 Found 256 hits of ki for UniProtKB: P35228
Found 256 hits of ki for UniProtKB: P35228 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50386178
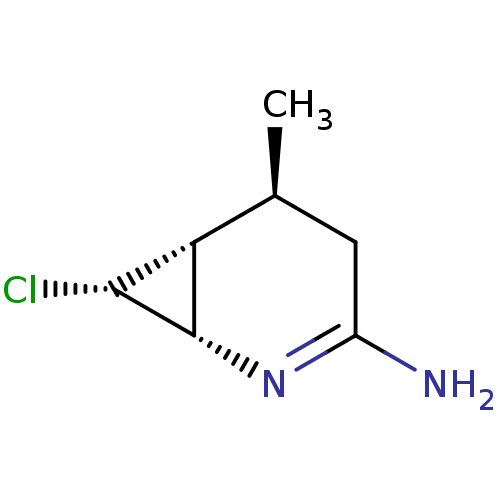
(CHEMBL1800346 | ONO-1714)Show SMILES C[C@H]1CC(N)=N[C@@H]2[C@H](Cl)[C@H]12 |r,c:4| Show InChI InChI=1S/C7H11ClN2/c1-3-2-4(9)10-7-5(3)6(7)8/h3,5-7H,2H2,1H3,(H2,9,10)/t3-,5?,6+,7-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Instituto Cajal (CSIC)
Curated by ChEMBL
| Assay Description
Inhibition of human iNOS |
Eur J Med Chem 54: 439-46 (2012)
Article DOI: 10.1016/j.ejmech.2012.05.031
BindingDB Entry DOI: 10.7270/Q2NC6280 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50386178

(CHEMBL1800346 | ONO-1714)Show SMILES C[C@H]1CC(N)=N[C@@H]2[C@H](Cl)[C@H]12 |r,c:4| Show InChI InChI=1S/C7H11ClN2/c1-3-2-4(9)10-7-5(3)6(7)8/h3,5-7H,2H2,1H3,(H2,9,10)/t3-,5?,6+,7-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.86 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Nycomed GmbH
Curated by ChEMBL
| Assay Description
Inhibition of human iNOS assessed as inhibition of [3H]L-arginine to [3H]L-citrulline conversion by scintillation counting |
Bioorg Med Chem Lett 21: 4228-32 (2011)
Article DOI: 10.1016/j.bmcl.2011.05.073
BindingDB Entry DOI: 10.7270/Q2H996GJ |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50111438
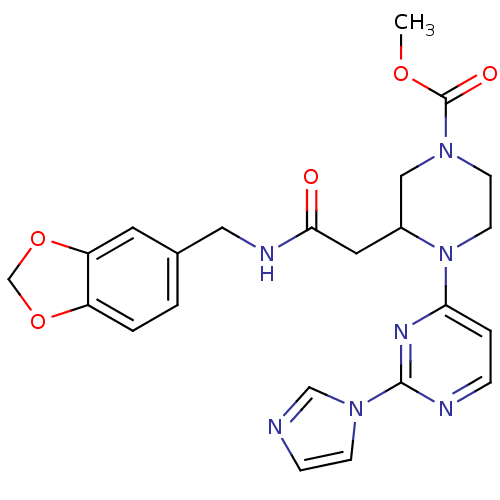
(3-{[(Benzo[1,3]dioxol-5-ylmethyl)-carbamoyl]-methy...)Show SMILES COC(=O)N1CCN(C(CC(=O)NCc2ccc3OCOc3c2)C1)c1ccnc(n1)-n1ccnc1 Show InChI InChI=1S/C23H25N7O5/c1-33-23(32)28-8-9-30(20-4-5-25-22(27-20)29-7-6-24-14-29)17(13-28)11-21(31)26-12-16-2-3-18-19(10-16)35-15-34-18/h2-7,10,14,17H,8-9,11-13,15H2,1H3,(H,26,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
PubMed
| 2.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Binding against the partially purified human Inducible nitric oxide synthase |
J Med Chem 45: 1543-58 (2002)
BindingDB Entry DOI: 10.7270/Q2CN74ND |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50065823
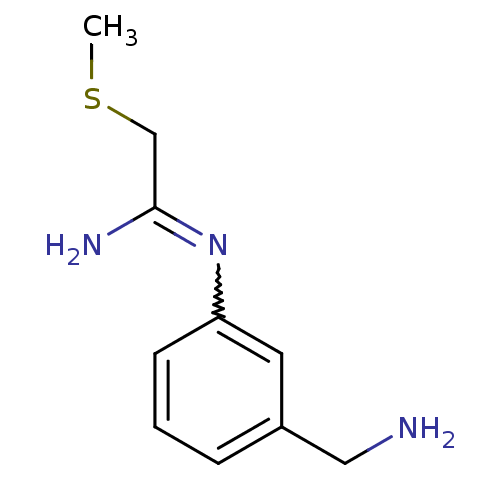
(CHEMBL555794 | N-(3-Aminomethyl-phenyl)-2-methylsu...)Show InChI InChI=1S/C10H15N3S/c1-14-7-10(12)13-9-4-2-3-8(5-9)6-11/h2-5H,6-7,11H2,1H3,(H2,12,13) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 21 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS) isoenzyme. |
J Med Chem 41: 2858-71 (1998)
Article DOI: 10.1021/jm980072p
BindingDB Entry DOI: 10.7270/Q2862H4X |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50064015
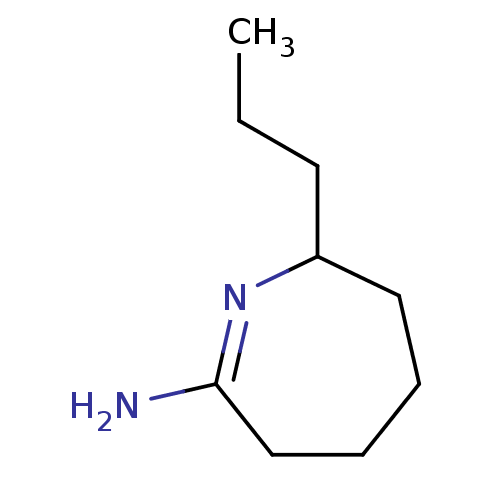
(7-Propyl-azepan-(2Z)-ylideneamine; hydrochloride |...)Show InChI InChI=1S/C9H18N2/c1-2-5-8-6-3-4-7-9(10)11-8/h8H,2-7H2,1H3,(H2,10,11) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibitory activity against L-arginine binding to Inducible nitric oxide synthase |
J Med Chem 41: 1361-6 (1998)
Article DOI: 10.1021/jm9704715
BindingDB Entry DOI: 10.7270/Q2348M29 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50065843
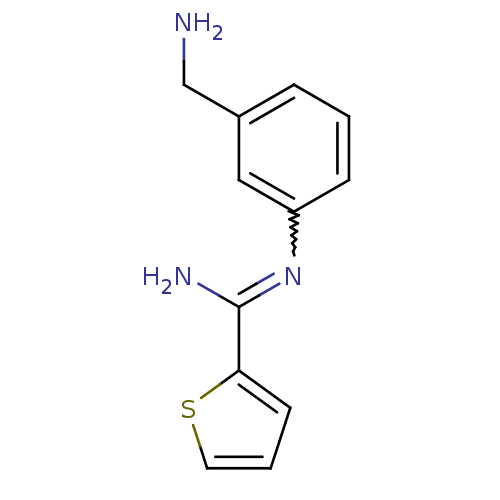
(CHEMBL553081 | CHEMBL555715 | N-(3-Aminomethyl-phe...)Show InChI InChI=1S/C12H13N3S/c13-8-9-3-1-4-10(7-9)15-12(14)11-5-2-6-16-11/h1-7H,8,13H2,(H2,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS) isoenzyme. |
J Med Chem 41: 2858-71 (1998)
Article DOI: 10.1021/jm980072p
BindingDB Entry DOI: 10.7270/Q2862H4X |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50065805
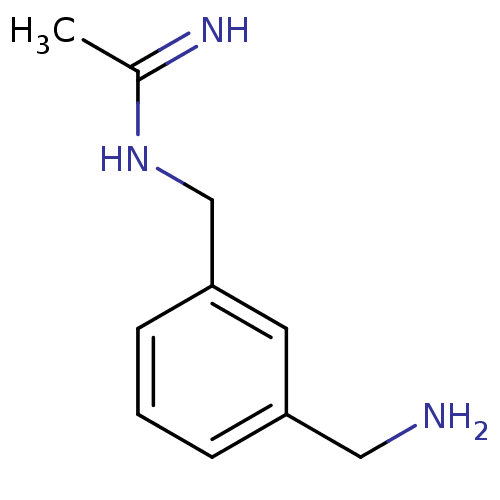
(CHEMBL544788 | N-(3-Aminomethyl-benzyl)-acetamidin...)Show InChI InChI=1S/C10H15N3/c1-8(12)13-7-10-4-2-3-9(5-10)6-11/h2-5H,6-7,11H2,1H3,(H2,12,13) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| 140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS) isoenzyme. |
J Med Chem 41: 2858-71 (1998)
Article DOI: 10.1021/jm980072p
BindingDB Entry DOI: 10.7270/Q2862H4X |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50065807
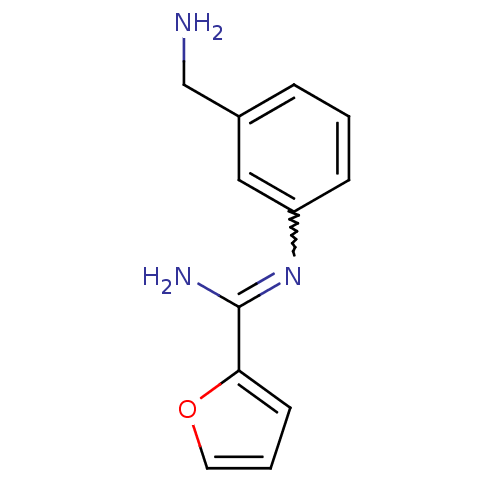
(CHEMBL552825 | N-(3-Aminomethyl-phenyl)-furan-2-ca...)Show InChI InChI=1S/C12H13N3O/c13-8-9-3-1-4-10(7-9)15-12(14)11-5-2-6-16-11/h1-7H,8,13H2,(H2,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS) isoenzyme. |
J Med Chem 41: 2858-71 (1998)
Article DOI: 10.1021/jm980072p
BindingDB Entry DOI: 10.7270/Q2862H4X |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Mus musculus (mouse)-Homo sapiens (Human)) | BDBM50136951
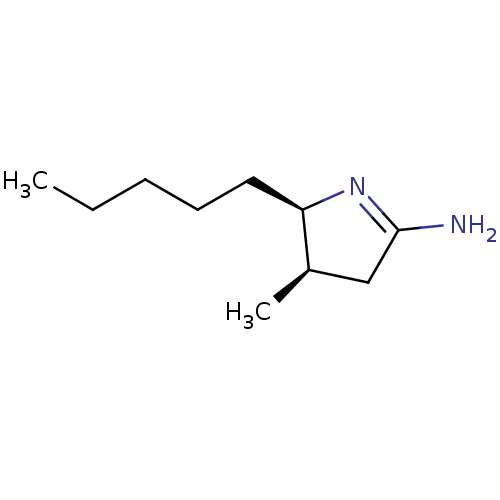
((4R,5R)-4-Methyl-5-pentyl-pyrrolidin-(2E)-ylidenea...)Show InChI InChI=1S/C10H20N2/c1-3-4-5-6-9-8(2)7-10(11)12-9/h8-9H,3-7H2,1-2H3,(H2,11,12)/t8-,9-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
Curated by ChEMBL
| Assay Description
Inhibitory activity against Inducible nitric oxide synthase |
J Med Chem 46: 5700-11 (2003)
Checked by Author
Article DOI: 10.1021/jm030301u
BindingDB Entry DOI: 10.7270/Q26D5TQV |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50330882
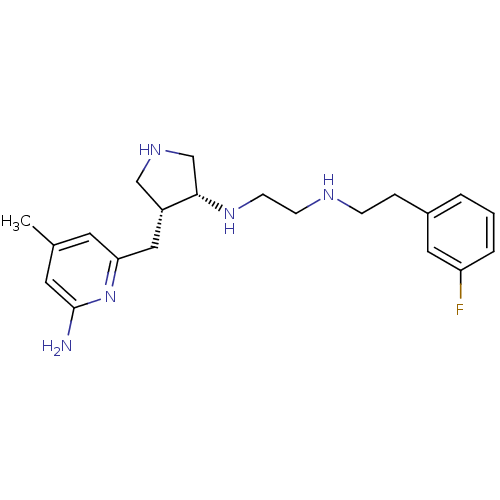
(CHEMBL1277951 | CHEMBL594682 | N1-((3R,4R)-4-((6-a...)Show SMILES Cc1cc(N)nc(C[C@@H]2CNC[C@@H]2NCCNCCc2cccc(F)c2)c1 |r| Show InChI InChI=1S/C21H30FN5/c1-15-9-19(27-21(23)10-15)12-17-13-25-14-20(17)26-8-7-24-6-5-16-3-2-4-18(22)11-16/h2-4,9-11,17,20,24-26H,5-8,12-14H2,1H3,(H2,23,27)/t17-,20+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 273 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
Rat and human nNOS, murine macrophage iNOS, and human eNOS were recombinant enzymes (expressed in E. coli and purified as reported previously in the ... |
US Patent US9783500 (2017)
BindingDB Entry DOI: 10.7270/Q2930WBP |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50072297
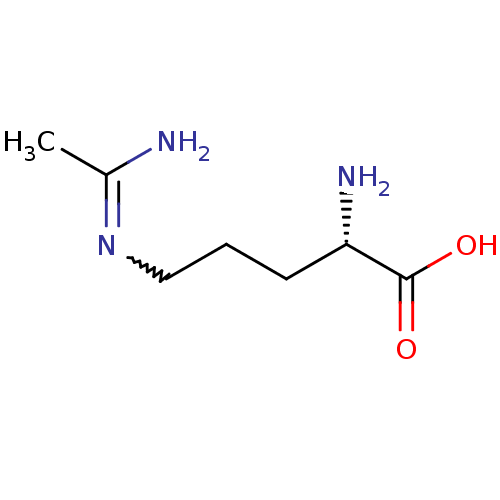
((S)-5-Acetimidoylamino-2-amino-pent | (S)-5-Acetim...)Show InChI InChI=1S/C7H15N3O2/c1-5(8)10-4-2-3-6(9)7(11)12/h6H,2-4,9H2,1H3,(H2,8,10)(H,11,12)/t6-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MMDB
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant for the inhibition of human Inducible nitric oxide synthase |
Bioorg Med Chem Lett 7: 1763-1768 (1997)
Article DOI: 10.1016/S0960-894X(97)00309-0
BindingDB Entry DOI: 10.7270/Q2TQ61J5 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50065813

(CHEMBL555584 | N-(3-Aminomethyl-phenyl)-2-fluoro-a...)Show InChI InChI=1S/C9H12FN3/c10-5-9(12)13-8-3-1-2-7(4-8)6-11/h1-4H,5-6,11H2,(H2,12,13) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS) isoenzyme. |
J Med Chem 41: 2858-71 (1998)
Article DOI: 10.1021/jm980072p
BindingDB Entry DOI: 10.7270/Q2862H4X |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50330882

(CHEMBL1277951 | CHEMBL594682 | N1-((3R,4R)-4-((6-a...)Show SMILES Cc1cc(N)nc(C[C@@H]2CNC[C@@H]2NCCNCCc2cccc(F)c2)c1 |r| Show InChI InChI=1S/C21H30FN5/c1-15-9-19(27-21(23)10-15)12-17-13-25-14-20(17)26-8-7-24-6-5-16-3-2-4-18(22)11-16/h2-4,9-11,17,20,24-26H,5-8,12-14H2,1H3,(H2,23,27)/t17-,20+/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 581 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
Rat and human nNOS, murine macrophage iNOS, and human eNOS were recombinant enzymes (expressed in E. coli and purified as reported previously in the ... |
US Patent US9783500 (2017)
BindingDB Entry DOI: 10.7270/Q2930WBP |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50289681
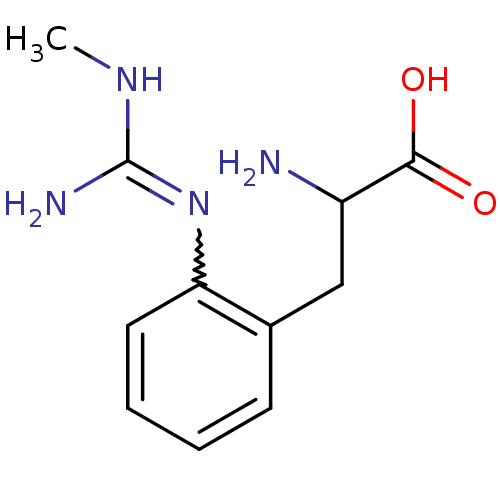
(2-Amino-3-[2-(N'-methyl-guanidino)-phenyl]-propion...)Show InChI InChI=1S/C11H16N4O2/c1-14-11(13)15-9-5-3-2-4-7(9)6-8(12)10(16)17/h2-5,8H,6,12H2,1H3,(H,16,17)(H3,13,14,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 600 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant for the inhibition of human Inducible nitric oxide synthase |
Bioorg Med Chem Lett 7: 1763-1768 (1997)
Article DOI: 10.1016/S0960-894X(97)00309-0
BindingDB Entry DOI: 10.7270/Q2TQ61J5 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50592473
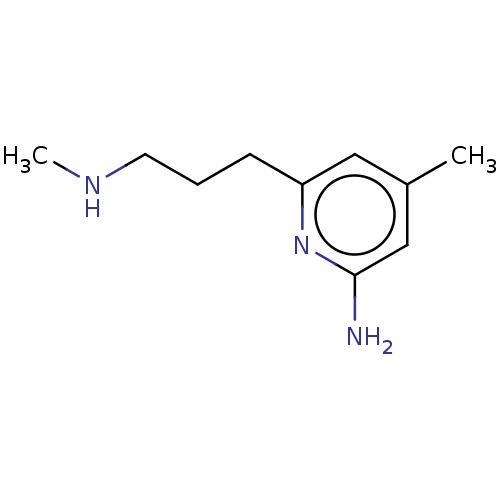
(CHEMBL5191295) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 669 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1016/j.bmc.2022.116878
BindingDB Entry DOI: 10.7270/Q2VQ36P6 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50225106
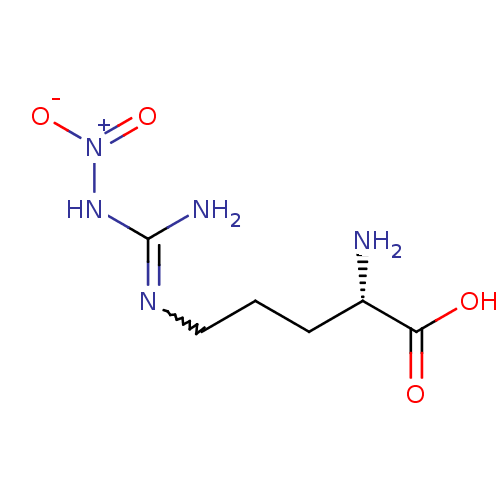
((2S)-2-amino-5-{[(E)-amino(nitroimino)methyl]amino...)Show SMILES N[C@@H](CCCNC(N)=N[N+]([O-])=O)C(O)=O |r,w:8.8| Show InChI InChI=1S/C6H13N5O4/c7-4(5(12)13)2-1-3-9-6(8)10-11(14)15/h4H,1-3,7H2,(H,12,13)(H3,8,9,10)/t4-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 670 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS) isoenzyme. |
J Med Chem 41: 2858-71 (1998)
Article DOI: 10.1021/jm980072p
BindingDB Entry DOI: 10.7270/Q2862H4X |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50341684
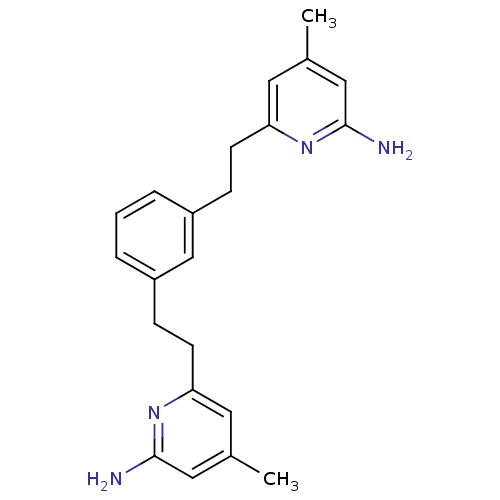
(6,6'-(2,2'-(1,3-Phenylene)bis(ethane-2,1-diyl))bis...)Show InChI InChI=1S/C22H26N4/c1-15-10-19(25-21(23)12-15)8-6-17-4-3-5-18(14-17)7-9-20-11-16(2)13-22(24)26-20/h3-5,10-14H,6-9H2,1-2H3,(H2,23,25)(H2,24,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| US Patent
| 682 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astex
| Assay Description
The NOSs isoform assays involved subjecting 3-8 to an oxyhemoglobin NO capture assay using a Biotek Gen5� microplate reader. IC50 values for each com... |
J Med Chem 52: 379-88 (2009)
BindingDB Entry DOI: 10.7270/Q21N83G1 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM275051

(US9878996, Compound 7)Show InChI InChI=1S/C18H20FN5/c19-16-5-1-3-15(13-16)4-2-8-20-9-6-17-7-10-22-18(23-17)24-12-11-21-14-24/h1,3,5,7,10-14,20H,2,4,6,8-9H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| US Patent
| 770 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
Assays were performed at the NIMH Psychoactive Drug Screening Program (PDSP) at UNC-Chapel Hill. |
US Patent US9878996 (2018)
BindingDB Entry DOI: 10.7270/Q2PN97PN |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50230993
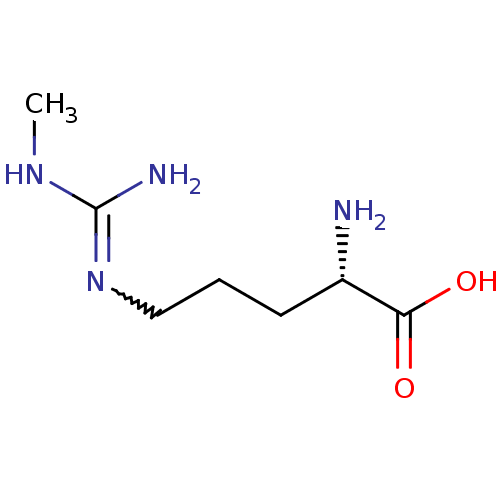
((2S)-2-amino-5-[(N-methylcarbamimidoyl)amino]penta...)Show InChI InChI=1S/C7H16N4O2/c1-10-7(9)11-4-2-3-5(8)6(12)13/h5H,2-4,8H2,1H3,(H,12,13)(H3,9,10,11)/t5-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 860 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant for the inhibition of human Inducible nitric oxide synthase |
Bioorg Med Chem Lett 7: 1763-1768 (1997)
Article DOI: 10.1016/S0960-894X(97)00309-0
BindingDB Entry DOI: 10.7270/Q2TQ61J5 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50230993

((2S)-2-amino-5-[(N-methylcarbamimidoyl)amino]penta...)Show InChI InChI=1S/C7H16N4O2/c1-10-7(9)11-4-2-3-5(8)6(12)13/h5H,2-4,8H2,1H3,(H,12,13)(H3,9,10,11)/t5-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 860 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS) isoenzyme. |
J Med Chem 41: 2858-71 (1998)
Article DOI: 10.1021/jm980072p
BindingDB Entry DOI: 10.7270/Q2862H4X |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50058459
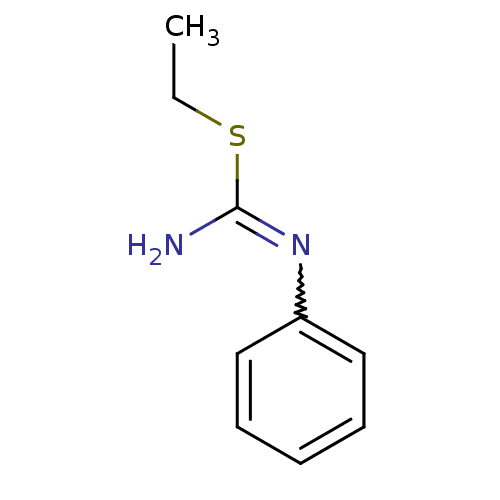
(2-Ethyl-1-phenyl-isothiourea; hydriodide | CHEMBL4...)Show InChI InChI=1S/C9H12N2S/c1-2-12-9(10)11-8-6-4-3-5-7-8/h3-7H,2H2,1H3,(H2,10,11) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS). |
J Med Chem 40: 1901-5 (1997)
Article DOI: 10.1021/jm960785c
BindingDB Entry DOI: 10.7270/Q2SJ1M94 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM275050
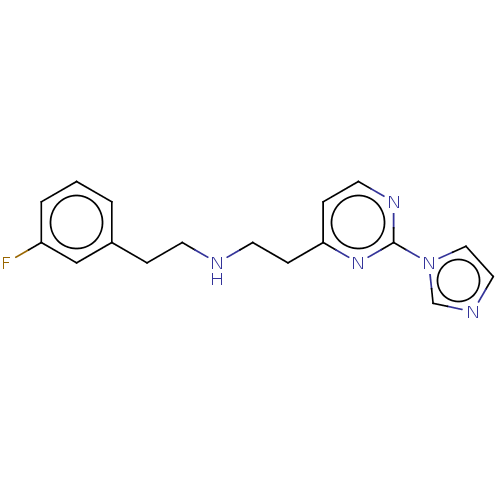
(US9878996, Compound 6)Show InChI InChI=1S/C17H18FN5/c18-15-3-1-2-14(12-15)4-7-19-8-5-16-6-9-21-17(22-16)23-11-10-20-13-23/h1-3,6,9-13,19H,4-5,7-8H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| US Patent
| 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
Assays were performed at the NIMH Psychoactive Drug Screening Program (PDSP) at UNC-Chapel Hill. |
US Patent US9878996 (2018)
BindingDB Entry DOI: 10.7270/Q2PN97PN |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50526363
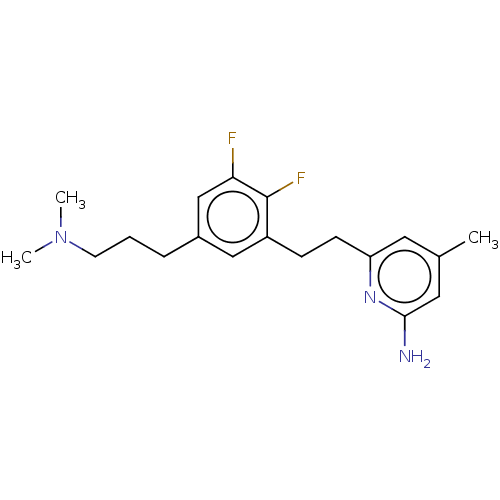
(CHEMBL4440481)Show InChI InChI=1S/C19H25F2N3/c1-13-9-16(23-18(22)10-13)7-6-15-11-14(5-4-8-24(2)3)12-17(20)19(15)21/h9-12H,4-8H2,1-3H3,(H2,22,23) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 1.31E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
Curated by ChEMBL
| Assay Description
Inhibition of human iNOS expressed in Escherichia coli using L-arginine as substrate in presence of human oxyhemoglobin after 6 mins by hemoglobin NO... |
J Med Chem 62: 2690-2707 (2019)
Article DOI: 10.1021/acs.jmedchem.8b02032
BindingDB Entry DOI: 10.7270/Q2H998NM |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50058448
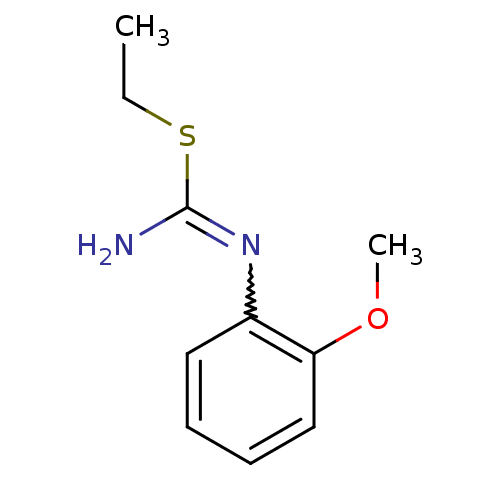
(2-Ethyl-1-(2-methoxy-phenyl)-isothiourea; hydrochl...)Show InChI InChI=1S/C10H14N2OS/c1-3-14-10(11)12-8-6-4-5-7-9(8)13-2/h4-7H,3H2,1-2H3,(H2,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS). |
J Med Chem 40: 1901-5 (1997)
Article DOI: 10.1021/jm960785c
BindingDB Entry DOI: 10.7270/Q2SJ1M94 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50341682
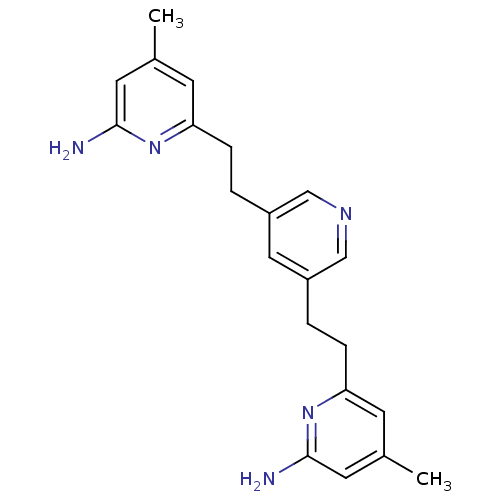
(6,6'-(2,2'-(Pyridine-3,5-diyl)bis(ethane-2,1-diyl)...)Show InChI InChI=1S/C21H25N5/c1-14-7-18(25-20(22)9-14)5-3-16-11-17(13-24-12-16)4-6-19-8-15(2)10-21(23)26-19/h7-13H,3-6H2,1-2H3,(H2,22,25)(H2,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| US Patent
| 1.45E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astex
| Assay Description
The NOSs isoform assays involved subjecting 3-8 to an oxyhemoglobin NO capture assay using a Biotek Gen5� microplate reader. IC50 values for each com... |
J Med Chem 52: 379-88 (2009)
BindingDB Entry DOI: 10.7270/Q21N83G1 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50341682

(6,6'-(2,2'-(Pyridine-3,5-diyl)bis(ethane-2,1-diyl)...)Show InChI InChI=1S/C21H25N5/c1-14-7-18(25-20(22)9-14)5-3-16-11-17(13-24-12-16)4-6-19-8-15(2)10-21(23)26-19/h7-13H,3-6H2,1-2H3,(H2,22,25)(H2,23,26) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 1.45E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California, Irvine
| Assay Description
Inhibition assay using nitric oxide synthases. |
Biochemistry 49: 10803-10 (2010)
Article DOI: 10.1021/bi1013479
BindingDB Entry DOI: 10.7270/Q2KS6Q51 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50065842
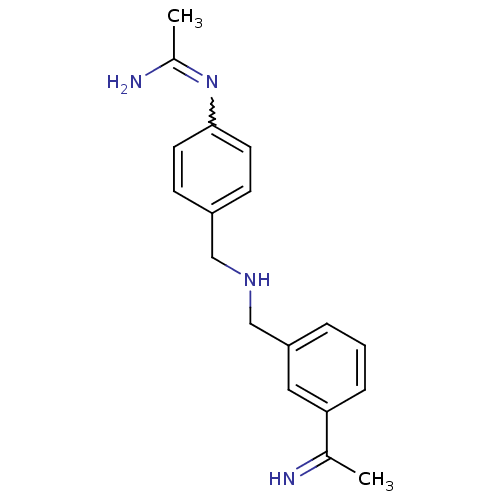
(CHEMBL540048 | N-(4-{[3-(1-Imino-ethyl)-benzylamin...)Show SMILES CC(N)=Nc1ccc(CNCc2cccc(c2)C(C)=N)cc1 |w:3.3| Show InChI InChI=1S/C18H22N4/c1-13(19)17-5-3-4-16(10-17)12-21-11-15-6-8-18(9-7-15)22-14(2)20/h3-10,19,21H,11-12H2,1-2H3,(H2,20,22) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS) isoenzyme. |
J Med Chem 41: 2858-71 (1998)
Article DOI: 10.1021/jm980072p
BindingDB Entry DOI: 10.7270/Q2862H4X |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50526359

(CHEMBL4450896)Show SMILES CN1CCC[C@H]1CCc1cc(F)c(F)c(CCc2cc(C)cc(N)n2)c1 |r| Show InChI InChI=1S/C21H27F2N3/c1-14-10-17(25-20(24)11-14)7-6-16-12-15(13-19(22)21(16)23)5-8-18-4-3-9-26(18)2/h10-13,18H,3-9H2,1-2H3,(H2,24,25)/t18-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 1.64E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
Curated by ChEMBL
| Assay Description
Inhibition of human iNOS expressed in Escherichia coli using L-arginine as substrate in presence of human oxyhemoglobin after 6 mins by hemoglobin NO... |
J Med Chem 62: 2690-2707 (2019)
Article DOI: 10.1021/acs.jmedchem.8b02032
BindingDB Entry DOI: 10.7270/Q2H998NM |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50526373
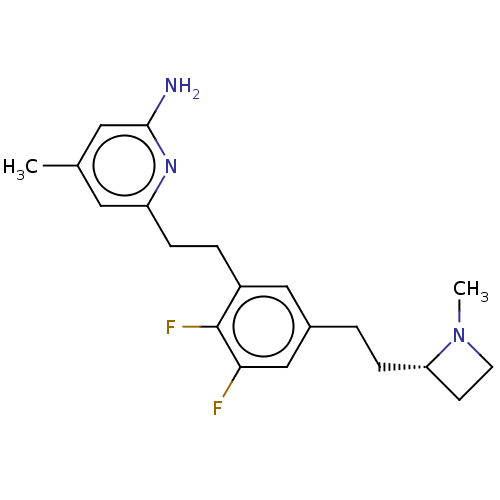
(CHEMBL4590333)Show SMILES CN1CC[C@@H]1CCc1cc(F)c(F)c(CCc2cc(C)cc(N)n2)c1 |r| Show InChI InChI=1S/C20H25F2N3/c1-13-9-16(24-19(23)10-13)5-4-15-11-14(12-18(21)20(15)22)3-6-17-7-8-25(17)2/h9-12,17H,3-8H2,1-2H3,(H2,23,24)/t17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 1.78E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
Curated by ChEMBL
| Assay Description
Inhibition of human iNOS expressed in Escherichia coli using L-arginine as substrate in presence of human oxyhemoglobin after 6 mins by hemoglobin NO... |
J Med Chem 62: 2690-2707 (2019)
Article DOI: 10.1021/acs.jmedchem.8b02032
BindingDB Entry DOI: 10.7270/Q2H998NM |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM275056
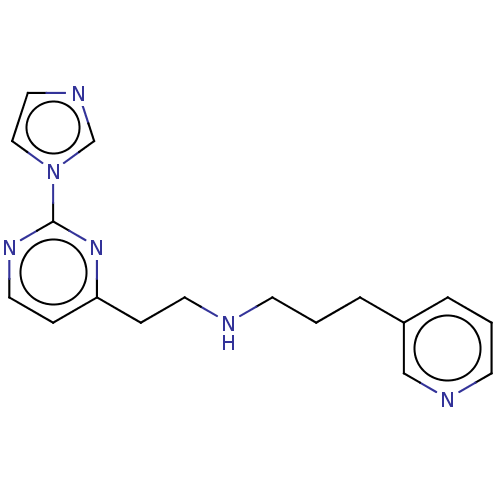
(US9878996, Compound 29)Show InChI InChI=1S/C17H20N6/c1(3-15-4-2-8-19-13-15)7-18-9-5-16-6-10-21-17(22-16)23-12-11-20-14-23/h2,4,6,8,10-14,18H,1,3,5,7,9H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| US Patent
| 1.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
Assays were performed at the NIMH Psychoactive Drug Screening Program (PDSP) at UNC-Chapel Hill. |
US Patent US9878996 (2018)
BindingDB Entry DOI: 10.7270/Q2PN97PN |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50441007
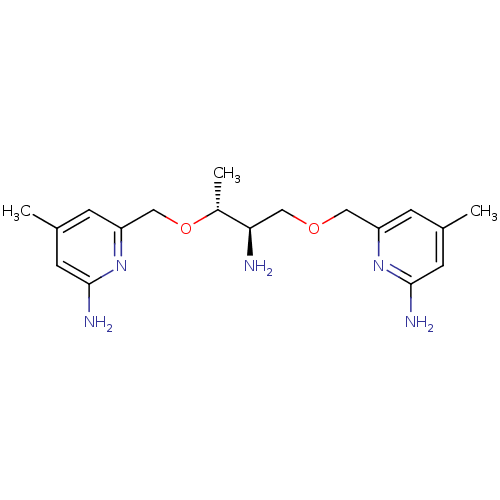
(CHEMBL2430149 | US9732037, Compound 19)Show SMILES C[C@@H](OCc1cc(C)cc(N)n1)[C@H](N)COCc1cc(C)cc(N)n1 |r| Show InChI InChI=1S/C18H27N5O2/c1-11-4-14(22-17(20)6-11)8-24-10-16(19)13(3)25-9-15-5-12(2)7-18(21)23-15/h4-7,13,16H,8-10,19H2,1-3H3,(H2,20,22)(H2,21,23)/t13-,16-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 1.86E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... |
US Patent US9732037 (2017)
BindingDB Entry DOI: 10.7270/Q26W9D6H |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50014713
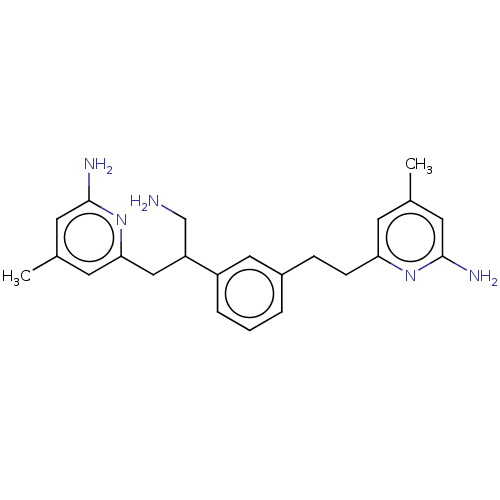
(CHEMBL3262022 | US10759791, Compound 6 | US9951014...)Show SMILES Cc1cc(N)nc(CCc2cccc(c2)C(CN)Cc2cc(C)cc(N)n2)c1 Show InChI InChI=1S/C23H29N5/c1-15-8-20(27-22(25)10-15)7-6-17-4-3-5-18(12-17)19(14-24)13-21-9-16(2)11-23(26)28-21/h3-5,8-12,19H,6-7,13-14,24H2,1-2H3,(H2,25,27)(H2,26,28) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 1.89E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astex
| Assay Description
The NOSs isoform assays involved subjecting 3-8 to an oxyhemoglobin NO capture assay using a Biotek Gen5� microplate reader. IC50 values for each com... |
J Med Chem 52: 379-88 (2009)
BindingDB Entry DOI: 10.7270/Q21N83G1 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50058443
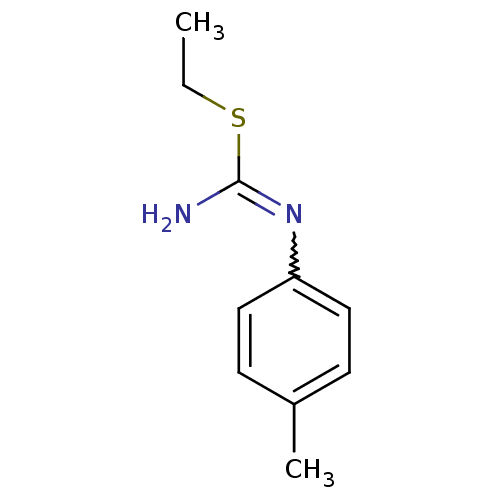
(2-Ethyl-1-p-tolyl-isothiourea; hydrochloride | CHE...)Show InChI InChI=1S/C10H14N2S/c1-3-13-10(11)12-9-6-4-8(2)5-7-9/h4-7H,3H2,1-2H3,(H2,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS). |
J Med Chem 40: 1901-5 (1997)
Article DOI: 10.1021/jm960785c
BindingDB Entry DOI: 10.7270/Q2SJ1M94 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM275053
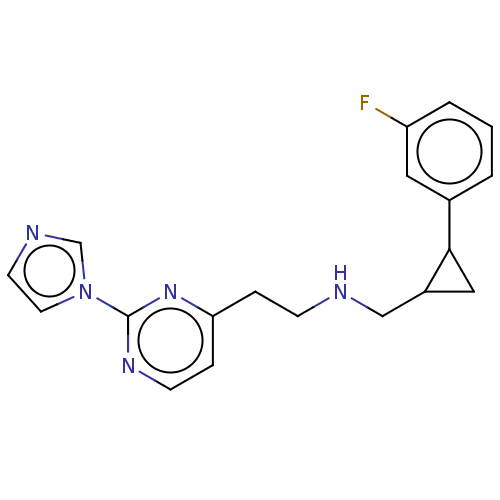
(US9878996, Compound 26)Show InChI InChI=1S/C19H20FN5/c20-16-3-1-2-14(10-16)18-11-15(18)12-21-6-4-17-5-7-23-19(24-17)25-9-8-22-13-25/h1-3,5,7-10,13,15,18,21H,4,6,11-12H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| US Patent
| 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
Assays were performed at the NIMH Psychoactive Drug Screening Program (PDSP) at UNC-Chapel Hill. |
US Patent US9878996 (2018)
BindingDB Entry DOI: 10.7270/Q2PN97PN |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50289677
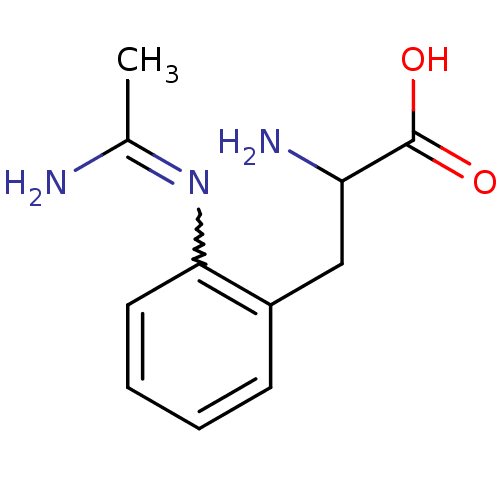
(3-(2-Acetimidoylamino-phenyl)-2-amino-propionic ac...)Show InChI InChI=1S/C11H15N3O2/c1-7(12)14-10-5-3-2-4-8(10)6-9(13)11(15)16/h2-5,9H,6,13H2,1H3,(H2,12,14)(H,15,16) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
| 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
Curated by ChEMBL
| Assay Description
Inhibitory constant for the inhibition of human Inducible nitric oxide synthase |
Bioorg Med Chem Lett 7: 1763-1768 (1997)
Article DOI: 10.1016/S0960-894X(97)00309-0
BindingDB Entry DOI: 10.7270/Q2TQ61J5 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50446249
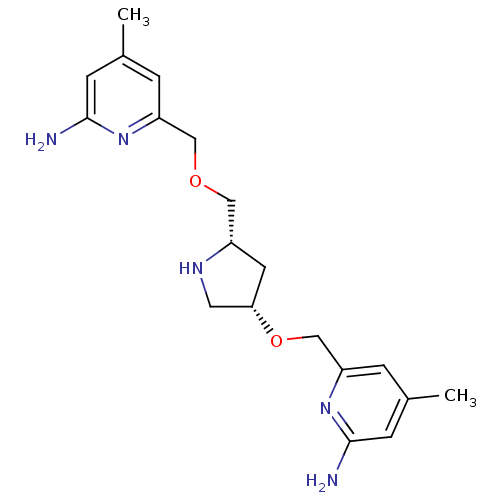
(CHEMBL3109189 | US9732037, Compound 8)Show SMILES Cc1cc(N)nc(COC[C@@H]2C[C@@H](CN2)OCc2cc(C)cc(N)n2)c1 |r| Show InChI InChI=1S/C19H27N5O2/c1-12-3-15(23-18(20)5-12)10-25-9-14-7-17(8-22-14)26-11-16-4-13(2)6-19(21)24-16/h3-6,14,17,22H,7-11H2,1-2H3,(H2,20,23)(H2,21,24)/t14-,17-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 1.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... |
US Patent US9732037 (2017)
BindingDB Entry DOI: 10.7270/Q26W9D6H |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50341681
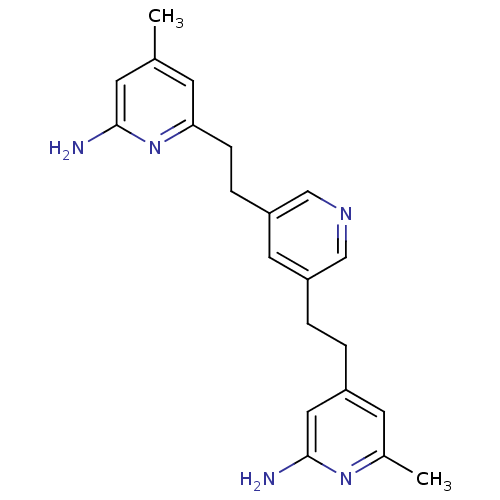
(4-(2-(5-(2-(6-Amino-4-methylpyridin-2-yl)ethyl)pyr...)Show InChI InChI=1S/C21H25N5/c1-14-7-19(26-20(22)8-14)6-5-18-10-17(12-24-13-18)4-3-16-9-15(2)25-21(23)11-16/h7-13H,3-6H2,1-2H3,(H2,22,26)(H2,23,25) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 1.91E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California, Irvine
| Assay Description
Inhibition assay using nitric oxide synthases. |
Biochemistry 49: 10803-10 (2010)
Article DOI: 10.1021/bi1013479
BindingDB Entry DOI: 10.7270/Q2KS6Q51 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50065841
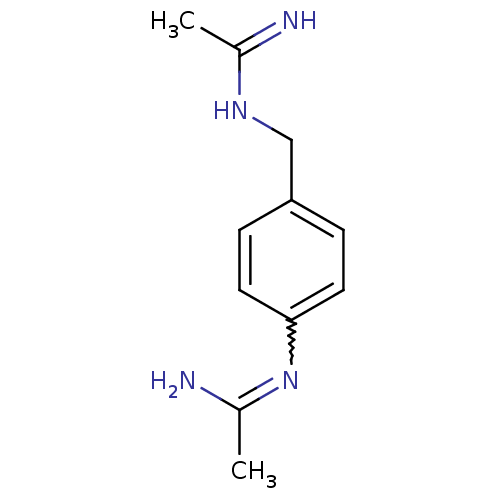
(CHEMBL545025 | N-[4-(Acetimidoylamino-methyl)-phen...)Show InChI InChI=1S/C11H16N4/c1-8(12)14-7-10-3-5-11(6-4-10)15-9(2)13/h3-6H,7H2,1-2H3,(H2,12,14)(H2,13,15) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS) isoenzyme. |
J Med Chem 41: 2858-71 (1998)
Article DOI: 10.1021/jm980072p
BindingDB Entry DOI: 10.7270/Q2862H4X |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM275052

(US9878996, Compound 25)Show InChI InChI=1S/C18H20ClN5/c19-16-5-1-3-15(13-16)4-2-8-20-9-6-17-7-10-22-18(23-17)24-12-11-21-14-24/h1,3,5,7,10-14,20H,2,4,6,8-9H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| US Patent
| 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
Assays were performed at the NIMH Psychoactive Drug Screening Program (PDSP) at UNC-Chapel Hill. |
US Patent US9878996 (2018)
BindingDB Entry DOI: 10.7270/Q2PN97PN |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50065833
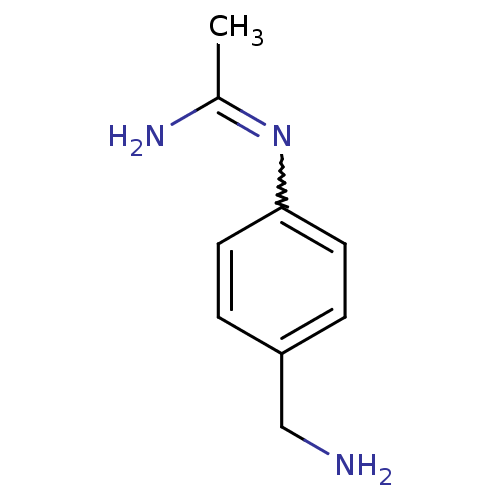
(CHEMBL542432 | N-(4-Aminomethyl-phenyl)-acetamidin...)Show InChI InChI=1S/C9H13N3/c1-7(11)12-9-4-2-8(6-10)3-5-9/h2-5H,6,10H2,1H3,(H2,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS) isoenzyme. |
J Med Chem 41: 2858-71 (1998)
Article DOI: 10.1021/jm980072p
BindingDB Entry DOI: 10.7270/Q2862H4X |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM344408
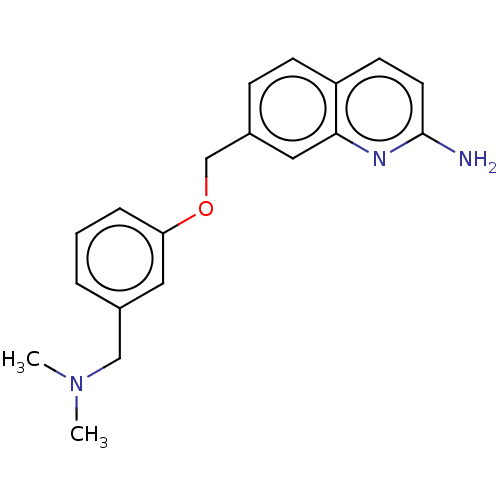
(US9783500, Compound 10 | US9783500, Compound 4)Show InChI InChI=1S/C19H21N3O/c1-22(2)12-14-4-3-5-17(10-14)23-13-15-6-7-16-8-9-19(20)21-18(16)11-15/h3-11H,12-13H2,1-2H3,(H2,20,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| US Patent
| 2.08E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
Rat and human nNOS, murine macrophage iNOS, and human eNOS were recombinant enzymes (expressed in E. coli and purified as reported previously in the ... |
US Patent US9783500 (2017)
BindingDB Entry DOI: 10.7270/Q2930WBP |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM152712
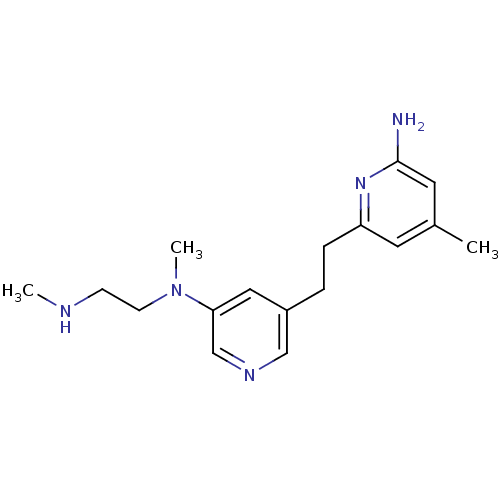
(4-methyl-6-[2-(5-{methyl[2-(methylamino)ethyl]amin...)Show InChI InChI=1S/C17H25N5/c1-13-8-15(21-17(18)9-13)5-4-14-10-16(12-20-11-14)22(3)7-6-19-2/h8-12,19H,4-7H2,1-3H3,(H2,18,21) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 2.15E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Astex
| Assay Description
The NOSs isoform assays involved subjecting 3-8 to an oxyhemoglobin NO capture assay using a Biotek Gen5� microplate reader. IC50 values for each com... |
J Med Chem 52: 379-88 (2009)
BindingDB Entry DOI: 10.7270/Q21N83G1 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50374677

(CHEMBL405231)Show InChI InChI=1S/C6H15N5O2/c7-4(5(12)13)2-1-3-10-6(8)11-9/h4H,1-3,7,9H2,(H,12,13)(H3,8,10,11)/t4-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of California
Curated by ChEMBL
| Assay Description
Inhibition of full length iNOS by high-throughput oxymyoglobin assay |
J Med Chem 51: 924-31 (2008)
Article DOI: 10.1021/jm701119v
BindingDB Entry DOI: 10.7270/Q2639QMP |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50526361
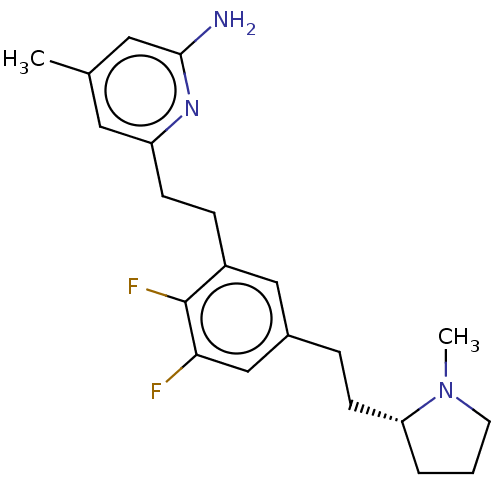
(CHEMBL4532683)Show SMILES CN1CCC[C@@H]1CCc1cc(F)c(F)c(CCc2cc(C)cc(N)n2)c1 |r| Show InChI InChI=1S/C21H27F2N3/c1-14-10-17(25-20(24)11-14)7-6-16-12-15(13-19(22)21(16)23)5-8-18-4-3-9-26(18)2/h10-13,18H,3-9H2,1-2H3,(H2,24,25)/t18-/m1/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
| Article
PubMed
| 2.22E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
Curated by ChEMBL
| Assay Description
Inhibition of human iNOS expressed in Escherichia coli using L-arginine as substrate in presence of human oxyhemoglobin after 6 mins by hemoglobin NO... |
J Med Chem 62: 2690-2707 (2019)
Article DOI: 10.1021/acs.jmedchem.8b02032
BindingDB Entry DOI: 10.7270/Q2H998NM |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50441008

(CHEMBL2430150 | US9732037, Compound 20)Show SMILES C[C@H](OCc1cc(C)cc(N)n1)[C@@H](N)COCc1cc(C)cc(N)n1 |r| Show InChI InChI=1S/C18H27N5O2/c1-11-4-14(22-17(20)6-11)8-24-10-16(19)13(3)25-9-15-5-12(2)7-18(21)23-15/h4-7,13,16H,8-10,19H2,1-3H3,(H2,20,22)(H2,21,23)/t13-,16-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
MMDB
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 2.26E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... |
US Patent US9732037 (2017)
BindingDB Entry DOI: 10.7270/Q26W9D6H |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50446252
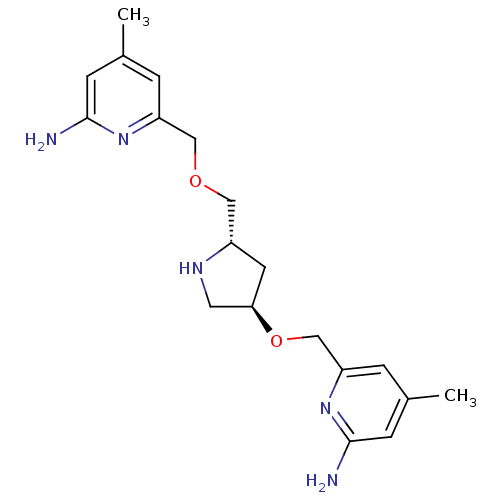
(CHEMBL3109186 | US9732037, Compound 6)Show SMILES Cc1cc(N)nc(COC[C@@H]2C[C@H](CN2)OCc2cc(C)cc(N)n2)c1 |r| Show InChI InChI=1S/C19H27N5O2/c1-12-3-15(23-18(20)5-12)10-25-9-14-7-17(8-22-14)26-11-16-4-13(2)6-19(21)24-16/h3-6,14,17,22H,7-11H2,1-2H3,(H2,20,23)(H2,21,24)/t14-,17+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| US Patent
| 2.27E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... |
US Patent US9732037 (2017)
BindingDB Entry DOI: 10.7270/Q26W9D6H |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50446248
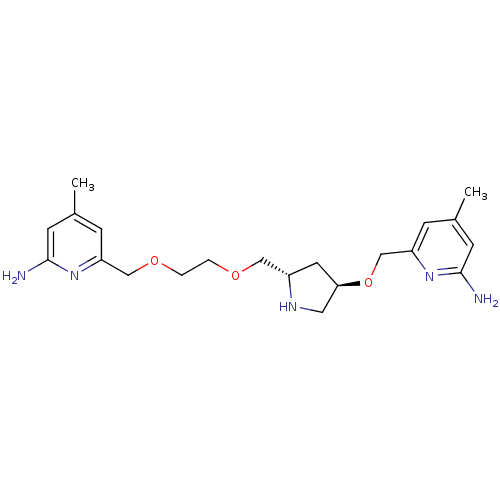
(CHEMBL3109190 | US9732037, Compound 10)Show SMILES Cc1cc(N)nc(COCCOC[C@@H]2C[C@H](CN2)OCc2cc(C)cc(N)n2)c1 |r| Show InChI InChI=1S/C21H31N5O3/c1-14-5-17(25-20(22)7-14)12-28-4-3-27-11-16-9-19(10-24-16)29-13-18-6-15(2)8-21(23)26-18/h5-8,16,19,24H,3-4,9-13H2,1-2H3,(H2,22,25)(H2,23,26)/t16-,19+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| US Patent
| 2.32E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University
US Patent
| Assay Description
NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... |
US Patent US9732037 (2017)
BindingDB Entry DOI: 10.7270/Q26W9D6H |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50058455
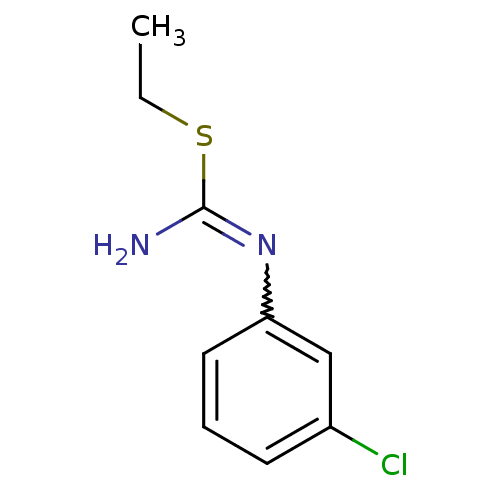
(1-(3-Chloro-phenyl)-2-ethyl-isothiourea; hydrochlo...)Show InChI InChI=1S/C9H11ClN2S/c1-2-13-9(11)12-8-5-3-4-7(10)6-8/h3-6H,2H2,1H3,(H2,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS). |
J Med Chem 40: 1901-5 (1997)
Article DOI: 10.1021/jm960785c
BindingDB Entry DOI: 10.7270/Q2SJ1M94 |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50065828
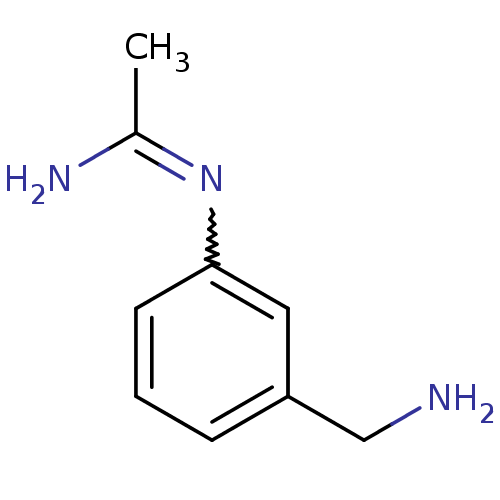
(CHEMBL542678 | N-(3-Aminomethyl-phenyl)-acetamidin...)Show InChI InChI=1S/C9H13N3/c1-7(11)12-9-4-2-3-8(5-9)6-10/h2-5H,6,10H2,1H3,(H2,11,12) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS) isoenzyme. |
J Med Chem 41: 2858-71 (1998)
Article DOI: 10.1021/jm980072p
BindingDB Entry DOI: 10.7270/Q2862H4X |
More data for this
Ligand-Target Pair | |
Nitric oxide synthase, inducible
(Homo sapiens (Human)) | BDBM50058444

(2-Ethyl-1-(4-hydroxy-phenyl)-isothiourea; hydriodi...)Show InChI InChI=1S/C9H12N2OS/c1-2-13-9(10)11-7-3-5-8(12)6-4-7/h3-6,12H,2H2,1H3,(H2,10,11) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Glaxo Wellcome Research and Development
Curated by ChEMBL
| Assay Description
Inhibitory activity against human inducible nitric oxide synthase (iNOS). |
J Med Chem 40: 1901-5 (1997)
Article DOI: 10.1021/jm960785c
BindingDB Entry DOI: 10.7270/Q2SJ1M94 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data