

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50269559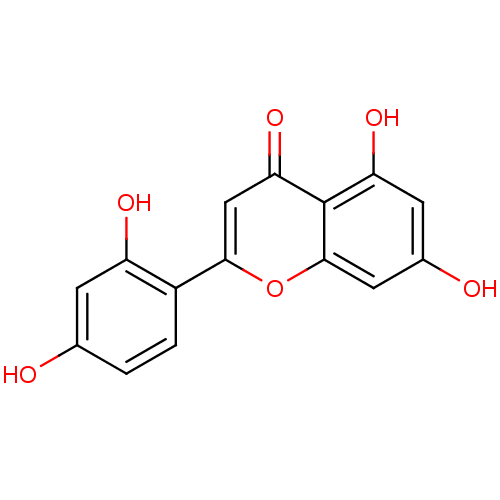 (2-(2,4-dihydroxyphenyl)-5,7-dihydroxy-4H-chromen-4...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 0.610 | -53.5 | n/a | n/a | n/a | n/a | n/a | n/a | 30 |
Gyeongsang National University | Assay Description Mushroom tyrosinase using either L-DOPA or L-tyrosine as substrate. In spectrophotometric experiments, enzyme activity was monitored by dopachrome f... | J Enzyme Inhib Med Chem 23: 922-30 (2008) Article DOI: 10.1080/14756360701810207 BindingDB Entry DOI: 10.7270/Q2P849G3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50343137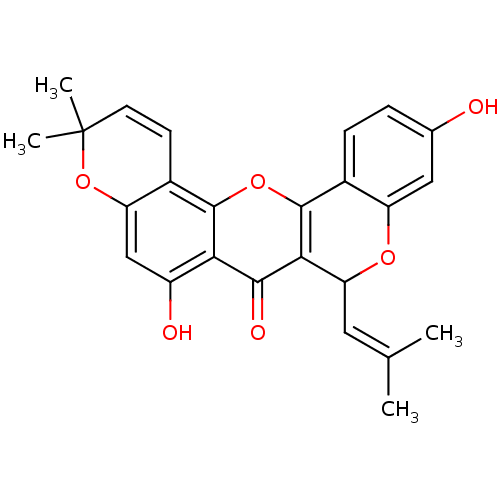 (CHEMBL1770313 | Cyclomorusin | Cycolmorusin, 2) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 46.4 | -42.6 | n/a | n/a | n/a | n/a | n/a | n/a | 30 |
Gyeongsang National University | Assay Description Mushroom tyrosinase using either L-DOPA or L-tyrosine as substrate. In spectrophotometric experiments, enzyme activity was monitored by dopachrome f... | J Enzyme Inhib Med Chem 23: 922-30 (2008) Article DOI: 10.1080/14756360701810207 BindingDB Entry DOI: 10.7270/Q2P849G3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50254430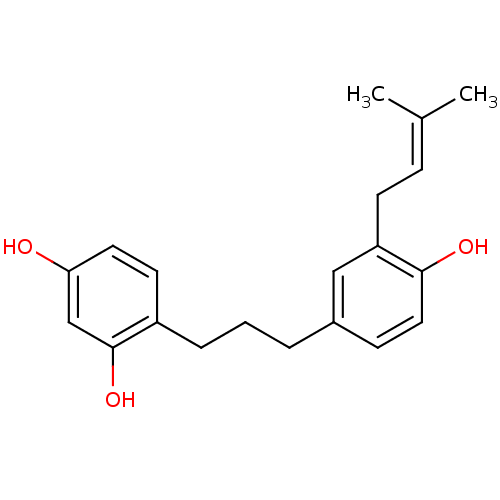 (CHEMBL468906 | broussonin C) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem | Article PubMed | 48.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Inhibition of mushroom tyrosinase by kinetic based assay | Bioorg Med Chem 17: 35-41 (2008) Article DOI: 10.1016/j.bmc.2008.11.022 BindingDB Entry DOI: 10.7270/Q2GH9HTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM91591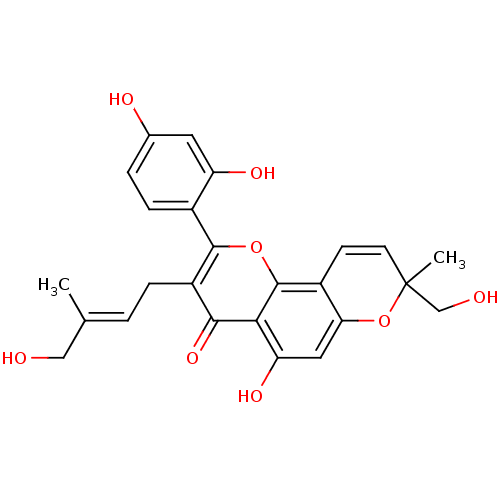 (Mormin, 1) | UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 49.2 | -42.4 | n/a | n/a | n/a | n/a | n/a | n/a | 30 |
Gyeongsang National University | Assay Description Mushroom tyrosinase using either L-DOPA or L-tyrosine as substrate. In spectrophotometric experiments, enzyme activity was monitored by dopachrome f... | J Enzyme Inhib Med Chem 23: 922-30 (2008) Article DOI: 10.1080/14756360701810207 BindingDB Entry DOI: 10.7270/Q2P849G3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50278443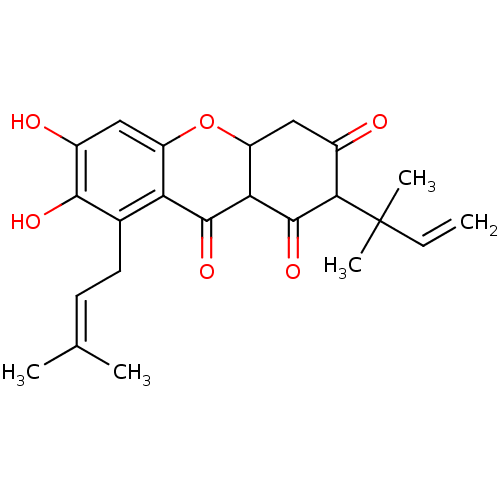 (1,3,6,7-tetrahydroxy-2-(3-methylbut-2-enyl)-8-(2-m...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 58 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by Lineweaver-Burke plot and Dixon plot | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50242015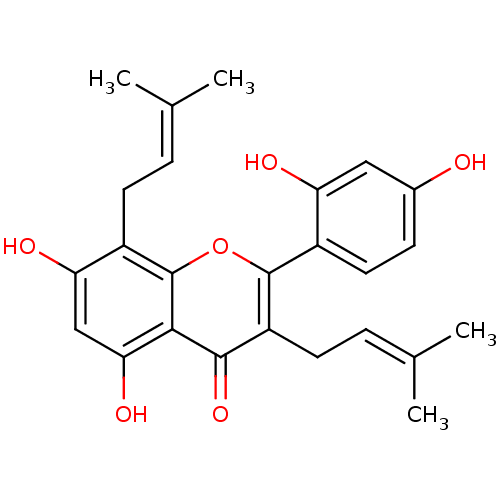 (CHEMBL518543 | Kuwanon C, 4 | kuwanon C) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 80.5 | -41.2 | n/a | n/a | n/a | n/a | n/a | n/a | 30 |
Gyeongsang National University | Assay Description Mushroom tyrosinase using either L-DOPA or L-tyrosine as substrate. In spectrophotometric experiments, enzyme activity was monitored by dopachrome f... | J Enzyme Inhib Med Chem 23: 922-30 (2008) Article DOI: 10.1080/14756360701810207 BindingDB Entry DOI: 10.7270/Q2P849G3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50175018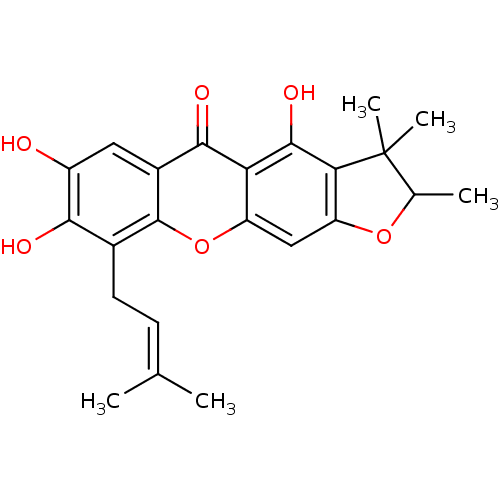 (4,7,8-trihydroxy-2,3,3-trimethyl-9-(3-methylbut-2-...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 98 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by Lineweaver-Burke plot and Dixon plot | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50378020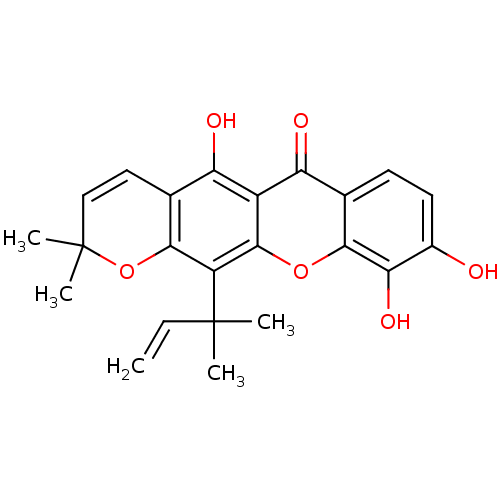 (MACLURAXANTHONE) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem | Article PubMed | 103 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by Lineweaver-Burke plot and Dixon plot | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50278443 (1,3,6,7-tetrahydroxy-2-(3-methylbut-2-enyl)-8-(2-m...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 127 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Apparent binding affinity at Clostridium perfringens neuraminidase by fluorimetry | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50278346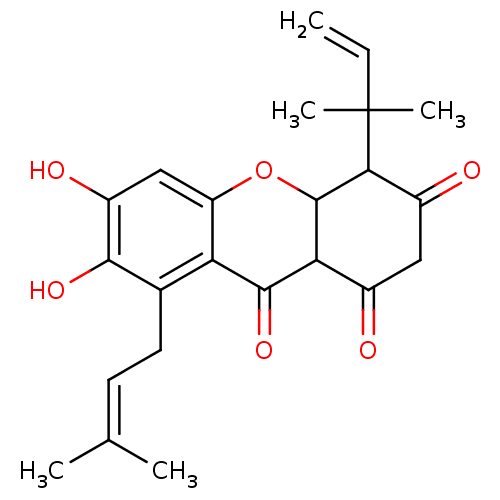 (CHEMBL470844 | Cudratricusxanthone) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 136 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by Lineweaver-Burke plot and Dixon plot | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50175013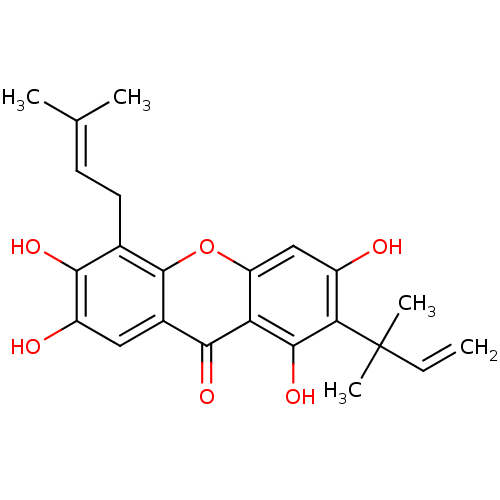 (1,3,6,7-tetrahydroxy-5-(3-methylbut-2-enyl)-2-(2-m...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 138 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by Lineweaver-Burke plot and Dixon plot | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50175019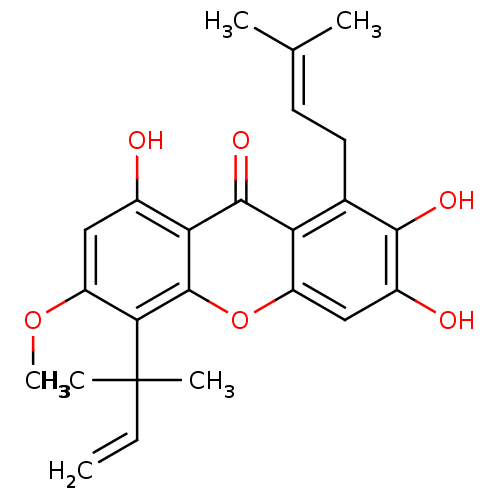 (2,3,8-trihydroxy-6-methoxy-1-(3-methylbut-2-enyl)-...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 143 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by Lineweaver-Burke plot and Dixon plot | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50325675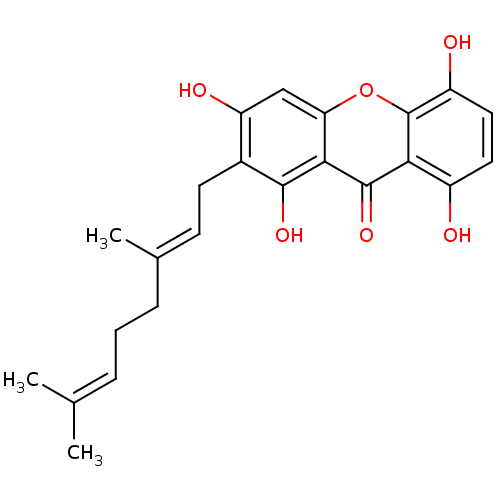 (CHEMBL480158 | smeathxanthone A) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by fluorometry | Bioorg Med Chem 18: 6258-64 (2010) Article DOI: 10.1016/j.bmc.2010.07.033 BindingDB Entry DOI: 10.7270/Q2BK1CK4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50251001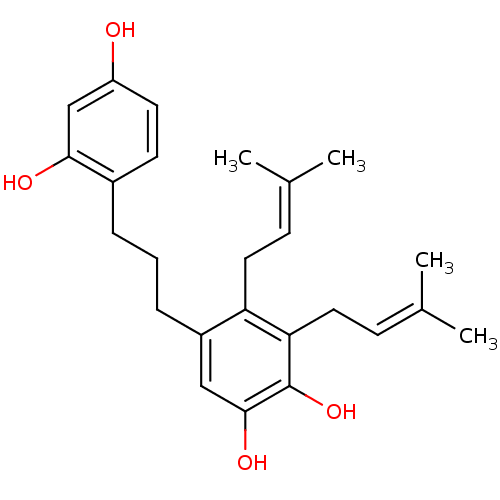 (CHEMBL457677 | Kazinol F) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 181 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Inhibition of mushroom tyrosinase by kinetic based assay | Bioorg Med Chem 17: 35-41 (2008) Article DOI: 10.1016/j.bmc.2008.11.022 BindingDB Entry DOI: 10.7270/Q2GH9HTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50278346 (CHEMBL470844 | Cudratricusxanthone) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 214 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Apparent binding affinity at Clostridium perfringens neuraminidase by fluorimetry | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50254430 (CHEMBL468906 | broussonin C) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem | Article PubMed | 230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Inhibition of monophenolase activity of mushroom tyrosinase using as L-tyrosine substrate by Lineweaver-Burk plot based kinetic assay | Bioorg Med Chem 17: 35-41 (2008) Article DOI: 10.1016/j.bmc.2008.11.022 BindingDB Entry DOI: 10.7270/Q2GH9HTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50254430 (CHEMBL468906 | broussonin C) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem | Article PubMed | 290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Inhibition of diphenolase activity of mushroom tyrosinase using as L-DOPA substrate by Lineweaver-Burk plot based kinetic assay | Bioorg Med Chem 17: 35-41 (2008) Article DOI: 10.1016/j.bmc.2008.11.022 BindingDB Entry DOI: 10.7270/Q2GH9HTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50251001 (CHEMBL457677 | Kazinol F) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Inhibition of monophenolase activity of mushroom tyrosinase using as L-tyrosine substrate by Lineweaver-Burk plot based kinetic assay | Bioorg Med Chem 17: 35-41 (2008) Article DOI: 10.1016/j.bmc.2008.11.022 BindingDB Entry DOI: 10.7270/Q2GH9HTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50175019 (2,3,8-trihydroxy-6-methoxy-1-(3-methylbut-2-enyl)-...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 436 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Apparent binding affinity at Clostridium perfringens neuraminidase by fluorimetry | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50378020 (MACLURAXANTHONE) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem | Article PubMed | 477 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Apparent binding affinity at Clostridium perfringens neuraminidase by fluorimetry | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50175018 (4,7,8-trihydroxy-2,3,3-trimethyl-9-(3-methylbut-2-...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 510 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Apparent binding affinity at Clostridium perfringens neuraminidase by fluorimetry | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50175013 (1,3,6,7-tetrahydroxy-5-(3-methylbut-2-enyl)-2-(2-m...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 568 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Apparent binding affinity at Clostridium perfringens neuraminidase by fluorimetry | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50251001 (CHEMBL457677 | Kazinol F) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 770 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Inhibition of diphenolase activity of mushroom tyrosinase using as L-DOPA substrate by Lineweaver-Burk plot based kinetic assay | Bioorg Med Chem 17: 35-41 (2008) Article DOI: 10.1016/j.bmc.2008.11.022 BindingDB Entry DOI: 10.7270/Q2GH9HTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50241453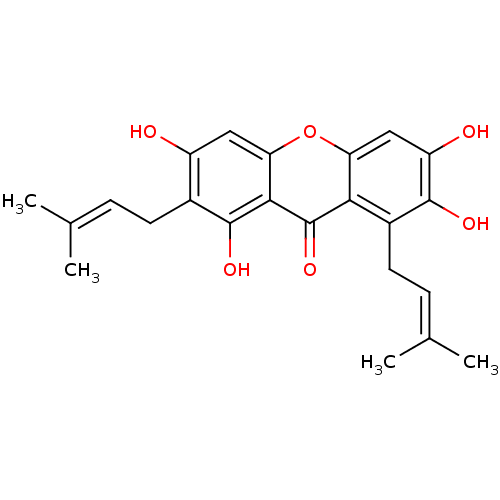 (1,3,6,7-tetrahydroxy-2,8-bis(3-methylbut-2-enyl)-9...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by fluorometry | Bioorg Med Chem 18: 6258-64 (2010) Article DOI: 10.1016/j.bmc.2010.07.033 BindingDB Entry DOI: 10.7270/Q2BK1CK4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Replicase polyprotein 1ab (Human SARS coronavirus (SARS-CoV) (Severe acute re...) | BDBM50481948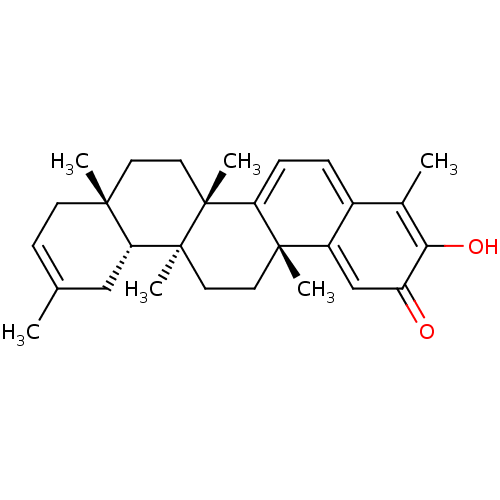 (Iguesterin | acs.jmedchem.1c00409_ST.224) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | 800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology Curated by ChEMBL | Assay Description Inhibition of 3C-like protease of SARS coronavirus assessed as concentration of FRET peptide for 60 mins by dixon plot | Bioorg Med Chem Lett 20: 1873-6 (2010) Article DOI: 10.1016/j.bmcl.2010.01.152 BindingDB Entry DOI: 10.7270/Q28P63CF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50254431 (5'-(2-methylbut-3-en-2-yl)-6''-(3-methylbut-2-enyl...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Inhibition of mushroom tyrosinase by kinetic based assay | Bioorg Med Chem 17: 35-41 (2008) Article DOI: 10.1016/j.bmc.2008.11.022 BindingDB Entry DOI: 10.7270/Q2GH9HTH | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50278394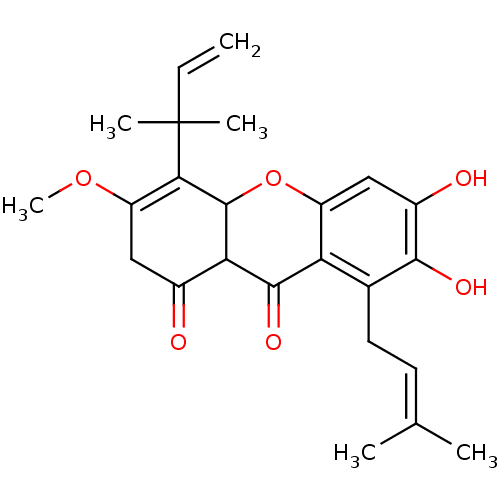 (CHEMBL469813 | Cudratricusxanthone F) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 950 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by Lineweaver-Burke plot and Dixon plot | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxylic ester hydrolase (Equus caballus (Horse)) | BDBM50380205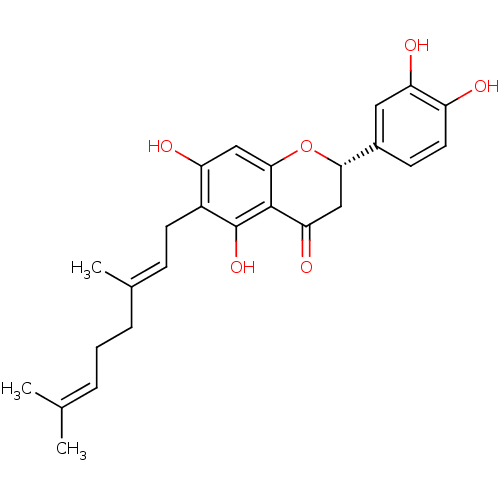 (NYMPHAEOL A) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 1.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Mixed type inhibition of equine BChE using butyrylthiocholine iodide as substrate by Lineweaver-Burk double-reciprocal-plot and dixon plot analysis | Bioorg Med Chem 20: 2595-602 (2012) Article DOI: 10.1016/j.bmc.2012.02.044 BindingDB Entry DOI: 10.7270/Q22808MC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Beta-secretase 1 (Homo sapiens (Human)) | BDBM50242015 (CHEMBL518543 | Kuwanon C, 4 | kuwanon C) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Gyeongsang National University Curated by ChEMBL | Assay Description Noncompetitive inhibition of human recombinant BACE-1 by Dixon plot analysis | Bioorg Med Chem Lett 21: 2945-8 (2011) Article DOI: 10.1016/j.bmcl.2011.03.060 BindingDB Entry DOI: 10.7270/Q2QR4XF7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50278394 (CHEMBL469813 | Cudratricusxanthone F) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.23E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Apparent binding affinity at Clostridium perfringens neuraminidase by fluorimetry | Bioorg Med Chem 17: 2744-50 (2009) Article DOI: 10.1016/j.bmc.2009.02.042 BindingDB Entry DOI: 10.7270/Q2V69KG5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Replicase polyprotein 1ab (Human SARS coronavirus (SARS-CoV) (Severe acute re...) | BDBM50259983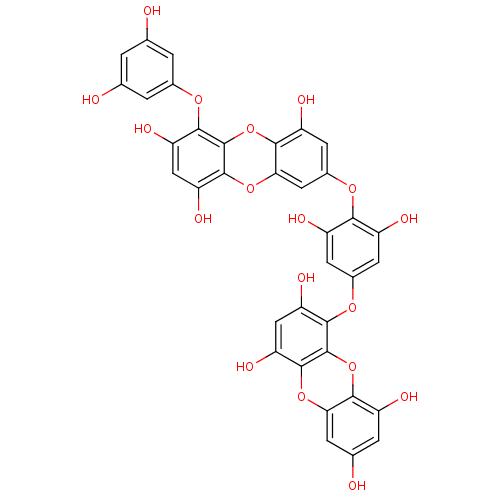 (CHEMBL508791 | US10106521, Compound Dieckol | diec...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology Curated by ChEMBL | Assay Description Competitive inhibition of C-terminal His6-tagged recombinant SARS coronavirus 3C-like protease trans-cleavage activity expressed in Escherichia coli ... | Bioorg Med Chem 21: 3730-7 (2013) Article DOI: 10.1016/j.bmc.2013.04.026 BindingDB Entry DOI: 10.7270/Q24J0J2W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50068224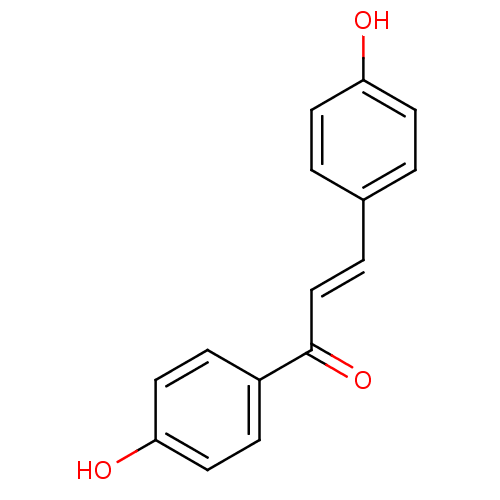 ((E)-1,3-Bis-(4-hydroxy-phenyl)-propenone | 1,3-bis...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Functional Crop Curated by ChEMBL | Assay Description Competitive inhibition of monophenolase activity of mushroom tyrosinase using as L-tyrosine substrate by Lineweaver-Burk plot analysis | Eur J Med Chem 45: 2010-7 (2010) Article DOI: 10.1016/j.ejmech.2010.01.049 BindingDB Entry DOI: 10.7270/Q2H70FZM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Replicase polyprotein 1ab (Human SARS coronavirus (SARS-CoV) (Severe acute re...) | BDBM50481947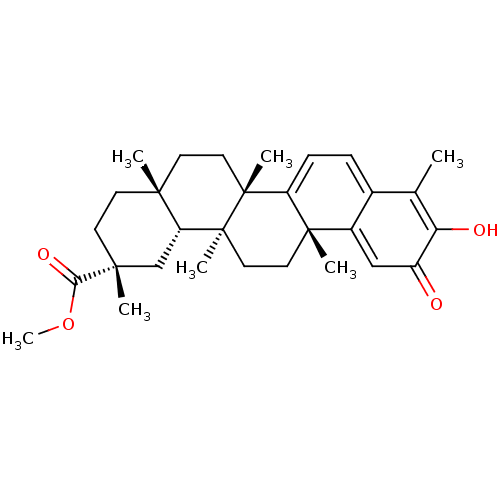 (CHEBI:8416 | GNF-Pf-476 | PRISTIMERIN | Pristimeri...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase MCE KEGG PC cid PC sid UniChem | Article PubMed | 3.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology Curated by ChEMBL | Assay Description Inhibition of 3C-like protease of SARS coronavirus assessed as concentration of FRET peptide for 60 mins by dixon plot | Bioorg Med Chem Lett 20: 1873-6 (2010) Article DOI: 10.1016/j.bmcl.2010.01.152 BindingDB Entry DOI: 10.7270/Q28P63CF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-glucosidase MAL12 (Saccharomyces cerevisiae) | BDBM84968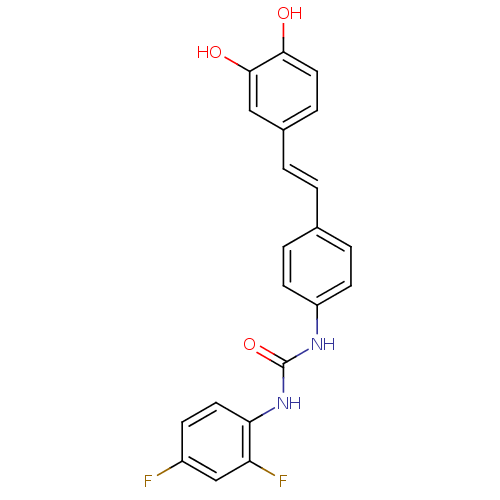 (Urea derivative, 12) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 3.20E+3 | n/a | 8.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Gyeongsang National University | Assay Description All enzymatic activities were determined by using the appropriate substrate (p-nitrophenyl-alpha-D-glucopyranoside, p-nitrophenyl-beta-D-gulcopyranos... | Chembiochem 11: 2125-31 (2010) Article DOI: 10.1002/cbic.201000376 BindingDB Entry DOI: 10.7270/Q2542M33 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50250510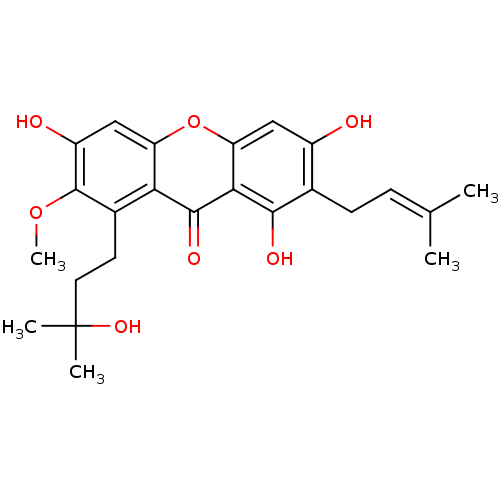 (CHEMBL462879 | GARCINONE D) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem | Article PubMed | 3.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by fluorometry | Bioorg Med Chem 18: 6258-64 (2010) Article DOI: 10.1016/j.bmc.2010.07.033 BindingDB Entry DOI: 10.7270/Q2BK1CK4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50311743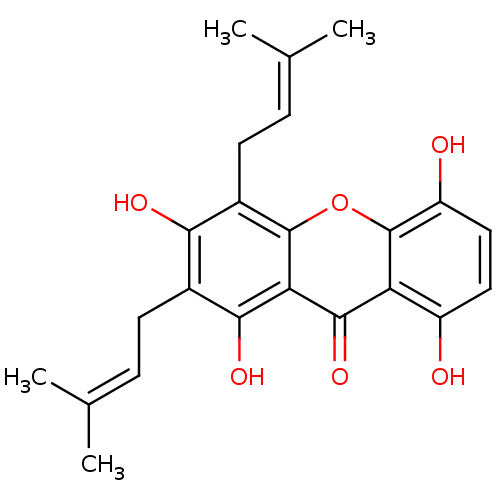 (CHEMBL487992 | GARTANIN) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 3.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by fluorometry | Bioorg Med Chem 18: 6258-64 (2010) Article DOI: 10.1016/j.bmc.2010.07.033 BindingDB Entry DOI: 10.7270/Q2BK1CK4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Polyphenol oxidase 2 (Agaricus bisporus (Common mushroom)) | BDBM50174839 (1-(4-aminophenyl)-3-(4-hydroxyphenyl)prop-2-en-1-o...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Department of Functional Crop Curated by ChEMBL | Assay Description Competitive inhibition of monophenolase activity of mushroom tyrosinase using as L-tyrosine substrate by Lineweaver-Burk plot analysis | Eur J Med Chem 45: 2010-7 (2010) Article DOI: 10.1016/j.ejmech.2010.01.049 BindingDB Entry DOI: 10.7270/Q2H70FZM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM412955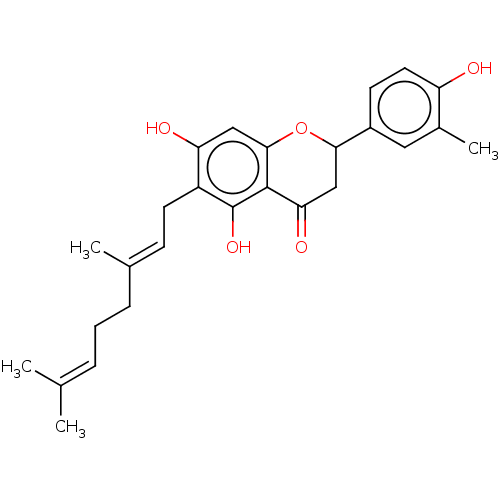 (US10406136, Compound 8) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | UniChem | US Patent | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INDUSTRY-ACADEMIC COOPERATION FOUNDATION GYEONGSANG NATIONAL UNIVERSITY US Patent | Assay Description Specifically, in order to determine IC50 of the compounds for neuraminidase, 0.01 U/ml of neuraminidase (EC. 3.2.1.8, C. perfringens, SIGMA, N2876) a... | US Patent US10406136 (2019) BindingDB Entry DOI: 10.7270/Q2NS0X86 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Replicase polyprotein 1ab (Human SARS coronavirus (SARS-CoV) (Severe acute re...) | BDBM50071055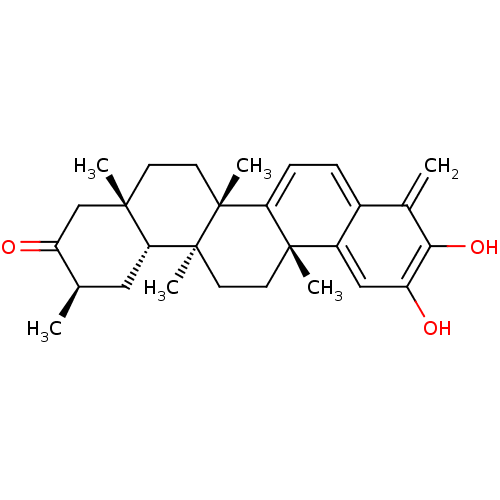 ((6bS,8aS,11R,12aR,12bS,14aR)-3-Hydroxy-4,6b,8a,11,...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology Curated by ChEMBL | Assay Description Inhibition of 3C-like protease of SARS coronavirus assessed as concentration of FRET peptide for 60 mins by dixon plot | Bioorg Med Chem Lett 20: 1873-6 (2010) Article DOI: 10.1016/j.bmcl.2010.01.152 BindingDB Entry DOI: 10.7270/Q28P63CF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Replicase polyprotein 1ab (Human SARS coronavirus (SARS-CoV) (Severe acute re...) | BDBM50071058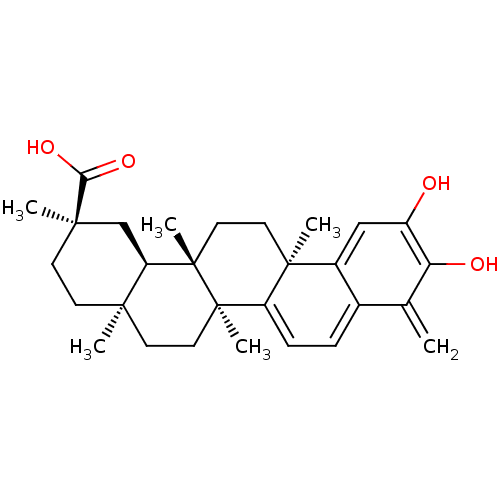 ((2R,4aS,6aS,12bR,14aS,14bR)-10-Hydroxy-2,4a,6a,9,1...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 4.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology Curated by ChEMBL | Assay Description Inhibition of 3C-like protease of SARS coronavirus assessed as concentration of FRET peptide for 60 mins by dixon plot | Bioorg Med Chem Lett 20: 1873-6 (2010) Article DOI: 10.1016/j.bmcl.2010.01.152 BindingDB Entry DOI: 10.7270/Q28P63CF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxylic ester hydrolase (Equus caballus (Horse)) | BDBM50380201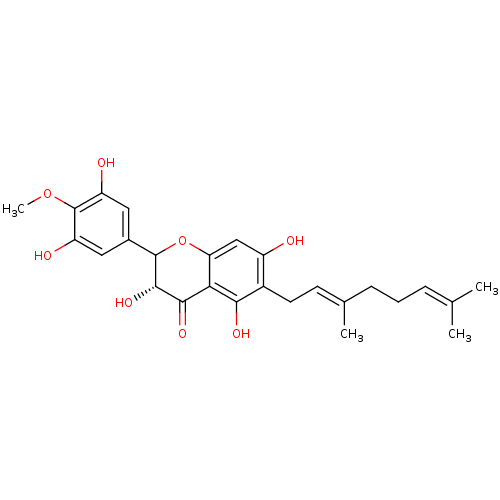 (CHEMBL2011403) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Mixed type inhibition of equine BChE using butyrylthiocholine iodide as substrate by Lineweaver-Burk double-reciprocal-plot and dixon plot analysis | Bioorg Med Chem 20: 2595-602 (2012) Article DOI: 10.1016/j.bmc.2012.02.044 BindingDB Entry DOI: 10.7270/Q22808MC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-glucosidase MAL12 (Saccharomyces cerevisiae) | BDBM84969 (Urea derivative, 13) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 4.60E+3 | n/a | 1.43E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Gyeongsang National University | Assay Description All enzymatic activities were determined by using the appropriate substrate (p-nitrophenyl-alpha-D-glucopyranoside, p-nitrophenyl-beta-D-gulcopyranos... | Chembiochem 11: 2125-31 (2010) Article DOI: 10.1002/cbic.201000376 BindingDB Entry DOI: 10.7270/Q2542M33 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50214969 (1,3,6-Trihydroxy-7-methoxy-2,8-bis-(3-methyl-but-2...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE KEGG PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 5.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by fluorometry | Bioorg Med Chem 18: 6258-64 (2010) Article DOI: 10.1016/j.bmc.2010.07.033 BindingDB Entry DOI: 10.7270/Q2BK1CK4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50311741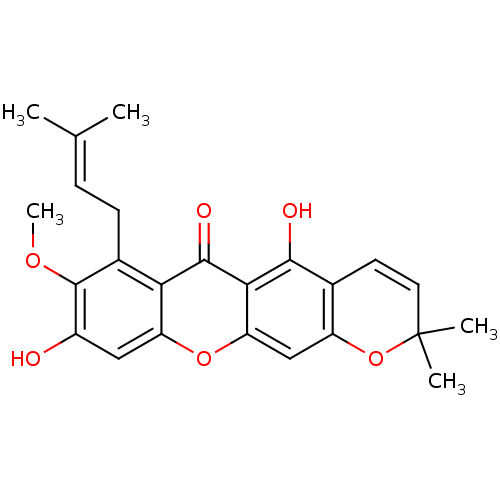 (1,6-dihydroxy-7-methoxy-8-(3-methylbut-2-enyl)-6',...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 5.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by fluorometry | Bioorg Med Chem 18: 6258-64 (2010) Article DOI: 10.1016/j.bmc.2010.07.033 BindingDB Entry DOI: 10.7270/Q2BK1CK4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50325676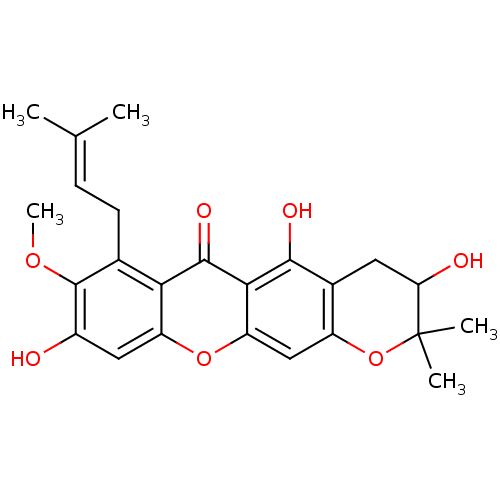 (CHEMBL1224250 | mangostanol) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 6.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by fluorometry | Bioorg Med Chem 18: 6258-64 (2010) Article DOI: 10.1016/j.bmc.2010.07.033 BindingDB Entry DOI: 10.7270/Q2BK1CK4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50325678 (4,8,10-trihydroxy-9-(3-methylbut-2-enyl)-2-(prop-1...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 6.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by fluorometry | Bioorg Med Chem 18: 6258-64 (2010) Article DOI: 10.1016/j.bmc.2010.07.033 BindingDB Entry DOI: 10.7270/Q2BK1CK4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Alpha-glucosidase MAL12 (Saccharomyces cerevisiae) | BDBM84967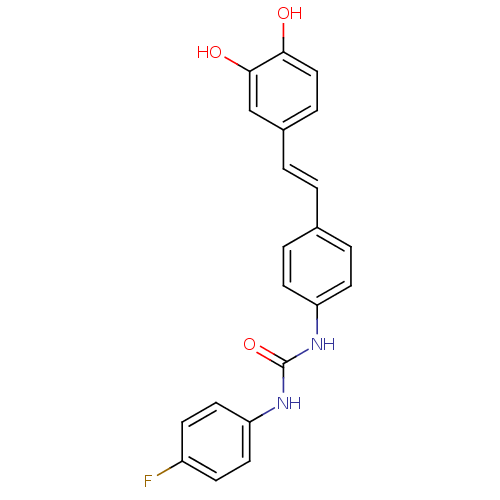 (Urea derivative, 11) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 7.20E+3 | n/a | 1.98E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Gyeongsang National University | Assay Description All enzymatic activities were determined by using the appropriate substrate (p-nitrophenyl-alpha-D-glucopyranoside, p-nitrophenyl-beta-D-gulcopyranos... | Chembiochem 11: 2125-31 (2010) Article DOI: 10.1002/cbic.201000376 BindingDB Entry DOI: 10.7270/Q2542M33 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxylic ester hydrolase (Equus caballus (Horse)) | BDBM50380204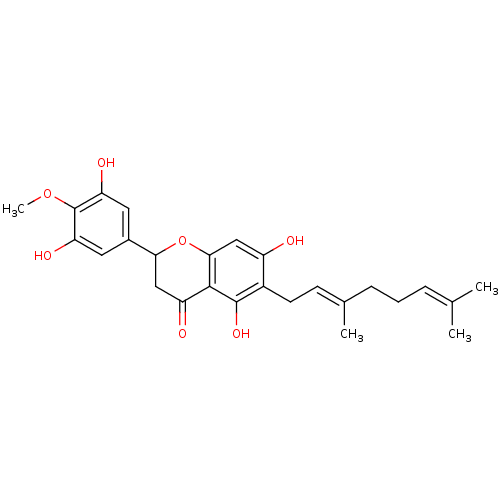 (CHEMBL2011405 | US10406136, Compound 6) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Mixed type inhibition of equine BChE using butyrylthiocholine iodide as substrate by Lineweaver-Burk double-reciprocal-plot and dixon plot analysis | Bioorg Med Chem 20: 2595-602 (2012) Article DOI: 10.1016/j.bmc.2012.02.044 BindingDB Entry DOI: 10.7270/Q22808MC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase (Clostridium perfringens) | BDBM50325677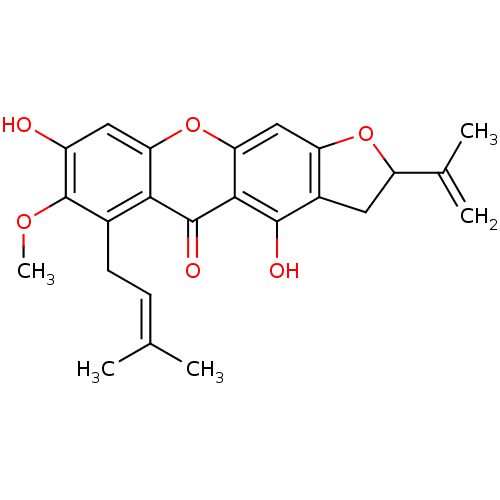 (4,8-dihydroxy-7-methoxy-6-(3-methylbut-2-enyl)-2-(...) | UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 7.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Graduate School of Gyeongsang National University Curated by ChEMBL | Assay Description Competitive inhibition of Clostridium perfringens neuraminidase by fluorometry | Bioorg Med Chem 18: 6258-64 (2010) Article DOI: 10.1016/j.bmc.2010.07.033 BindingDB Entry DOI: 10.7270/Q2BK1CK4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Replicase polyprotein 1ab (Human SARS coronavirus (SARS-CoV) (Severe acute re...) | BDBM50259982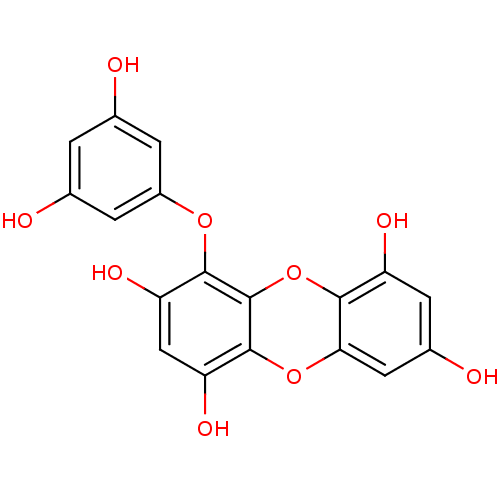 (CHEMBL471187 | US10106521, Compound Eckol | eckol) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 8.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Korea Research Institute of Bioscience and Biotechnology Curated by ChEMBL | Assay Description Competitive inhibition of C-terminal His6-tagged recombinant SARS coronavirus 3C-like protease trans-cleavage activity expressed in Escherichia coli ... | Bioorg Med Chem 21: 3730-7 (2013) Article DOI: 10.1016/j.bmc.2013.04.026 BindingDB Entry DOI: 10.7270/Q24J0J2W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 352 total ) | Next | Last >> |