

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315314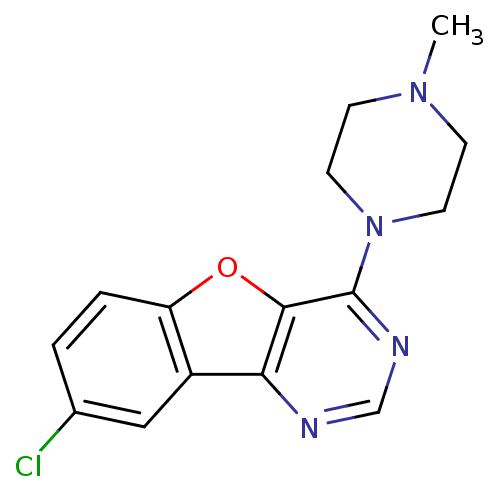 (8-chloro-4-(4-methylpiperazin-1-yl)benzofuro[3,2-d...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 19 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine human recombinant histamine H4 receptor | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM22914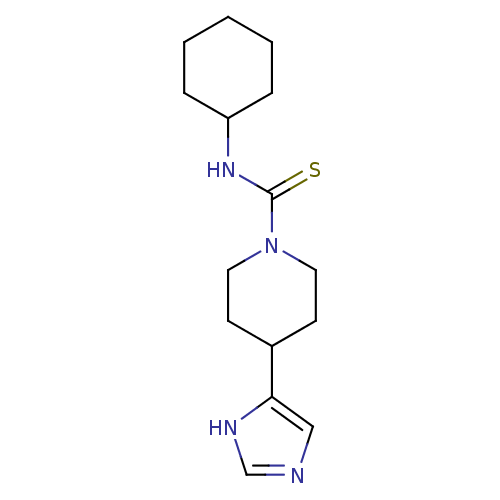 (CHEMBL260374 | N-cyclohexyl-4-(1H-imidazol-5-yl)pi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | 27 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine human recombinant histamine H4 receptor | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM22566 (5-chloro-2-[(4-methylpiperazin-1-yl)carbonyl]-1H-i...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | 38 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine human recombinant histamine H4 receptor | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315348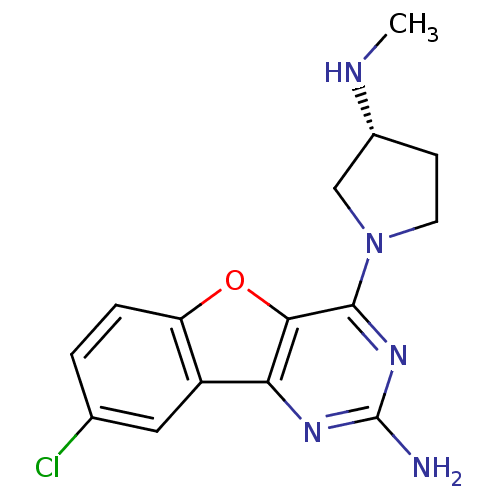 ((R)-8-chloro-4-(3-(methylamino)pyrrolidin-1-yl)ben...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315348 ((R)-8-chloro-4-(3-(methylamino)pyrrolidin-1-yl)ben...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Inverse agonist activity at human histamine H4 receptor assessed as inhibition of [35S]GTPgammaS binding after 15 mins by scintillation proximity ass... | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096734 ((S)-4-[(S)-3-Carboxy-2-(3-carboxy-propionylamino)-...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 with a Y181C mutation in RT (reverse ... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096726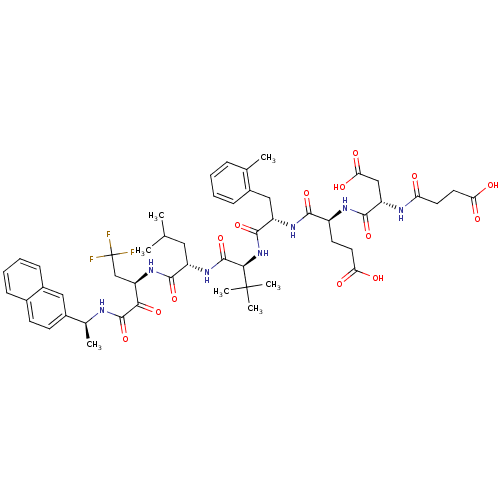 (4-[3-Carboxy-2-(3-carboxy-propionylamino)-propiony...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Binding affinity towards 5-hydroxytryptamine 3 receptor | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096729 ((S)-4-((S)-1-{(S)-1-[(S)-1-((R)-1-Benzylaminooxaly...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Binding affinity towards 5-hydroxytryptamine 3 receptor | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096730 (4-[3-Carboxy-2-(3-carboxy-propionylamino)-propiony...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 variant with Y181C plus K103N mutatio... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315349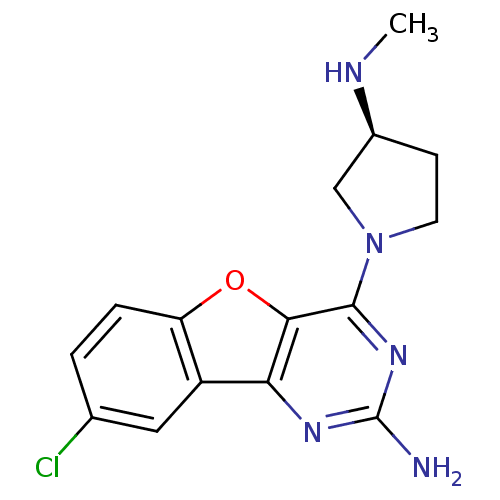 ((S)-8-chloro-4-(3-(methylamino)pyrrolidin-1-yl)ben...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Inverse agonist activity at human histamine H4 receptor assessed as inhibition of [35S]GTPgammaS binding after 15 mins by scintillation proximity ass... | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096727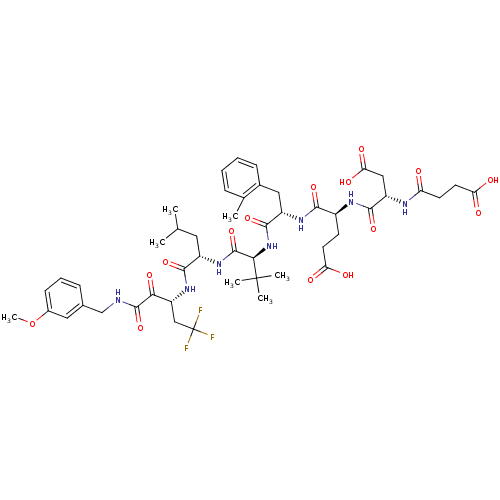 ((S)-4-[(S)-3-Carboxy-2-(3-carboxy-propionylamino)-...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 variant with Y181C plus K103N mutatio... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096728 ((S)-4-((S)-1-{(S)-1-[(S)-1-((S)-1-Amino-2-carbamoy...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 with a Y181C mutation in RT (reverse ... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096733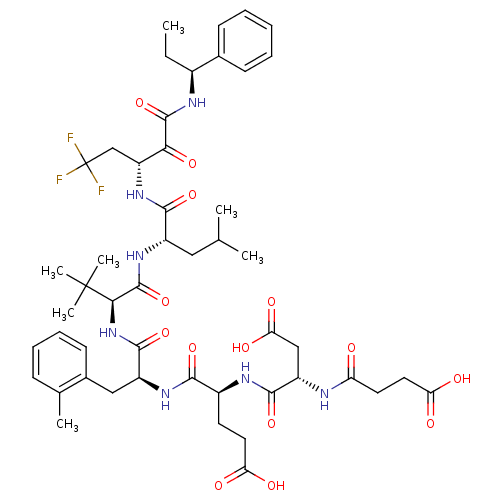 (4-[3-Carboxy-2-(3-carboxy-propionylamino)-propiony...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Binding affinity towards 5-hydroxytryptamine 3 receptor | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096725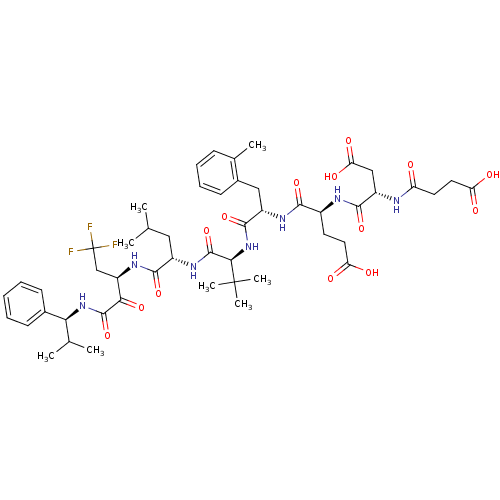 (4-[3-Carboxy-2-(3-carboxy-propionylamino)-propiony...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against delta receptor of (endomorphin 2) in mouse vas deferens was determined | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096732 (4-[3-Carboxy-2-(3-carboxy-propionylamino)-propiony...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 with a Y181C mutation in RT (reverse ... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096724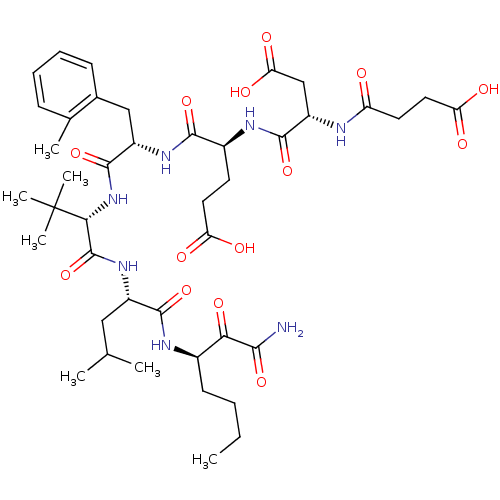 ((S)-4-((S)-1-{(S)-1-[(S)-1-((R)-1-Aminooxalyl-pent...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 variant with Y181C plus K103N mutatio... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315349 ((S)-8-chloro-4-(3-(methylamino)pyrrolidin-1-yl)ben...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315314 (8-chloro-4-(4-methylpiperazin-1-yl)benzofuro[3,2-d...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315333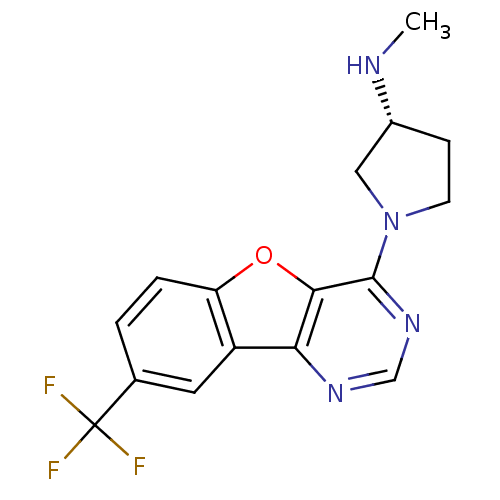 ((R)-N-methyl-1-(8-(trifluoromethyl)benzofuro[3,2-d...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315323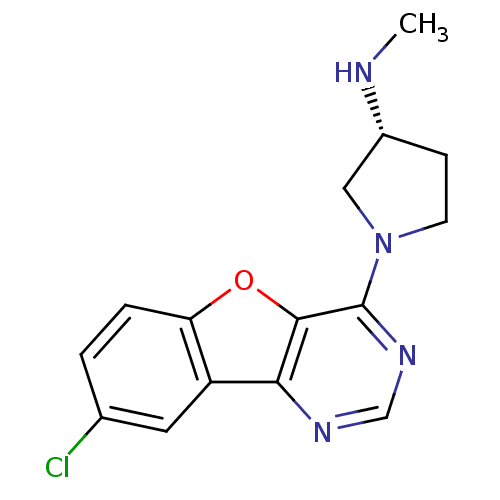 ((R)-1-(8-chlorobenzofuro[3,2-d]pyrimidin-4-yl)-N-m...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315339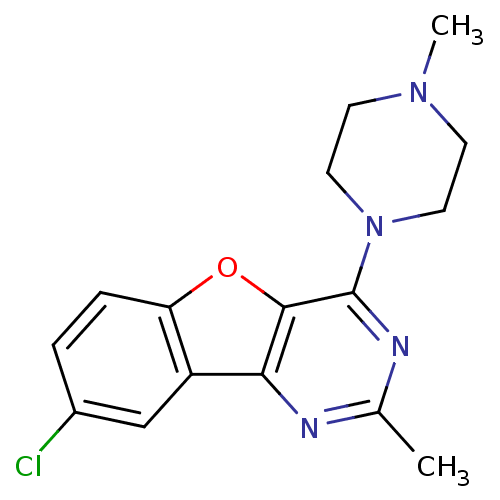 (8-chloro-2-methyl-4-(4-methylpiperazin-1-yl)benzof...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315342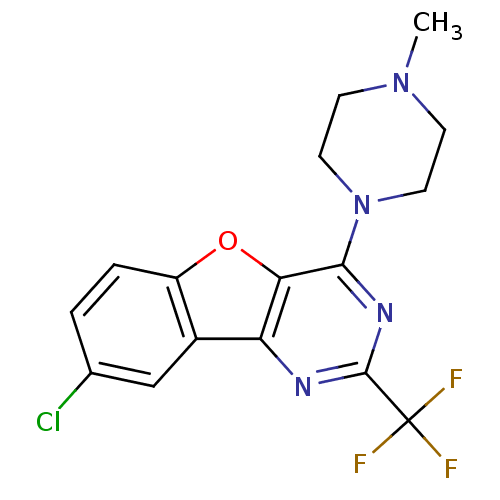 (8-chloro-4-(4-methylpiperazin-1-yl)-2-(trifluorome...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 39 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Inverse agonist activity at human histamine H4 receptor assessed as inhibition of [35S]GTPgammaS binding after 15 mins by scintillation proximity ass... | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315323 ((R)-1-(8-chlorobenzofuro[3,2-d]pyrimidin-4-yl)-N-m...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Inverse agonist activity at human histamine H4 receptor assessed as inhibition of [35S]GTPgammaS binding after 15 mins by scintillation proximity ass... | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315317 (8-chloro-4-((1S,4S)-5-methyl-2,5-diazabicyclo[2.2....) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Inverse agonist activity at human histamine H4 receptor assessed as inhibition of [35S]GTPgammaS binding after 15 mins by scintillation proximity ass... | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315331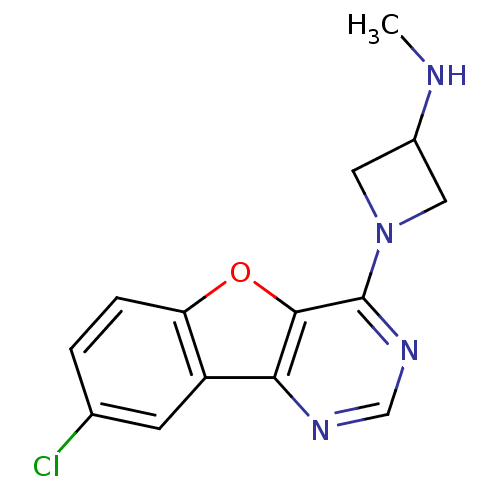 (1-(8-chlorobenzofuro[3,2-d]pyrimidin-4-yl)-N-methy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 76 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Inverse agonist activity at human histamine H4 receptor assessed as inhibition of [35S]GTPgammaS binding after 15 mins by scintillation proximity ass... | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315342 (8-chloro-4-(4-methylpiperazin-1-yl)-2-(trifluorome...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 83 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315332 ((R)-N-methyl-1-(8-methylbenzofuro[3,2-d]pyrimidin-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 86 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315331 (1-(8-chlorobenzofuro[3,2-d]pyrimidin-4-yl)-N-methy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 87 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315347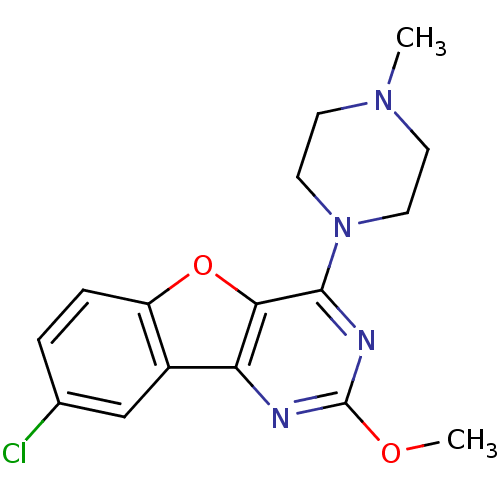 (8-chloro-2-methoxy-4-(4-methylpiperazin-1-yl)benzo...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315322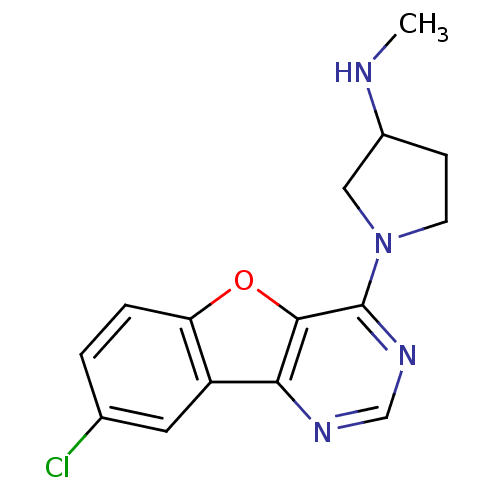 (1-(8-chlorobenzofuro[3,2-d]pyrimidin-4-yl)-N-methy...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 95 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315317 (8-chloro-4-((1S,4S)-5-methyl-2,5-diazabicyclo[2.2....) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 111 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315325 (1-(8-chlorobenzofuro[3,2-d]pyrimidin-4-yl)pyrrolid...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 126 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315318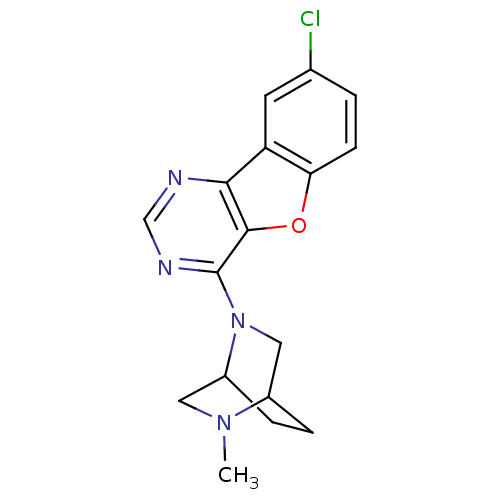 (8-chloro-4-(5-methyl-2,5-diazabicyclo[2.2.2]octan-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 169 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315338 ((R)-1-(7-chlorobenzofuro[3,2-d]pyrimidin-4-yl)-N-m...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 171 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315320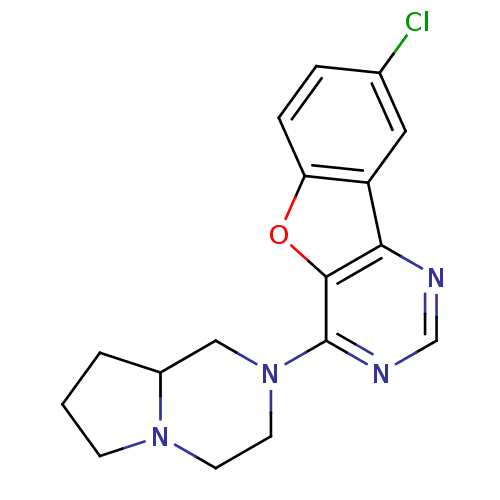 (8-chloro-4-(hexahydropyrrolo[1,2-a]pyrazin-2(1H)-y...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 236 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315324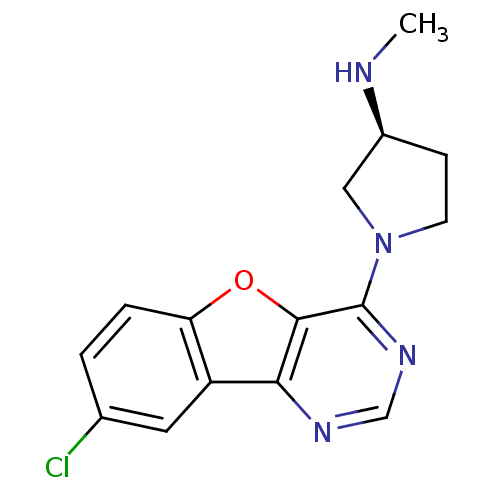 ((S)-1-(8-chlorobenzofuro[3,2-d]pyrimidin-4-yl)-N-m...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 285 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315324 ((S)-1-(8-chlorobenzofuro[3,2-d]pyrimidin-4-yl)-N-m...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 292 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Inverse agonist activity at human histamine H4 receptor assessed as inhibition of [35S]GTPgammaS binding after 15 mins by scintillation proximity ass... | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Chymotrypsin-C (Homo sapiens (Human)) | BDBM50096724 ((S)-4-((S)-1-{(S)-1-[(S)-1-((R)-1-Aminooxalyl-pent...) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Compound was tested for inhibition of Serine protease chymotrypsin | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315344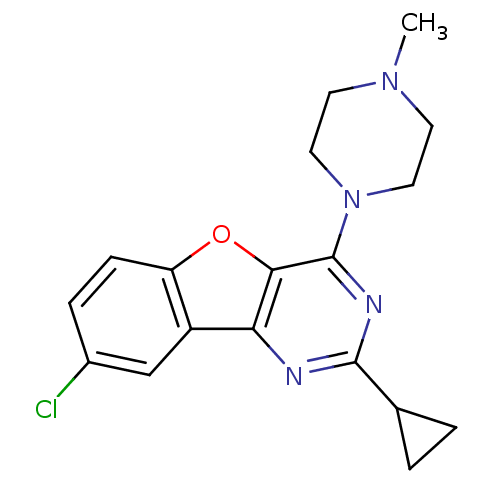 (8-chloro-2-cyclopropyl-4-(4-methylpiperazin-1-yl)b...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 310 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315319 (8-chloro-4-(8-methyl-3,8-diazabicyclo[3.2.1]octan-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 440 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315346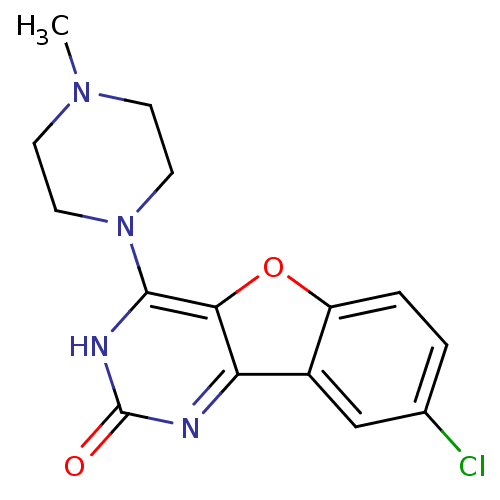 (8-chloro-4-(4-methylpiperazin-1-yl)benzofuro[3,2-d...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Genome polyprotein/Non-structural protein 4A (Hepatitis C virus) | BDBM50096731 (4-[3-Carboxy-2-(3-carboxy-propionylamino)-propiony...) | PDB UniProtKB/TrEMBL GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Roche Discover Welwyn Curated by ChEMBL | Assay Description Inhibitory activity against non-nucleoside reverse transcriptase inhibitors (NNRTI) -resistant HIV-1 strain A17 variant with Y181C plus K103N mutatio... | Bioorg Med Chem Lett 11: 355-7 (2001) BindingDB Entry DOI: 10.7270/Q2WQ0322 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315328 (8-chloro-4-(tetrahydro-1H-pyrrolo[3,4-b]pyridin-6(...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315327 (1-(8-chlorobenzofuro[3,2-d]pyrimidin-4-yl)-N,3-dim...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315340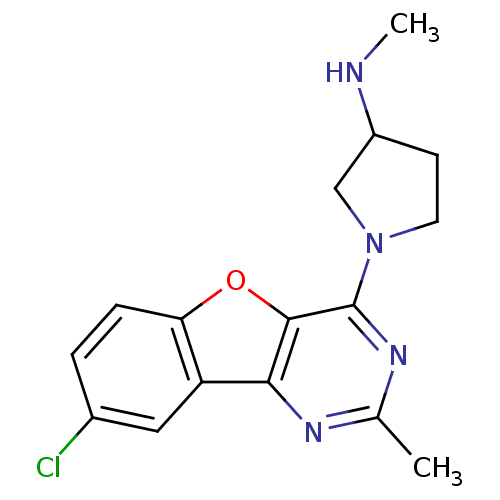 (1-(8-chloro-2-methylbenzofuro[3,2-d]pyrimidin-4-yl...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315315 (8-chloro-4-(3,4-dimethylpiperazin-1-yl)benzofuro[3...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H3 receptor (Homo sapiens (Human)) | BDBM50315348 ((R)-8-chloro-4-(3-(methylamino)pyrrolidin-1-yl)ben...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 5.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Binding affinity to histamine H3 receptor | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315336 (4-(3-(methylamino)pyrrolidin-1-yl)benzofuro[3,2-d]...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315345 (8-chloro-4-(4-methylpiperazin-1-yl)benzofuro[3,2-d...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histamine H4 receptor (Homo sapiens (Human)) | BDBM50315334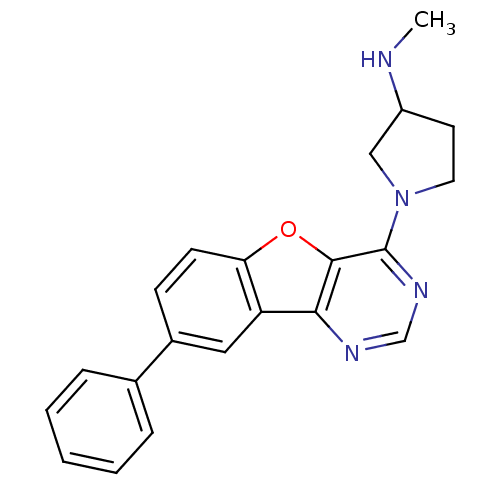 (CHEMBL1089166 | N-methyl-1-(8-phenylbenzofuro[3,2-...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Argenta Discovery Ltd. Curated by ChEMBL | Assay Description Displacement of [3H]histamine dihydrochloride from human histamine H4 receptor after 2.5 hrs by scintillation proximity assay | Bioorg Med Chem Lett 20: 2516-9 (2010) Article DOI: 10.1016/j.bmcl.2010.02.097 BindingDB Entry DOI: 10.7270/Q2CN74V3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 63 total ) | Next | Last >> |