 Found 822 hits with Last Name = 'ramnauth' and Initial = 'j'
Found 822 hits with Last Name = 'ramnauth' and Initial = 'j' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM50176062
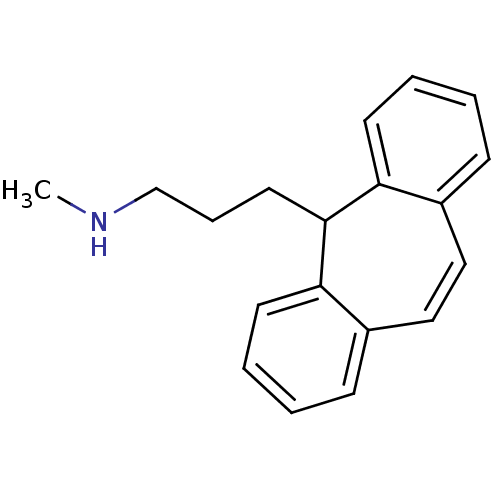
(3-(5H-dibenzo[a,d][7]annulen-5-yl)-N-methylpropan-...)Show InChI InChI=1S/C19H21N/c1-20-14-6-11-19-17-9-4-2-7-15(17)12-13-16-8-3-5-10-18(16)19/h2-5,7-10,12-13,19-20H,6,11,14H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
| DrugBank
Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]nisoxetine from human NET expressed in CHO cells after 120 mins by scintillation counting |
J Med Chem 55: 3488-501 (2012)
Article DOI: 10.1021/jm300138g
BindingDB Entry DOI: 10.7270/Q2KW5H2V |
More data for this
Ligand-Target Pair | |
Sodium-dependent noradrenaline transporter
(Homo sapiens (Human)) | BDBM50176062

(3-(5H-dibenzo[a,d][7]annulen-5-yl)-N-methylpropan-...)Show InChI InChI=1S/C19H21N/c1-20-14-6-11-19-17-9-4-2-7-15(17)12-13-16-8-3-5-10-18(16)19/h2-5,7-10,12-13,19-20H,6,11,14H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
| DrugBank
Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]nisoxetine from human NET expressed in CHO cells after 120 mins by scintillation counting |
J Med Chem 55: 3488-501 (2012)
Article DOI: 10.1021/jm300138g
BindingDB Entry DOI: 10.7270/Q2KW5H2V |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
(Escherichia coli) | BDBM50481103

(CHEMBL583197)Show SMILES CN(Cc1sc2ccccc2c1C)C(=O)\C=C\c1cnc2NCCNCc2c1 Show InChI InChI=1S/C22H24N4OS/c1-15-18-5-3-4-6-19(18)28-20(15)14-26(2)21(27)8-7-16-11-17-13-23-9-10-24-22(17)25-12-16/h3-8,11-12,23H,9-10,13-14H2,1-2H3,(H,24,25)/b8-7+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Affinium Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Escherichia coli FabI |
Bioorg Med Chem Lett 19: 5359-62 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.094
BindingDB Entry DOI: 10.7270/Q2571FT3 |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
(Escherichia coli) | BDBM50481102
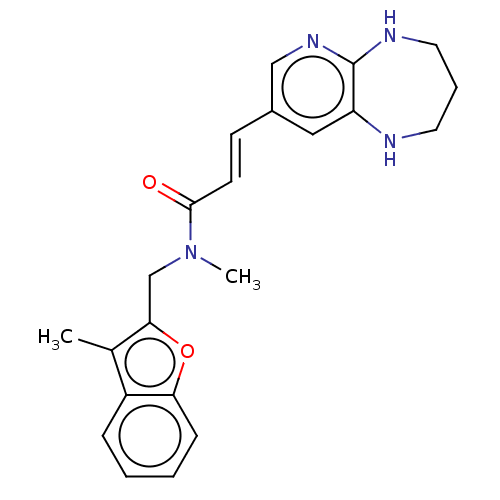
(CHEMBL567520)Show SMILES CN(Cc1oc2ccccc2c1C)C(=O)\C=C\c1cnc2NCCCNc2c1 Show InChI InChI=1S/C22H24N4O2/c1-15-17-6-3-4-7-19(17)28-20(15)14-26(2)21(27)9-8-16-12-18-22(25-13-16)24-11-5-10-23-18/h3-4,6-9,12-13,23H,5,10-11,14H2,1-2H3,(H,24,25)/b9-8+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Affinium Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Escherichia coli FabI |
Bioorg Med Chem Lett 19: 5359-62 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.094
BindingDB Entry DOI: 10.7270/Q2571FT3 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM111496
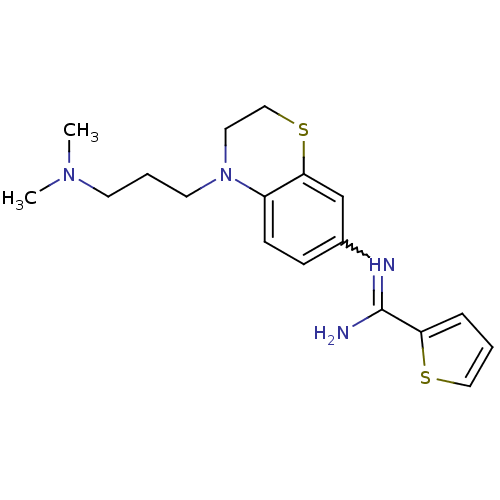
(US8618286, 22)Show SMILES CN(C)CCCN1CCSc2cc(ccc12)N=C(N)c1cccs1 |w:16.17| Show InChI InChI=1S/C18H24N4S2/c1-21(2)8-4-9-22-10-12-24-17-13-14(6-7-15(17)22)20-18(19)16-5-3-11-23-16/h3,5-7,11,13H,4,8-10,12H2,1-2H3,(H2,19,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
Primary stock solutions of test compounds at a concentration of 6 mM were prepared from the 2 to 5 mg powder. The primary stock solutions of each tes... |
US Patent US8618286 (2013)
BindingDB Entry DOI: 10.7270/Q2C53JH7 |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
(Escherichia coli) | BDBM50481105
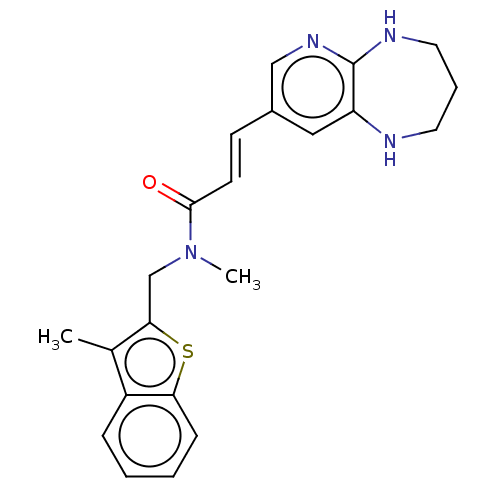
(CHEMBL567313)Show SMILES CN(Cc1sc2ccccc2c1C)C(=O)\C=C\c1cnc2NCCCNc2c1 Show InChI InChI=1S/C22H24N4OS/c1-15-17-6-3-4-7-19(17)28-20(15)14-26(2)21(27)9-8-16-12-18-22(25-13-16)24-11-5-10-23-18/h3-4,6-9,12-13,23H,5,10-11,14H2,1-2H3,(H,24,25)/b9-8+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Affinium Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Escherichia coli FabI |
Bioorg Med Chem Lett 19: 5359-62 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.094
BindingDB Entry DOI: 10.7270/Q2571FT3 |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
(Escherichia coli) | BDBM50481097

(CHEMBL565647)Show SMILES CN(Cc1oc2ccccc2c1C)C(=O)\C=C\c1cnc2NCCNCc2c1 Show InChI InChI=1S/C22H24N4O2/c1-15-18-5-3-4-6-19(18)28-20(15)14-26(2)21(27)8-7-16-11-17-13-23-9-10-24-22(17)25-12-16/h3-8,11-12,23H,9-10,13-14H2,1-2H3,(H,24,25)/b8-7+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Affinium Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Escherichia coli FabI |
Bioorg Med Chem Lett 19: 5359-62 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.094
BindingDB Entry DOI: 10.7270/Q2571FT3 |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
(Escherichia coli) | BDBM50481100

(CHEMBL576646)Show SMILES CN(Cc1oc2ccccc2c1C)C(=O)\C=C\c1cnc2NCCCNCc2c1 Show InChI InChI=1S/C23H26N4O2/c1-16-19-6-3-4-7-20(19)29-21(16)15-27(2)22(28)9-8-17-12-18-14-24-10-5-11-25-23(18)26-13-17/h3-4,6-9,12-13,24H,5,10-11,14-15H2,1-2H3,(H,25,26)/b9-8+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Affinium Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Escherichia coli FabI |
Bioorg Med Chem Lett 19: 5359-62 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.094
BindingDB Entry DOI: 10.7270/Q2571FT3 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50352593

(CHEMBL1825174)Show SMILES CN1CCC[C@H]1CCn1ccc2cc(ccc12)N=C(N)c1cccs1 |r,w:17.19| Show InChI InChI=1S/C20H24N4S/c1-23-10-2-4-17(23)9-12-24-11-8-15-14-16(6-7-18(15)24)22-20(21)19-5-3-13-25-19/h3,5-8,11,13-14,17H,2,4,9-10,12H2,1H3,(H2,21,22)/t17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in Sf9 cells assessed as conversion of [3H]-L-arginine to [3H]-L-citrulline by radiometric method |
Bioorg Med Chem Lett 21: 5301-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.022
BindingDB Entry DOI: 10.7270/Q2FN16KP |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
(Escherichia coli) | BDBM50481101
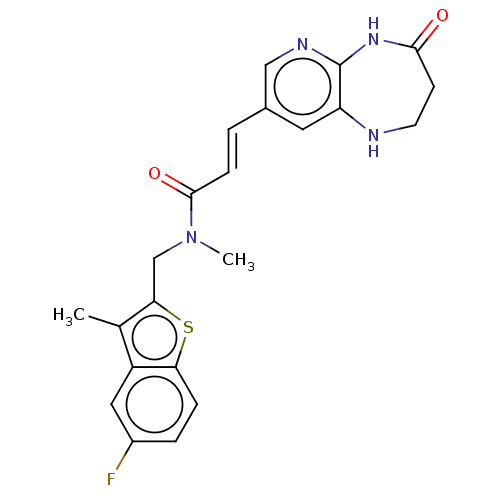
(CHEMBL567084)Show SMILES CN(Cc1sc2ccc(F)cc2c1C)C(=O)\C=C\c1cnc2NC(=O)CCNc2c1 Show InChI InChI=1S/C22H21FN4O2S/c1-13-16-10-15(23)4-5-18(16)30-19(13)12-27(2)21(29)6-3-14-9-17-22(25-11-14)26-20(28)7-8-24-17/h3-6,9-11,24H,7-8,12H2,1-2H3,(H,25,26,28)/b6-3+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Affinium Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Escherichia coli FabI |
Bioorg Med Chem Lett 19: 5359-62 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.094
BindingDB Entry DOI: 10.7270/Q2571FT3 |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
(Escherichia coli) | BDBM50481094
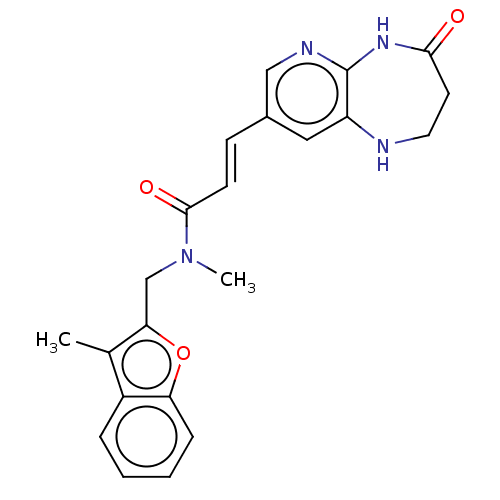
(CHEMBL567931)Show SMILES CN(Cc1oc2ccccc2c1C)C(=O)\C=C\c1cnc2NC(=O)CCNc2c1 Show InChI InChI=1S/C22H22N4O3/c1-14-16-5-3-4-6-18(16)29-19(14)13-26(2)21(28)8-7-15-11-17-22(24-12-15)25-20(27)9-10-23-17/h3-8,11-12,23H,9-10,13H2,1-2H3,(H,24,25,27)/b8-7+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Affinium Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Escherichia coli FabI |
Bioorg Med Chem Lett 19: 5359-62 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.094
BindingDB Entry DOI: 10.7270/Q2571FT3 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM111497
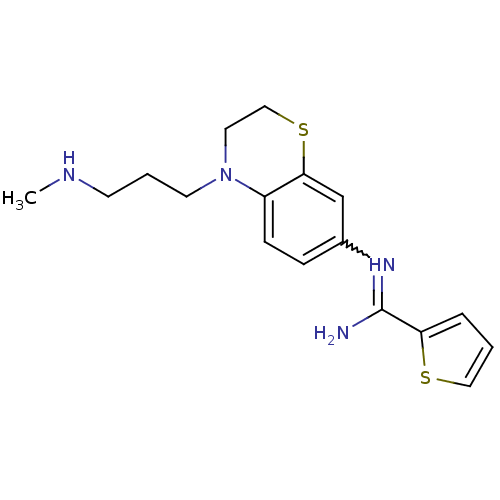
(US8618286, 23)Show SMILES CNCCCN1CCSc2cc(ccc12)N=C(N)c1cccs1 |w:15.16| Show InChI InChI=1S/C17H22N4S2/c1-19-7-3-8-21-9-11-23-16-12-13(5-6-14(16)21)20-17(18)15-4-2-10-22-15/h2,4-6,10,12,19H,3,7-9,11H2,1H3,(H2,18,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
Primary stock solutions of test compounds at a concentration of 6 mM were prepared from the 2 to 5 mg powder. The primary stock solutions of each tes... |
US Patent US8618286 (2013)
BindingDB Entry DOI: 10.7270/Q2C53JH7 |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
(Escherichia coli) | BDBM50481096

(CHEMBL567710)Show SMILES CN(Cc1oc2ccccc2c1C)C(=O)\C=C\c1cnc2NC(=O)CNCc2c1 Show InChI InChI=1S/C22H22N4O3/c1-14-17-5-3-4-6-18(17)29-19(14)13-26(2)21(28)8-7-15-9-16-11-23-12-20(27)25-22(16)24-10-15/h3-10,23H,11-13H2,1-2H3,(H,24,25,27)/b8-7+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
Affinium Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Escherichia coli FabI |
Bioorg Med Chem Lett 19: 5359-62 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.094
BindingDB Entry DOI: 10.7270/Q2571FT3 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50206074

((+/-)-N-{2-[2-(1-methyl-pyrrolidin-2-yl)-ethylamin...)Show SMILES CN1CCCC1CCNc1nc2cc(ccc2s1)N=C(N)c1cccs1 |w:18.20| Show InChI InChI=1S/C19H23N5S2/c1-24-10-2-4-14(24)8-9-21-19-23-15-12-13(6-7-16(15)26-19)22-18(20)17-5-3-11-25-17/h3,5-7,11-12,14H,2,4,8-10H2,1H3,(H2,20,22)(H,21,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human neuronal NOS activity |
Bioorg Med Chem Lett 17: 2540-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.02.011
BindingDB Entry DOI: 10.7270/Q2WM1F6B |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
(Escherichia coli) | BDBM50481098

(CHEMBL567311)Show SMILES CN(Cc1oc2ccccc2c1C)C(=O)\C=C\c1cnc2NCCC(=O)NCc2c1 Show InChI InChI=1S/C23H24N4O3/c1-15-18-5-3-4-6-19(18)30-20(15)14-27(2)22(29)8-7-16-11-17-13-25-21(28)9-10-24-23(17)26-12-16/h3-8,11-12H,9-10,13-14H2,1-2H3,(H,24,26)(H,25,28)/b8-7+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Affinium Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Escherichia coli FabI |
Bioorg Med Chem Lett 19: 5359-62 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.094
BindingDB Entry DOI: 10.7270/Q2571FT3 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM111490
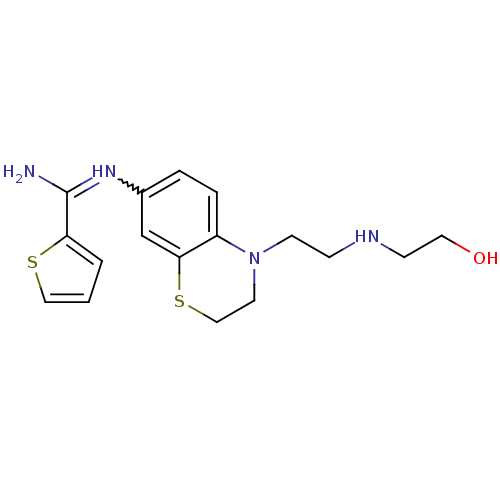
(US8618286, 16)Show SMILES NC(=Nc1ccc2N(CCNCCO)CCSc2c1)c1cccs1 |w:2.2| Show InChI InChI=1S/C17H22N4OS2/c18-17(15-2-1-10-23-15)20-13-3-4-14-16(12-13)24-11-8-21(14)7-5-19-6-9-22/h1-4,10,12,19,22H,5-9,11H2,(H2,18,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 34 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
Primary stock solutions of test compounds at a concentration of 6 mM were prepared from the 2 to 5 mg powder. The primary stock solutions of each tes... |
US Patent US8618286 (2013)
BindingDB Entry DOI: 10.7270/Q2C53JH7 |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
(Escherichia coli) | BDBM50481099
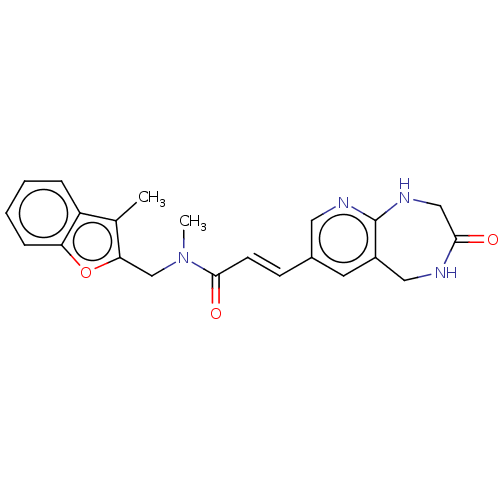
(CHEMBL570866)Show SMILES CN(Cc1oc2ccccc2c1C)C(=O)\C=C\c1cnc2NCC(=O)NCc2c1 Show InChI InChI=1S/C22H22N4O3/c1-14-17-5-3-4-6-18(17)29-19(14)13-26(2)21(28)8-7-15-9-16-11-23-20(27)12-25-22(16)24-10-15/h3-10H,11-13H2,1-2H3,(H,23,27)(H,24,25)/b8-7+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
Affinium Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Escherichia coli FabI |
Bioorg Med Chem Lett 19: 5359-62 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.094
BindingDB Entry DOI: 10.7270/Q2571FT3 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50401280
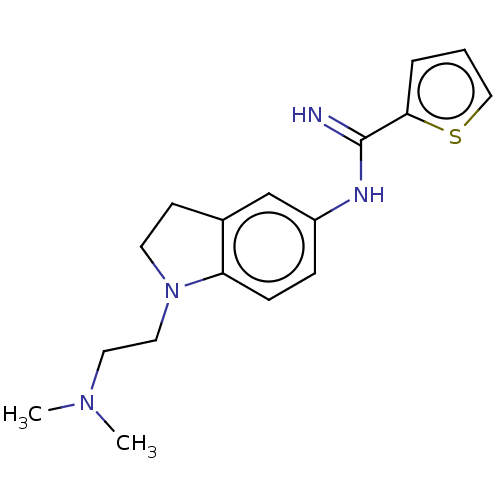
(CHEMBL3216124)Show InChI InChI=1S/C17H22N4S/c1-20(2)9-10-21-8-7-13-12-14(5-6-15(13)21)19-17(18)16-4-3-11-22-16/h3-6,11-12H,7-10H2,1-2H3,(H2,18,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human nNOS expressed in baculovirus-infected Sf9 cells assessed as conversion of [3H]-larginine to [3H]-L-citrulline preinc... |
Eur J Med Chem 55: 94-107 (2012)
Article DOI: 10.1016/j.ejmech.2012.07.006
BindingDB Entry DOI: 10.7270/Q2GQ6ZWQ |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine 1D receptor
(Bos taurus (Bovine)) | BDBM106707
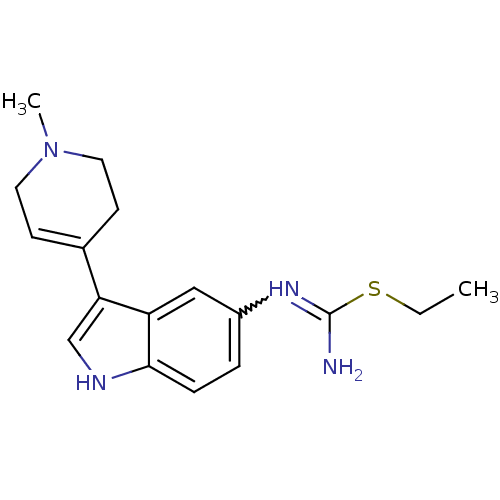
(CHEMBL1957358 | US8586620, 67)Show SMILES CCSC(N)=Nc1ccc2[nH]cc(C3=CCN(C)CC3)c2c1 |w:5.5,t:13| Show InChI InChI=1S/C17H22N4S/c1-3-22-17(18)20-13-4-5-16-14(10-13)15(11-19-16)12-6-8-21(2)9-7-12/h4-6,10-11,19H,3,7-9H2,1-2H3,(H2,18,20) | PDB
Reactome pathway
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 48 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
5-HT1D binding assays (agonist radioligand)were performed using bovine caudate membranes according to the methods of Heuring and Peroutka (J. Neurosc... |
US Patent US8586620 (2013)
BindingDB Entry DOI: 10.7270/Q2348J0K |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Rattus norvegicus (rat)) | BDBM50206073

(CHEMBL233652 | N-[2-(2-pyridin-2-yl-ethylamino)-be...)Show SMILES NC(=Nc1ccc2nc(NCCc3ccccn3)sc2c1)c1cccs1 |w:2.2| Show InChI InChI=1S/C19H17N5S2/c20-18(16-5-3-11-25-16)23-14-6-7-15-17(12-14)26-19(24-15)22-10-8-13-4-1-2-9-21-13/h1-7,9,11-12H,8,10H2,(H2,20,23)(H,22,24) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of rat neuronal NOS activity |
Bioorg Med Chem Lett 17: 2540-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.02.011
BindingDB Entry DOI: 10.7270/Q2WM1F6B |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine 1D receptor
(Bos taurus (Bovine)) | BDBM106699
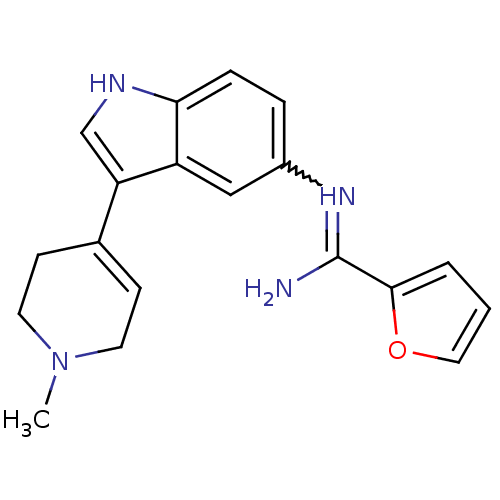
(CHEMBL1957350 | US8586620, 46)Show SMILES CN1CCC(=CC1)c1c[nH]c2ccc(cc12)N=C(N)c1ccco1 |w:16.18,c:4| Show InChI InChI=1S/C19H20N4O/c1-23-8-6-13(7-9-23)16-12-21-17-5-4-14(11-15(16)17)22-19(20)18-3-2-10-24-18/h2-6,10-12,21H,7-9H2,1H3,(H2,20,22) | PDB
Reactome pathway
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
5-HT1D binding assays (agonist radioligand)were performed using bovine caudate membranes according to the methods of Heuring and Peroutka (J. Neurosc... |
US Patent US8586620 (2013)
BindingDB Entry DOI: 10.7270/Q2348J0K |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM111498
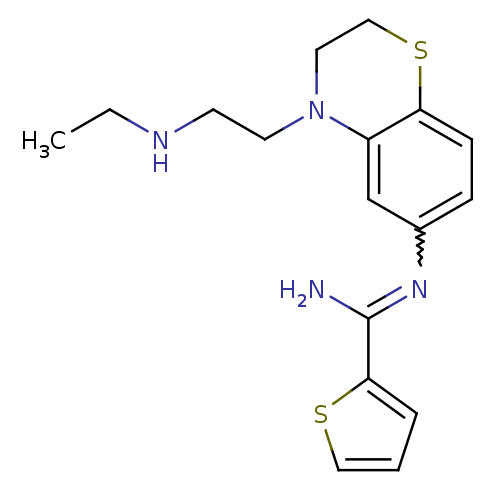
(US8618286, 24)Show SMILES CCNCCN1CCSc2ccc(cc12)N=C(N)c1cccs1 |w:15.16| Show InChI InChI=1S/C17H22N4S2/c1-2-19-7-8-21-9-11-23-15-6-5-13(12-14(15)21)20-17(18)16-4-3-10-22-16/h3-6,10,12,19H,2,7-9,11H2,1H3,(H2,18,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 50 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
Primary stock solutions of test compounds at a concentration of 6 mM were prepared from the 2 to 5 mg powder. The primary stock solutions of each tes... |
US Patent US8618286 (2013)
BindingDB Entry DOI: 10.7270/Q2C53JH7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine 1D receptor
(Bos taurus (Bovine)) | BDBM106698
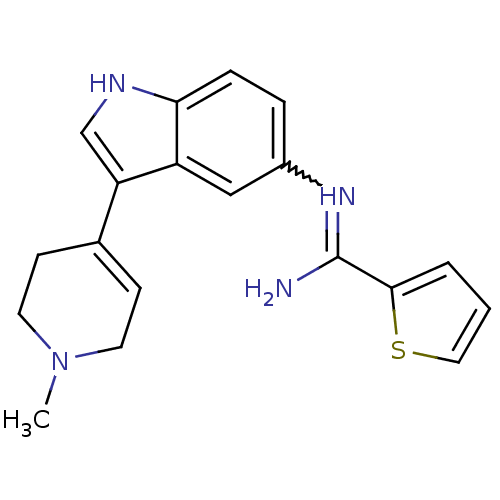
(CHEMBL1957349 | US8586620, 42)Show SMILES CN1CCC(=CC1)c1c[nH]c2ccc(cc12)N=C(N)c1cccs1 |w:16.18,c:4| Show InChI InChI=1S/C19H20N4S/c1-23-8-6-13(7-9-23)16-12-21-17-5-4-14(11-15(16)17)22-19(20)18-3-2-10-24-18/h2-6,10-12,21H,7-9H2,1H3,(H2,20,22) | PDB
Reactome pathway
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 51 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
5-HT1D binding assays (agonist radioligand)were performed using bovine caudate membranes according to the methods of Heuring and Peroutka (J. Neurosc... |
US Patent US8586620 (2013)
BindingDB Entry DOI: 10.7270/Q2348J0K |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine 1D receptor
(Bos taurus (Bovine)) | BDBM106706

(CHEMBL1957356 | US8586620, 64)Show SMILES CN1CCC(=CC1)c1c[nH]c2ccc(NC(=S)NC(=O)c3ccccc3)cc12 |c:4| Show InChI InChI=1S/C22H22N4OS/c1-26-11-9-15(10-12-26)19-14-23-20-8-7-17(13-18(19)20)24-22(28)25-21(27)16-5-3-2-4-6-16/h2-9,13-14,23H,10-12H2,1H3,(H2,24,25,27,28) | PDB
Reactome pathway
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| US Patent
| n/a | n/a | 56 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
5-HT1D binding assays (agonist radioligand)were performed using bovine caudate membranes according to the methods of Heuring and Peroutka (J. Neurosc... |
US Patent US8586620 (2013)
BindingDB Entry DOI: 10.7270/Q2348J0K |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine 1D receptor
(Bos taurus (Bovine)) | BDBM50005835
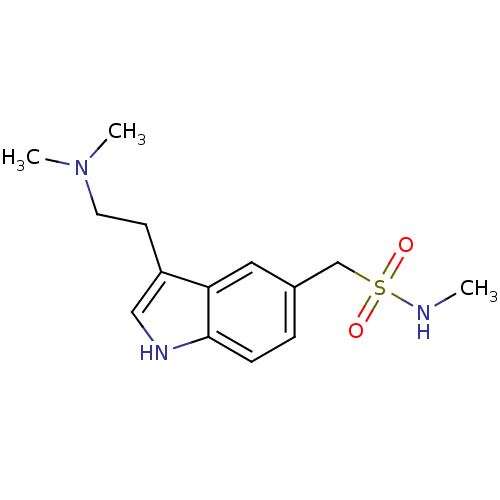
((3-[2-(dimethylamino)ethyl]-1H-indol-5-yl)-N-methy...)Show InChI InChI=1S/C14H21N3O2S/c1-15-20(18,19)10-11-4-5-14-13(8-11)12(9-16-14)6-7-17(2)3/h4-5,8-9,15-16H,6-7,10H2,1-3H3 | PDB
Reactome pathway
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
| US Patent
| n/a | n/a | 59 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
5-HT1D binding assays (agonist radioligand)were performed using bovine caudate membranes according to the methods of Heuring and Peroutka (J. Neurosc... |
US Patent US8586620 (2013)
BindingDB Entry DOI: 10.7270/Q2348J0K |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM111482

(US8618286, 8)Show SMILES CNCCN1CCSc2cc(ccc12)N=C(N)c1cccs1 |w:14.15| Show InChI InChI=1S/C16H20N4S2/c1-18-6-7-20-8-10-22-15-11-12(4-5-13(15)20)19-16(17)14-3-2-9-21-14/h2-5,9,11,18H,6-8,10H2,1H3,(H2,17,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
Primary stock solutions of test compounds at a concentration of 6 mM were prepared from the 2 to 5 mg powder. The primary stock solutions of each tes... |
US Patent US8618286 (2013)
BindingDB Entry DOI: 10.7270/Q2C53JH7 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50352592
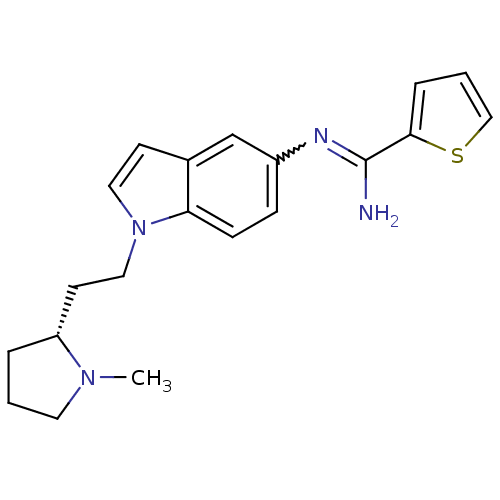
(CHEMBL1825173)Show SMILES CN1CCC[C@@H]1CCn1ccc2cc(ccc12)N=C(N)c1cccs1 |r,w:17.19| Show InChI InChI=1S/C20H24N4S/c1-23-10-2-4-17(23)9-12-24-11-8-15-14-16(6-7-18(15)24)22-20(21)19-5-3-13-25-19/h3,5-8,11,13-14,17H,2,4,9-10,12H2,1H3,(H2,21,22)/t17-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 60 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in Sf9 cells assessed as conversion of [3H]-L-arginine to [3H]-L-citrulline by radiometric method |
Bioorg Med Chem Lett 21: 5301-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.022
BindingDB Entry DOI: 10.7270/Q2FN16KP |
More data for this
Ligand-Target Pair | |
Enoyl-[acyl-carrier-protein] reductase [NADH] FabI
(Escherichia coli) | BDBM50481092
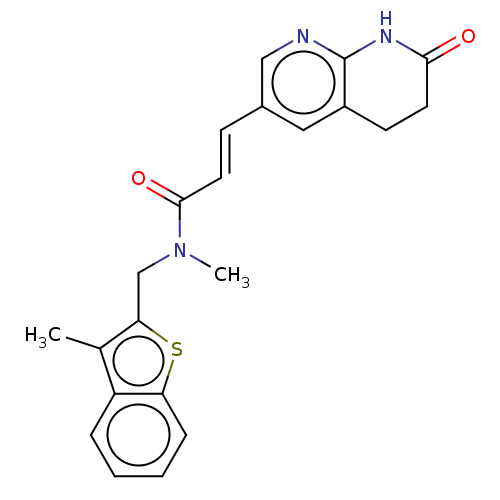
(CHEMBL566655)Show SMILES CN(Cc1sc2ccccc2c1C)C(=O)\C=C\c1cnc2NC(=O)CCc2c1 Show InChI InChI=1S/C22H21N3O2S/c1-14-17-5-3-4-6-18(17)28-19(14)13-25(2)21(27)10-7-15-11-16-8-9-20(26)24-22(16)23-12-15/h3-7,10-12H,8-9,13H2,1-2H3,(H,23,24,26)/b10-7+ | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 64 | n/a | n/a | n/a | n/a | n/a | n/a |
Affinium Pharmaceuticals Inc.
Curated by ChEMBL
| Assay Description
Inhibition of Escherichia coli FabI |
Bioorg Med Chem Lett 19: 5359-62 (2009)
Article DOI: 10.1016/j.bmcl.2009.07.094
BindingDB Entry DOI: 10.7270/Q2571FT3 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50352591

(CHEMBL1825172)Show SMILES CN1CCCC1CCn1ccc2cc(ccc12)N=C(N)c1cccs1 |w:17.19| Show InChI InChI=1S/C20H24N4S/c1-23-10-2-4-17(23)9-12-24-11-8-15-14-16(6-7-18(15)24)22-20(21)19-5-3-13-25-19/h3,5-8,11,13-14,17H,2,4,9-10,12H2,1H3,(H2,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in Sf9 cells assessed as conversion of [3H]-L-arginine to [3H]-L-citrulline by radiometric method |
Bioorg Med Chem Lett 21: 5301-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.022
BindingDB Entry DOI: 10.7270/Q2FN16KP |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50365335
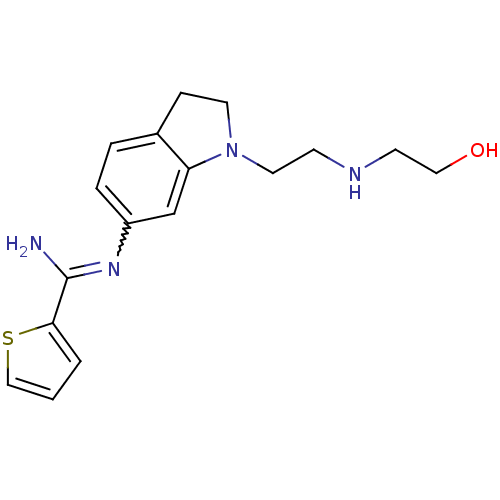
(CHEMBL1955937)Show InChI InChI=1S/C17H22N4OS/c18-17(16-2-1-11-23-16)20-14-4-3-13-5-8-21(15(13)12-14)9-6-19-7-10-22/h1-4,11-12,19,22H,5-10H2,(H2,18,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in baculovirus infected insect Sf9 cells assessed as conversion of [3H]L-arginine to [3H]L-citrulline ... |
J Med Chem 55: 943-55 (2012)
Article DOI: 10.1021/jm201564u
BindingDB Entry DOI: 10.7270/Q20P10HM |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM111484
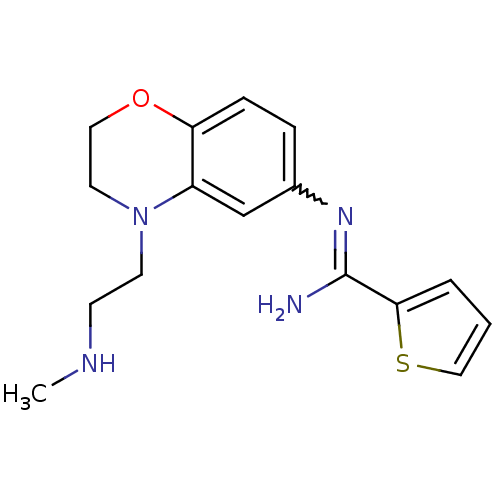
(US8618286, 10)Show SMILES CNCCN1CCOc2ccc(cc12)N=C(N)c1cccs1 |w:14.15| Show InChI InChI=1S/C16H20N4OS/c1-18-6-7-20-8-9-21-14-5-4-12(11-13(14)20)19-16(17)15-3-2-10-22-15/h2-5,10-11,18H,6-9H2,1H3,(H2,17,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
Primary stock solutions of test compounds at a concentration of 6 mM were prepared from the 2 to 5 mg powder. The primary stock solutions of each tes... |
US Patent US8618286 (2013)
BindingDB Entry DOI: 10.7270/Q2C53JH7 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50401282
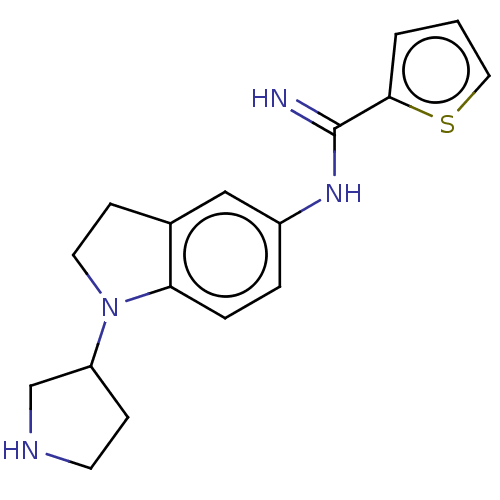
(CHEMBL3216566)Show InChI InChI=1S/C17H20N4S/c18-17(16-2-1-9-22-16)20-13-3-4-15-12(10-13)6-8-21(15)14-5-7-19-11-14/h1-4,9-10,14,19H,5-8,11H2,(H2,18,20) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human nNOS expressed in baculovirus-infected Sf9 cells assessed as conversion of [3H]-larginine to [3H]-L-citrulline preinc... |
Eur J Med Chem 55: 94-107 (2012)
Article DOI: 10.1016/j.ejmech.2012.07.006
BindingDB Entry DOI: 10.7270/Q2GQ6ZWQ |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM111502

(US8618286, 28)Show SMILES CNCCN1CCSc2ccc(cc12)N=C(N)c1cccs1 |w:14.15| Show InChI InChI=1S/C16H20N4S2/c1-18-6-7-20-8-10-22-14-5-4-12(11-13(14)20)19-16(17)15-3-2-9-21-15/h2-5,9,11,18H,6-8,10H2,1H3,(H2,17,19) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
Primary stock solutions of test compounds at a concentration of 6 mM were prepared from the 2 to 5 mg powder. The primary stock solutions of each tes... |
US Patent US8618286 (2013)
BindingDB Entry DOI: 10.7270/Q2C53JH7 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50379744
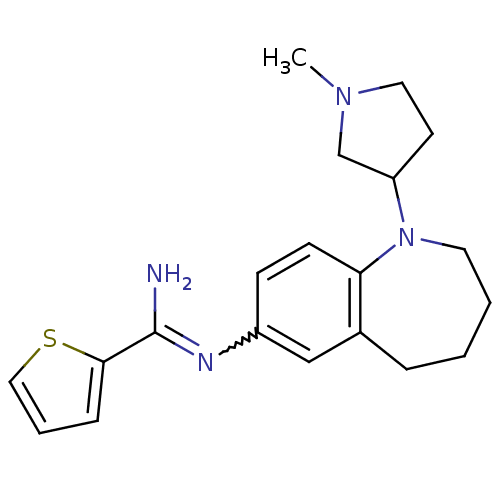
(CHEMBL2011136)Show SMILES CN1CCC(C1)N1CCCCc2cc(ccc12)N=C(N)c1cccs1 |w:17.19| Show InChI InChI=1S/C20H26N4S/c1-23-11-9-17(14-23)24-10-3-2-5-15-13-16(7-8-18(15)24)22-20(21)19-6-4-12-25-19/h4,6-8,12-13,17H,2-3,5,9-11,14H2,1H3,(H2,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 80 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in baculovirus infected insect sf9 cells assessed as conversion of [3H]-L-arginine into [3H]-L-citrull... |
Bioorg Med Chem Lett 22: 2510-3 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.004
BindingDB Entry DOI: 10.7270/Q2639QRG |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50357692

(CHEMBL1915288)Show SMILES NC(=Nc1ccc2N(CCCN3CCCC3)CCCc2c1)c1cccs1 |w:2.2| Show InChI InChI=1S/C21H28N4S/c22-21(20-7-4-15-26-20)23-18-8-9-19-17(16-18)6-3-13-25(19)14-5-12-24-10-1-2-11-24/h4,7-9,15-16H,1-3,5-6,10-14H2,(H2,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 86 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in baculovirus infected Sf9 cells assessed as conversion of [3H]L-arginine to [3H]L-citrulline preincu... |
J Med Chem 54: 5562-75 (2011)
Article DOI: 10.1021/jm200648s
BindingDB Entry DOI: 10.7270/Q2222V58 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Rattus norvegicus (rat)) | BDBM50206064

(CHEMBL233857 | N-[2-(1-benzyl-piperidin-4-ylamino)...)Show SMILES NC(=Nc1ccc2nc(NC3CCN(Cc4ccccc4)CC3)sc2c1)c1cccs1 |w:2.2| Show InChI InChI=1S/C24H25N5S2/c25-23(21-7-4-14-30-21)26-19-8-9-20-22(15-19)31-24(28-20)27-18-10-12-29(13-11-18)16-17-5-2-1-3-6-17/h1-9,14-15,18H,10-13,16H2,(H2,25,26)(H,27,28) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of rat neuronal NOS activity |
Bioorg Med Chem Lett 17: 2540-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.02.011
BindingDB Entry DOI: 10.7270/Q2WM1F6B |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50352590
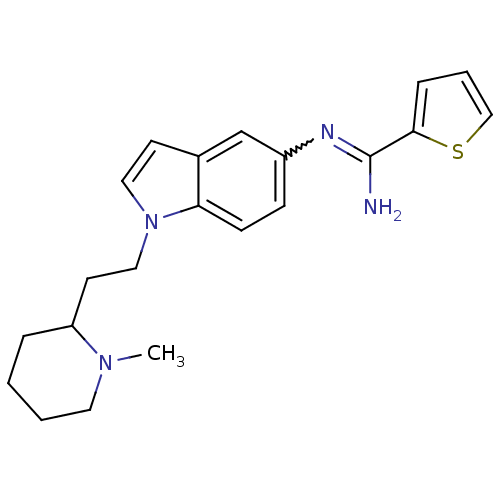
(CHEMBL1825171)Show SMILES CN1CCCCC1CCn1ccc2cc(ccc12)N=C(N)c1cccs1 |w:18.20| Show InChI InChI=1S/C21H26N4S/c1-24-11-3-2-5-18(24)10-13-25-12-9-16-15-17(7-8-19(16)25)23-21(22)20-6-4-14-26-20/h4,6-9,12,14-15,18H,2-3,5,10-11,13H2,1H3,(H2,22,23) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in Sf9 cells assessed as conversion of [3H]-L-arginine to [3H]-L-citrulline by radiometric method |
Bioorg Med Chem Lett 21: 5301-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.022
BindingDB Entry DOI: 10.7270/Q2FN16KP |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50357690
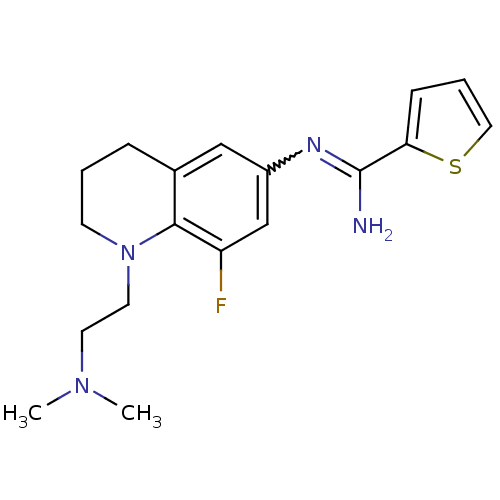
(CHEMBL1915286)Show SMILES CN(C)CCN1CCCc2cc(cc(F)c12)N=C(N)c1cccs1 |w:16.17| Show InChI InChI=1S/C18H23FN4S/c1-22(2)8-9-23-7-3-5-13-11-14(12-15(19)17(13)23)21-18(20)16-6-4-10-24-16/h4,6,10-12H,3,5,7-9H2,1-2H3,(H2,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 93 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in baculovirus infected Sf9 cells assessed as conversion of [3H]L-arginine to [3H]L-citrulline preincu... |
J Med Chem 54: 5562-75 (2011)
Article DOI: 10.1021/jm200648s
BindingDB Entry DOI: 10.7270/Q2222V58 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50394745

(CHEMBL2165821)Show InChI InChI=1S/C19H26N4S/c1-3-22(2)11-12-23-10-4-6-15-14-16(8-9-17(15)23)21-19(20)18-7-5-13-24-18/h5,7-9,13-14H,3-4,6,10-12H2,1-2H3,(H2,20,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 97 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human nNOS expressed in Sf9 cells assessed as reduction in conversion of [3H]L-arginine to [3H]L-citrulline preincubated for 15 mins by... |
J Med Chem 55: 2882-93 (2012)
Article DOI: 10.1021/jm3000449
BindingDB Entry DOI: 10.7270/Q24F1RV1 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50357687
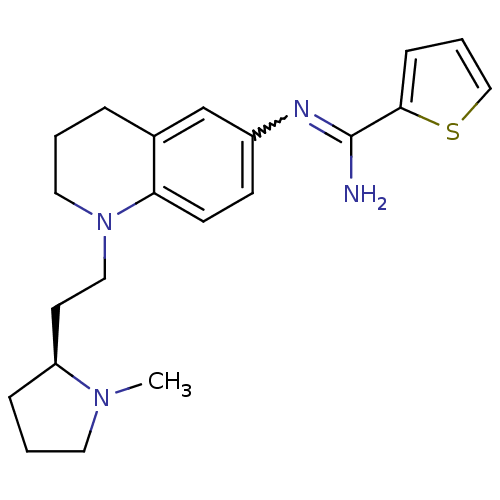
(CHEMBL1956112)Show SMILES CN1CCC[C@H]1CCN1CCCc2cc(ccc12)N=C(N)c1cccs1 |r,w:18.20| Show InChI InChI=1S/C21H28N4S/c1-24-11-3-6-18(24)10-13-25-12-2-5-16-15-17(8-9-19(16)25)23-21(22)20-7-4-14-26-20/h4,7-9,14-15,18H,2-3,5-6,10-13H2,1H3,(H2,22,23)/t18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 98 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in baculovirus infected Sf9 cells assessed as conversion of [3H]L-arginine to [3H]L-citrulline preincu... |
J Med Chem 54: 5562-75 (2011)
Article DOI: 10.1021/jm200648s
BindingDB Entry DOI: 10.7270/Q2222V58 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50206065

(CHEMBL233655 | N-{2-[1-(3-methoxy-benzyl)-piperidi...)Show SMILES COc1cccc(CN2CCC(CC2)Nc2nc3ccc(cc3s2)N=C(N)c2cccs2)c1 |w:24.26| Show InChI InChI=1S/C25H27N5OS2/c1-31-20-5-2-4-17(14-20)16-30-11-9-18(10-12-30)28-25-29-21-8-7-19(15-23(21)33-25)27-24(26)22-6-3-13-32-22/h2-8,13-15,18H,9-12,16H2,1H3,(H2,26,27)(H,28,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human neuronal NOS activity |
Bioorg Med Chem Lett 17: 2540-4 (2007)
Article DOI: 10.1016/j.bmcl.2007.02.011
BindingDB Entry DOI: 10.7270/Q2WM1F6B |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50379740

(CHEMBL2011132)Show SMILES CNCCN1CCCCc2cc(ccc12)N=C(N)c1cccs1 |w:15.16| Show InChI InChI=1S/C18H24N4S/c1-20-9-11-22-10-3-2-5-14-13-15(7-8-16(14)22)21-18(19)17-6-4-12-23-17/h4,6-8,12-13,20H,2-3,5,9-11H2,1H3,(H2,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in baculovirus infected insect sf9 cells assessed as conversion of [3H]-L-arginine into [3H]-L-citrull... |
Bioorg Med Chem Lett 22: 2510-3 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.004
BindingDB Entry DOI: 10.7270/Q2639QRG |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50365334
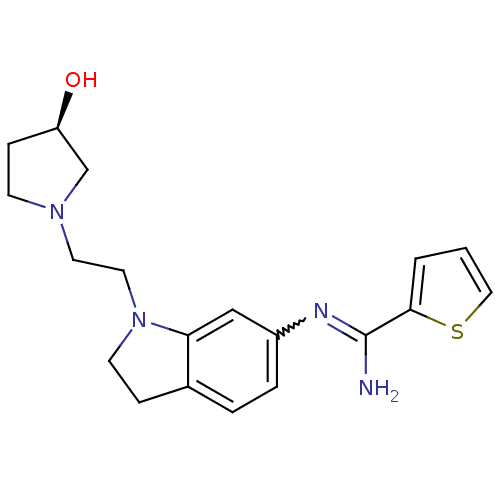
(CHEMBL1955936)Show SMILES NC(=Nc1ccc2CCN(CCN3CC[C@@H](O)C3)c2c1)c1cccs1 |r,w:2.2| Show InChI InChI=1S/C19H24N4OS/c20-19(18-2-1-11-25-18)21-15-4-3-14-5-8-23(17(14)12-15)10-9-22-7-6-16(24)13-22/h1-4,11-12,16,24H,5-10,13H2,(H2,20,21)/t16-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in baculovirus infected insect Sf9 cells assessed as conversion of [3H]L-arginine to [3H]L-citrulline ... |
J Med Chem 55: 943-55 (2012)
Article DOI: 10.1021/jm201564u
BindingDB Entry DOI: 10.7270/Q20P10HM |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM111503

(US8618286, 29)Show SMILES NC(=Nc1ccc2SCCN(C3CCNCC3)c2c1)c1cccs1 |w:2.2| Show InChI InChI=1S/C18H22N4S2/c19-18(17-2-1-10-23-17)21-13-3-4-16-15(12-13)22(9-11-24-16)14-5-7-20-8-6-14/h1-4,10,12,14,20H,5-9,11H2,(H2,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| US Patent
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
Primary stock solutions of test compounds at a concentration of 6 mM were prepared from the 2 to 5 mg powder. The primary stock solutions of each tes... |
US Patent US8618286 (2013)
BindingDB Entry DOI: 10.7270/Q2C53JH7 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1B
(Rattus norvegicus (Rat)) | BDBM50005835

((3-[2-(dimethylamino)ethyl]-1H-indol-5-yl)-N-methy...)Show InChI InChI=1S/C14H21N3O2S/c1-15-20(18,19)10-11-4-5-14-13(8-11)12(9-16-14)6-7-17(2)3/h4-5,8-9,15-16H,6-7,10H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
| US Patent
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon, Inc.
US Patent
| Assay Description
5-HT1D binding assays (agonist radioligand)were performed using bovine caudate membranes according to the methods of Heuring and Peroutka (J. Neurosc... |
US Patent US8586620 (2013)
BindingDB Entry DOI: 10.7270/Q2348J0K |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50401271

(CHEMBL2203713 | CHEMBL3216328)Show InChI InChI=1S/C18H22N4S/c19-18(17-2-1-11-23-17)21-14-3-4-16-13(12-14)7-10-22(16)15-5-8-20-9-6-15/h1-4,11-12,15,20H,5-10H2,(H2,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human nNOS expressed in baculovirus-infected Sf9 cells assessed as conversion of [3H]-larginine to [3H]-L-citrulline preinc... |
Eur J Med Chem 55: 94-107 (2012)
Article DOI: 10.1016/j.ejmech.2012.07.006
BindingDB Entry DOI: 10.7270/Q2GQ6ZWQ |
More data for this
Ligand-Target Pair | |
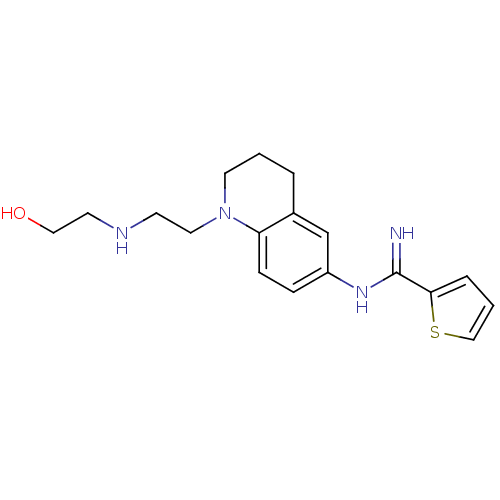
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50394739
(CHEMBL2165827)Show InChI InChI=1S/C18H24N4OS/c19-18(17-4-2-12-24-17)21-15-5-6-16-14(13-15)3-1-9-22(16)10-7-20-8-11-23/h2,4-6,12-13,20,23H,1,3,7-11H2,(H2,19,21) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 115 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human nNOS expressed in Sf9 cells assessed as reduction in conversion of [3H]L-arginine to [3H]L-citrulline preincubated for 15 mins by... |
J Med Chem 55: 2882-93 (2012)
Article DOI: 10.1021/jm3000449
BindingDB Entry DOI: 10.7270/Q24F1RV1 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50352595

(CHEMBL1825176)Show SMILES CN1CCCC(CC1)n1ccc2cc(ccc12)N=C(N)c1cccs1 |w:17.19| Show InChI InChI=1S/C20H24N4S/c1-23-10-2-4-17(9-11-23)24-12-8-15-14-16(6-7-18(15)24)22-20(21)19-5-3-13-25-19/h3,5-8,12-14,17H,2,4,9-11H2,1H3,(H2,21,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in Sf9 cells assessed as conversion of [3H]-L-arginine to [3H]-L-citrulline by radiometric method |
Bioorg Med Chem Lett 21: 5301-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.07.022
BindingDB Entry DOI: 10.7270/Q2FN16KP |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Homo sapiens (Human)) | BDBM50355311
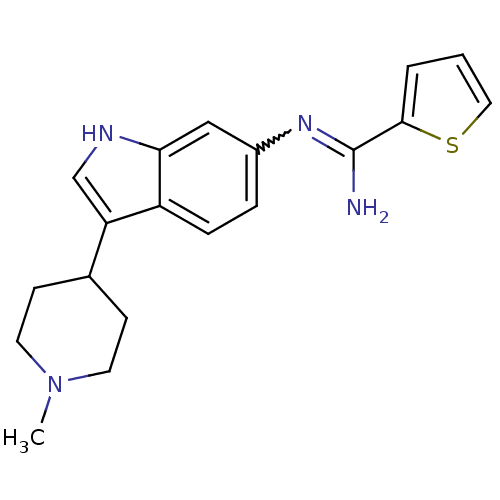
(CHEMBL1835114)Show SMILES CN1CCC(CC1)c1c[nH]c2cc(ccc12)N=C(N)c1cccs1 |w:16.18| Show InChI InChI=1S/C19H22N4S/c1-23-8-6-13(7-9-23)16-12-21-17-11-14(4-5-15(16)17)22-19(20)18-3-2-10-24-18/h2-5,10-13,21H,6-9H2,1H3,(H2,20,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant nNOS expressed in Sf9 cells assessed as inhibition of conversion of [3H]-L-arginine to [3H]-L-citrulline after 45 min... |
J Med Chem 54: 7408-16 (2011)
Article DOI: 10.1021/jm201063u
BindingDB Entry DOI: 10.7270/Q2JQ1201 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, brain
(Rattus norvegicus (rat)) | BDBM50379737
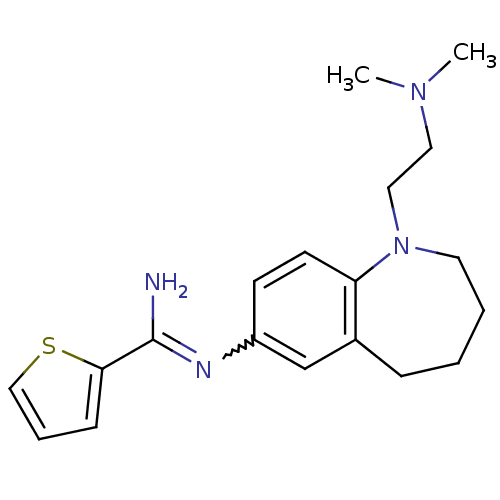
(CHEMBL2011128)Show SMILES CN(C)CCN1CCCCc2cc(ccc12)N=C(N)c1cccs1 |w:16.17| Show InChI InChI=1S/C19H26N4S/c1-22(2)11-12-23-10-4-3-6-15-14-16(8-9-17(15)23)21-19(20)18-7-5-13-24-18/h5,7-9,13-14H,3-4,6,10-12H2,1-2H3,(H2,20,21) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
NeurAxon Inc.
Curated by ChEMBL
| Assay Description
Inhibition of rat nNOS |
Bioorg Med Chem Lett 22: 2510-3 (2012)
Article DOI: 10.1016/j.bmcl.2012.02.004
BindingDB Entry DOI: 10.7270/Q2639QRG |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data