113,138)(H,114,137)(H,115,142)(H,116,146)(H,117,149)(H,118,141)(H,119,140)(H,120,145)(H,121,144)(H,122,143)(H4,96,97,102)(H4,98,99,103)(H4,100,101,104)/t45-,46-,47+,48+,49+,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,67-,68-,69-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.70E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50210049
(CHEMBL3885101)Show SMILES CC(C)C[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]1CCCN1C(=O)CO)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCCNC(=O)CO)C(N)=O |r| Show InChI InChI=1S/C61H103N19O15/c1-35(2)30-42(75-53(89)43(31-37-16-6-5-7-17-37)76-55(91)44(32-81)77-52(88)39(20-12-26-69-60(64)65)72-56(92)45-22-14-28-79(45)48(85)34-83)54(90)78-49(36(3)4)58(94)73-40(21-13-27-70-61(66)67)51(87)74-41(19-8-10-24-62)59(95)80-29-15-23-46(80)57(93)71-38(50(63)86)18-9-11-25-68-47(84)33-82/h5-7,16-17,35-36,38-46,49,81-83H,8-15,18-34,62H2,1-4H3,(H2,63,86)(H,68,84)(H,71,93)(H,72,92)(H,73,94)(H,74,87)(H,75,89)(H,76,91)(H,77,88)(H,78,90)(H4,64,65,69)(H4,66,67,70)/t38-,39-,40-,41-,42-,43-,44-,45-,46-,49-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.90E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... |
Bioorg Med Chem 25: 1227-1234 (2017)
Article DOI: 10.1016/j.bmc.2016.12.033
BindingDB Entry DOI: 10.7270/Q2X92D9V |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50210050

(CHEMBL3884529)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CO)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]2CCCN2C(=O)OCCCCCCOC(=O)NCCCC[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC1=O)C(C)C)C(=O)N1CCC[C@H]1C(N)=O |r| Show InChI InChI=1S/C59H97N17O14/c1-35(2)32-41-51(82)74-46(36(3)4)54(85)69-39(22-15-27-66-57(63)64)48(79)70-40(55(86)75-28-16-23-44(75)47(60)78)20-10-11-25-67-58(87)89-30-12-5-6-13-31-90-59(88)76-29-17-24-45(76)53(84)68-38(21-14-26-65-56(61)62)49(80)73-43(34-77)52(83)72-42(50(81)71-41)33-37-18-8-7-9-19-37/h7-9,18-19,35-36,38-46,77H,5-6,10-17,20-34H2,1-4H3,(H2,60,78)(H,67,87)(H,68,84)(H,69,85)(H,70,79)(H,71,81)(H,72,83)(H,73,80)(H,74,82)(H4,61,62,65)(H4,63,64,66)/t38-,39-,40-,41-,42-,43-,44-,45-,46-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... |
Bioorg Med Chem 25: 1227-1234 (2017)
Article DOI: 10.1016/j.bmc.2016.12.033
BindingDB Entry DOI: 10.7270/Q2X92D9V |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50210050

(CHEMBL3884529)Show SMILES CC(C)C[C@@H]1NC(=O)[C@H](Cc2ccccc2)NC(=O)[C@H](CO)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H]2CCCN2C(=O)OCCCCCCOC(=O)NCCCC[C@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC1=O)C(C)C)C(=O)N1CCC[C@H]1C(N)=O |r| Show InChI InChI=1S/C59H97N17O14/c1-35(2)32-41-51(82)74-46(36(3)4)54(85)69-39(22-15-27-66-57(63)64)48(79)70-40(55(86)75-28-16-23-44(75)47(60)78)20-10-11-25-67-58(87)89-30-12-5-6-13-31-90-59(88)76-29-17-24-45(76)53(84)68-38(21-14-26-65-56(61)62)49(80)73-43(34-77)52(83)72-42(50(81)71-41)33-37-18-8-7-9-19-37/h7-9,18-19,35-36,38-46,77H,5-6,10-17,20-34H2,1-4H3,(H2,60,78)(H,67,87)(H,68,84)(H,69,85)(H,70,79)(H,71,81)(H,72,83)(H,73,80)(H,74,82)(H4,61,62,65)(H4,63,64,66)/t38-,39-,40-,41-,42-,43-,44-,45-,46-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >3.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincubated for 10 mins foll... |
Bioorg Med Chem 25: 1227-1234 (2017)
Article DOI: 10.1016/j.bmc.2016.12.033
BindingDB Entry DOI: 10.7270/Q2X92D9V |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240119

(CHEMBL4104579)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C92H167N35O28S/c1-45(2)40-61(72(98)138)122-80(146)58(27-29-64(96)133)118-76(142)53(21-11-14-33-94)116-78(144)56(24-17-36-106-91(101)102)120-85(151)63-26-19-38-127(63)89(155)48(5)112-75(141)52(20-10-13-32-93)113-67(136)42-108-66(135)41-109-86(152)69(49(6)130)124-84(150)62(43-128)123-79(145)54(22-12-15-34-95)117-77(143)55(23-16-35-105-90(99)100)114-74(140)47(4)111-87(153)70(50(7)131)125-83(149)59(28-30-65(97)134)119-81(147)60(31-39-156-9)121-88(154)71(51(8)132)126-82(148)57(25-18-37-107-92(103)104)115-73(139)46(3)110-68(137)44-129/h45-63,69-71,128-132H,10-44,93-95H2,1-9H3,(H2,96,133)(H2,97,134)(H2,98,138)(H,108,135)(H,109,152)(H,110,137)(H,111,153)(H,112,141)(H,113,136)(H,114,140)(H,115,139)(H,116,144)(H,117,143)(H,118,142)(H,119,147)(H,120,151)(H,121,154)(H,122,146)(H,123,145)(H,124,150)(H,125,149)(H,126,148)(H4,99,100,105)(H4,101,102,106)(H4,103,104,107)/t46-,47-,48-,49+,50+,51+,52-,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,69-,70-,71-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240117

(CHEMBL4069633)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCNC(=O)CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C94H169N35O30S/c1-46(2)40-62(74(99)142)124-82(150)59(27-29-65(97)136)120-78(146)55(22-12-15-34-106-69(140)44-131)119-80(148)57(24-17-36-108-93(102)103)122-87(155)64-26-19-38-129(64)91(159)49(5)114-77(145)53(20-10-13-32-95)115-68(139)42-110-67(138)41-111-88(156)71(50(6)133)126-86(154)63(43-130)125-81(149)54(21-11-14-33-96)118-79(147)56(23-16-35-107-92(100)101)116-76(144)48(4)113-89(157)72(51(7)134)127-85(153)60(28-30-66(98)137)121-83(151)61(31-39-160-9)123-90(158)73(52(8)135)128-84(152)58(25-18-37-109-94(104)105)117-75(143)47(3)112-70(141)45-132/h46-64,71-73,130-135H,10-45,95-96H2,1-9H3,(H2,97,136)(H2,98,137)(H2,99,142)(H,106,140)(H,110,138)(H,111,156)(H,112,141)(H,113,157)(H,114,145)(H,115,139)(H,116,144)(H,117,143)(H,118,147)(H,119,148)(H,120,146)(H,121,151)(H,122,155)(H,123,158)(H,124,150)(H,125,149)(H,126,154)(H,127,153)(H,128,152)(H4,100,101,107)(H4,102,103,108)(H4,104,105,109)/t47-,48-,49-,50+,51+,52+,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,71-,72-,73-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240116
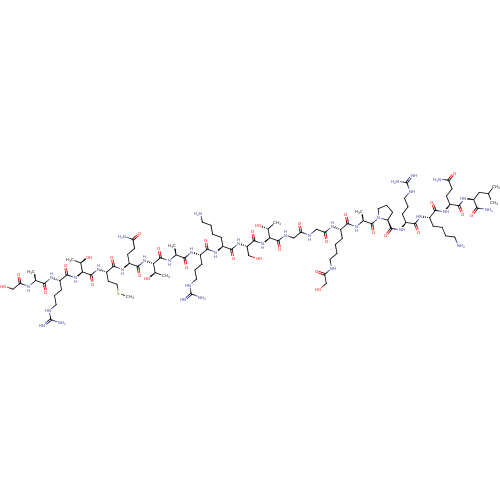
(CHEMBL4081041)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)CO)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCNC(=O)CO)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C94H169N35O30S/c1-46(2)40-62(74(99)142)124-82(150)59(27-29-65(97)136)120-78(146)54(20-10-13-32-95)118-80(148)57(24-17-36-108-93(102)103)122-87(155)64-26-19-38-129(64)91(159)49(5)114-77(145)53(22-12-15-34-106-69(140)44-131)115-68(139)42-110-67(138)41-111-88(156)71(50(6)133)126-86(154)63(43-130)125-81(149)55(21-11-14-33-96)119-79(147)56(23-16-35-107-92(100)101)116-76(144)48(4)113-89(157)72(51(7)134)127-85(153)60(28-30-66(98)137)121-83(151)61(31-39-160-9)123-90(158)73(52(8)135)128-84(152)58(25-18-37-109-94(104)105)117-75(143)47(3)112-70(141)45-132/h46-64,71-73,130-135H,10-45,95-96H2,1-9H3,(H2,97,136)(H2,98,137)(H2,99,142)(H,106,140)(H,110,138)(H,111,156)(H,112,141)(H,113,157)(H,114,145)(H,115,139)(H,116,144)(H,117,143)(H,118,148)(H,119,147)(H,120,146)(H,121,151)(H,122,155)(H,123,158)(H,124,150)(H,125,149)(H,126,154)(H,127,153)(H,128,152)(H4,100,101,107)(H4,102,103,108)(H4,104,105,109)/t47-,48-,49-,50+,51+,52+,53-,54-,55-,56-,57-,58-,59-,60-,61-,62-,63-,64-,71-,72-,73-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Lysine-specific histone demethylase 1A
(Homo sapiens (Human)) | BDBM50240120

(CHEMBL4099300)Show SMILES CSCC[C@H](NC(=O)[C@@H](NC(=O)[C@@H](N)CCCNC(N)=N)[C@@H](C)O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(C)C)C(N)=O |r| Show InChI InChI=1S/C87H160N34O25S/c1-43(2)39-58(68(94)130)116-76(138)55(26-28-61(92)126)112-72(134)51(21-10-13-32-89)110-74(136)54(24-17-36-103-87(99)100)114-80(142)60-25-18-37-121(60)84(146)45(4)107-71(133)50(20-9-12-31-88)108-64(129)41-104-63(128)40-105-81(143)65(46(5)123)119-79(141)59(42-122)117-75(137)52(22-11-14-33-90)111-73(135)53(23-16-35-102-86(97)98)109-69(131)44(3)106-82(144)66(47(6)124)120-78(140)56(27-29-62(93)127)113-77(139)57(30-38-147-8)115-83(145)67(48(7)125)118-70(132)49(91)19-15-34-101-85(95)96/h43-60,65-67,122-125H,9-42,88-91H2,1-8H3,(H2,92,126)(H2,93,127)(H2,94,130)(H,104,128)(H,105,143)(H,106,144)(H,107,133)(H,108,129)(H,109,131)(H,110,136)(H,111,135)(H,112,134)(H,113,139)(H,114,142)(H,115,145)(H,116,138)(H,117,137)(H,118,132)(H,119,141)(H,120,140)(H4,95,96,101)(H4,97,98,102)(H4,99,100,103)/t44-,45-,46+,47+,48+,49-,50-,51-,52-,53-,54-,55-,56-,57-,58-,59-,60-,65-,66-,67-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| >5.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nagoya City University
Curated by ChEMBL
| Assay Description
Inhibition of human LSD1 (172 to 833 residues) assessed as reduction in H2O2 production using H3K4me2 (1 to 20 residues) peptide as substrate preincu... |
Bioorg Med Chem 25: 2617-2624 (2017)
Article DOI: 10.1016/j.bmc.2017.03.016
BindingDB Entry DOI: 10.7270/Q2KH0QG1 |
More data for this
Ligand-Target Pair | |
Endothelin-1 receptor
(Homo sapiens (Human)) | BDBM50119678
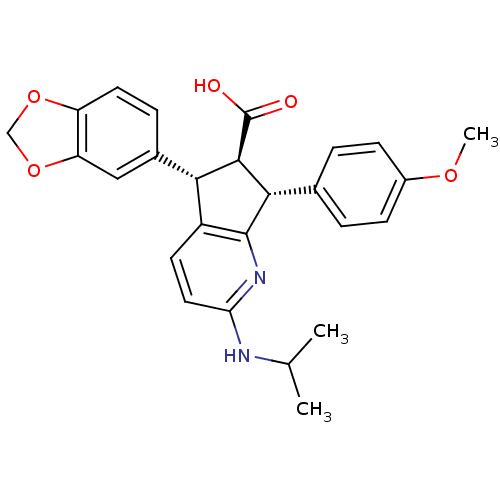
((R)-5-Benzo[1,3]dioxol-5-yl-2-isopropylamino-7-(4-...)Show SMILES COc1ccc(cc1)[C@H]1[C@@H]([C@H](c2ccc(NC(C)C)nc12)c1ccc2OCOc2c1)C(O)=O Show InChI InChI=1S/C26H26N2O5/c1-14(2)27-21-11-9-18-22(16-6-10-19-20(12-16)33-13-32-19)24(26(29)30)23(25(18)28-21)15-4-7-17(31-3)8-5-15/h4-12,14,22-24H,13H2,1-3H3,(H,27,28)(H,29,30)/t22-,23-,24+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against I-labeled ET-1 binding to Endothelin A receptor |
Bioorg Med Chem Lett 12: 3041-5 (2002)
BindingDB Entry DOI: 10.7270/Q2KK9B3N |
More data for this
Ligand-Target Pair | |
Endothelin-1 receptor
(Homo sapiens (Human)) | BDBM50119676
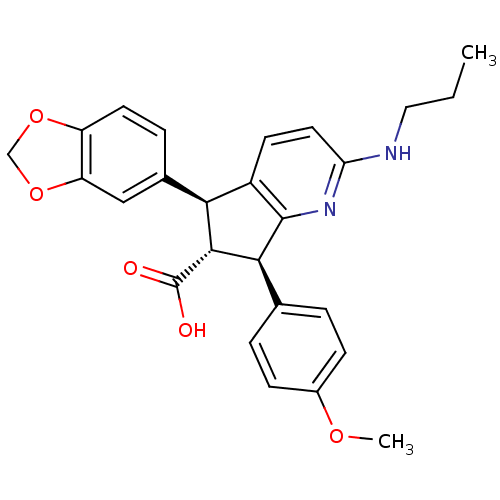
((R)-5-Benzo[1,3]dioxol-5-yl-7-(4-methoxy-phenyl)-2...)Show SMILES CCCNc1ccc2[C@@H]([C@H]([C@@H](c2n1)c1ccc(OC)cc1)C(O)=O)c1ccc2OCOc2c1 Show InChI InChI=1S/C26H26N2O5/c1-3-12-27-21-11-9-18-22(16-6-10-19-20(13-16)33-14-32-19)24(26(29)30)23(25(18)28-21)15-4-7-17(31-2)8-5-15/h4-11,13,22-24H,3,12,14H2,1-2H3,(H,27,28)(H,29,30)/t22-,23-,24+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.120 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against I-labeled ET-1 binding to Endothelin A receptor |
Bioorg Med Chem Lett 12: 3041-5 (2002)
BindingDB Entry DOI: 10.7270/Q2KK9B3N |
More data for this
Ligand-Target Pair | |
Endothelin-1 receptor
(Homo sapiens (Human)) | BDBM50119665
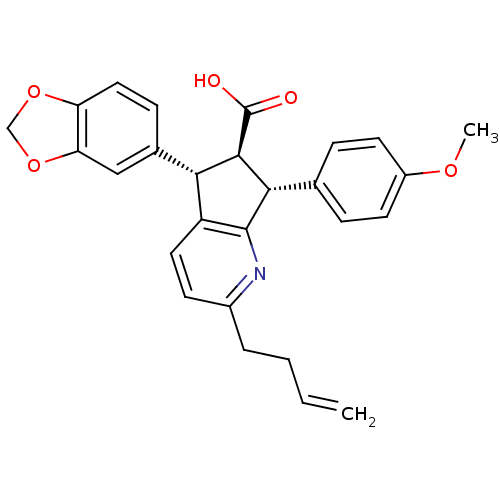
(5-Benzo[1,3]dioxol-5-yl-2-but-3-enyl-7-(4-methoxy-...)Show SMILES COc1ccc(cc1)[C@H]1[C@@H]([C@H](c2ccc(CCC=C)nc12)c1ccc2OCOc2c1)C(O)=O Show InChI InChI=1S/C27H25NO5/c1-3-4-5-18-9-12-20-23(17-8-13-21-22(14-17)33-15-32-21)25(27(29)30)24(26(20)28-18)16-6-10-19(31-2)11-7-16/h3,6-14,23-25H,1,4-5,15H2,2H3,(H,29,30)/t23-,24-,25+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against I-labeled ET-1 binding to Endothelin A receptor |
Bioorg Med Chem Lett 12: 3041-5 (2002)
BindingDB Entry DOI: 10.7270/Q2KK9B3N |
More data for this
Ligand-Target Pair | |
Endothelin-1 receptor
(Homo sapiens (Human)) | BDBM50119671

(5-Benzo[1,3]dioxol-5-yl-2-butyl-7-(4-methoxy-pheny...)Show SMILES CCCCc1ccc2[C@@H]([C@H]([C@@H](c2n1)c1ccc(OC)cc1)C(O)=O)c1ccc2OCOc2c1 Show InChI InChI=1S/C27H27NO5/c1-3-4-5-18-9-12-20-23(17-8-13-21-22(14-17)33-15-32-21)25(27(29)30)24(26(20)28-18)16-6-10-19(31-2)11-7-16/h6-14,23-25H,3-5,15H2,1-2H3,(H,29,30)/t23-,24-,25+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against I-labeled ET-1 binding to Endothelin A receptor |
Bioorg Med Chem Lett 12: 3041-5 (2002)
BindingDB Entry DOI: 10.7270/Q2KK9B3N |
More data for this
Ligand-Target Pair | |
Endothelin-1 receptor
(Homo sapiens (Human)) | BDBM50119663

(5-Benzo[1,3]dioxol-5-yl-2-isobutyl-7-(4-methoxy-ph...)Show SMILES COc1ccc(cc1)[C@H]1[C@@H]([C@H](c2ccc(CC(C)C)nc12)c1ccc2OCOc2c1)C(O)=O Show InChI InChI=1S/C27H27NO5/c1-15(2)12-18-7-10-20-23(17-6-11-21-22(13-17)33-14-32-21)25(27(29)30)24(26(20)28-18)16-4-8-19(31-3)9-5-16/h4-11,13,15,23-25H,12,14H2,1-3H3,(H,29,30)/t23-,24-,25+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.320 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against I-labeled ET-1 binding to Endothelin A receptor |
Bioorg Med Chem Lett 12: 3041-5 (2002)
BindingDB Entry DOI: 10.7270/Q2KK9B3N |
More data for this
Ligand-Target Pair | |
Endothelin-1 receptor
(Homo sapiens (Human)) | BDBM50119670
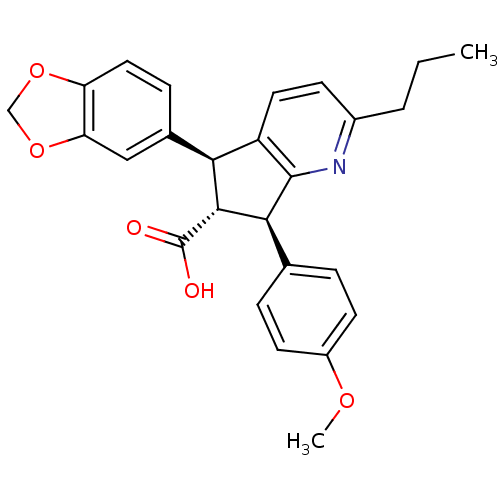
(5-Benzo[1,3]dioxol-5-yl-7-(4-methoxy-phenyl)-2-pro...)Show SMILES CCCc1ccc2[C@@H]([C@H]([C@@H](c2n1)c1ccc(OC)cc1)C(O)=O)c1ccc2OCOc2c1 Show InChI InChI=1S/C26H25NO5/c1-3-4-17-8-11-19-22(16-7-12-20-21(13-16)32-14-31-20)24(26(28)29)23(25(19)27-17)15-5-9-18(30-2)10-6-15/h5-13,22-24H,3-4,14H2,1-2H3,(H,28,29)/t22-,23-,24+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.370 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against I-labeled ET-1 binding to Endothelin A receptor |
Bioorg Med Chem Lett 12: 3041-5 (2002)
BindingDB Entry DOI: 10.7270/Q2KK9B3N |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50505569
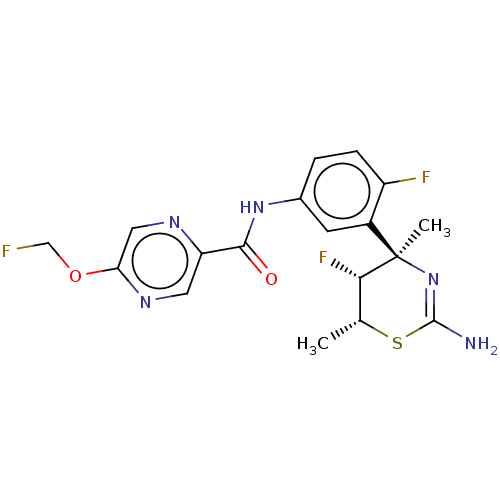
(CHEMBL4557670)Show SMILES C[C@H]1SC(N)=N[C@@](C)([C@H]1F)c1cc(NC(=O)c2cnc(OCF)cn2)ccc1F |r,c:4| Show InChI InChI=1S/C18H18F3N5O2S/c1-9-15(21)18(2,26-17(22)29-9)11-5-10(3-4-12(11)20)25-16(27)13-6-24-14(7-23-13)28-8-19/h3-7,9,15H,8H2,1-2H3,(H2,22,26)(H,25,27)/t9-,15+,18-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| n/a | n/a | 0.380 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 in human SH-SY5Y cells expressing human wild type amyloid precursor protein assessed as reduction in amyloidbeta40 production inc... |
J Med Chem 62: 9331-9337 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01140
BindingDB Entry DOI: 10.7270/Q2CJ8HRN |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase JAK3
(Homo sapiens (Human)) | BDBM50463838
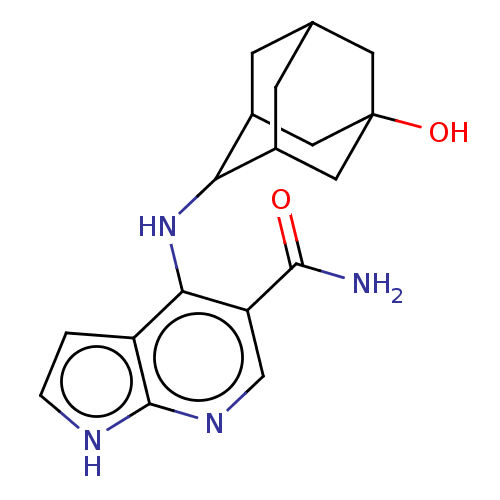
(CHEMBL4238926)Show SMILES NC(=O)c1cnc2[nH]ccc2c1NC1C2CC3CC1CC(O)(C3)C2 |TLB:13:14:22:17.18.19,12:13:22.16.17:19,THB:15:16:19:23.14.13,15:14:22.16.17:19,13:18:22:23.15.14,(62.43,-6.04,;61.1,-6.81,;61.1,-8.35,;59.76,-6.04,;59.75,-4.49,;58.42,-3.73,;57.08,-4.5,;55.61,-4.03,;54.71,-5.28,;55.62,-6.53,;57.09,-6.04,;58.42,-6.82,;58.42,-8.36,;57.14,-9.21,;57.12,-10.75,;56.11,-12.03,;54.69,-11.48,;54.69,-9.88,;55.72,-8.64,;54.38,-9.11,;54.39,-10.61,;53.04,-9.84,;53.19,-11.89,;55.71,-11.09,)| Show InChI InChI=1S/C18H22N4O2/c19-16(23)13-8-21-17-12(1-2-20-17)15(13)22-14-10-3-9-4-11(14)7-18(24,5-9)6-10/h1-2,8-11,14,24H,3-7H2,(H2,19,23)(H2,20,21,22) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 0.430 | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal His-tagged JAK3 catalytic domain (795 to 1124 residues) expressed in baculovirus expression system using B... |
Bioorg Med Chem 26: 4971-4983 (2018)
Article DOI: 10.1016/j.bmc.2018.08.005
BindingDB Entry DOI: 10.7270/Q2Z89G3M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Tyrosine-protein kinase JAK1
(Homo sapiens (Human)) | BDBM50463838

(CHEMBL4238926)Show SMILES NC(=O)c1cnc2[nH]ccc2c1NC1C2CC3CC1CC(O)(C3)C2 |TLB:13:14:22:17.18.19,12:13:22.16.17:19,THB:15:16:19:23.14.13,15:14:22.16.17:19,13:18:22:23.15.14,(62.43,-6.04,;61.1,-6.81,;61.1,-8.35,;59.76,-6.04,;59.75,-4.49,;58.42,-3.73,;57.08,-4.5,;55.61,-4.03,;54.71,-5.28,;55.62,-6.53,;57.09,-6.04,;58.42,-6.82,;58.42,-8.36,;57.14,-9.21,;57.12,-10.75,;56.11,-12.03,;54.69,-11.48,;54.69,-9.88,;55.72,-8.64,;54.38,-9.11,;54.39,-10.61,;53.04,-9.84,;53.19,-11.89,;55.71,-11.09,)| Show InChI InChI=1S/C18H22N4O2/c19-16(23)13-8-21-17-12(1-2-20-17)15(13)22-14-10-3-9-4-11(14)7-18(24,5-9)6-10/h1-2,8-11,14,24H,3-7H2,(H2,19,23)(H2,20,21,22) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal GST-tagged JAK1 catalytic domain (850 to 1154 residues) expressed in baculovirus expression system using B... |
Bioorg Med Chem 26: 4971-4983 (2018)
Article DOI: 10.1016/j.bmc.2018.08.005
BindingDB Entry DOI: 10.7270/Q2Z89G3M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Endothelin-1 receptor
(Homo sapiens (Human)) | BDBM50119673
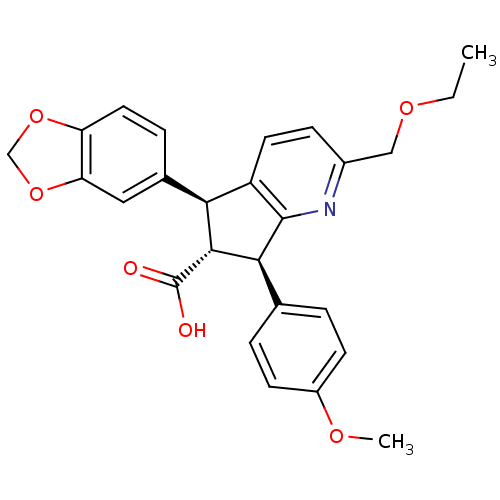
((R)-5-Benzo[1,3]dioxol-5-yl-2-ethoxymethyl-7-(4-me...)Show SMILES CCOCc1ccc2[C@@H]([C@H]([C@@H](c2n1)c1ccc(OC)cc1)C(O)=O)c1ccc2OCOc2c1 Show InChI InChI=1S/C26H25NO6/c1-3-31-13-17-7-10-19-22(16-6-11-20-21(12-16)33-14-32-20)24(26(28)29)23(25(19)27-17)15-4-8-18(30-2)9-5-15/h4-12,22-24H,3,13-14H2,1-2H3,(H,28,29)/t22-,23-,24+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.510 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against I-labeled ET-1 binding to Endothelin A receptor |
Bioorg Med Chem Lett 12: 3041-5 (2002)
BindingDB Entry DOI: 10.7270/Q2KK9B3N |
More data for this
Ligand-Target Pair | |
Endothelin-1 receptor
(Homo sapiens (Human)) | BDBM50119662
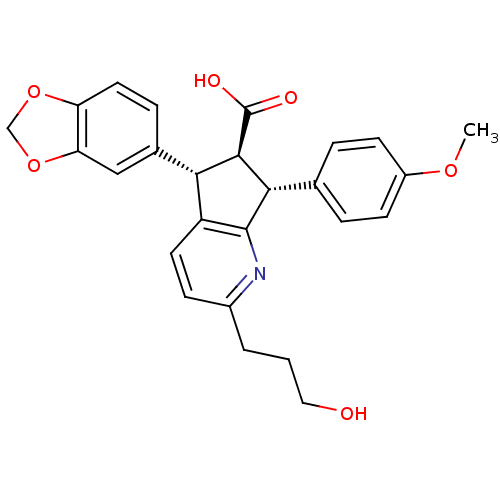
((R)-5-Benzo[1,3]dioxol-5-yl-2-(3-hydroxy-propyl)-7...)Show SMILES COc1ccc(cc1)[C@H]1[C@@H]([C@H](c2ccc(CCCO)nc12)c1ccc2OCOc2c1)C(O)=O Show InChI InChI=1S/C26H25NO6/c1-31-18-8-4-15(5-9-18)23-24(26(29)30)22(16-6-11-20-21(13-16)33-14-32-20)19-10-7-17(3-2-12-28)27-25(19)23/h4-11,13,22-24,28H,2-3,12,14H2,1H3,(H,29,30)/t22-,23-,24+/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.550 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute
Curated by ChEMBL
| Assay Description
Inhibitory activity of the compound against I-labeled ET-1 binding to Endothelin A receptor |
Bioorg Med Chem Lett 12: 3041-5 (2002)
BindingDB Entry DOI: 10.7270/Q2KK9B3N |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM204940
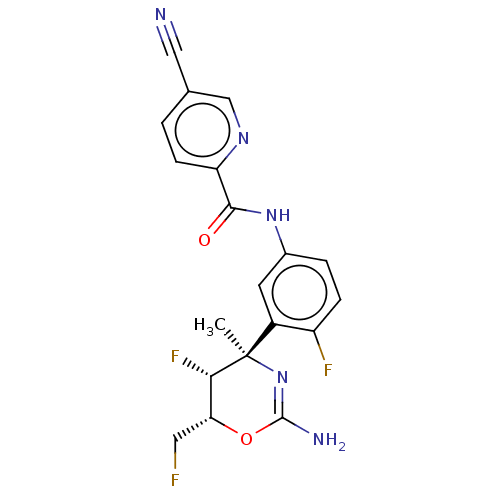
(I-25)Show SMILES C[C@@]1(N=C(N)O[C@H](CF)[C@@H]1F)c1cc(NC(=O)c2ccc(cn2)C#N)ccc1F |r,t:2| Show InChI InChI=1S/C19H16F3N5O2/c1-19(16(22)15(7-20)29-18(24)27-19)12-6-11(3-4-13(12)21)26-17(28)14-5-2-10(8-23)9-25-14/h2-6,9,15-16H,7H2,1H3,(H2,24,27)(H,26,28)/t15-,16+,19-/m1/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.580 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 in human SH-SY5Y cells harboring wild-type human beta-APP assessed as reduction in secreted amyloid beta (1 to 40) after 24 hrs b... |
J Med Chem 61: 5525-5546 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00011
BindingDB Entry DOI: 10.7270/Q2SB48DD |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase JAK3
(Homo sapiens (Human)) | BDBM50463842
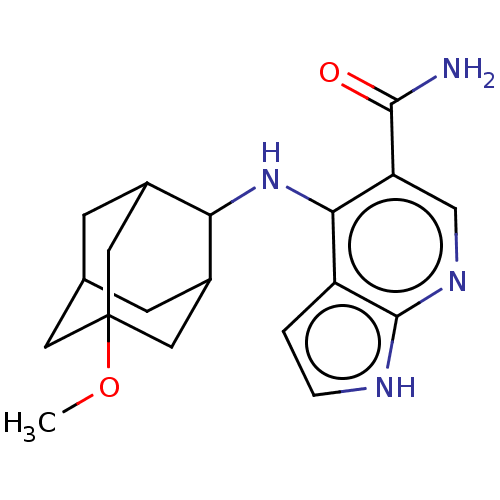
(CHEMBL4246788)Show SMILES COC12CC3CC(C1)C(Nc1c(cnc4[nH]ccc14)C(N)=O)C(C3)C2 |TLB:5:4:24:7.6.8,5:6:3.4.23:24,THB:8:6:3:23.22.24,8:22:3:7.5.6,9:8:3.4.23:24,(18.96,-21.75,;18.96,-23.29,;20.3,-24.06,;19.11,-25.34,;20.61,-24.93,;22.03,-25.48,;23.04,-24.2,;21.63,-24.54,;23.06,-22.66,;24.34,-21.81,;24.34,-20.27,;25.68,-19.49,;25.67,-17.94,;24.33,-17.18,;23,-17.96,;21.53,-17.49,;20.63,-18.74,;21.54,-19.98,;23,-19.5,;27.01,-20.26,;28.34,-19.49,;27.01,-21.8,;21.64,-22.09,;20.6,-23.33,;20.3,-22.56,)| Show InChI InChI=1S/C19H24N4O2/c1-25-19-6-10-4-11(7-19)15(12(5-10)8-19)23-16-13-2-3-21-18(13)22-9-14(16)17(20)24/h2-3,9-12,15H,4-8H2,1H3,(H2,20,24)(H2,21,22,23) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.630 | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal His-tagged JAK3 catalytic domain (795 to 1124 residues) expressed in baculovirus expression system using B... |
Bioorg Med Chem 26: 4971-4983 (2018)
Article DOI: 10.1016/j.bmc.2018.08.005
BindingDB Entry DOI: 10.7270/Q2Z89G3M |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase JAK3
(Homo sapiens (Human)) | BDBM50463838

(CHEMBL4238926)Show SMILES NC(=O)c1cnc2[nH]ccc2c1NC1C2CC3CC1CC(O)(C3)C2 |TLB:13:14:22:17.18.19,12:13:22.16.17:19,THB:15:16:19:23.14.13,15:14:22.16.17:19,13:18:22:23.15.14,(62.43,-6.04,;61.1,-6.81,;61.1,-8.35,;59.76,-6.04,;59.75,-4.49,;58.42,-3.73,;57.08,-4.5,;55.61,-4.03,;54.71,-5.28,;55.62,-6.53,;57.09,-6.04,;58.42,-6.82,;58.42,-8.36,;57.14,-9.21,;57.12,-10.75,;56.11,-12.03,;54.69,-11.48,;54.69,-9.88,;55.72,-8.64,;54.38,-9.11,;54.39,-10.61,;53.04,-9.84,;53.19,-11.89,;55.71,-11.09,)| Show InChI InChI=1S/C18H22N4O2/c19-16(23)13-8-21-17-12(1-2-20-17)15(13)22-14-10-3-9-4-11(14)7-18(24,5-9)6-10/h1-2,8-11,14,24H,3-7H2,(H2,19,23)(H2,20,21,22) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 0.710 | n/a | n/a | n/a | n/a | n/a | n/a |
Astellas Pharma Inc.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human N-terminal His-tagged JAK3 catalytic domain (795 to 1124 residues) expressed in baculovirus expression system using B... |
Bioorg Med Chem 26: 4971-4983 (2018)
Article DOI: 10.1016/j.bmc.2018.08.005
BindingDB Entry DOI: 10.7270/Q2Z89G3M |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Endothelin receptor type B
(Homo sapiens (Human)) | BDBM50284769

((S)-2-((R)-3-(2-Bromo-1H-indol-3-yl)-2-{(S)-2-cycl...)Show SMILES CCCC[C@H](NC(=O)[C@@H](Cc1c(Br)[nH]c2ccccc12)NC(=O)[C@@H](NC(=O)N1[C@@H](C)CCC[C@H]1C)C1CC1)C(O)=O Show InChI InChI=1S/C30H42BrN5O5/c1-4-5-12-23(29(39)40)33-27(37)24(16-21-20-11-6-7-13-22(20)32-26(21)31)34-28(38)25(19-14-15-19)35-30(41)36-17(2)9-8-10-18(36)3/h6-7,11,13,17-19,23-25,32H,4-5,8-10,12,14-16H2,1-3H3,(H,33,37)(H,34,38)(H,35,41)(H,39,40)/t17-,18+,23-,24+,25-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 0.800 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Functional inhibition of ET-1 induced [Ca2+] increase in human Girardi heart (hGH) cells, which express Endothelin B receptor |
Bioorg Med Chem Lett 5: 1483-1488 (1995)
Article DOI: 10.1016/0960-894X(95)00237-N
BindingDB Entry DOI: 10.7270/Q27D2VM3 |
More data for this
Ligand-Target Pair | |
Endothelin receptor type B
(Sus scrofa) | BDBM50284769

((S)-2-((R)-3-(2-Bromo-1H-indol-3-yl)-2-{(S)-2-cycl...)Show SMILES CCCC[C@H](NC(=O)[C@@H](Cc1c(Br)[nH]c2ccccc12)NC(=O)[C@@H](NC(=O)N1[C@@H](C)CCC[C@H]1C)C1CC1)C(O)=O Show InChI InChI=1S/C30H42BrN5O5/c1-4-5-12-23(29(39)40)33-27(37)24(16-21-20-11-6-7-13-22(20)32-26(21)31)34-28(38)25(19-14-15-19)35-30(41)36-17(2)9-8-10-18(36)3/h6-7,11,13,17-19,23-25,32H,4-5,8-10,12,14-16H2,1-3H3,(H,33,37)(H,34,38)(H,35,41)(H,39,40)/t17-,18+,23-,24+,25-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| n/a | n/a | 0.810 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory activity against 125 I-labeled ET-1 binding to Endothelin B receptor in porcine cerebellum membranes |
Bioorg Med Chem Lett 5: 1483-1488 (1995)
Article DOI: 10.1016/0960-894X(95)00237-N
BindingDB Entry DOI: 10.7270/Q27D2VM3 |
More data for this
Ligand-Target Pair | |
Endothelin receptor type B
(Sus scrofa) | BDBM50051385
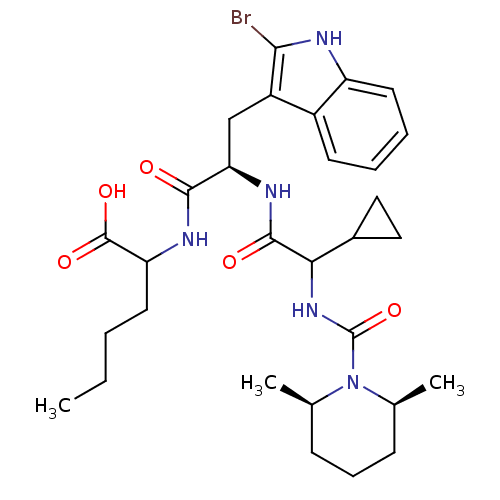
(2-((R)-3-(2-Bromo-1H-indol-3-yl)-2-{2-cyclopropyl-...)Show SMILES CCCCC(NC(=O)[C@@H](Cc1c(Br)[nH]c2ccccc12)NC(=O)C(NC(=O)N1[C@@H](C)CCC[C@H]1C)C1CC1)C(O)=O Show InChI InChI=1S/C30H42BrN5O5/c1-4-5-12-23(29(39)40)33-27(37)24(16-21-20-11-6-7-13-22(20)32-26(21)31)34-28(38)25(19-14-15-19)35-30(41)36-17(2)9-8-10-18(36)3/h6-7,11,13,17-19,23-25,32H,4-5,8-10,12,14-16H2,1-3H3,(H,33,37)(H,34,38)(H,35,41)(H,39,40)/t17-,18+,23?,24-,25?/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.810 | n/a | n/a | n/a | n/a | n/a | n/a |
Tsukuba Research Institute
Curated by ChEMBL
| Assay Description
Binding affinity towards endothelin B receptor in porcine cerebellum membranes |
J Med Chem 39: 2313-30 (1996)
Article DOI: 10.1021/jm9600914
BindingDB Entry DOI: 10.7270/Q2XS5W2G |
More data for this
Ligand-Target Pair | |
Beta-secretase 1
(Homo sapiens (Human)) | BDBM50432632

(CHEMBL2347211)Show SMILES C[C@]1(CCSC(N)=N1)c1cc(NC(=O)c2ccc(Cl)cn2)ccc1F |r,c:6| Show InChI InChI=1S/C17H16ClFN4OS/c1-17(6-7-25-16(20)23-17)12-8-11(3-4-13(12)19)22-15(24)14-5-2-10(18)9-21-14/h2-5,8-9H,6-7H2,1H3,(H2,20,23)(H,22,24)/t17-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.840 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibition of BACE1 (unknown origin) assessed as reduction in amyloid beta production by cell based assay |
J Med Chem 61: 5122-5137 (2018)
Article DOI: 10.1021/acs.jmedchem.8b00002
BindingDB Entry DOI: 10.7270/Q27W6FSQ |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data





















































