 Found 10565 hits with Last Name = 'gu' and Initial = 'w'
Found 10565 hits with Last Name = 'gu' and Initial = 'w' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
5-hydroxytryptamine receptor 1B
(Homo sapiens (Human)) | BDBM50280055
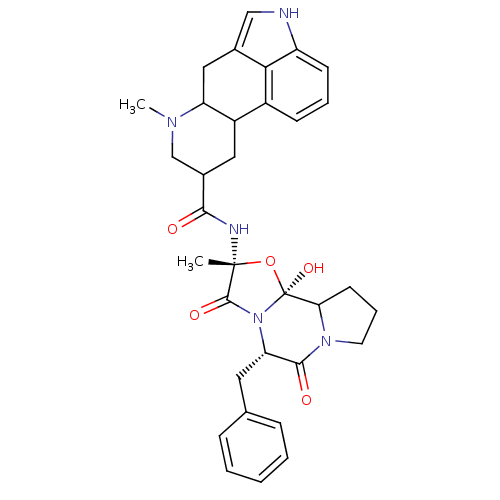
(50285557 | 7-Methyl-4,6,6a,7,8,9,10,10a-octahydro-...)Show SMILES CN1CC(CC2C1Cc1c[nH]c3cccc2c13)C(=O)N[C@]1(C)O[C@@]2(O)C3CCCN3C(=O)[C@H](Cc3ccccc3)N2C1=O Show InChI InChI=1S/C33H37N5O5/c1-32(35-29(39)21-15-23-22-10-6-11-24-28(22)20(17-34-24)16-25(23)36(2)18-21)31(41)38-26(14-19-8-4-3-5-9-19)30(40)37-13-7-12-27(37)33(38,42)43-32/h3-6,8-11,17,21,23,25-27,34,42H,7,12-16,18H2,1-2H3,(H,35,39)/t21?,23?,25?,26-,27?,32+,33-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.00600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Veterans Affairs Medical Center
Curated by PDSP Ki Database
| |
Biochem Biophys Res Commun 184: 752-9 (1992)
Article DOI: 10.1016/0006-291x(92)90654-4
BindingDB Entry DOI: 10.7270/Q2ST7NB1 |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50036480

(2,6-Dichloro-3-(2-morpholin-4-yl-ethoxy)-benzoic a...)Show SMILES CC(C)c1cccc2c1C(=O)N(COC(=O)c1c(Cl)ccc(OCCN3CCOCC3)c1Cl)S2(=O)=O Show InChI InChI=1S/C24H26Cl2N2O7S/c1-15(2)16-4-3-5-19-20(16)23(29)28(36(19,31)32)14-35-24(30)21-17(25)6-7-18(22(21)26)34-13-10-27-8-11-33-12-9-27/h3-7,15H,8-14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.00800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sterling Winthrop Inc.
Curated by ChEMBL
| Assay Description
Potency of inhibition against human leukocyte elastase (HLE) expressed as an apparent binding constant |
J Med Chem 38: 739-44 (1995)
BindingDB Entry DOI: 10.7270/Q2W66JTD |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50036478

(3-Carboxymethoxy-2,6-dichloro-benzoic acid 4-isopr...)Show SMILES COc1cc2c(C(=O)N(COC(=O)c3c(Cl)ccc(OCC(O)=O)c3Cl)S2(=O)=O)c(c1)C(C)C Show InChI InChI=1S/C21H19Cl2NO9S/c1-10(2)12-6-11(31-3)7-15-17(12)20(27)24(34(15,29)30)9-33-21(28)18-13(22)4-5-14(19(18)23)32-8-16(25)26/h4-7,10H,8-9H2,1-3H3,(H,25,26) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sterling Winthrop Inc.
Curated by ChEMBL
| Assay Description
Potency of inhibition against human leukocyte elastase (HLE) expressed as an apparent binding constant |
J Med Chem 38: 739-44 (1995)
BindingDB Entry DOI: 10.7270/Q2W66JTD |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50036476

(2,6-Dichloro-3-(4-methyl-piperazine-1-sulfonyl)-be...)Show SMILES COc1cc2c(C(=O)N(COC(=O)c3c(Cl)ccc(c3Cl)S(=O)(=O)N3CCN(C)CC3)S2(=O)=O)c(c1)C(C)C Show InChI InChI=1S/C24H27Cl2N3O8S2/c1-14(2)16-11-15(36-4)12-19-20(16)23(30)29(39(19,34)35)13-37-24(31)21-17(25)5-6-18(22(21)26)38(32,33)28-9-7-27(3)8-10-28/h5-6,11-12,14H,7-10,13H2,1-4H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sterling Winthrop Inc.
Curated by ChEMBL
| Assay Description
Potency of inhibition against human leukocyte elastase (HLE) expressed as an apparent binding constant |
J Med Chem 38: 739-44 (1995)
BindingDB Entry DOI: 10.7270/Q2W66JTD |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50036481

(2,6-Dichloro-3-[(2-dimethylamino-ethyl)-methyl-sul...)Show SMILES COc1cc2c(C(=O)N(COC(=O)c3c(Cl)ccc(c3Cl)S(=O)(=O)N(C)CCN(C)C)S2(=O)=O)c(c1)C(C)C Show InChI InChI=1S/C24H29Cl2N3O8S2/c1-14(2)16-11-15(36-6)12-19-20(16)23(30)29(39(19,34)35)13-37-24(31)21-17(25)7-8-18(22(21)26)38(32,33)28(5)10-9-27(3)4/h7-8,11-12,14H,9-10,13H2,1-6H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sterling Winthrop Inc.
Curated by ChEMBL
| Assay Description
Potency of inhibition against human leukocyte elastase (HLE) expressed as an apparent binding constant |
J Med Chem 38: 739-44 (1995)
BindingDB Entry DOI: 10.7270/Q2W66JTD |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50036477

(2,6-Dichloro-3-(2-morpholin-4-yl-ethoxy)-benzoic a...)Show SMILES COc1cc2c(C(=O)N(COC(=O)c3c(Cl)ccc(OCCN4CCOCC4)c3Cl)S2(=O)=O)c(c1)C(C)C Show InChI InChI=1S/C25H28Cl2N2O8S/c1-15(2)17-12-16(34-3)13-20-21(17)24(30)29(38(20,32)33)14-37-25(31)22-18(26)4-5-19(23(22)27)36-11-8-28-6-9-35-10-7-28/h4-5,12-13,15H,6-11,14H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sterling Winthrop Inc.
Curated by ChEMBL
| Assay Description
Potency of inhibition against human leukocyte elastase (HLE) expressed as an apparent binding constant |
J Med Chem 38: 739-44 (1995)
BindingDB Entry DOI: 10.7270/Q2W66JTD |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50036475

(2,6-Dichloro-3-(2-pyrrolidin-1-yl-ethoxy)-benzoic ...)Show SMILES COc1cc2c(C(=O)N(COC(=O)c3c(Cl)ccc(OCCN4CCCC4)c3Cl)S2(=O)=O)c(c1)C(C)C Show InChI InChI=1S/C25H28Cl2N2O7S/c1-15(2)17-12-16(34-3)13-20-21(17)24(30)29(37(20,32)33)14-36-25(31)22-18(26)6-7-19(23(22)27)35-11-10-28-8-4-5-9-28/h6-7,12-13,15H,4-5,8-11,14H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sterling Winthrop Inc.
Curated by ChEMBL
| Assay Description
Potency of inhibition against human leukocyte elastase (HLE) expressed as an apparent binding constant |
J Med Chem 38: 739-44 (1995)
BindingDB Entry DOI: 10.7270/Q2W66JTD |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50029699
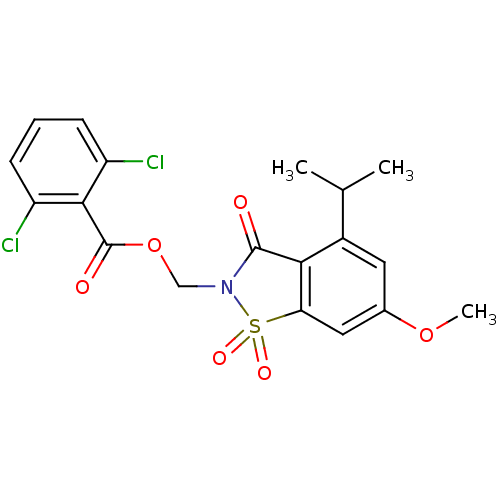
(2,6-Dichloro-benzoic acid 4-isopropyl-6-methoxy-1,...)Show SMILES COc1cc2c(C(=O)N(COC(=O)c3c(Cl)cccc3Cl)S2(=O)=O)c(c1)C(C)C Show InChI InChI=1S/C19H17Cl2NO6S/c1-10(2)12-7-11(27-3)8-15-16(12)18(23)22(29(15,25)26)9-28-19(24)17-13(20)5-4-6-14(17)21/h4-8,10H,9H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Evaluated for inhibitory activity against Human leukocyte elastase (HLE) |
Bioorg Med Chem Lett 5: 331-336 (1995)
Article DOI: 10.1016/0960-894X(95)00030-W
BindingDB Entry DOI: 10.7270/Q2SX6D60 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50112518
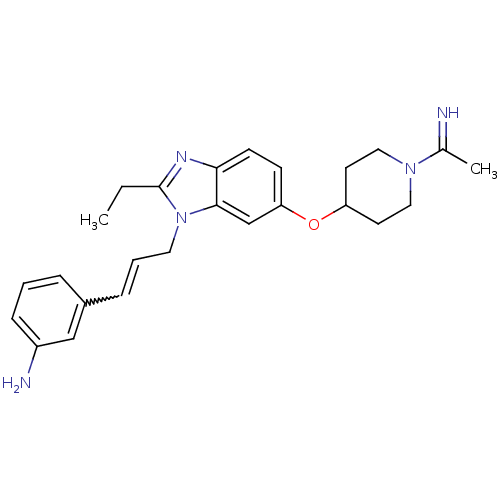
(3-(3-{2-Ethyl-6-[1-(1-imino-ethyl)-piperidin-4-ylo...)Show SMILES CCc1nc2ccc(OC3CCN(CC3)C(C)=N)cc2n1CC=Cc1cccc(N)c1 |w:23.26| Show InChI InChI=1S/C25H31N5O/c1-3-25-28-23-10-9-22(31-21-11-14-29(15-12-21)18(2)26)17-24(23)30(25)13-5-7-19-6-4-8-20(27)16-19/h4-10,16-17,21,26H,3,11-15,27H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Tested in vitro for the inhibitory potency against Coagulation factor Xa |
Bioorg Med Chem Lett 12: 1311-4 (2002)
BindingDB Entry DOI: 10.7270/Q2X066CP |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50286326
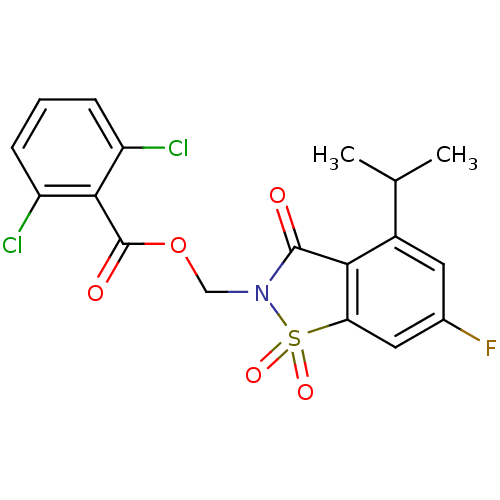
(2,6-Dichloro-benzoic acid 6-fluoro-4-isopropyl-1,1...)Show SMILES CC(C)c1cc(F)cc2c1C(=O)N(COC(=O)c1c(Cl)cccc1Cl)S2(=O)=O Show InChI InChI=1S/C18H14Cl2FNO5S/c1-9(2)11-6-10(21)7-14-15(11)17(23)22(28(14,25)26)8-27-18(24)16-12(19)4-3-5-13(16)20/h3-7,9H,8H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Evaluated for inhibitory activity against Human leukocyte elastase (HLE) |
Bioorg Med Chem Lett 5: 331-336 (1995)
Article DOI: 10.1016/0960-894X(95)00030-W
BindingDB Entry DOI: 10.7270/Q2SX6D60 |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50039631
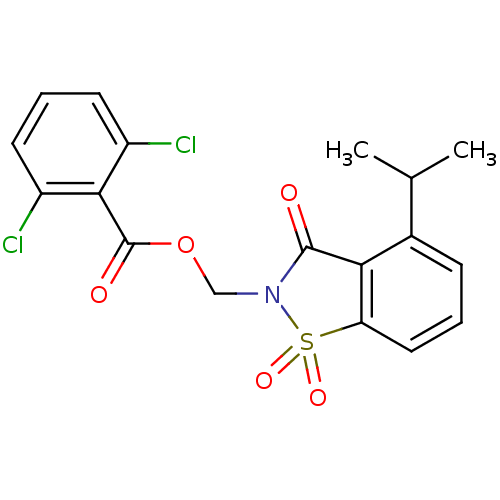
(2,6-Dichloro-benzoic acid 4-isopropyl-1,1,3-trioxo...)Show SMILES CC(C)c1cccc2c1C(=O)N(COC(=O)c1c(Cl)cccc1Cl)S2(=O)=O Show InChI InChI=1S/C18H15Cl2NO5S/c1-10(2)11-5-3-8-14-15(11)17(22)21(27(14,24)25)9-26-18(23)16-12(19)6-4-7-13(16)20/h3-8,10H,9H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Evaluated for inhibitory activity against Human leukocyte elastase (HLE) |
Bioorg Med Chem Lett 5: 331-336 (1995)
Article DOI: 10.1016/0960-894X(95)00030-W
BindingDB Entry DOI: 10.7270/Q2SX6D60 |
More data for this
Ligand-Target Pair | |
Sigma non-opioid intracellular receptor 1
(Cavia porcellus (Guinea pig)) | BDBM50179280
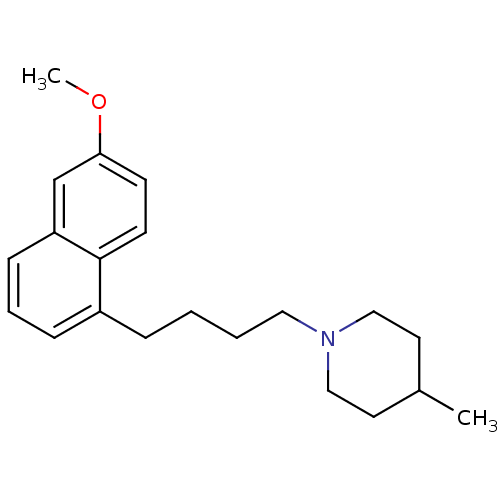
(1-(4-(6-methoxynaphthalen-1-yl)butyl)-4-methylpipe...)Show InChI InChI=1S/C21H29NO/c1-17-11-14-22(15-12-17)13-4-3-6-18-7-5-8-19-16-20(23-2)9-10-21(18)19/h5,7-10,16-17H,3-4,6,11-15H2,1-2H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ Degli Studi di Bari ALDO MORO
Curated by ChEMBL
| Assay Description
Displacement of [3H]-pentazocine from sigma 1 receptor in guinea pig brain membrane after 120 mins |
Eur J Med Chem 108: 577-85 (2016)
Article DOI: 10.1016/j.ejmech.2015.12.014
BindingDB Entry DOI: 10.7270/Q22Z17C3 |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50105327
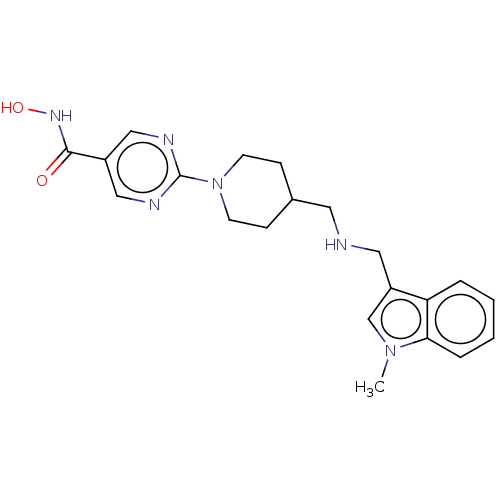
(JNJ-26481585 | Quisinostat)Show SMILES Cn1cc(CNCC2CCN(CC2)c2ncc(cn2)C(=O)NO)c2ccccc12 Show InChI InChI=1S/C21H26N6O2/c1-26-14-17(18-4-2-3-5-19(18)26)11-22-10-15-6-8-27(9-7-15)21-23-12-16(13-24-21)20(28)25-29/h2-5,12-15,22,29H,6-11H2,1H3,(H,25,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St Jude Children's Research Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human HDAC1 |
Bioorg Med Chem 23: 5151-5 (2015)
Article DOI: 10.1016/j.bmc.2014.12.066
BindingDB Entry DOI: 10.7270/Q2B859V9 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1B
(Homo sapiens (Human)) | BDBM81497
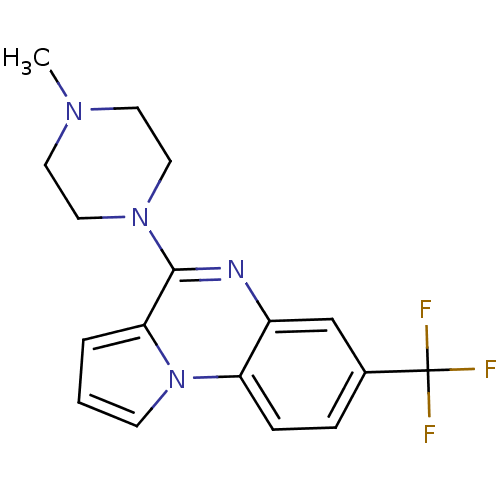
(4-(4-Methyl-piperazin-1-yl)-7-trifluoromethyl-pyrr...)Show InChI InChI=1S/C17H17F3N4/c1-22-7-9-23(10-8-22)16-15-3-2-6-24(15)14-5-4-12(17(18,19)20)11-13(14)21-16/h2-6,11H,7-10H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Veterans Affairs Medical Center
Curated by PDSP Ki Database
| |
Biochem Biophys Res Commun 184: 752-9 (1992)
Article DOI: 10.1016/0006-291x(92)90654-4
BindingDB Entry DOI: 10.7270/Q2ST7NB1 |
More data for this
Ligand-Target Pair | |
Polyamine deacetylase HDAC10
(Homo sapiens (Human)) | BDBM50105327

(JNJ-26481585 | Quisinostat)Show SMILES Cn1cc(CNCC2CCN(CC2)c2ncc(cn2)C(=O)NO)c2ccccc12 Show InChI InChI=1S/C21H26N6O2/c1-26-14-17(18-4-2-3-5-19(18)26)11-22-10-15-6-8-27(9-7-15)21-23-12-16(13-24-21)20(28)25-29/h2-5,12-15,22,29H,6-11H2,1H3,(H,25,28) | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.0500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St Jude Children's Research Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human HDAC10 |
Bioorg Med Chem 23: 5151-5 (2015)
Article DOI: 10.1016/j.bmc.2014.12.066
BindingDB Entry DOI: 10.7270/Q2B859V9 |
More data for this
Ligand-Target Pair | |
Histone deacetylase 2
(Homo sapiens (Human)) | BDBM50105327

(JNJ-26481585 | Quisinostat)Show SMILES Cn1cc(CNCC2CCN(CC2)c2ncc(cn2)C(=O)NO)c2ccccc12 Show InChI InChI=1S/C21H26N6O2/c1-26-14-17(18-4-2-3-5-19(18)26)11-22-10-15-6-8-27(9-7-15)21-23-12-16(13-24-21)20(28)25-29/h2-5,12-15,22,29H,6-11H2,1H3,(H,25,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.0600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St Jude Children's Research Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human HDAC2 |
Bioorg Med Chem 23: 5151-5 (2015)
Article DOI: 10.1016/j.bmc.2014.12.066
BindingDB Entry DOI: 10.7270/Q2B859V9 |
More data for this
Ligand-Target Pair | |
Histone deacetylase 11
(Homo sapiens (Human)) | BDBM50105327

(JNJ-26481585 | Quisinostat)Show SMILES Cn1cc(CNCC2CCN(CC2)c2ncc(cn2)C(=O)NO)c2ccccc12 Show InChI InChI=1S/C21H26N6O2/c1-26-14-17(18-4-2-3-5-19(18)26)11-22-10-15-6-8-27(9-7-15)21-23-12-16(13-24-21)20(28)25-29/h2-5,12-15,22,29H,6-11H2,1H3,(H,25,28) | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St Jude Children's Research Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human HDAC11 |
Bioorg Med Chem 23: 5151-5 (2015)
Article DOI: 10.1016/j.bmc.2014.12.066
BindingDB Entry DOI: 10.7270/Q2B859V9 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50112502
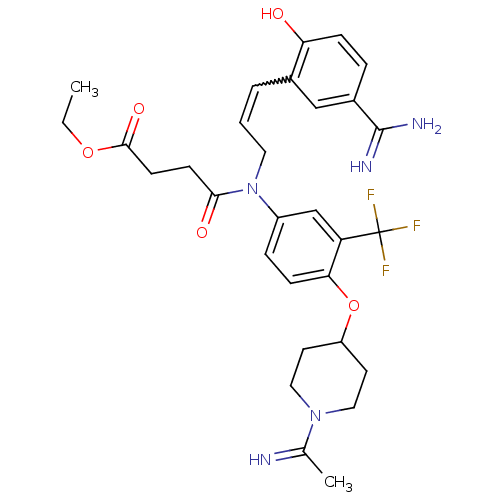
(CHEMBL26299 | N-[3-(5-Carbamimidoyl-2-hydroxy-phen...)Show SMILES CCOC(=O)CCC(=O)N(CC=Cc1cc(ccc1O)C(N)=N)c1ccc(OC2CCN(CC2)C(C)=N)c(c1)C(F)(F)F |w:12.12| Show InChI InChI=1S/C30H36F3N5O5/c1-3-42-28(41)11-10-27(40)38(14-4-5-20-17-21(29(35)36)6-8-25(20)39)22-7-9-26(24(18-22)30(31,32)33)43-23-12-15-37(16-13-23)19(2)34/h4-9,17-18,23,34,39H,3,10-16H2,1-2H3,(H3,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Competitive inhibition of coagulation factor Xa |
Bioorg Med Chem Lett 12: 1307-10 (2002)
BindingDB Entry DOI: 10.7270/Q21R6PV8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50112506
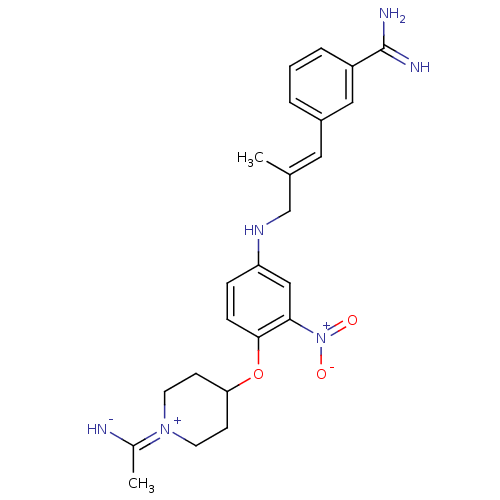
(3-(3-{4-[1-(1-Imino-ethyl)-piperidin-4-yloxy]-3-ni...)Show SMILES C\C(CNc1ccc(OC2CC[N+](CC2)=C(C)[NH-])c(c1)[N+]([O-])=O)=C/c1cccc(c1)C(N)=N |(8.92,-4.79,;7.58,-5.56,;6.23,-4.79,;6.23,-3.25,;4.91,-2.48,;3.57,-3.25,;2.24,-2.48,;2.24,-.94,;.91,-.17,;-.44,-.94,;-1.76,-.16,;-3.09,-.92,;-3.1,-2.46,;-1.76,-3.24,;-.44,-2.47,;-4.43,-3.23,;-5.78,-2.46,;-4.45,-4.77,;3.57,-.17,;4.91,-.94,;3.55,1.37,;4.9,2.14,;2.22,2.14,;7.56,-7.1,;8.89,-7.87,;8.91,-9.41,;10.25,-10.18,;11.58,-9.4,;11.57,-7.84,;10.22,-7.09,;12.9,-7.07,;12.88,-5.53,;14.23,-7.82,)| Show InChI InChI=1S/C24H30N6O3/c1-16(12-18-4-3-5-19(13-18)24(26)27)15-28-20-6-7-23(22(14-20)30(31)32)33-21-8-10-29(11-9-21)17(2)25/h3-7,12-14,21,25,28H,8-11,15H2,1-2H3,(H3,26,27)/b16-12+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Competitive inhibition of coagulation factor Xa |
Bioorg Med Chem Lett 12: 1307-10 (2002)
BindingDB Entry DOI: 10.7270/Q21R6PV8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50112503

(7-({4-[1-(1-Imino-ethyl)-piperidin-4-yloxy]-3-nitr...)Show SMILES CC([NH-])=[N+]1CCC(CC1)Oc1ccc(NCc2ccc3ccc(cc3c2)C(N)=N)cc1[N+]([O-])=O |(-2.07,-4.18,;-.74,-4.97,;-.74,-6.51,;.59,-4.2,;.61,-2.66,;1.94,-1.9,;3.27,-2.68,;3.27,-4.21,;1.94,-4.98,;4.6,-1.91,;5.93,-2.68,;5.93,-4.22,;7.26,-4.99,;8.61,-4.22,;9.95,-4.99,;9.95,-6.53,;11.28,-7.29,;11.28,-8.83,;12.61,-9.6,;13.94,-8.83,;15.28,-9.59,;16.62,-8.8,;16.6,-7.26,;15.27,-6.49,;13.94,-7.28,;12.61,-6.52,;17.93,-6.47,;17.92,-4.93,;19.27,-7.24,;8.61,-2.67,;7.26,-1.9,;7.26,-.36,;8.59,.41,;5.93,.4,)| Show InChI InChI=1S/C25H28N6O3/c1-16(26)30-10-8-22(9-11-30)34-24-7-6-21(14-23(24)31(32)33)29-15-17-2-3-18-4-5-19(25(27)28)13-20(18)12-17/h2-7,12-14,22,26,29H,8-11,15H2,1H3,(H3,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.0800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Competitive inhibition of coagulation factor Xa |
Bioorg Med Chem Lett 12: 1307-10 (2002)
BindingDB Entry DOI: 10.7270/Q21R6PV8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50112513
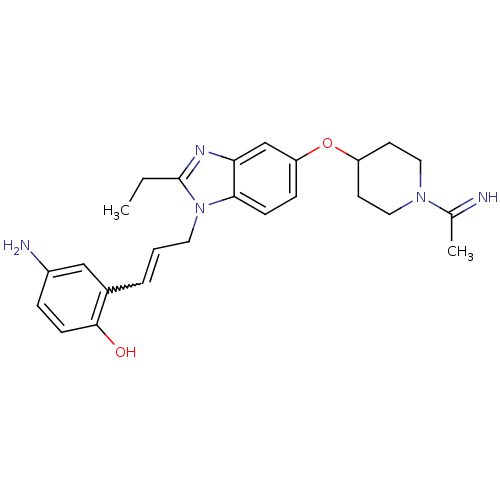
(4-Amino-2-(3-{2-ethyl-5-[1-(1-imino-ethyl)-piperid...)Show SMILES CCc1nc2cc(OC3CCN(CC3)C(C)=N)ccc2n1CC=Cc1cc(N)ccc1O |w:23.26| Show InChI InChI=1S/C25H31N5O2/c1-3-25-28-22-16-21(32-20-10-13-29(14-11-20)17(2)26)7-8-23(22)30(25)12-4-5-18-15-19(27)6-9-24(18)31/h4-9,15-16,20,26,31H,3,10-14,27H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Tested in vitro for the inhibitory potency against Coagulation factor Xa |
Bioorg Med Chem Lett 12: 1311-4 (2002)
BindingDB Entry DOI: 10.7270/Q2X066CP |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50112491
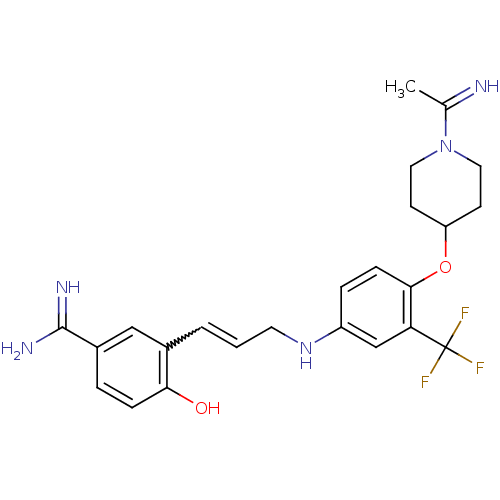
(4-Hydroxy-3-(3-{4-[1-(1-imino-ethyl)-piperidin-4-y...)Show SMILES CC(=N)N1CCC(CC1)Oc1ccc(NCC=Cc2cc(ccc2O)C(N)=N)cc1C(F)(F)F |w:17.18| Show InChI InChI=1S/C24H28F3N5O2/c1-15(28)32-11-8-19(9-12-32)34-22-7-5-18(14-20(22)24(25,26)27)31-10-2-3-16-13-17(23(29)30)4-6-21(16)33/h2-7,13-14,19,28,31,33H,8-12H2,1H3,(H3,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Competitive inhibition of coagulation factor Xa |
Bioorg Med Chem Lett 12: 1307-10 (2002)
BindingDB Entry DOI: 10.7270/Q21R6PV8 |
More data for this
Ligand-Target Pair | |
Histone deacetylase 2
(Homo sapiens (Human)) | BDBM50123957
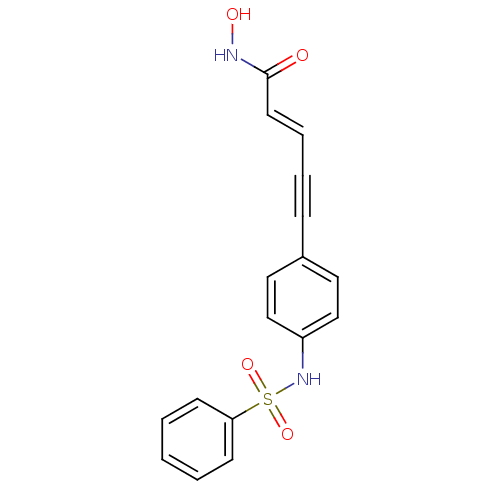
((E)-5-(3-Benzenesulfonylamino-phenyl)-pent-2-en-4-...)Show SMILES ONC(=O)\C=C\C#Cc1ccc(NS(=O)(=O)c2ccccc2)cc1 Show InChI InChI=1S/C17H14N2O4S/c20-17(18-21)9-5-4-6-14-10-12-15(13-11-14)19-24(22,23)16-7-2-1-3-8-16/h1-3,5,7-13,19,21H,(H,18,20)/b9-5+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St Jude Children's Research Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human HDAC2 |
Bioorg Med Chem 23: 5151-5 (2015)
Article DOI: 10.1016/j.bmc.2014.12.066
BindingDB Entry DOI: 10.7270/Q2B859V9 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50112497

(CHEMBL26240 | N-[3-(5-Carbamimidoyl-2-hydroxy-phen...)Show SMILES CC(=N)N1CCC(CC1)Oc1ccc(cc1C(F)(F)F)N(CC=Cc1cc(ccc1O)C(N)=N)C(=O)CCC(O)=O |w:23.25| Show InChI InChI=1S/C28H32F3N5O5/c1-17(32)35-13-10-21(11-14-35)41-24-7-5-20(16-22(24)28(29,30)31)36(25(38)8-9-26(39)40)12-2-3-18-15-19(27(33)34)4-6-23(18)37/h2-7,15-16,21,32,37H,8-14H2,1H3,(H3,33,34)(H,39,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Competitive inhibition of coagulation factor Xa |
Bioorg Med Chem Lett 12: 1307-10 (2002)
BindingDB Entry DOI: 10.7270/Q21R6PV8 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50112524
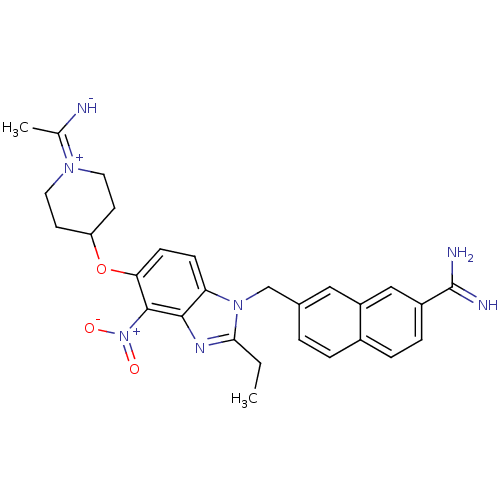
(7-{2-Ethyl-5-[1-(1-imino-ethyl)-piperidin-4-yloxy]...)Show SMILES CCc1nc2c(c(OC3CCN(CC3)C(C)=N)ccc2n1Cc1ccc2ccc(cc2c1)C(N)=N)[N+]([O-])=O Show InChI InChI=1S/C28H31N7O3/c1-3-25-32-26-23(34(25)16-18-4-5-19-6-7-20(28(30)31)15-21(19)14-18)8-9-24(27(26)35(36)37)38-22-10-12-33(13-11-22)17(2)29/h4-9,14-15,22,29H,3,10-13,16H2,1-2H3,(H3,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Tested in vitro for the inhibitory potency against Coagulation factor Xa |
Bioorg Med Chem Lett 12: 1311-4 (2002)
BindingDB Entry DOI: 10.7270/Q2X066CP |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50112521
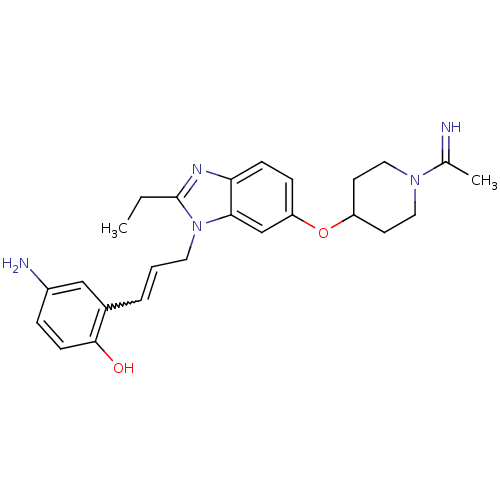
(4-Amino-2-(3-{2-ethyl-6-[1-(1-imino-ethyl)-piperid...)Show SMILES CCc1nc2ccc(OC3CCN(CC3)C(C)=N)cc2n1CC=Cc1cc(N)ccc1O |w:23.26| Show InChI InChI=1S/C25H31N5O2/c1-3-25-28-22-8-7-21(32-20-10-13-29(14-11-20)17(2)26)16-23(22)30(25)12-4-5-18-15-19(27)6-9-24(18)31/h4-9,15-16,20,26,31H,3,10-14,27H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Tested in vitro for the inhibitory potency against Coagulation factor Xa |
Bioorg Med Chem Lett 12: 1311-4 (2002)
BindingDB Entry DOI: 10.7270/Q2X066CP |
More data for this
Ligand-Target Pair | |
Histone deacetylase 8
(Homo sapiens (Human)) | BDBM50123957

((E)-5-(3-Benzenesulfonylamino-phenyl)-pent-2-en-4-...)Show SMILES ONC(=O)\C=C\C#Cc1ccc(NS(=O)(=O)c2ccccc2)cc1 Show InChI InChI=1S/C17H14N2O4S/c20-17(18-21)9-5-4-6-14-10-12-15(13-11-14)19-24(22,23)16-7-2-1-3-8-16/h1-3,5,7-13,19,21H,(H,18,20)/b9-5+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St Jude Children's Research Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human HDAC8 |
Bioorg Med Chem 23: 5151-5 (2015)
Article DOI: 10.1016/j.bmc.2014.12.066
BindingDB Entry DOI: 10.7270/Q2B859V9 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM17280

(1-[2-(5-carbamimidoyl-2-hydroxyphenoxy)-3,5-difluo...)Show SMILES CN1CCN=C1c1cccc(Oc2nc(Oc3cc(ccc3O)C(N)=N)c(F)c(N3CCCC(C3)C(O)=O)c2F)c1 |c:4| Show InChI InChI=1S/C28H28F2N6O5/c1-35-11-9-33-25(35)16-4-2-6-18(12-16)40-26-21(29)23(36-10-3-5-17(14-36)28(38)39)22(30)27(34-26)41-20-13-15(24(31)32)7-8-19(20)37/h2,4,6-8,12-13,17,37H,3,5,9-11,14H2,1H3,(H3,31,32)(H,38,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.110 | -56.3 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Berlex
| Assay Description
The enzyme activities were determined kinetically as the initial rate of cleavage of a peptide p-nitroanilide. Km for enzyme and substrate was determ... |
Acta Crystallogr D Biol Crystallogr 55: 1395-404 (1999)
Article DOI: 10.1107/s0907444999007350
BindingDB Entry DOI: 10.7270/Q2H1308B |
More data for this
Ligand-Target Pair | |
Sigma non-opioid intracellular receptor 1
(Cavia porcellus (Guinea pig)) | BDBM50299711
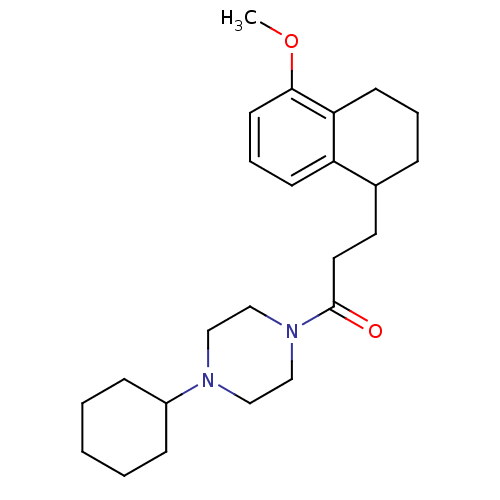
(4-Cyclohexyl-1-[3-(5-methoxy-1,2,3,4-tetrahydronap...)Show SMILES COc1cccc2C(CCC(=O)N3CCN(CC3)C3CCCCC3)CCCc12 Show InChI InChI=1S/C24H36N2O2/c1-28-23-12-6-10-21-19(7-5-11-22(21)23)13-14-24(27)26-17-15-25(16-18-26)20-8-3-2-4-9-20/h6,10,12,19-20H,2-5,7-9,11,13-18H2,1H3 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ Degli Studi di Bari ALDO MORO
Curated by ChEMBL
| Assay Description
Displacement of [3H]-pentazocine from sigma 1 receptor in guinea pig brain membrane after 120 mins |
Eur J Med Chem 108: 577-85 (2016)
Article DOI: 10.1016/j.ejmech.2015.12.014
BindingDB Entry DOI: 10.7270/Q22Z17C3 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM17284
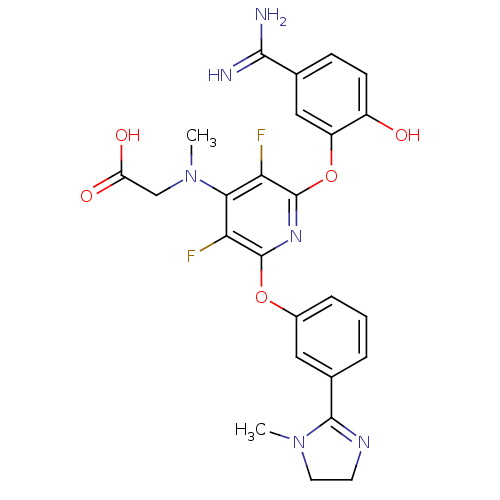
(2-{[2-(5-carbamimidoyl-2-hydroxyphenoxy)-3,5-diflu...)Show SMILES CN(CC(O)=O)c1c(F)c(Oc2cccc(c2)C2=NCCN2C)nc(Oc2cc(ccc2O)C(N)=N)c1F |t:18| Show InChI InChI=1S/C25H24F2N6O5/c1-32-9-8-30-23(32)14-4-3-5-15(10-14)37-24-19(26)21(33(2)12-18(35)36)20(27)25(31-24)38-17-11-13(22(28)29)6-7-16(17)34/h3-7,10-11,34H,8-9,12H2,1-2H3,(H3,28,29)(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
Article
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Inhibitory potency was measured against human coagulation factor X |
J Med Chem 41: 3557-62 (1998)
Article DOI: 10.1021/jm980280h
BindingDB Entry DOI: 10.7270/Q2KS6QPR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Coagulation factor X
(Homo sapiens (Human)) | BDBM17284

(2-{[2-(5-carbamimidoyl-2-hydroxyphenoxy)-3,5-diflu...)Show SMILES CN(CC(O)=O)c1c(F)c(Oc2cccc(c2)C2=NCCN2C)nc(Oc2cc(ccc2O)C(N)=N)c1F |t:18| Show InChI InChI=1S/C25H24F2N6O5/c1-32-9-8-30-23(32)14-4-3-5-15(10-14)37-24-19(26)21(33(2)12-18(35)36)20(27)25(31-24)38-17-11-13(22(28)29)6-7-16(17)34/h3-7,10-11,34H,8-9,12H2,1-2H3,(H3,28,29)(H,35,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| MMDB
PDB
PubMed
| 0.110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
In vitro inhibition of human coagulation factor Xa (Xa) in a purified enzyme system. |
J Med Chem 42: 5415-25 (2000)
BindingDB Entry DOI: 10.7270/Q2TT4Q5S |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Melatonin receptor type 1B
(Homo sapiens (Human)) | BDBM9019
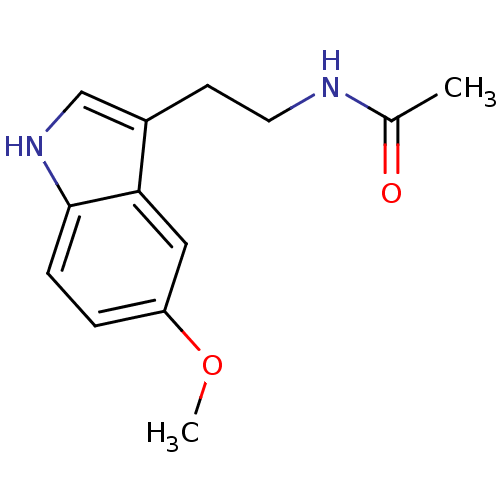
(CHEMBL45 | Melatonin | N-[2-(5-methoxy-1H-indol-3-...)Show InChI InChI=1S/C13H16N2O2/c1-9(16)14-6-5-10-8-15-13-4-3-11(17-2)7-12(10)13/h3-4,7-8,15H,5-6H2,1-2H3,(H,14,16) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of 2-[1251]-iodomelatonin from human MT2 receptor expressed in CHO cells incubated for 120 mins by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00627
BindingDB Entry DOI: 10.7270/Q2Z03CZJ |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50112500
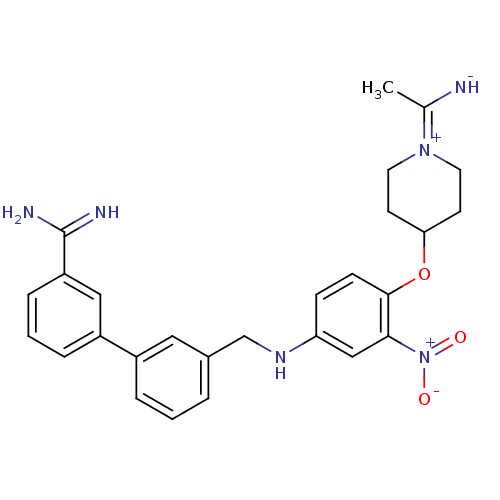
(3'-({4-[1-(1-Imino-ethyl)-piperidin-4-yloxy]-3-nit...)Show SMILES CC([NH-])=[N+]1CCC(CC1)Oc1ccc(NCc2cccc(c2)-c2cccc(c2)C(N)=N)cc1[N+]([O-])=O |(-4.64,-1.92,;-3.23,-2.6,;-3.08,-4.13,;-2,-1.75,;-2.14,-.22,;-.91,.63,;.5,-.06,;.64,-1.56,;-.61,-2.43,;1.73,.79,;3.12,.12,;3.29,-1.4,;4.68,-2.08,;5.93,-1.21,;7.32,-1.89,;7.48,-3.42,;8.87,-4.09,;10.1,-3.21,;11.49,-3.88,;11.66,-5.42,;10.42,-6.28,;9.03,-5.61,;10.59,-7.79,;9.35,-8.66,;9.51,-10.19,;10.9,-10.86,;12.14,-10,;11.98,-8.47,;13.52,-10.71,;14.79,-9.91,;13.61,-12.25,;5.76,.32,;4.36,.98,;4.2,2.52,;5.43,3.37,;2.81,3.18,)| Show InChI InChI=1S/C27H30N6O3/c1-18(28)32-12-10-24(11-13-32)36-26-9-8-23(16-25(26)33(34)35)31-17-19-4-2-5-20(14-19)21-6-3-7-22(15-21)27(29)30/h2-9,14-16,24,28,31H,10-13,17H2,1H3,(H3,29,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Competitive inhibition of coagulation factor Xa |
Bioorg Med Chem Lett 12: 1307-10 (2002)
BindingDB Entry DOI: 10.7270/Q21R6PV8 |
More data for this
Ligand-Target Pair | |
Melatonin receptor type 1A
(Homo sapiens (Human)) | BDBM9019

(CHEMBL45 | Melatonin | N-[2-(5-methoxy-1H-indol-3-...)Show InChI InChI=1S/C13H16N2O2/c1-9(16)14-6-5-10-8-15-13-4-3-11(17-2)7-12(10)13/h3-4,7-8,15H,5-6H2,1-2H3,(H,14,16) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of 2-[1251]-iodomelatonin from human MT1 receptor expressed in CHO cells incubated for 60 mins by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00627
BindingDB Entry DOI: 10.7270/Q2Z03CZJ |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 4
(GUINEA PIG) | BDBM50181844
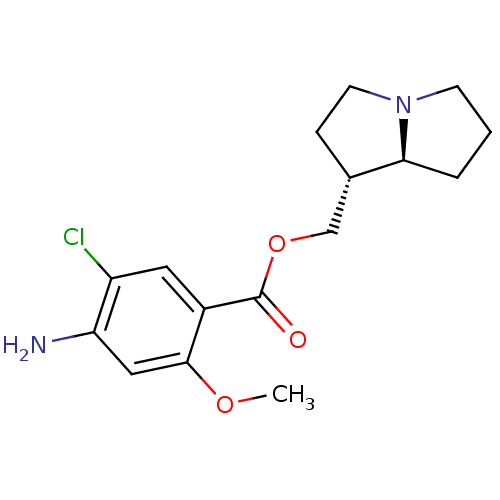
((1R,7aS)-hexahydro-1H-pyrrolizin-1-ylmethyl 4-amin...)Show SMILES COc1cc(N)c(Cl)cc1C(=O)OC[C@@H]1CCN2CCC[C@@H]12 Show InChI InChI=1S/C16H21ClN2O3/c1-21-15-8-13(18)12(17)7-11(15)16(20)22-9-10-4-6-19-5-2-3-14(10)19/h7-8,10,14H,2-6,9,18H2,1H3/t10-,14-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.183 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]GR-113808 from 5HT4 receptor in guinea pig striatum |
J Med Chem 49: 1125-39 (2006)
Article DOI: 10.1021/jm0509501
BindingDB Entry DOI: 10.7270/Q2W096Q0 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 4
(GUINEA PIG) | BDBM50181840
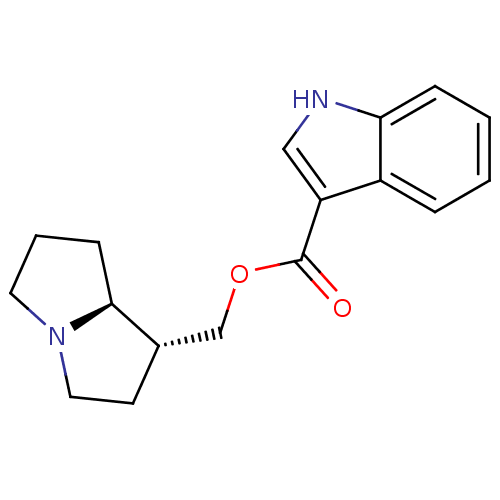
((1R,7aS)-hexahydro-1H-pyrrolizin-1-ylmethyl 1-Meth...)Show SMILES O=C(OC[C@@H]1CCN2CCC[C@@H]12)c1c[nH]c2ccccc12 Show InChI InChI=1S/C17H20N2O2/c20-17(14-10-18-15-5-2-1-4-13(14)15)21-11-12-7-9-19-8-3-6-16(12)19/h1-2,4-5,10,12,16,18H,3,6-9,11H2/t12-,16-/m0/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.183 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Displacement of [3H]GR-113808 from 5HT4 receptor in guinea pig striatum |
J Med Chem 49: 1125-39 (2006)
Article DOI: 10.1021/jm0509501
BindingDB Entry DOI: 10.7270/Q2W096Q0 |
More data for this
Ligand-Target Pair | |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM50072775
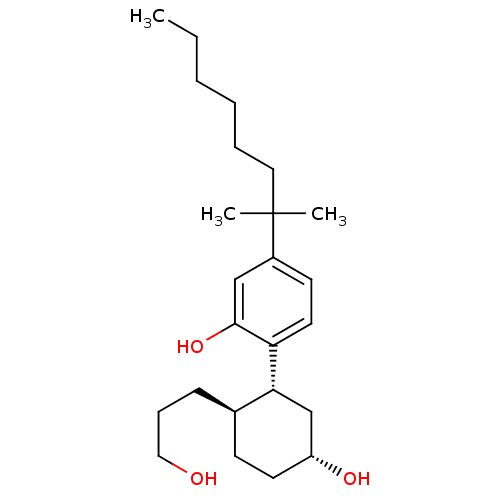
(2-((1R,2R,5R)-5-hydroxy-2-(3-hydroxypropyl)cyclohe...)Show SMILES CCCCCCC(C)(C)c1ccc([C@@H]2C[C@H](O)CC[C@H]2CCCO)c(O)c1 |r| Show InChI InChI=1S/C24H40O3/c1-4-5-6-7-14-24(2,3)19-11-13-21(23(27)16-19)22-17-20(26)12-10-18(22)9-8-15-25/h11,13,16,18,20,22,25-27H,4-10,12,14-15,17H2,1-3H3/t18-,20-,22-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
The University of Mississippi
Curated by ChEMBL
| Assay Description
Agonist activity at recombinant human full length CB2 receptor expressed in HEK293 cell membranes after 2 hrs by [35S]GTP-gammaS binding assay |
Eur J Med Chem 143: 983-996 (2018)
Article DOI: 10.1016/j.ejmech.2017.11.043
BindingDB Entry DOI: 10.7270/Q2D50QN8 |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Cannabinoid receptor 2
(Homo sapiens (Human)) | BDBM603932

((+)-trans-Ethyl 2-ethyl-2-{[6-({-2-[(fluoromethoxy...)Show SMILES CCOC(=O)C(CC)(CC)NC(=O)c1ccc(N2CC(C2)OC)c(OC[C@H]2C[C@@H]2COCF)n1 |r| | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
BindingDB Entry DOI: 10.7270/Q2K93CHD |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1B
(Homo sapiens (Human)) | BDBM78940
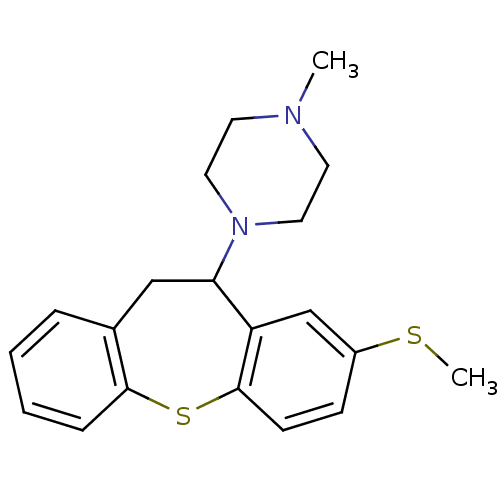
(METHIOTHEPIN | MLS000859918 | Methiothepin mesylat...)Show InChI InChI=1S/C20H24N2S2/c1-21-9-11-22(12-10-21)18-13-15-5-3-4-6-19(15)24-20-8-7-16(23-2)14-17(18)20/h3-8,14,18H,9-13H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Veterans Affairs Medical Center
Curated by PDSP Ki Database
| |
Biochem Biophys Res Commun 184: 752-9 (1992)
Article DOI: 10.1016/0006-291x(92)90654-4
BindingDB Entry DOI: 10.7270/Q2ST7NB1 |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50036482
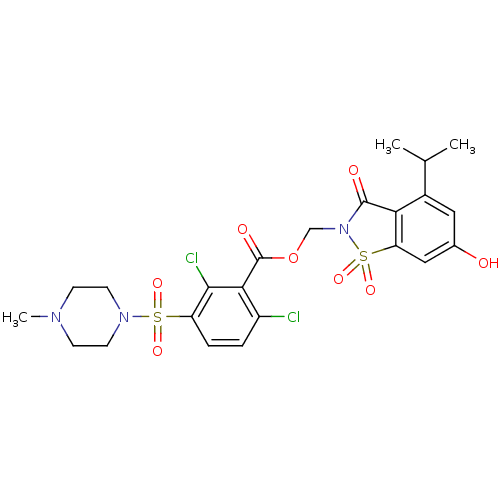
(2,6-Dichloro-3-(4-methyl-piperazine-1-sulfonyl)-be...)Show SMILES CC(C)c1cc(O)cc2c1C(=O)N(COC(=O)c1c(Cl)ccc(c1Cl)S(=O)(=O)N1CCN(C)CC1)S2(=O)=O Show InChI InChI=1S/C23H25Cl2N3O8S2/c1-13(2)15-10-14(29)11-18-19(15)22(30)28(38(18,34)35)12-36-23(31)20-16(24)4-5-17(21(20)25)37(32,33)27-8-6-26(3)7-9-27/h4-5,10-11,13,29H,6-9,12H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Sterling Winthrop Inc.
Curated by ChEMBL
| Assay Description
Potency of inhibition against human leukocyte elastase (HLE) expressed as an apparent binding constant |
J Med Chem 38: 739-44 (1995)
BindingDB Entry DOI: 10.7270/Q2W66JTD |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50112516
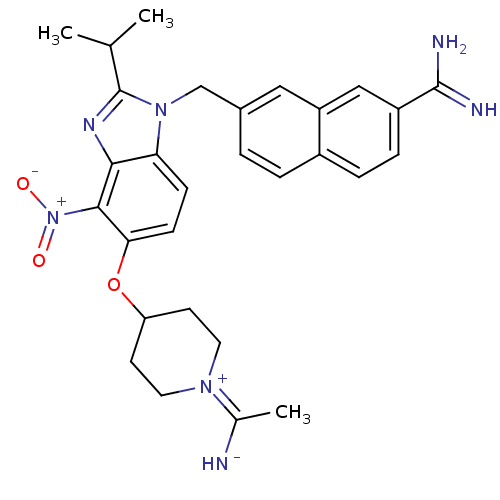
(7-{5-[1-(1-Imino-ethyl)-piperidin-4-yloxy]-2-isopr...)Show SMILES CC(C)c1nc2c(c(OC3CCN(CC3)C(C)=N)ccc2n1Cc1ccc2ccc(cc2c1)C(N)=N)[N+]([O-])=O Show InChI InChI=1S/C29H33N7O3/c1-17(2)29-33-26-24(35(29)16-19-4-5-20-6-7-21(28(31)32)15-22(20)14-19)8-9-25(27(26)36(37)38)39-23-10-12-34(13-11-23)18(3)30/h4-9,14-15,17,23,30H,10-13,16H2,1-3H3,(H3,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Tested in vitro for the inhibitory potency against Coagulation factor Xa |
Bioorg Med Chem Lett 12: 1311-4 (2002)
BindingDB Entry DOI: 10.7270/Q2X066CP |
More data for this
Ligand-Target Pair | |
Histone deacetylase 1
(Homo sapiens (Human)) | BDBM50005711
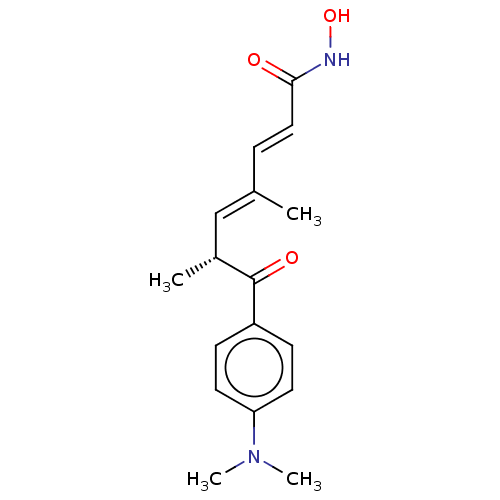
(CHEBI:46024 | GNF-Pf-1011 | TRICHOSTATIN | Trichos...)Show SMILES C[C@H](\C=C(/C)\C=C\C(=O)NO)C(=O)c1ccc(cc1)N(C)C |r| Show InChI InChI=1S/C17H22N2O3/c1-12(5-10-16(20)18-22)11-13(2)17(21)14-6-8-15(9-7-14)19(3)4/h5-11,13,22H,1-4H3,(H,18,20)/b10-5+,12-11+/t13-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
St Jude Children's Research Hospital
Curated by ChEMBL
| Assay Description
Inhibition of human HDAC1 |
Bioorg Med Chem 23: 5151-5 (2015)
Article DOI: 10.1016/j.bmc.2014.12.066
BindingDB Entry DOI: 10.7270/Q2B859V9 |
More data for this
Ligand-Target Pair | |
Neutrophil elastase
(Homo sapiens (Human)) | BDBM50286333
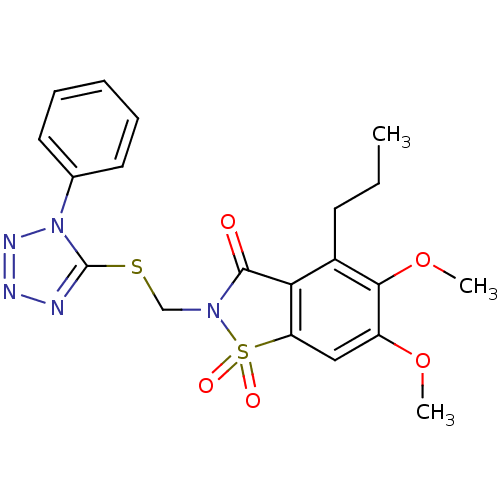
(5,6-Dimethoxy-1,1-dioxo-2-(1-phenyl-1H-tetrazol-5-...)Show SMILES CCCc1c2C(=O)N(CSc3nnnn3-c3ccccc3)S(=O)(=O)c2cc(OC)c1OC Show InChI InChI=1S/C20H21N5O5S2/c1-4-8-14-17-16(11-15(29-2)18(14)30-3)32(27,28)24(19(17)26)12-31-20-21-22-23-25(20)13-9-6-5-7-10-13/h5-7,9-11H,4,8,12H2,1-3H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Evaluated for inhibitory activity against Human leukocyte elastase (HLE) |
Bioorg Med Chem Lett 5: 331-336 (1995)
Article DOI: 10.1016/0960-894X(95)00030-W
BindingDB Entry DOI: 10.7270/Q2SX6D60 |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 3A
(RAT) | BDBM50056419

(4-Amino-N-(1-aza-bicyclo[2.2.2]oct-3-yl)-5-chloro-...)Show SMILES COc1cc(N)c(Cl)cc1C(=O)N[C@@H]1CN2CCC1CC2 |wD:13.13,TLB:12:13:17.16:19.20,(11.11,-14.18,;11.11,-12.63,;9.77,-11.88,;8.43,-12.66,;7.09,-11.89,;5.75,-12.66,;7.09,-10.33,;5.76,-9.57,;8.43,-9.56,;9.75,-10.36,;11.09,-9.59,;11.11,-8.04,;12.44,-10.36,;13.77,-9.59,;14.8,-8.58,;16.63,-8.19,;17.94,-9.45,;16.9,-10.39,;15.57,-9.1,;15.88,-7.77,;16.88,-7.14,)| Show InChI InChI=1S/C15H20ClN3O2/c1-21-14-7-12(17)11(16)6-10(14)15(20)18-13-8-19-4-2-9(13)3-5-19/h6-7,9,13H,2-5,8,17H2,1H3,(H,18,20)/t13-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Compound was evaluated in vivo for the antagonistic activity towards 5-hydroxytryptamine 3 receptor |
Bioorg Med Chem Lett 2: 1613-1618 (1992)
Article DOI: 10.1016/S0960-894X(00)80441-2
BindingDB Entry DOI: 10.7270/Q2J67HFB |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 3A/3B
(Rattus norvegicus-RAT) | BDBM50368604
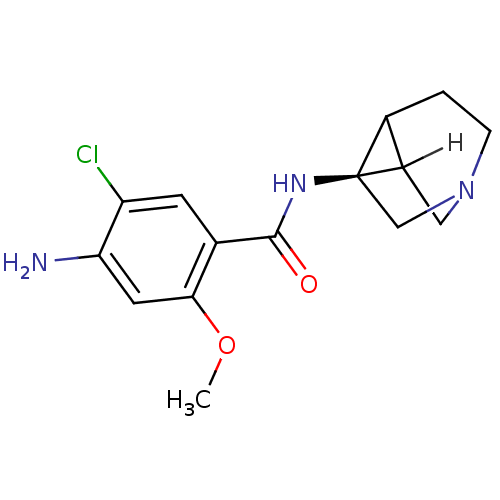
(CHEMBL1907770)Show SMILES [H]C12CN3C[C@]1(NC(=O)c1cc(Cl)c(N)cc1OC)C2CC3 |r| Show InChI InChI=1S/C15H18ClN3O2/c1-21-13-5-12(17)11(16)4-8(13)14(20)18-15-7-19-3-2-9(15)10(15)6-19/h4-5,9-10H,2-3,6-7,17H2,1H3,(H,18,20)/t9?,10?,15-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Searle Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of [3H]GR-65630 binding to 5-hydroxytryptamine 3 receptor |
J Med Chem 35: 1486-9 (1992)
BindingDB Entry DOI: 10.7270/Q2CC11B3 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50066635
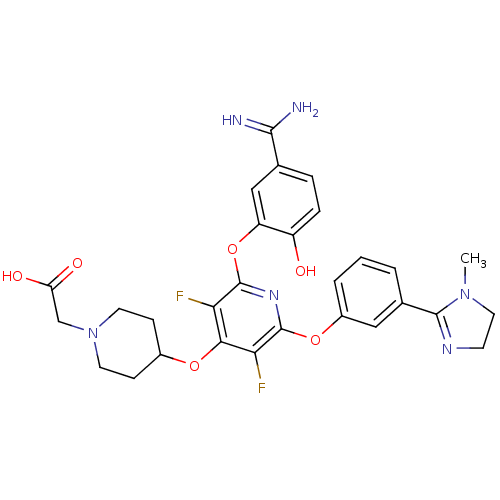
((4-{2-(5-Carbamimidoyl-2-hydroxy-phenoxy)-3,5-difl...)Show SMILES CN1CCN=C1c1cccc(Oc2nc(Oc3cc(ccc3O)C(N)=N)c(F)c(OC3CCN(CC(O)=O)CC3)c2F)c1 |c:4| Show InChI InChI=1S/C29H30F2N6O6/c1-36-12-9-34-27(36)17-3-2-4-19(13-17)42-28-23(30)25(41-18-7-10-37(11-8-18)15-22(39)40)24(31)29(35-28)43-21-14-16(26(32)33)5-6-20(21)38/h2-6,13-14,18,38H,7-12,15H2,1H3,(H3,32,33)(H,39,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Inhibitory potency was measured against human coagulation factor X |
J Med Chem 41: 3557-62 (1998)
Article DOI: 10.1021/jm980280h
BindingDB Entry DOI: 10.7270/Q2KS6QPR |
More data for this
Ligand-Target Pair | |
Melatonin receptor type 1B
(Homo sapiens (Human)) | BDBM50575514
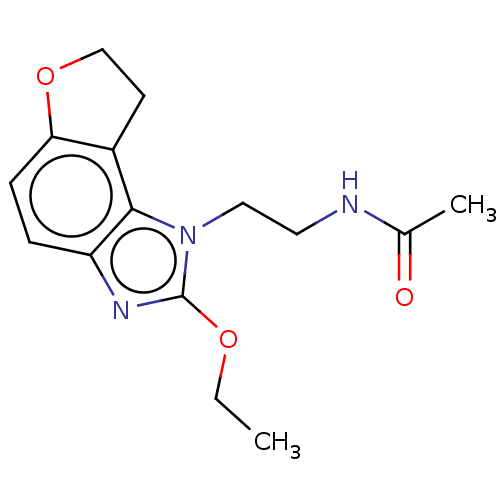
(CHEMBL4852440) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Displacement of 2-[1251]-iodomelatonin from human MT2 receptor expressed in CHO cells incubated for 120 mins by scintillation counting analysis |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.0c00627
BindingDB Entry DOI: 10.7270/Q2Z03CZJ |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 1B
(Homo sapiens (Human)) | BDBM30712
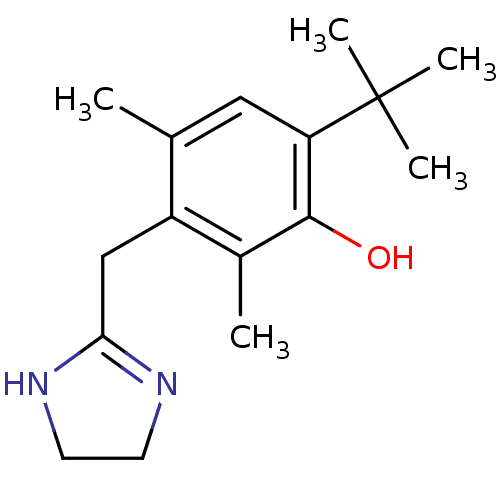
(6-tert-butyl-3-(2-imidazolin-2-ylmethyl)-2,4-dimet...)Show InChI InChI=1S/C16H24N2O/c1-10-8-13(16(3,4)5)15(19)11(2)12(10)9-14-17-6-7-18-14/h8,19H,6-7,9H2,1-5H3,(H,17,18) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Veterans Affairs Medical Center
Curated by PDSP Ki Database
| |
Biochem Biophys Res Commun 184: 752-9 (1992)
Article DOI: 10.1016/0006-291x(92)90654-4
BindingDB Entry DOI: 10.7270/Q2ST7NB1 |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50066619

(({2-(5-Carbamimidoyl-2-hydroxy-phenoxy)-3,5-difluo...)Show SMILES CCOC(=O)CN(C)c1c(F)c(Oc2cccc(c2)C2=NCCN2C)nc(Oc2cc(ccc2O)C(N)=N)c1F |t:20| Show InChI InChI=1S/C27H28F2N6O5/c1-4-38-20(37)14-35(3)23-21(28)26(39-17-7-5-6-16(12-17)25-32-10-11-34(25)2)33-27(22(23)29)40-19-13-15(24(30)31)8-9-18(19)36/h5-9,12-13,36H,4,10-11,14H2,1-3H3,(H3,30,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Inhibitory potency was measured against human coagulation factor X |
J Med Chem 41: 3557-62 (1998)
Article DOI: 10.1021/jm980280h
BindingDB Entry DOI: 10.7270/Q2KS6QPR |
More data for this
Ligand-Target Pair | |
Coagulation factor X
(Homo sapiens (Human)) | BDBM50112488
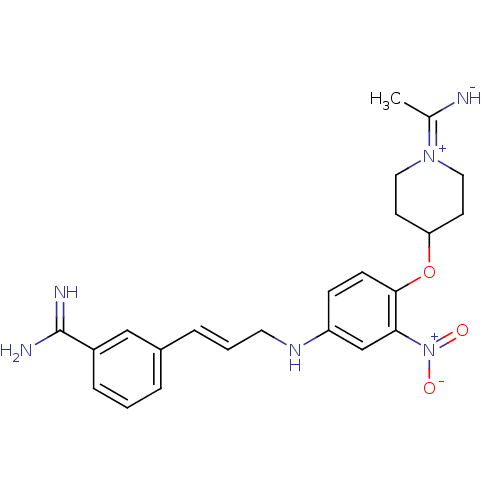
(3-(3-{4-[1-(1-Imino-ethyl)-piperidin-4-yloxy]-3-ni...)Show SMILES CC([NH-])=[N+]1CCC(CC1)Oc1ccc(NC\C=C\c2cccc(c2)C(N)=N)cc1[N+]([O-])=O |(-5.78,-2.46,;-4.43,-3.23,;-4.45,-4.77,;-3.1,-2.46,;-3.1,-.92,;-1.77,-.16,;-.44,-.94,;-.44,-2.47,;-1.77,-3.24,;.91,-.17,;2.24,-.94,;2.24,-2.48,;3.57,-3.25,;4.91,-2.48,;6.24,-3.25,;6.24,-4.79,;7.58,-5.56,;7.57,-7.1,;8.9,-7.87,;8.91,-9.41,;10.25,-10.18,;11.58,-9.4,;11.57,-7.85,;10.23,-7.09,;12.9,-7.07,;12.89,-5.53,;14.24,-7.82,;4.91,-.94,;3.57,-.17,;3.55,1.37,;4.9,2.14,;2.22,2.14,)| Show InChI InChI=1S/C23H28N6O3/c1-16(24)28-12-9-20(10-13-28)32-22-8-7-19(15-21(22)29(30)31)27-11-3-5-17-4-2-6-18(14-17)23(25)26/h2-8,14-15,20,24,27H,9-13H2,1H3,(H3,25,26)/b5-3+ | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Berlex Biosciences
Curated by ChEMBL
| Assay Description
Competitive inhibition of coagulation factor Xa |
Bioorg Med Chem Lett 12: 1307-10 (2002)
BindingDB Entry DOI: 10.7270/Q21R6PV8 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data