

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8143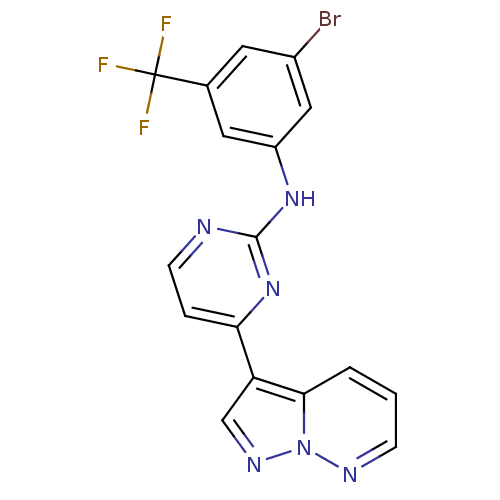 (N-[3-bromo-5-(trifluoromethyl)phenyl]-4-{pyrazolo[...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 0.300 | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Homo sapiens (Human)) | BDBM50086433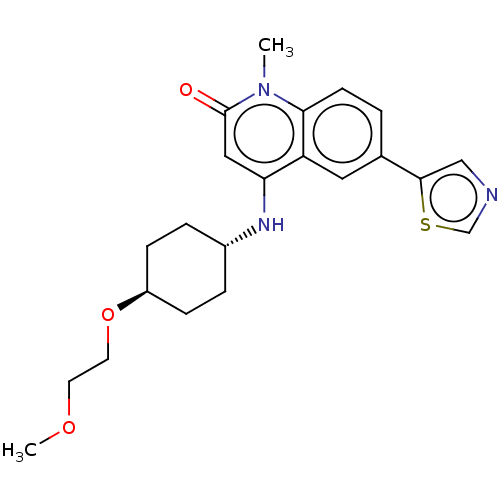 (CHEMBL3426034) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of wild type fully glycosylated human recombinant CD38-catalyzed NAD hydrolysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Homo sapiens (Human)) | BDBM50086438 (CHEMBL3426039) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of wild type fully glycosylated human recombinant CD38-catalyzed NAD hydrolysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Homo sapiens (Human)) | BDBM50086434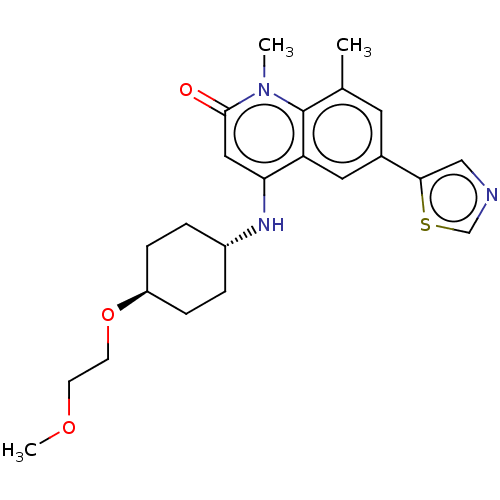 (CHEMBL3426035) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of wild type fully glycosylated human recombinant CD38-catalyzed NAD hydrolysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8138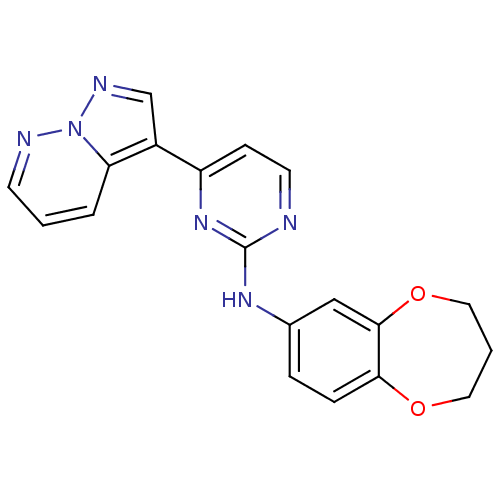 (N-(3,4-dihydro-2H-1,5-benzodioxepin-7-yl)-4-{pyraz...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1 | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8145 (N-(3,5-dichlorophenyl)-4-{pyrazolo[1,5-a]pyridazin...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1 | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8146 (N-(3,5-dimethylphenyl)-4-{pyrazolo[1,5-a]pyridazin...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1 | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8136 (N-(2,3-dihydro-1,4-benzodioxin-6-yl)-4-{pyrazolo[1...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 1 | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8144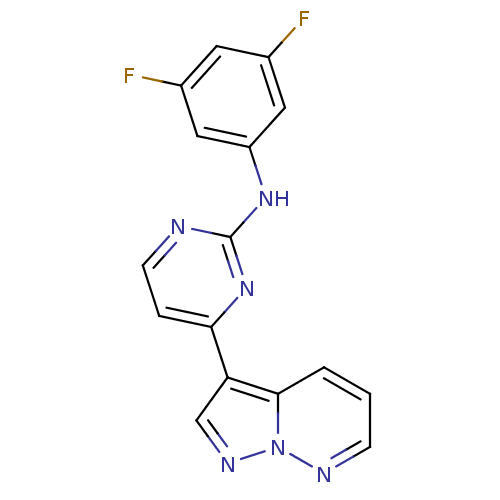 (N-(3,5-difluorophenyl)-4-{pyrazolo[1,5-a]pyridazin...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 2 | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8130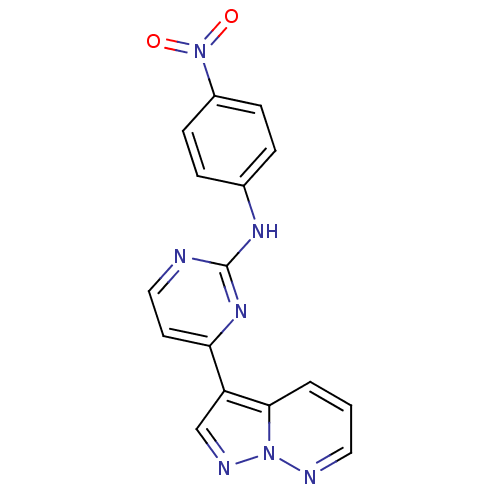 (N-(4-nitrophenyl)-4-{pyrazolo[1,5-a]pyridazin-3-yl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | 2 | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8129 (4-[(4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidin-2-y...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 2 | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8171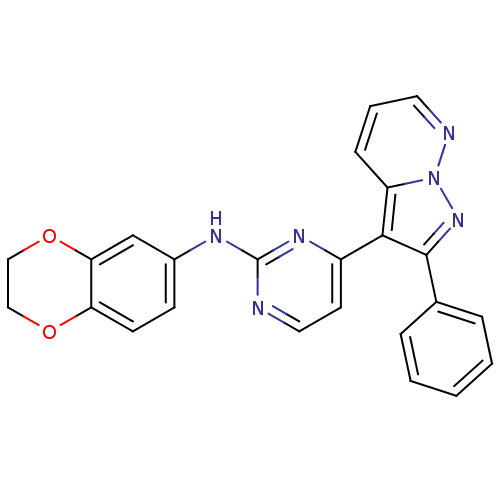 (N-(2,3-dihydro-1,4-benzodioxin-6-yl)-4-{2-phenylpy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 2 | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8135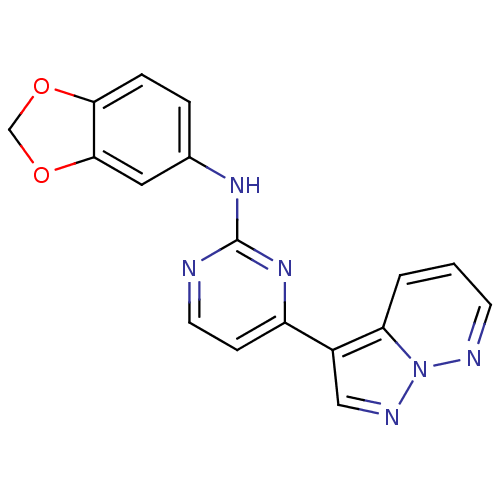 (N-(2H-1,3-benzodioxol-5-yl)-4-{pyrazolo[1,5-a]pyri...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 2 | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8142 (N-[3-methoxy-5-(trifluoromethyl)phenyl]-4-{pyrazol...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 2 | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8140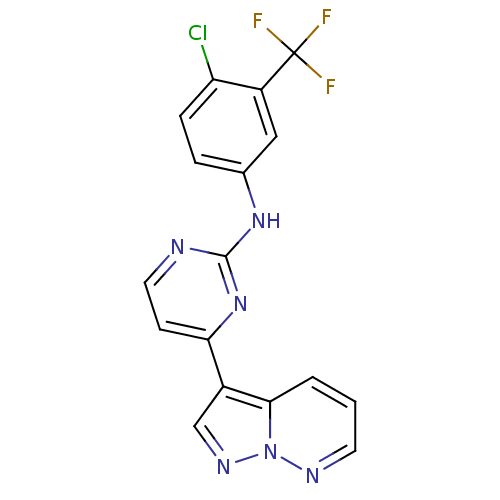 (N-[4-chloro-3-(trifluoromethyl)phenyl]-4-{pyrazolo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | 2 | n/a | <10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Homo sapiens (Human)) | BDBM50086425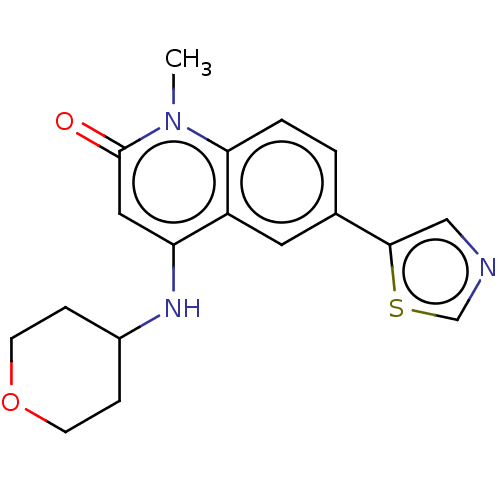 (CHEMBL3426030) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of wild type fully glycosylated human recombinant CD38-catalyzed NAD hydrolysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086336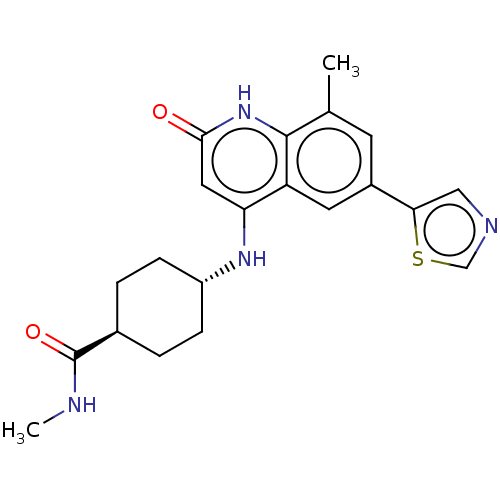 (CHEMBL3426065) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | <0.180 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086432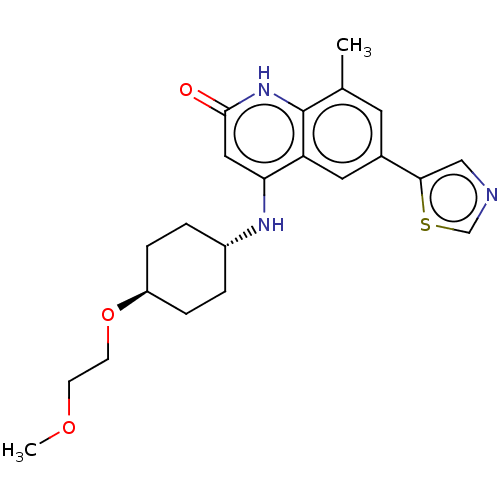 (CHEMBL3426033) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.620 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086436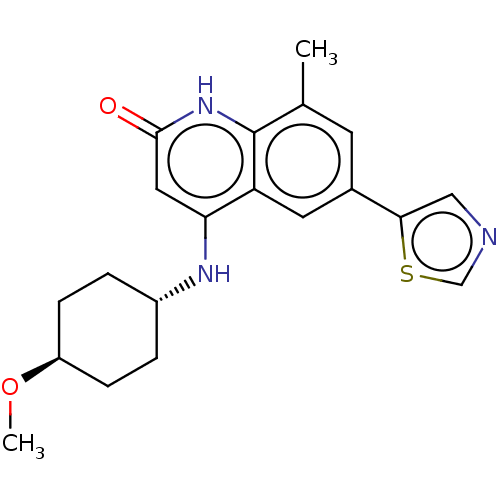 (CHEMBL3426037) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.650 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086334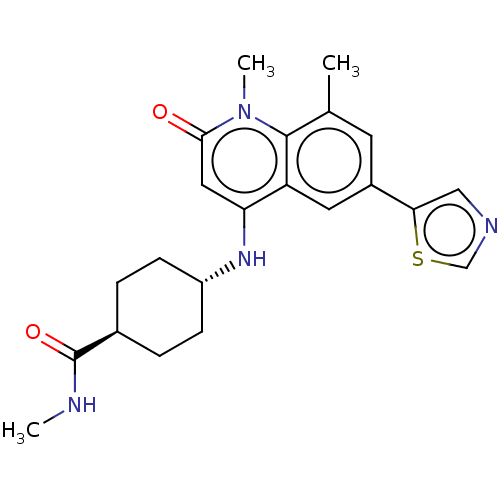 (CHEMBL3426067) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 0.780 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086337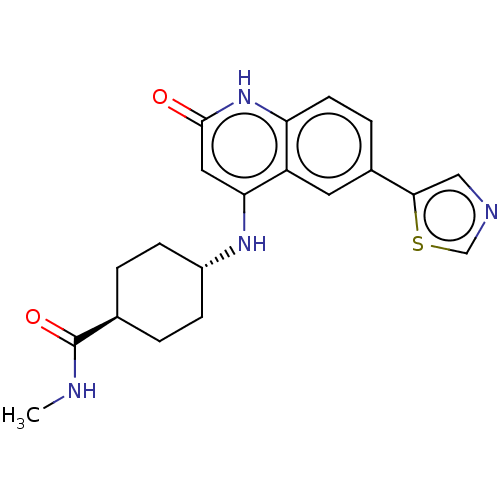 (CHEMBL3426064) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclin-A2/Cyclin-dependent kinase 2 (Homo sapiens (Human)) | BDBM8129 (4-[(4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidin-2-y...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | 7.2 | 22 |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclin-A2/Cyclin-dependent kinase 2 (Homo sapiens (Human)) | BDBM8139 (N-(3,4-dichlorophenyl)-4-{pyrazolo[1,5-a]pyridazin...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | 7.2 | 22 |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclin-A2/Cyclin-dependent kinase 2 (Homo sapiens (Human)) | BDBM8144 (N-(3,5-difluorophenyl)-4-{pyrazolo[1,5-a]pyridazin...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086350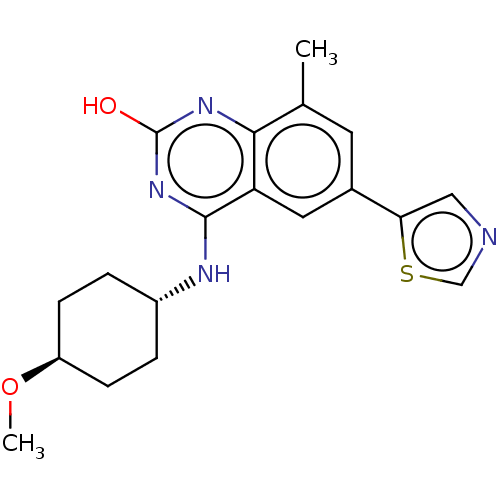 (CHEMBL3426058) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086335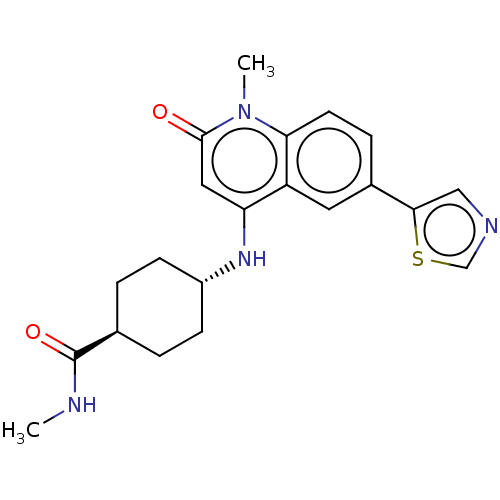 (CHEMBL3426066) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086431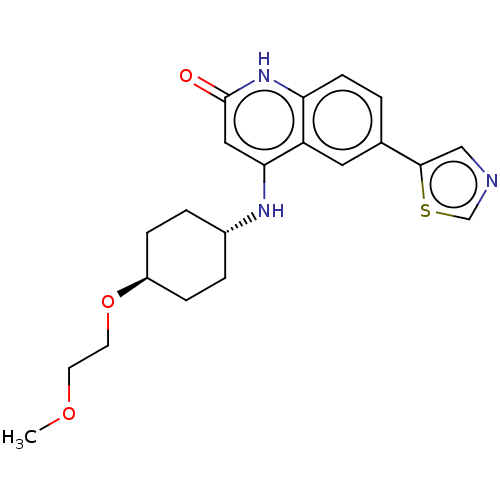 (CHEMBL3426032) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086438 (CHEMBL3426039) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Homo sapiens (Human)) | BDBM50086336 (CHEMBL3426065) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of human CD38 extracellular domain expressed in Pichia pastoris assessed as NAD hydrolysis by colorimetric-based assay | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086434 (CHEMBL3426035) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086437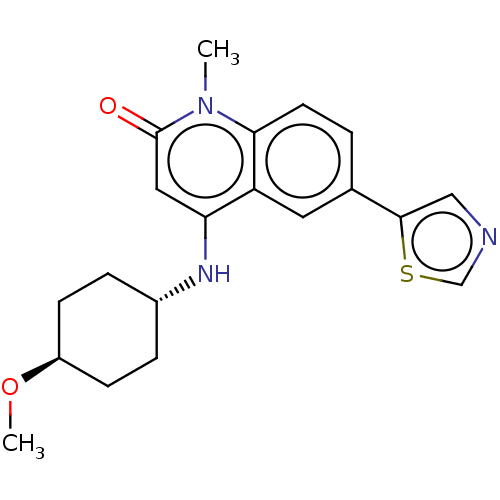 (CHEMBL3426038) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086433 (CHEMBL3426034) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.90 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclin-A2/Cyclin-dependent kinase 2 (Homo sapiens (Human)) | BDBM8128 (N-(3-methoxyphenyl)-4-{pyrazolo[1,5-a]pyridazin-3-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | 7.2 | 22 |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclin-A2/Cyclin-dependent kinase 2 (Homo sapiens (Human)) | BDBM8137 (N-(3,4-difluorophenyl)-4-{pyrazolo[1,5-a]pyridazin...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 2 | n/a | n/a | n/a | n/a | 7.2 | 22 |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086439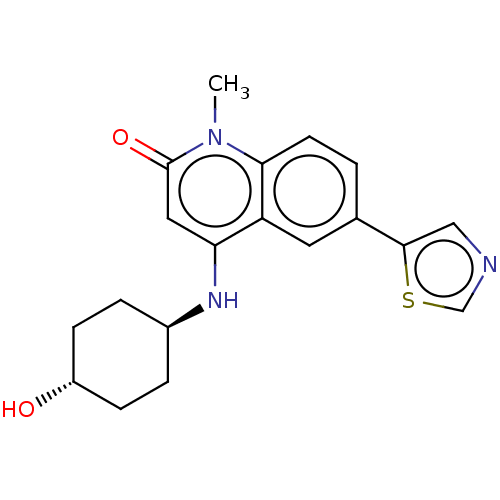 (CHEMBL3426040) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8688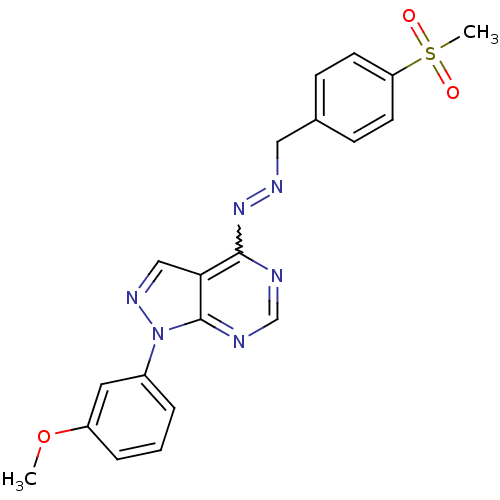 ((1E)-1-[(4-methanesulfonylphenyl)methylidene]-2-[1...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.51 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-33P] ATP. Af... | Bioorg Med Chem Lett 14: 2121-5 (2004) Article DOI: 10.1016/j.bmcl.2004.02.036 BindingDB Entry DOI: 10.7270/Q2NV9GF5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8686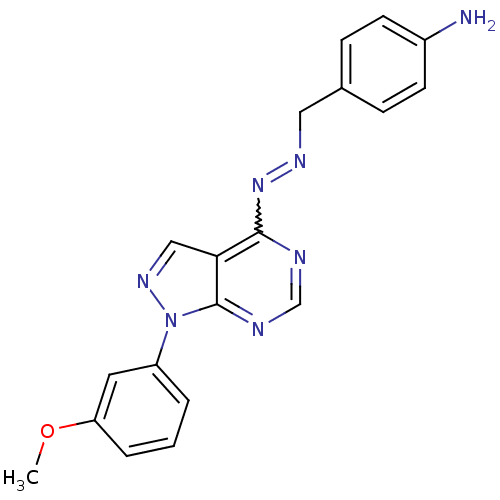 (4-[(1E)-{2-[1-(3-methoxyphenyl)-1H-pyrazolo[3,4-d]...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.51 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-33P] ATP. Af... | Bioorg Med Chem Lett 14: 2121-5 (2004) Article DOI: 10.1016/j.bmcl.2004.02.036 BindingDB Entry DOI: 10.7270/Q2NV9GF5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclin-A2/Cyclin-dependent kinase 2 (Homo sapiens (Human)) | BDBM8146 (N-(3,5-dimethylphenyl)-4-{pyrazolo[1,5-a]pyridazin...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8674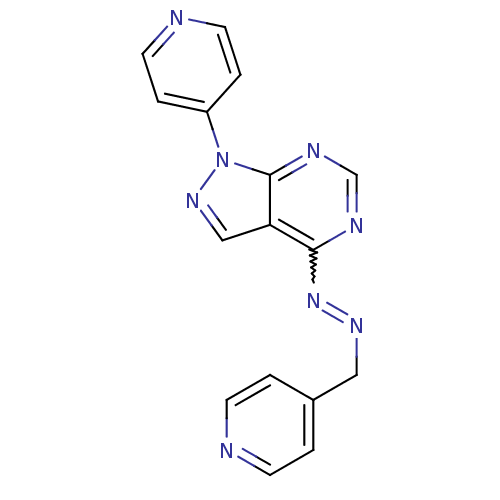 (4-{4-[(2E)-2-(pyridin-4-ylmethylidene)hydrazin-1-y...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.16 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-33P] ATP. Af... | Bioorg Med Chem Lett 14: 2121-5 (2004) Article DOI: 10.1016/j.bmcl.2004.02.036 BindingDB Entry DOI: 10.7270/Q2NV9GF5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Homo sapiens (Human)) | BDBM50086337 (CHEMBL3426064) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of human CD38 extracellular domain expressed in Pichia pastoris assessed as NAD hydrolysis by colorimetric-based assay | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Homo sapiens (Human)) | BDBM50086436 (CHEMBL3426037) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.80 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of human CD38 extracellular domain expressed in Pichia pastoris assessed as NAD hydrolysis by colorimetric-based assay | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Homo sapiens (Human)) | BDBM50086432 (CHEMBL3426033) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.90 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of human CD38 extracellular domain expressed in Pichia pastoris assessed as NAD hydrolysis by colorimetric-based assay | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8695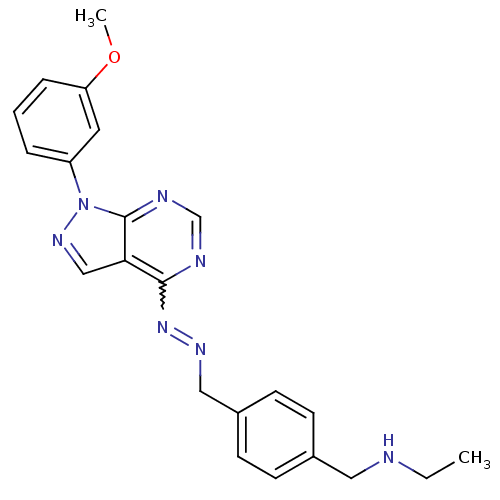 (ethyl({4-[(1E)-{2-[1-(3-methoxyphenyl)-1H-pyrazolo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.98 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-33P] ATP. Af... | Bioorg Med Chem Lett 14: 2121-5 (2004) Article DOI: 10.1016/j.bmcl.2004.02.036 BindingDB Entry DOI: 10.7270/Q2NV9GF5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8687 ((1E)-1-[(3-methanesulfonylphenyl)methylidene]-2-[1...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.98 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-33P] ATP. Af... | Bioorg Med Chem Lett 14: 2121-5 (2004) Article DOI: 10.1016/j.bmcl.2004.02.036 BindingDB Entry DOI: 10.7270/Q2NV9GF5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086351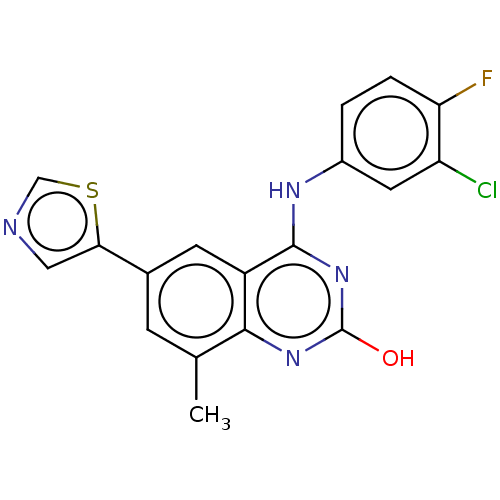 (CHEMBL3426057) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086355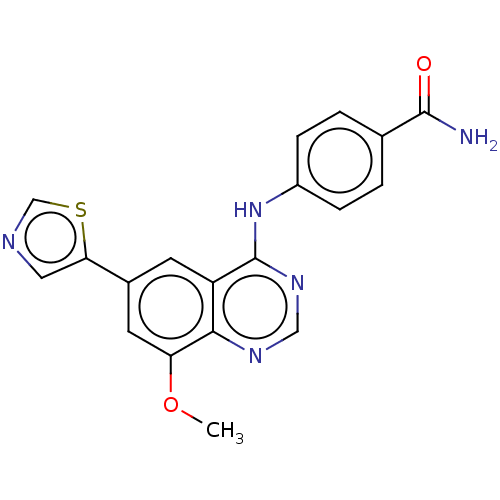 (CHEMBL3426053) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Mus musculus) | BDBM50086424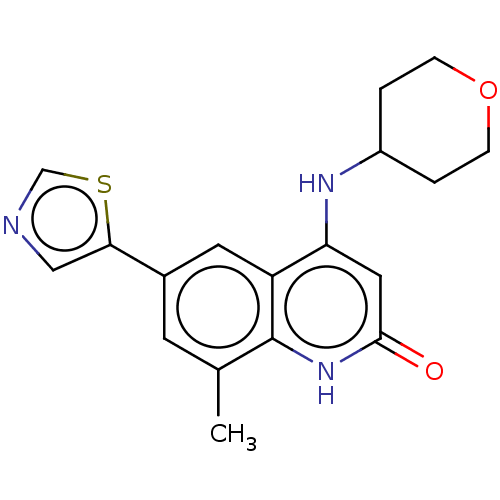 (CHEMBL3426029) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of mouse recombinant CD38 extracellular domain expressed in CHO CGE cells assessed as NAD hydrolysis by fluorescence plate reader analysis | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 (Homo sapiens (Human)) | BDBM50086335 (CHEMBL3426066) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a |
GlaxoSmithKline Curated by ChEMBL | Assay Description Inhibition of human CD38 extracellular domain expressed in Pichia pastoris assessed as NAD hydrolysis by colorimetric-based assay | J Med Chem 58: 3548-71 (2015) Article DOI: 10.1021/jm502009h BindingDB Entry DOI: 10.7270/Q2NV9M00 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cyclin-A2/Cyclin-dependent kinase 2 (Homo sapiens (Human)) | BDBM8126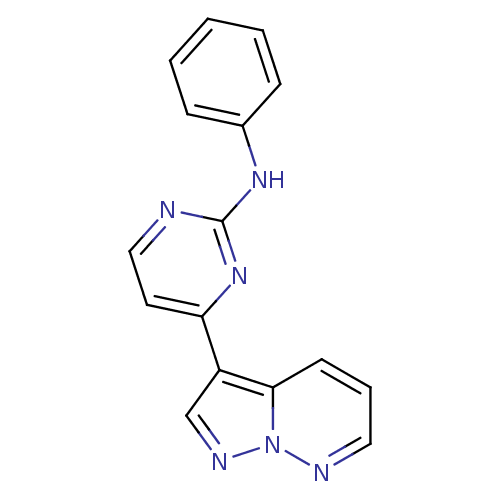 (N-phenyl-4-{pyrazolo[1,5-a]pyridazin-3-yl}pyrimidi...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | 7.2 | 22 |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-32P] ATP. Af... | J Med Chem 47: 4716-30 (2004) Article DOI: 10.1021/jm040063i BindingDB Entry DOI: 10.7270/Q2VM49HJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen synthase kinase-3 beta (Homo sapiens (Human)) | BDBM8540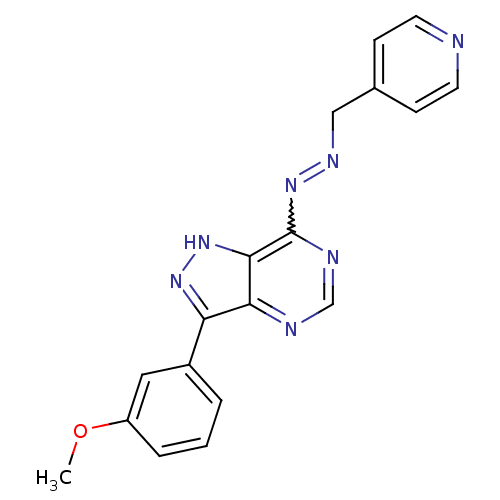 (4-[(1E)-{2-[3-(3-methoxyphenyl)-1H-pyrazolo[4,3-d]...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | Article PubMed | n/a | n/a | 5.01 | n/a | n/a | n/a | n/a | 7.2 | 22 |
GlaxoSmithKline | Assay Description The biochemical activity of compounds was determined by incubation with specific enzyme and substrate in the presence 2.5 uM ATP/ [gamma-33P] ATP. Af... | Bioorg Med Chem Lett 16: 2091-4 (2006) Article DOI: 10.1016/j.bmcl.2006.01.057 BindingDB Entry DOI: 10.7270/Q25X274X | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 546 total ) | Next | Last >> |