 Found 256 hits with Last Name = 'chapman' and Initial = 's'
Found 256 hits with Last Name = 'chapman' and Initial = 's' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM31883

(9-cis-retinoic acid (9cRA) | ALL-TRANS-RETINOIC AC...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(O)=O |c:4| Show InChI InChI=1S/C20H28O2/c1-15(8-6-9-16(2)14-19(21)22)11-12-18-17(3)10-7-13-20(18,4)5/h6,8-9,11-12,14H,7,10,13H2,1-5H3,(H,21,22)/b9-6+,12-11+,15-8+,16-14+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| MMDB
PubMed
| 0.700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Ohio State University
Curated by ChEMBL
| Assay Description
Binding affinity against Retinoic acid receptor gamma |
Bioorg Med Chem Lett 11: 1583-6 (2001)
BindingDB Entry DOI: 10.7270/Q20R9NQW |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50092055
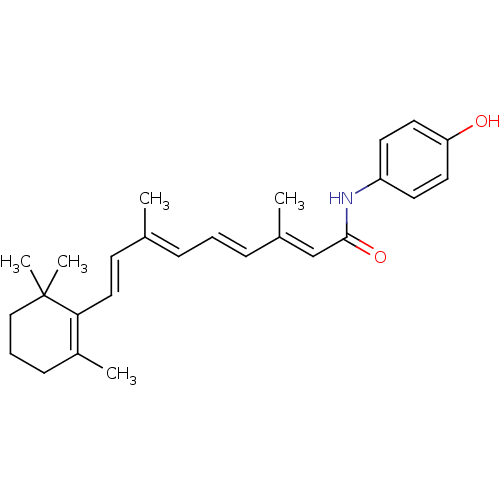
((2E,4E,6E,8E)-3,7-Dimethyl-9-(2,6,6-trimethyl-cycl...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=C/C(/C)=C/C(=O)Nc1ccc(O)cc1 |c:4| Show InChI InChI=1S/C26H33NO2/c1-19(11-16-24-21(3)10-7-17-26(24,4)5)8-6-9-20(2)18-25(29)27-22-12-14-23(28)15-13-22/h6,8-9,11-16,18,28H,7,10,17H2,1-5H3,(H,27,29)/b9-6+,16-11+,19-8+,20-18+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| >400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Ohio State University
Curated by ChEMBL
| Assay Description
Binding affinity against Retinoic acid receptor gamma |
Bioorg Med Chem Lett 11: 1583-6 (2001)
BindingDB Entry DOI: 10.7270/Q20R9NQW |
More data for this
Ligand-Target Pair | |
Retinoic acid receptor gamma
(Homo sapiens (Human)) | BDBM50100957
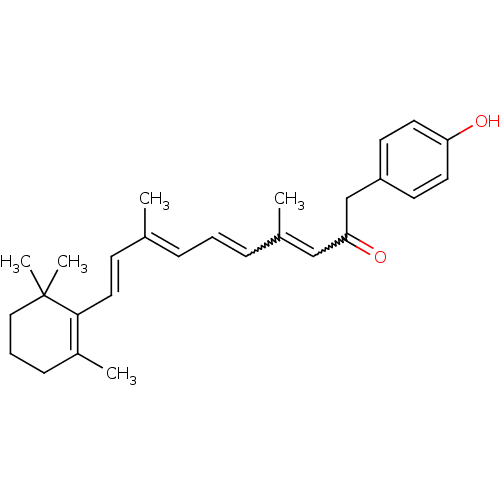
((3E,5E,7E,9E)-1-(4-Hydroxy-phenyl)-4,8-dimethyl-10...)Show SMILES C\C(\C=C\C1=C(C)CCCC1(C)C)=C/C=CC(C)=CC(=O)Cc1ccc(O)cc1 |w:18.19,15.16,c:4| Show InChI InChI=1S/C27H34O2/c1-20(11-16-26-22(3)10-7-17-27(26,4)5)8-6-9-21(2)18-25(29)19-23-12-14-24(28)15-13-23/h6,8-9,11-16,18,28H,7,10,17,19H2,1-5H3/b9-6?,16-11+,20-8+,21-18? | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| PubMed
| >4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The Ohio State University
Curated by ChEMBL
| Assay Description
Binding affinity against Retinoic acid receptor gamma |
Bioorg Med Chem Lett 11: 1583-6 (2001)
BindingDB Entry DOI: 10.7270/Q20R9NQW |
More data for this
Ligand-Target Pair | |
Squalene synthase
(Rattus norvegicus) | BDBM50029174
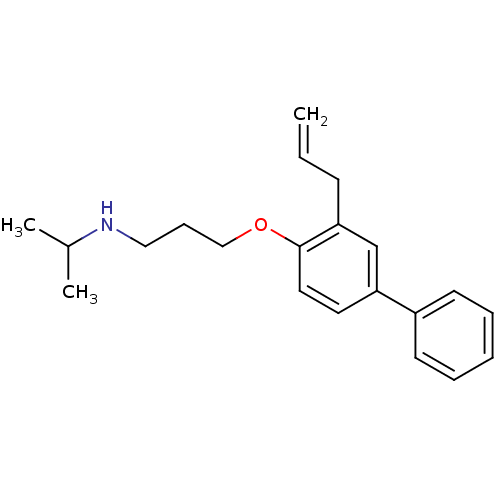
(CHEMBL131973 | N-(1-methylethyl)-3-[(3-prop-2-en-1...)Show InChI InChI=1S/C21H27NO/c1-4-9-20-16-19(18-10-6-5-7-11-18)12-13-21(20)23-15-8-14-22-17(2)3/h4-7,10-13,16-17,22H,1,8-9,14-15H2,2-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
| PubMed
| n/a | n/a | 0.00400 | n/a | n/a | n/a | n/a | n/a | n/a |
Zeneca Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of rat microsomal squalene synthase |
J Med Chem 38: 4157-60 (1995)
BindingDB Entry DOI: 10.7270/Q21V5D0D |
More data for this
Ligand-Target Pair | |
Squalene synthase
(Rattus norvegicus) | BDBM50029177

(CHEMBL134337 | Propionic acid 3-allyl-4-(3-isoprop...)Show InChI InChI=1S/C18H27NO3/c1-5-8-15-13-16(22-18(20)6-2)9-10-17(15)21-12-7-11-19-14(3)4/h5,9-10,13-14,19H,1,6-8,11-12H2,2-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a |
Zeneca Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of rat microsomal squalene synthase |
J Med Chem 38: 4157-60 (1995)
BindingDB Entry DOI: 10.7270/Q21V5D0D |
More data for this
Ligand-Target Pair | |
Squalene synthase
(Rattus norvegicus) | BDBM50029166
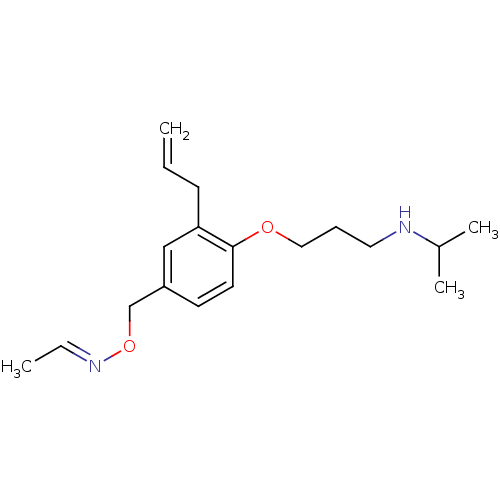
(Acetaldehyde O-[3-allyl-4-(3-isopropylamino-propox...)Show InChI InChI=1S/C18H28N2O2/c1-5-8-17-13-16(14-22-20-6-2)9-10-18(17)21-12-7-11-19-15(3)4/h5-6,9-10,13,15,19H,1,7-8,11-12,14H2,2-4H3/b20-6+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Zeneca Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of rat microsomal squalene synthase |
J Med Chem 38: 4157-60 (1995)
BindingDB Entry DOI: 10.7270/Q21V5D0D |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Rattus norvegicus (rat)) | BDBM50385398
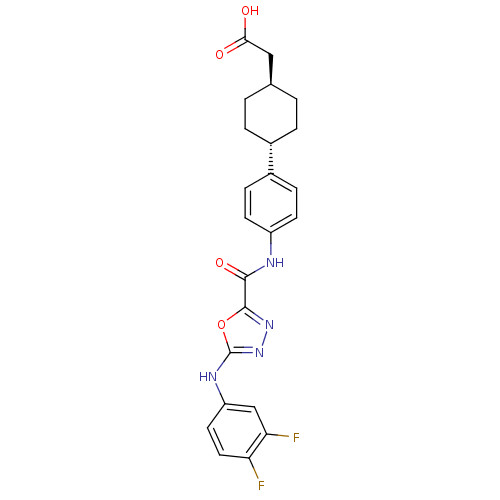
(CHEMBL2036730)Show SMILES OC(=O)C[C@H]1CC[C@@H](CC1)c1ccc(NC(=O)c2nnc(Nc3ccc(F)c(F)c3)o2)cc1 |r,wU:4.3,wD:7.10,(19.62,-39.47,;18.85,-40.8,;17.31,-40.79,;19.62,-42.14,;21.16,-42.14,;21.93,-40.81,;23.47,-40.82,;24.23,-42.16,;23.46,-43.49,;21.93,-43.48,;25.77,-42.17,;26.55,-40.84,;28.08,-40.85,;28.84,-42.19,;30.38,-42.2,;31.16,-40.87,;30.4,-39.53,;32.71,-40.88,;33.18,-39.41,;34.72,-39.41,;35.19,-40.87,;36.52,-41.64,;37.85,-40.86,;37.84,-39.33,;39.16,-38.55,;40.51,-39.31,;41.84,-38.53,;40.52,-40.85,;41.85,-41.61,;39.19,-41.63,;33.95,-41.78,;28.07,-43.51,;26.54,-43.51,)| Show InChI InChI=1S/C23H22F2N4O4/c24-18-10-9-17(12-19(18)25)27-23-29-28-22(33-23)21(32)26-16-7-5-15(6-8-16)14-3-1-13(2-4-14)11-20(30)31/h5-10,12-14H,1-4,11H2,(H,26,32)(H,27,29)(H,30,31)/t13-,14- | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of rat DGAT1 |
Bioorg Med Chem Lett 22: 3873-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.117
BindingDB Entry DOI: 10.7270/Q2T72JG4 |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50385398

(CHEMBL2036730)Show SMILES OC(=O)C[C@H]1CC[C@@H](CC1)c1ccc(NC(=O)c2nnc(Nc3ccc(F)c(F)c3)o2)cc1 |r,wU:4.3,wD:7.10,(19.62,-39.47,;18.85,-40.8,;17.31,-40.79,;19.62,-42.14,;21.16,-42.14,;21.93,-40.81,;23.47,-40.82,;24.23,-42.16,;23.46,-43.49,;21.93,-43.48,;25.77,-42.17,;26.55,-40.84,;28.08,-40.85,;28.84,-42.19,;30.38,-42.2,;31.16,-40.87,;30.4,-39.53,;32.71,-40.88,;33.18,-39.41,;34.72,-39.41,;35.19,-40.87,;36.52,-41.64,;37.85,-40.86,;37.84,-39.33,;39.16,-38.55,;40.51,-39.31,;41.84,-38.53,;40.52,-40.85,;41.85,-41.61,;39.19,-41.63,;33.95,-41.78,;28.07,-43.51,;26.54,-43.51,)| Show InChI InChI=1S/C23H22F2N4O4/c24-18-10-9-17(12-19(18)25)27-23-29-28-22(33-23)21(32)26-16-7-5-15(6-8-16)14-3-1-13(2-4-14)11-20(30)31/h5-10,12-14H,1-4,11H2,(H,26,32)(H,27,29)(H,30,31)/t13-,14- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of DGAT1-mediated triacylglycerol synthesis in human HuTu80 cells |
Bioorg Med Chem Lett 22: 3873-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.117
BindingDB Entry DOI: 10.7270/Q2T72JG4 |
More data for this
Ligand-Target Pair | |
Squalene synthase
(Rattus norvegicus) | BDBM50029159
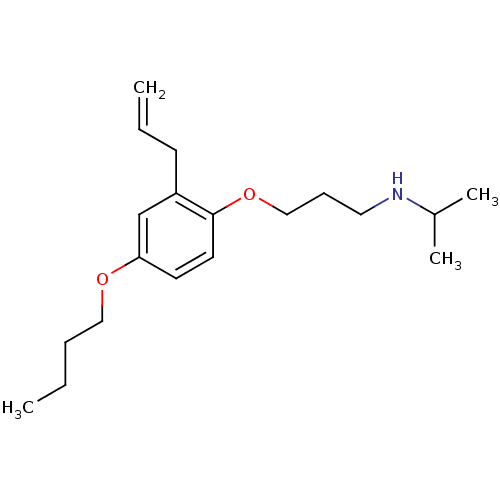
(CHEMBL132881 | [3-(2-Allyl-4-butoxy-phenoxy)-propy...)Show InChI InChI=1S/C19H31NO2/c1-5-7-13-21-18-10-11-19(17(15-18)9-6-2)22-14-8-12-20-16(3)4/h6,10-11,15-16,20H,2,5,7-9,12-14H2,1,3-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Zeneca Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of rat microsomal squalene synthase |
J Med Chem 38: 4157-60 (1995)
BindingDB Entry DOI: 10.7270/Q21V5D0D |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50385398

(CHEMBL2036730)Show SMILES OC(=O)C[C@H]1CC[C@@H](CC1)c1ccc(NC(=O)c2nnc(Nc3ccc(F)c(F)c3)o2)cc1 |r,wU:4.3,wD:7.10,(19.62,-39.47,;18.85,-40.8,;17.31,-40.79,;19.62,-42.14,;21.16,-42.14,;21.93,-40.81,;23.47,-40.82,;24.23,-42.16,;23.46,-43.49,;21.93,-43.48,;25.77,-42.17,;26.55,-40.84,;28.08,-40.85,;28.84,-42.19,;30.38,-42.2,;31.16,-40.87,;30.4,-39.53,;32.71,-40.88,;33.18,-39.41,;34.72,-39.41,;35.19,-40.87,;36.52,-41.64,;37.85,-40.86,;37.84,-39.33,;39.16,-38.55,;40.51,-39.31,;41.84,-38.53,;40.52,-40.85,;41.85,-41.61,;39.19,-41.63,;33.95,-41.78,;28.07,-43.51,;26.54,-43.51,)| Show InChI InChI=1S/C23H22F2N4O4/c24-18-10-9-17(12-19(18)25)27-23-29-28-22(33-23)21(32)26-16-7-5-15(6-8-16)14-3-1-13(2-4-14)11-20(30)31/h5-10,12-14H,1-4,11H2,(H,26,32)(H,27,29)(H,30,31)/t13-,14- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant DGAT1 expressed in baculovirus infected insect sf9 cells using [14C] oleoyl coenzyme A after 30 mins by scintillation... |
Bioorg Med Chem Lett 22: 3873-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.117
BindingDB Entry DOI: 10.7270/Q2T72JG4 |
More data for this
Ligand-Target Pair | |
Squalene synthase
(Rattus norvegicus) | BDBM50029171

(CHEMBL341371 | N-[3-Benzyl-4-(3-isopropylamino-pro...)Show InChI InChI=1S/C22H30N2O2/c1-4-22(25)24-20-11-12-21(26-14-8-13-23-17(2)3)19(16-20)15-18-9-6-5-7-10-18/h5-7,9-12,16-17,23H,4,8,13-15H2,1-3H3,(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 0.600 | n/a | n/a | n/a | n/a | n/a | n/a |
Zeneca Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of rat microsomal squalene synthase |
J Med Chem 38: 4157-60 (1995)
BindingDB Entry DOI: 10.7270/Q21V5D0D |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116610
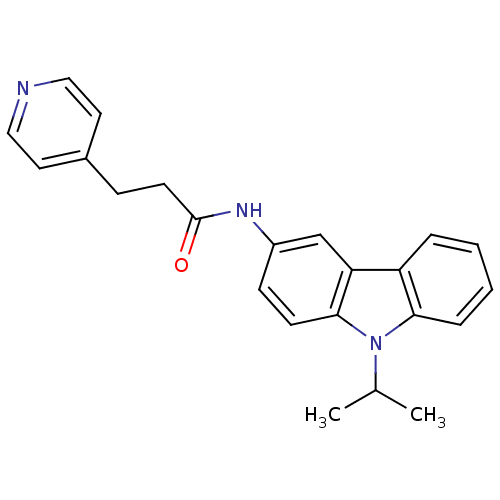
(CHEMBL119743 | N-(9-Isopropyl-9H-carbazol-3-yl)-3-...)Show InChI InChI=1S/C23H23N3O/c1-16(2)26-21-6-4-3-5-19(21)20-15-18(8-9-22(20)26)25-23(27)10-7-17-11-13-24-14-12-17/h3-6,8-9,11-16H,7,10H2,1-2H3,(H,25,27) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to the human Neuropeptide Y receptor Y5 (NPY5) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116590
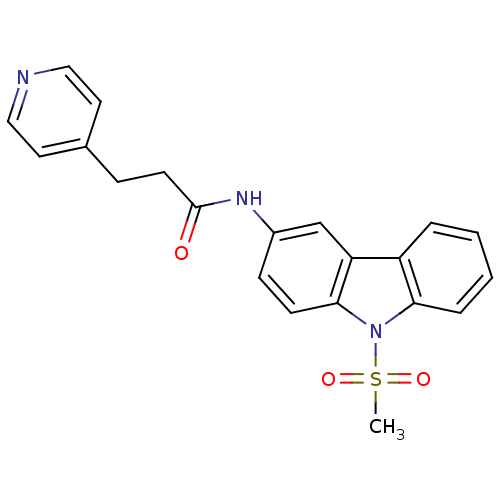
(CHEMBL325486 | N-(9-Methanesulfonyl-9H-carbazol-3-...)Show SMILES CS(=O)(=O)n1c2ccccc2c2cc(NC(=O)CCc3ccncc3)ccc12 Show InChI InChI=1S/C21H19N3O3S/c1-28(26,27)24-19-5-3-2-4-17(19)18-14-16(7-8-20(18)24)23-21(25)9-6-15-10-12-22-13-11-15/h2-5,7-8,10-14H,6,9H2,1H3,(H,23,25) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to the human Neuropeptide Y receptor Y5 (NPY5) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Squalene synthase
(Rattus norvegicus) | BDBM50029179
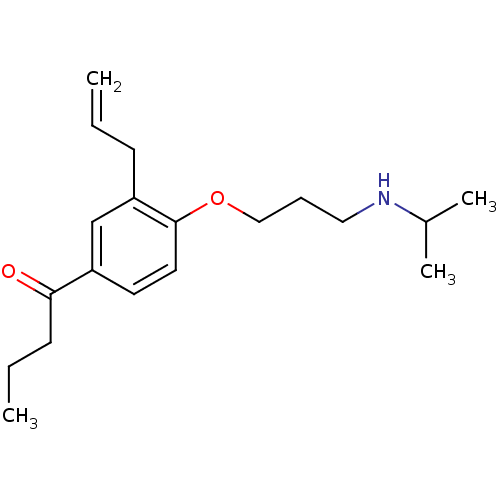
(1-[3-Allyl-4-(3-isopropylamino-propoxy)-phenyl]-bu...)Show InChI InChI=1S/C19H29NO2/c1-5-8-17-14-16(18(21)9-6-2)10-11-19(17)22-13-7-12-20-15(3)4/h5,10-11,14-15,20H,1,6-9,12-13H2,2-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Zeneca Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of rat microsomal squalene synthase |
J Med Chem 38: 4157-60 (1995)
BindingDB Entry DOI: 10.7270/Q21V5D0D |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Mus musculus (mouse)) | BDBM50385398

(CHEMBL2036730)Show SMILES OC(=O)C[C@H]1CC[C@@H](CC1)c1ccc(NC(=O)c2nnc(Nc3ccc(F)c(F)c3)o2)cc1 |r,wU:4.3,wD:7.10,(19.62,-39.47,;18.85,-40.8,;17.31,-40.79,;19.62,-42.14,;21.16,-42.14,;21.93,-40.81,;23.47,-40.82,;24.23,-42.16,;23.46,-43.49,;21.93,-43.48,;25.77,-42.17,;26.55,-40.84,;28.08,-40.85,;28.84,-42.19,;30.38,-42.2,;31.16,-40.87,;30.4,-39.53,;32.71,-40.88,;33.18,-39.41,;34.72,-39.41,;35.19,-40.87,;36.52,-41.64,;37.85,-40.86,;37.84,-39.33,;39.16,-38.55,;40.51,-39.31,;41.84,-38.53,;40.52,-40.85,;41.85,-41.61,;39.19,-41.63,;33.95,-41.78,;28.07,-43.51,;26.54,-43.51,)| Show InChI InChI=1S/C23H22F2N4O4/c24-18-10-9-17(12-19(18)25)27-23-29-28-22(33-23)21(32)26-16-7-5-15(6-8-16)14-3-1-13(2-4-14)11-20(30)31/h5-10,12-14H,1-4,11H2,(H,26,32)(H,27,29)(H,30,31)/t13-,14- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of mouse DGAT1 |
Bioorg Med Chem Lett 22: 3873-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.117
BindingDB Entry DOI: 10.7270/Q2T72JG4 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116619
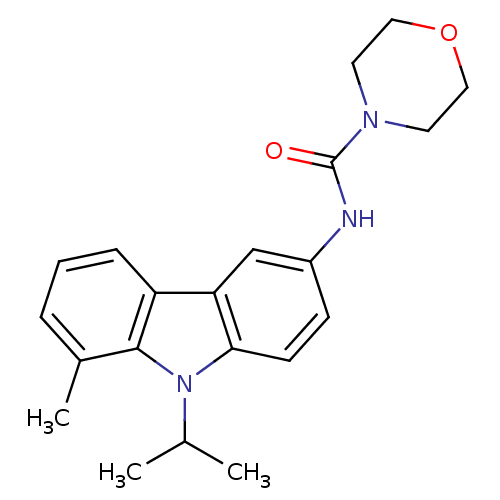
(CHEMBL117563 | Morpholine-4-carboxylic acid (9-iso...)Show SMILES CC(C)n1c2ccc(NC(=O)N3CCOCC3)cc2c2cccc(C)c12 Show InChI InChI=1S/C21H25N3O2/c1-14(2)24-19-8-7-16(22-21(25)23-9-11-26-12-10-23)13-18(19)17-6-4-5-15(3)20(17)24/h4-8,13-14H,9-12H2,1-3H3,(H,22,25) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to the human Neuropeptide Y receptor Y5 (NPY5) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116602
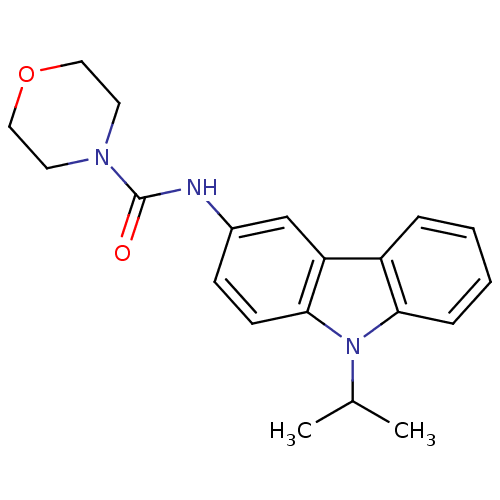
(CHEMBL419951 | Morpholine-4-carboxylic acid (9-iso...)Show InChI InChI=1S/C20H23N3O2/c1-14(2)23-18-6-4-3-5-16(18)17-13-15(7-8-19(17)23)21-20(24)22-9-11-25-12-10-22/h3-8,13-14H,9-12H2,1-2H3,(H,21,24) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to the human Neuropeptide Y receptor Y5 (NPY5) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116600
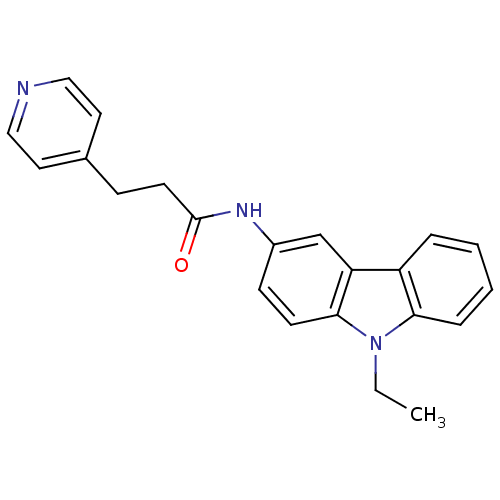
(CHEMBL325475 | N-(9-Ethyl-9H-carbazol-3-yl)-3-pyri...)Show InChI InChI=1S/C22H21N3O/c1-2-25-20-6-4-3-5-18(20)19-15-17(8-9-21(19)25)24-22(26)10-7-16-11-13-23-14-12-16/h3-6,8-9,11-15H,2,7,10H2,1H3,(H,24,26) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to the human Neuropeptide Y receptor Y5 (NPY5) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116605
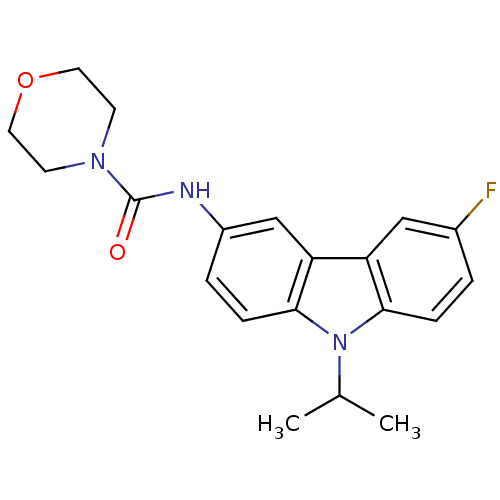
(CHEMBL432628 | Morpholine-4-carboxylic acid (6-flu...)Show SMILES CC(C)n1c2ccc(F)cc2c2cc(NC(=O)N3CCOCC3)ccc12 Show InChI InChI=1S/C20H22FN3O2/c1-13(2)24-18-5-3-14(21)11-16(18)17-12-15(4-6-19(17)24)22-20(25)23-7-9-26-10-8-23/h3-6,11-13H,7-10H2,1-2H3,(H,22,25) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to the human Neuropeptide Y receptor Y5 (NPY5) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116617
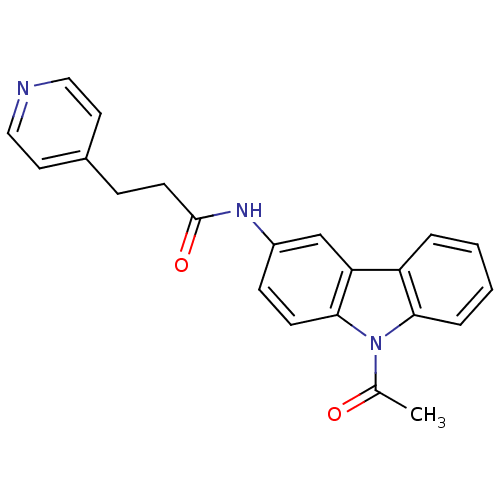
(CHEMBL116210 | N-(9-Acetyl-9H-carbazol-3-yl)-3-pyr...)Show SMILES CC(=O)n1c2ccccc2c2cc(NC(=O)CCc3ccncc3)ccc12 Show InChI InChI=1S/C22H19N3O2/c1-15(26)25-20-5-3-2-4-18(20)19-14-17(7-8-21(19)25)24-22(27)9-6-16-10-12-23-13-11-16/h2-5,7-8,10-14H,6,9H2,1H3,(H,24,27) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to the human Neuropeptide Y receptor Y5 (NPY5) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Squalene synthase
(Rattus norvegicus) | BDBM50029163
(CHEMBL340992 | Propionic acid 4-(3-isopropylamino-...)Show InChI InChI=1S/C15H23NO3/c1-4-15(17)19-14-8-6-13(7-9-14)18-11-5-10-16-12(2)3/h6-9,12,16H,4-5,10-11H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Zeneca Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of rat microsomal squalene synthase |
J Med Chem 38: 4157-60 (1995)
BindingDB Entry DOI: 10.7270/Q21V5D0D |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116592
(CHEMBL325226 | Morpholine-4-carboxylic acid (9-iso...)Show SMILES CC(C)n1c2ccccc2c2c(C)c(NC(=O)N3CCOCC3)ccc12 Show InChI InChI=1S/C21H25N3O2/c1-14(2)24-18-7-5-4-6-16(18)20-15(3)17(8-9-19(20)24)22-21(25)23-10-12-26-13-11-23/h4-9,14H,10-13H2,1-3H3,(H,22,25) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to the human Neuropeptide Y receptor Y5 (NPY5) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50385439
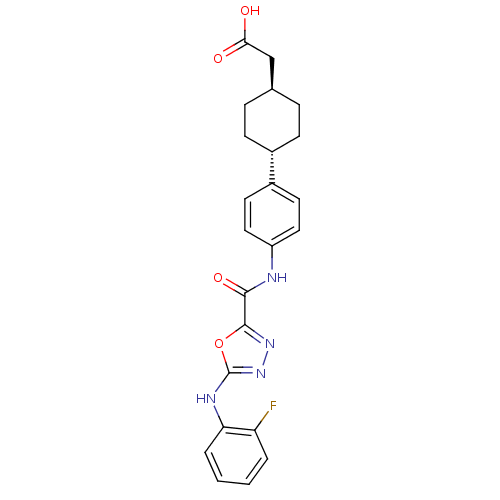
(CHEMBL2036745)Show SMILES OC(=O)C[C@H]1CC[C@@H](CC1)c1ccc(NC(=O)c2nnc(Nc3ccccc3F)o2)cc1 |r,wU:7.10,wD:4.3,(45.29,-27.64,;44.52,-28.98,;42.98,-28.97,;45.28,-30.31,;46.82,-30.32,;47.6,-31.66,;49.13,-31.66,;49.9,-30.34,;49.14,-29,;47.6,-28.99,;51.43,-30.34,;52.21,-29.01,;53.75,-29.02,;54.51,-30.36,;56.05,-30.37,;56.83,-29.04,;56.07,-27.71,;58.38,-29.05,;58.85,-27.59,;60.38,-27.59,;60.86,-29.05,;62.19,-29.81,;63.52,-29.04,;63.51,-27.5,;64.83,-26.72,;66.18,-27.49,;66.19,-29.03,;64.86,-29.81,;64.86,-31.35,;59.62,-29.96,;53.74,-31.69,;52.2,-31.69,)| Show InChI InChI=1S/C23H23FN4O4/c24-18-3-1-2-4-19(18)26-23-28-27-22(32-23)21(31)25-17-11-9-16(10-12-17)15-7-5-14(6-8-15)13-20(29)30/h1-4,9-12,14-15H,5-8,13H2,(H,25,31)(H,26,28)(H,29,30)/t14-,15- | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.40 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant DGAT1 expressed in baculovirus infected insect sf9 cells using [14C] oleoyl coenzyme A after 30 mins by scintillation... |
Bioorg Med Chem Lett 22: 3873-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.117
BindingDB Entry DOI: 10.7270/Q2T72JG4 |
More data for this
Ligand-Target Pair | |
Squalene synthase
(Rattus norvegicus) | BDBM50029170
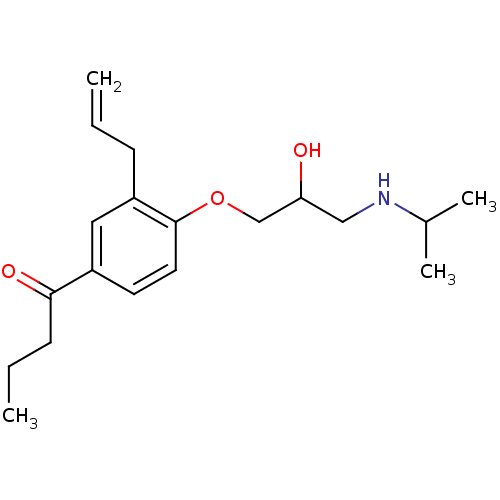
(1-[3-Allyl-4-(2-hydroxy-3-isopropylamino-propoxy)-...)Show InChI InChI=1S/C19H29NO3/c1-5-7-16-11-15(18(22)8-6-2)9-10-19(16)23-13-17(21)12-20-14(3)4/h5,9-11,14,17,20-21H,1,6-8,12-13H2,2-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Zeneca Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of rat microsomal squalene synthase |
J Med Chem 38: 4157-60 (1995)
BindingDB Entry DOI: 10.7270/Q21V5D0D |
More data for this
Ligand-Target Pair | |
Squalene synthase
(Rattus norvegicus) | BDBM50029161
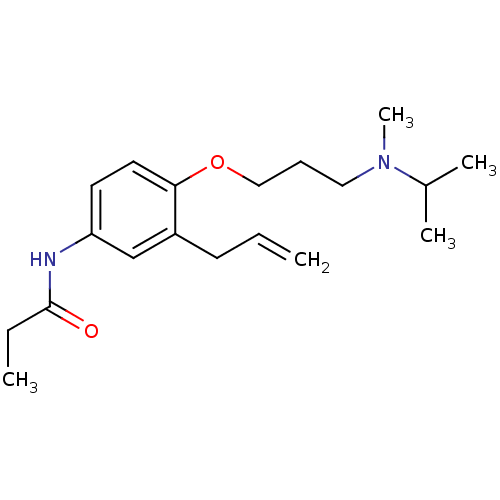
(CHEMBL433864 | N-{3-Allyl-4-[3-(isopropyl-methyl-a...)Show InChI InChI=1S/C19H30N2O2/c1-6-9-16-14-17(20-19(22)7-2)10-11-18(16)23-13-8-12-21(5)15(3)4/h6,10-11,14-15H,1,7-9,12-13H2,2-5H3,(H,20,22) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Zeneca Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of rat microsomal squalene synthase |
J Med Chem 38: 4157-60 (1995)
BindingDB Entry DOI: 10.7270/Q21V5D0D |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50385429
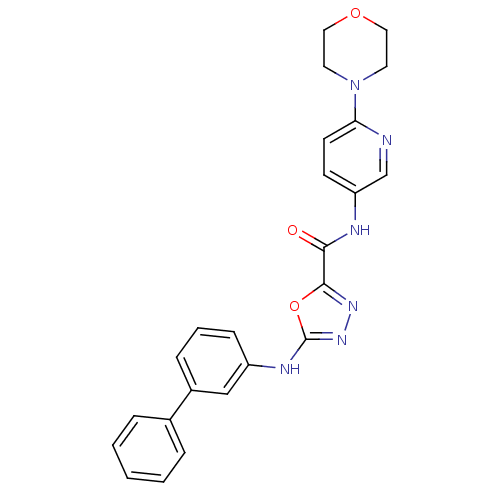
(CHEMBL2036734)Show SMILES O=C(Nc1ccc(nc1)N1CCOCC1)c1nnc(Nc2cccc(c2)-c2ccccc2)o1 Show InChI InChI=1S/C24H22N6O3/c31-22(26-20-9-10-21(25-16-20)30-11-13-32-14-12-30)23-28-29-24(33-23)27-19-8-4-7-18(15-19)17-5-2-1-3-6-17/h1-10,15-16H,11-14H2,(H,26,31)(H,27,29) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.70 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant DGAT1 expressed in baculovirus infected insect sf9 cells using [14C] oleoyl coenzyme A after 30 mins by scintillation... |
Bioorg Med Chem Lett 22: 3873-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.117
BindingDB Entry DOI: 10.7270/Q2T72JG4 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116592

(CHEMBL325226 | Morpholine-4-carboxylic acid (9-iso...)Show SMILES CC(C)n1c2ccccc2c2c(C)c(NC(=O)N3CCOCC3)ccc12 Show InChI InChI=1S/C21H25N3O2/c1-14(2)24-18-7-5-4-6-16(18)20-15(3)17(8-9-19(20)24)22-21(25)23-10-12-26-13-11-23/h4-9,14H,10-13H2,1-3H3,(H,22,25) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Compound was evaluated for functional antagonism of Neuropeptide Y receptor Y5 activity in cellular Ca flux |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50385424
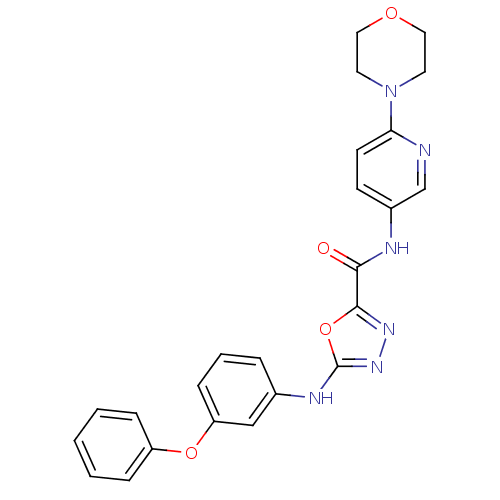
(CHEMBL2036596)Show SMILES O=C(Nc1ccc(nc1)N1CCOCC1)c1nnc(Nc2cccc(Oc3ccccc3)c2)o1 Show InChI InChI=1S/C24H22N6O4/c31-22(26-18-9-10-21(25-16-18)30-11-13-32-14-12-30)23-28-29-24(34-23)27-17-5-4-8-20(15-17)33-19-6-2-1-3-7-19/h1-10,15-16H,11-14H2,(H,26,31)(H,27,29) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.60 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant DGAT1 expressed in baculovirus infected insect sf9 cells using [14C] oleoyl coenzyme A after 30 mins by scintillation... |
Bioorg Med Chem Lett 22: 3873-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.117
BindingDB Entry DOI: 10.7270/Q2T72JG4 |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50385412
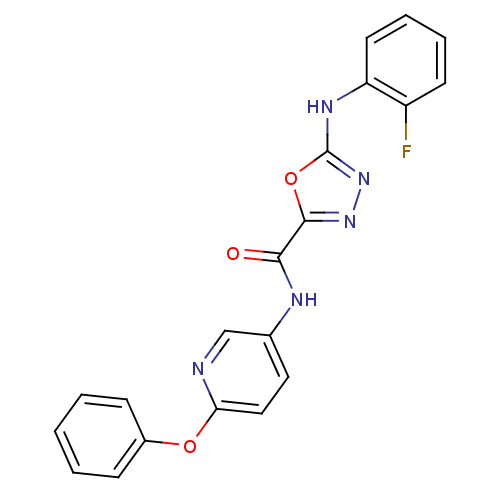
(CHEMBL2036583)Show SMILES Fc1ccccc1Nc1nnc(o1)C(=O)Nc1ccc(Oc2ccccc2)nc1 Show InChI InChI=1S/C20H14FN5O3/c21-15-8-4-5-9-16(15)24-20-26-25-19(29-20)18(27)23-13-10-11-17(22-12-13)28-14-6-2-1-3-7-14/h1-12H,(H,23,27)(H,24,26) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6.70 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant DGAT1 expressed in baculovirus infected insect sf9 cells using [14C] oleoyl coenzyme A after 30 mins by scintillation... |
Bioorg Med Chem Lett 22: 3873-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.117
BindingDB Entry DOI: 10.7270/Q2T72JG4 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116597
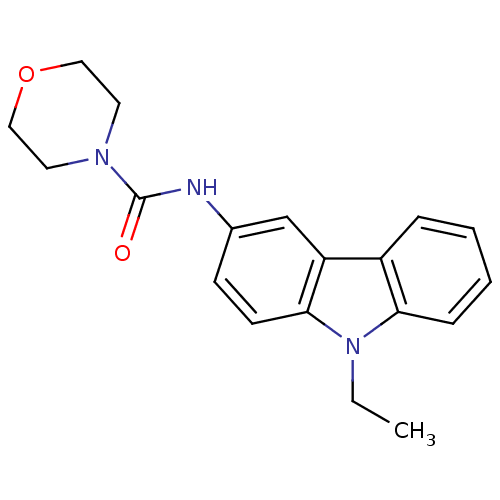
(CHEMBL119247 | Morpholine-4-carboxylic acid (9-eth...)Show InChI InChI=1S/C19H21N3O2/c1-2-22-17-6-4-3-5-15(17)16-13-14(7-8-18(16)22)20-19(23)21-9-11-24-12-10-21/h3-8,13H,2,9-12H2,1H3,(H,20,23) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to the human Neuropeptide Y receptor Y5 (NPY5) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Rat 6B) | BDBM50116600

(CHEMBL325475 | N-(9-Ethyl-9H-carbazol-3-yl)-3-pyri...)Show InChI InChI=1S/C22H21N3O/c1-2-25-20-6-4-3-5-18(20)19-15-17(8-9-21(19)25)24-22(26)10-7-16-11-13-23-14-12-16/h3-6,8-9,11-15H,2,7,10H2,1H3,(H,24,26) | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity against rat Neuropeptide Y receptor Y5 |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116608
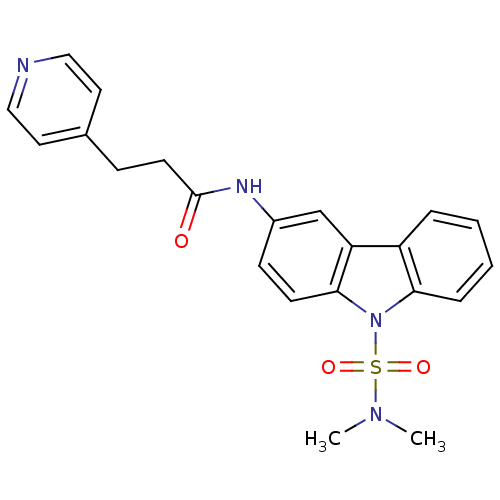
(CHEMBL117922 | N-(9-Dimethylsulfamoyl-9H-carbazol-...)Show SMILES CN(C)S(=O)(=O)n1c2ccccc2c2cc(NC(=O)CCc3ccncc3)ccc12 Show InChI InChI=1S/C22H22N4O3S/c1-25(2)30(28,29)26-20-6-4-3-5-18(20)19-15-17(8-9-21(19)26)24-22(27)10-7-16-11-13-23-14-12-16/h3-6,8-9,11-15H,7,10H2,1-2H3,(H,24,27) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to the human Neuropeptide Y receptor Y5 (NPY5) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
D-amino-acid oxidase
(Homo sapiens (Human)) | BDBM50605601
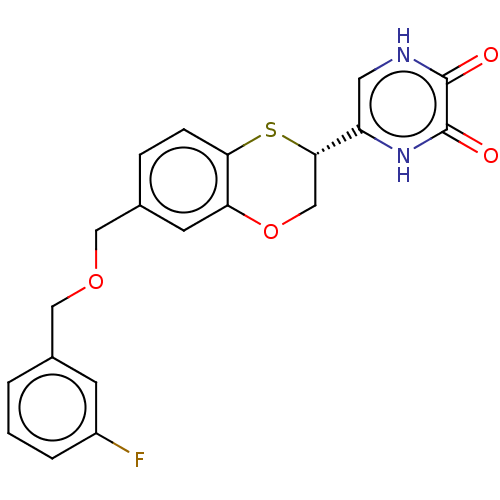
(CHEMBL5184138)Show SMILES Fc1cccc(COCc2ccc3S[C@@H](COc3c2)c2c[nH]c(=O)c(=O)[nH]2)c1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00118
BindingDB Entry DOI: 10.7270/Q2MG7TK7 |
More data for this
Ligand-Target Pair | |
D-amino-acid oxidase
(Homo sapiens (Human)) | BDBM50605598

(CHEMBL5188193)Show SMILES C[C@]1(COc2cc(Cl)ccc2S1)c1c[nH]c(=O)c(=O)[nH]1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00118
BindingDB Entry DOI: 10.7270/Q2MG7TK7 |
More data for this
Ligand-Target Pair | |
D-amino-acid oxidase
(Homo sapiens (Human)) | BDBM50605597
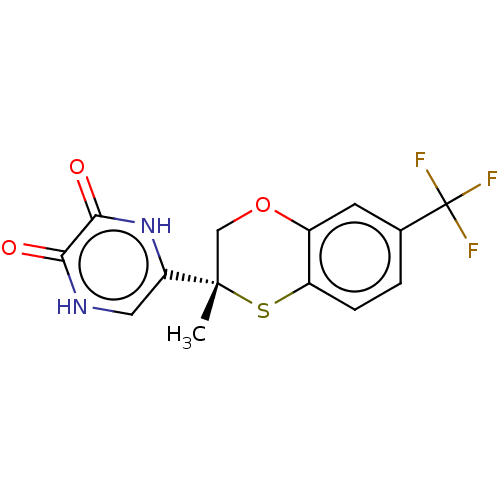
(CHEMBL5204161)Show SMILES C[C@]1(COc2cc(ccc2S1)C(F)(F)F)c1c[nH]c(=O)c(=O)[nH]1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00118
BindingDB Entry DOI: 10.7270/Q2MG7TK7 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116600

(CHEMBL325475 | N-(9-Ethyl-9H-carbazol-3-yl)-3-pyri...)Show InChI InChI=1S/C22H21N3O/c1-2-25-20-6-4-3-5-18(20)19-15-17(8-9-21(19)25)24-22(26)10-7-16-11-13-23-14-12-16/h3-6,8-9,11-15H,2,7,10H2,1H3,(H,24,26) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Compound was evaluated in vitro for human Neuropeptide Y receptor Y5 functional antagonism (reporter gene assay) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
D-amino-acid oxidase
(Homo sapiens (Human)) | BDBM50605555
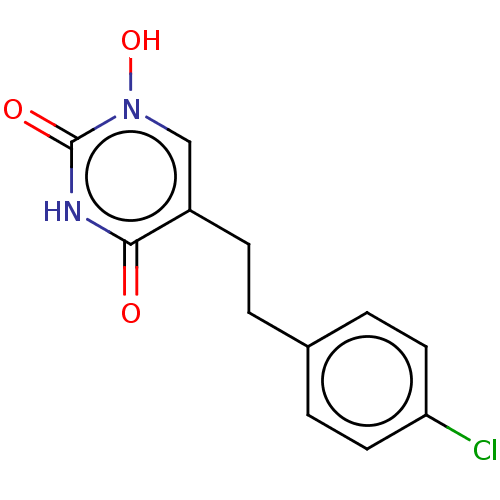
(CHEMBL5173250) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00118
BindingDB Entry DOI: 10.7270/Q2MG7TK7 |
More data for this
Ligand-Target Pair | |
Squalene synthase
(Rattus norvegicus) | BDBM50029172
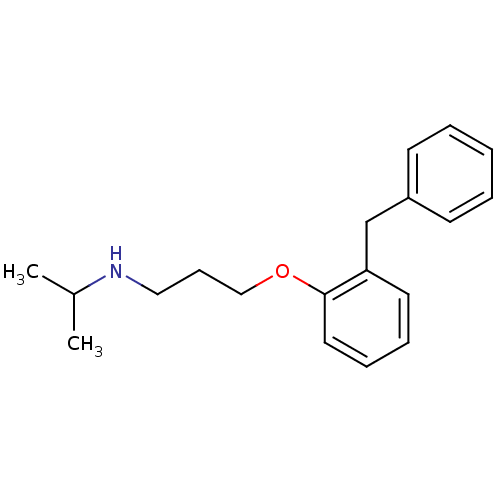
(CHEMBL131930 | [3-(2-Benzyl-phenoxy)-propyl]-isopr...)Show InChI InChI=1S/C19H25NO/c1-16(2)20-13-8-14-21-19-12-7-6-11-18(19)15-17-9-4-3-5-10-17/h3-7,9-12,16,20H,8,13-15H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Zeneca Pharmaceuticals
Curated by ChEMBL
| Assay Description
Inhibition of rat microsomal squalene synthase |
J Med Chem 38: 4157-60 (1995)
BindingDB Entry DOI: 10.7270/Q21V5D0D |
More data for this
Ligand-Target Pair | |
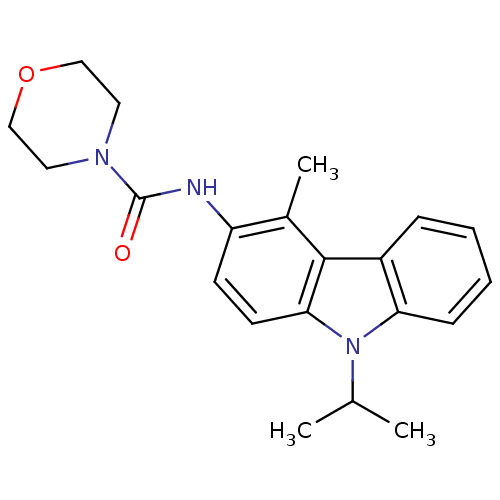
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116607
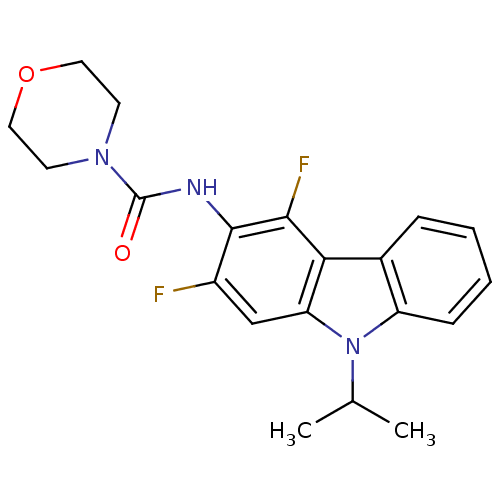
(CHEMBL325639 | Morpholine-4-carboxylic acid (2,4-d...)Show SMILES CC(C)n1c2ccccc2c2c(F)c(NC(=O)N3CCOCC3)c(F)cc12 Show InChI InChI=1S/C20H21F2N3O2/c1-12(2)25-15-6-4-3-5-13(15)17-16(25)11-14(21)19(18(17)22)23-20(26)24-7-9-27-10-8-24/h3-6,11-12H,7-10H2,1-2H3,(H,23,26) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to the human Neuropeptide Y receptor Y5 (NPY5) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50385425

(CHEMBL2036597)Show SMILES O=C(Nc1ccc(nc1)N1CCOCC1)c1nnc(Nc2cccc(OCc3ccccc3)c2)o1 Show InChI InChI=1S/C25H24N6O4/c32-23(27-20-9-10-22(26-16-20)31-11-13-33-14-12-31)24-29-30-25(35-24)28-19-7-4-8-21(15-19)34-17-18-5-2-1-3-6-18/h1-10,15-16H,11-14,17H2,(H,27,32)(H,28,30) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant DGAT1 expressed in baculovirus infected insect sf9 cells using [14C] oleoyl coenzyme A after 30 mins by scintillation... |
Bioorg Med Chem Lett 22: 3873-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.117
BindingDB Entry DOI: 10.7270/Q2T72JG4 |
More data for this
Ligand-Target Pair | |
D-amino-acid oxidase
(Homo sapiens (Human)) | BDBM50605593
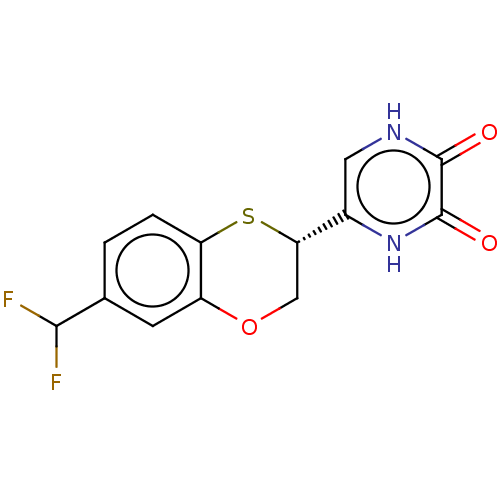
(CHEMBL5179642)Show SMILES FC(F)c1ccc2S[C@@H](COc2c1)c1c[nH]c(=O)c(=O)[nH]1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00118
BindingDB Entry DOI: 10.7270/Q2MG7TK7 |
More data for this
Ligand-Target Pair | |
Neuropeptide Y receptor type 5
(Homo sapiens (Human)) | BDBM50116603
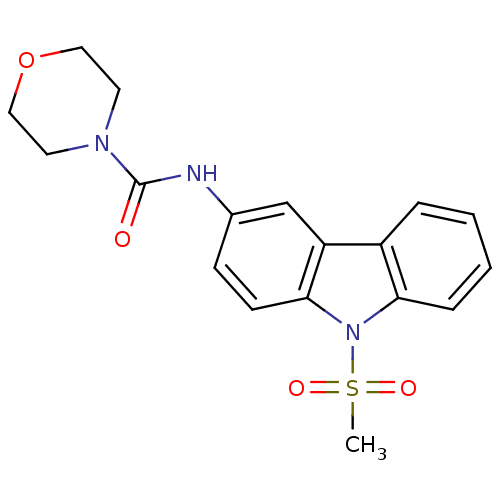
(CHEMBL443809 | Morpholine-4-carboxylic acid (9-met...)Show SMILES CS(=O)(=O)n1c2ccccc2c2cc(NC(=O)N3CCOCC3)ccc12 Show InChI InChI=1S/C18H19N3O4S/c1-26(23,24)21-16-5-3-2-4-14(16)15-12-13(6-7-17(15)21)19-18(22)20-8-10-25-11-9-20/h2-7,12H,8-11H2,1H3,(H,19,22) | KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 15 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Binding affinity to the human Neuropeptide Y receptor Y5 (NPY5) |
J Med Chem 45: 3509-23 (2002)
BindingDB Entry DOI: 10.7270/Q27S7N3K |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50385415
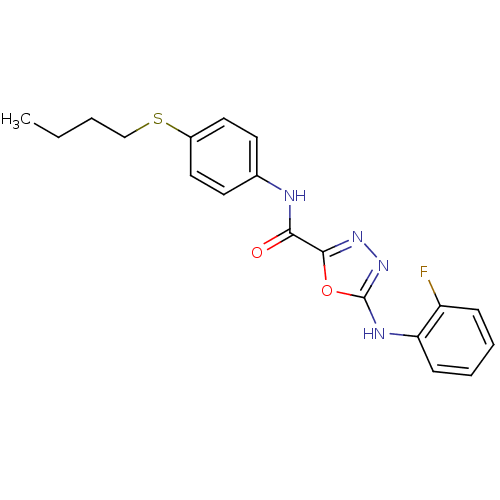
(CHEMBL2036586)Show InChI InChI=1S/C19H19FN4O2S/c1-2-3-12-27-14-10-8-13(9-11-14)21-17(25)18-23-24-19(26-18)22-16-7-5-4-6-15(16)20/h4-11H,2-3,12H2,1H3,(H,21,25)(H,22,24) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant DGAT1 expressed in baculovirus infected insect sf9 cells using [14C] oleoyl coenzyme A after 30 mins by scintillation... |
Bioorg Med Chem Lett 22: 3873-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.117
BindingDB Entry DOI: 10.7270/Q2T72JG4 |
More data for this
Ligand-Target Pair | |
D-amino-acid oxidase
(Homo sapiens (Human)) | BDBM210802
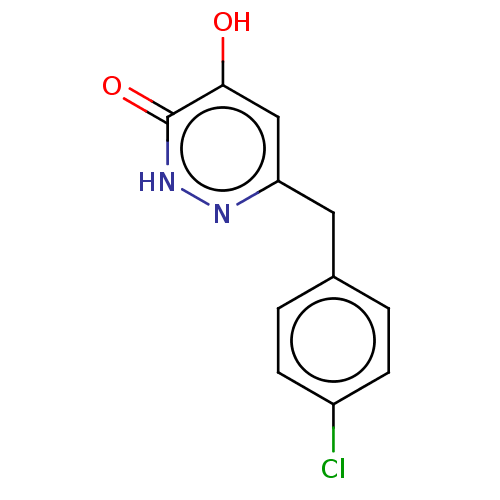
(US10463663, Example 40 | US11129828, Example 40 | ...)Show InChI InChI=1S/C11H9ClN2O2/c12-8-3-1-7(2-4-8)5-9-6-10(15)11(16)14-13-9/h1-4,6H,5H2,(H,13,15)(H,14,16) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00118
BindingDB Entry DOI: 10.7270/Q2MG7TK7 |
More data for this
Ligand-Target Pair | |
D-amino-acid oxidase
(Homo sapiens (Human)) | BDBM50605594

(CHEMBL5186374)Show SMILES O=c1[nH]cc([nH]c1=O)[C@@H]1COc2cc(ccc2S1)C#N |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00118
BindingDB Entry DOI: 10.7270/Q2MG7TK7 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
D-amino-acid oxidase
(Homo sapiens (Human)) | BDBM50605599

(CHEMBL5178413)Show SMILES C[C@]1(COc2cc(ccc2S1)C#N)c1c[nH]c(=O)c(=O)[nH]1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 20 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00118
BindingDB Entry DOI: 10.7270/Q2MG7TK7 |
More data for this
Ligand-Target Pair | |
D-amino-acid oxidase
(Homo sapiens (Human)) | BDBM50605592

(CHEMBL5179643)Show SMILES Clc1ccc2S[C@@H](COc2c1)c1c[nH]c(=O)c(=O)[nH]1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00118
BindingDB Entry DOI: 10.7270/Q2MG7TK7 |
More data for this
Ligand-Target Pair | |
Diacylglycerol O-acyltransferase 1
(Homo sapiens (Human)) | BDBM50385403
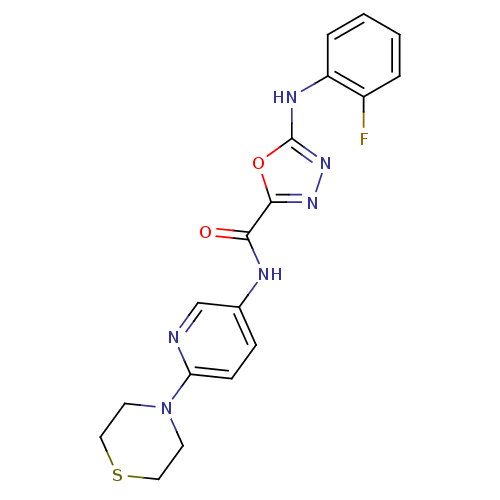
(CHEMBL2036574)Show SMILES Fc1ccccc1Nc1nnc(o1)C(=O)Nc1ccc(nc1)N1CCSCC1 Show InChI InChI=1S/C18H17FN6O2S/c19-13-3-1-2-4-14(13)22-18-24-23-17(27-18)16(26)21-12-5-6-15(20-11-12)25-7-9-28-10-8-25/h1-6,11H,7-10H2,(H,21,26)(H,22,24) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant DGAT1 expressed in baculovirus infected insect sf9 cells using [14C] oleoyl coenzyme A after 30 mins by scintillation... |
Bioorg Med Chem Lett 22: 3873-8 (2012)
Article DOI: 10.1016/j.bmcl.2012.04.117
BindingDB Entry DOI: 10.7270/Q2T72JG4 |
More data for this
Ligand-Target Pair | |
D-amino-acid oxidase
(Homo sapiens (Human)) | BDBM50605595

(CHEMBL5174518)Show SMILES CS(=O)(=O)c1ccc2S[C@@H](COc2c1)c1c[nH]c(=O)c(=O)[nH]1 |r| | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00118
BindingDB Entry DOI: 10.7270/Q2MG7TK7 |
More data for this
Ligand-Target Pair | |
D-amino-acid oxidase
(Homo sapiens (Human)) | BDBM50605589
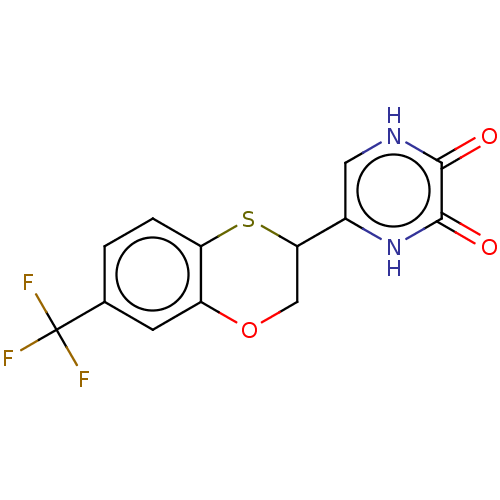
(CHEMBL5196542)Show SMILES FC(F)(F)c1ccc2SC(COc2c1)c1c[nH]c(=O)c(=O)[nH]1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00118
BindingDB Entry DOI: 10.7270/Q2MG7TK7 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data