 Found 781 hits with Last Name = 'mcpherson' and Initial = 'k'
Found 781 hits with Last Name = 'mcpherson' and Initial = 'k' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Dipeptidyl peptidase 4
(Rattus norvegicus (rat)) | BDBM50338518
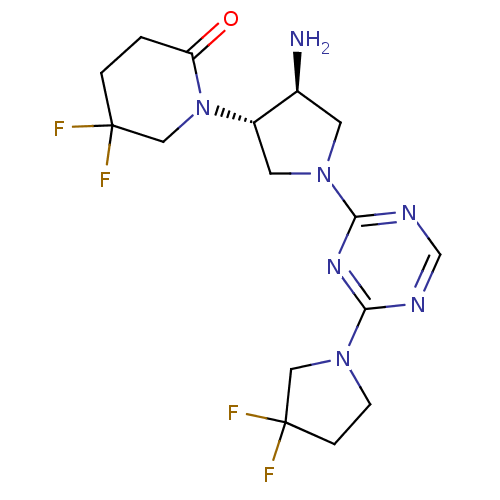
(1-((3S,4S)-4-amino-1-(4-(3,3-difluoropyrrolidin-1-...)Show SMILES N[C@H]1CN(C[C@@H]1N1CC(F)(F)CCC1=O)c1ncnc(n1)N1CCC(F)(F)C1 |r| Show InChI InChI=1S/C16H21F4N7O/c17-15(18)2-1-12(28)27(8-15)11-6-26(5-10(11)21)14-23-9-22-13(24-14)25-4-3-16(19,20)7-25/h9-11H,1-8,21H2/t10-,11-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of DPP4 in rat plasma |
Bioorg Med Chem Lett 21: 1810-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.055
BindingDB Entry DOI: 10.7270/Q28W3DM7 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50338518

(1-((3S,4S)-4-amino-1-(4-(3,3-difluoropyrrolidin-1-...)Show SMILES N[C@H]1CN(C[C@@H]1N1CC(F)(F)CCC1=O)c1ncnc(n1)N1CCC(F)(F)C1 |r| Show InChI InChI=1S/C16H21F4N7O/c17-15(18)2-1-12(28)27(8-15)11-6-26(5-10(11)21)14-23-9-22-13(24-14)25-4-3-16(19,20)7-25/h9-11H,1-8,21H2/t10-,11-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 14 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 21: 1810-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.055
BindingDB Entry DOI: 10.7270/Q28W3DM7 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 3
(Homo sapiens (Human)) | BDBM50338518

(1-((3S,4S)-4-amino-1-(4-(3,3-difluoropyrrolidin-1-...)Show SMILES N[C@H]1CN(C[C@@H]1N1CC(F)(F)CCC1=O)c1ncnc(n1)N1CCC(F)(F)C1 |r| Show InChI InChI=1S/C16H21F4N7O/c17-15(18)2-1-12(28)27(8-15)11-6-26(5-10(11)21)14-23-9-22-13(24-14)25-4-3-16(19,20)7-25/h9-11H,1-8,21H2/t10-,11-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of DPP3 |
Bioorg Med Chem Lett 21: 1810-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.055
BindingDB Entry DOI: 10.7270/Q28W3DM7 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 9
(Homo sapiens (Human)) | BDBM50338518

(1-((3S,4S)-4-amino-1-(4-(3,3-difluoropyrrolidin-1-...)Show SMILES N[C@H]1CN(C[C@@H]1N1CC(F)(F)CCC1=O)c1ncnc(n1)N1CCC(F)(F)C1 |r| Show InChI InChI=1S/C16H21F4N7O/c17-15(18)2-1-12(28)27(8-15)11-6-26(5-10(11)21)14-23-9-22-13(24-14)25-4-3-16(19,20)7-25/h9-11H,1-8,21H2/t10-,11-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of DPP9 |
Bioorg Med Chem Lett 21: 1810-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.055
BindingDB Entry DOI: 10.7270/Q28W3DM7 |
More data for this
Ligand-Target Pair | |
Prolyl endopeptidase
(Homo sapiens (Human)) | BDBM50338518

(1-((3S,4S)-4-amino-1-(4-(3,3-difluoropyrrolidin-1-...)Show SMILES N[C@H]1CN(C[C@@H]1N1CC(F)(F)CCC1=O)c1ncnc(n1)N1CCC(F)(F)C1 |r| Show InChI InChI=1S/C16H21F4N7O/c17-15(18)2-1-12(28)27(8-15)11-6-26(5-10(11)21)14-23-9-22-13(24-14)25-4-3-16(19,20)7-25/h9-11H,1-8,21H2/t10-,11-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of POP |
Bioorg Med Chem Lett 21: 1810-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.055
BindingDB Entry DOI: 10.7270/Q28W3DM7 |
More data for this
Ligand-Target Pair | |
Prolyl endopeptidase FAP
(Homo sapiens (Human)) | BDBM50338518

(1-((3S,4S)-4-amino-1-(4-(3,3-difluoropyrrolidin-1-...)Show SMILES N[C@H]1CN(C[C@@H]1N1CC(F)(F)CCC1=O)c1ncnc(n1)N1CCC(F)(F)C1 |r| Show InChI InChI=1S/C16H21F4N7O/c17-15(18)2-1-12(28)27(8-15)11-6-26(5-10(11)21)14-23-9-22-13(24-14)25-4-3-16(19,20)7-25/h9-11H,1-8,21H2/t10-,11-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of FAP |
Bioorg Med Chem Lett 21: 1810-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.055
BindingDB Entry DOI: 10.7270/Q28W3DM7 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 2
(Homo sapiens (Human)) | BDBM50338518

(1-((3S,4S)-4-amino-1-(4-(3,3-difluoropyrrolidin-1-...)Show SMILES N[C@H]1CN(C[C@@H]1N1CC(F)(F)CCC1=O)c1ncnc(n1)N1CCC(F)(F)C1 |r| Show InChI InChI=1S/C16H21F4N7O/c17-15(18)2-1-12(28)27(8-15)11-6-26(5-10(11)21)14-23-9-22-13(24-14)25-4-3-16(19,20)7-25/h9-11H,1-8,21H2/t10-,11-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of DPP2 |
Bioorg Med Chem Lett 21: 1810-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.055
BindingDB Entry DOI: 10.7270/Q28W3DM7 |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50314930
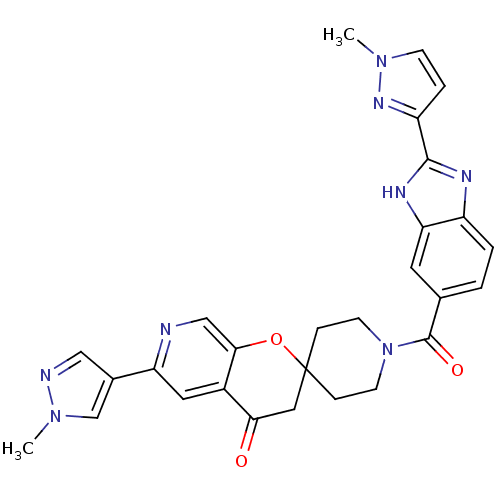
(1-(2-(1-methyl-1H-pyrazol-3-yl)-1H-benzo[d]imidazo...)Show SMILES Cn1ccc(n1)-c1nc2ccc(cc2[nH]1)C(=O)N1CCC2(CC1)CC(=O)c1cc(ncc1O2)-c1cnn(C)c1 Show InChI InChI=1S/C28H26N8O3/c1-34-8-5-21(33-34)26-31-20-4-3-17(11-23(20)32-26)27(38)36-9-6-28(7-10-36)13-24(37)19-12-22(29-15-25(19)39-28)18-14-30-35(2)16-18/h3-5,8,11-12,14-16H,6-7,9-10,13H2,1-2H3,(H,31,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.210 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACC2 expressed in CHO cells after 1 hr by fluorescence reader |
Bioorg Med Chem Lett 20: 2383-8 (2010)
Article DOI: 10.1016/j.bmcl.2009.04.091
BindingDB Entry DOI: 10.7270/Q22N52DW |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50314931
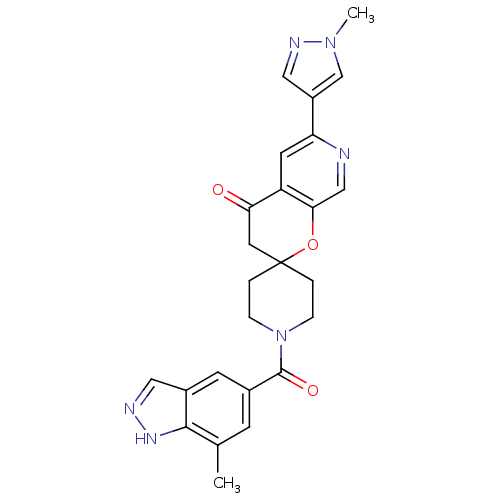
(1-(7-methyl-1H-indazole-5-carbonyl)-6'-(1-methyl-1...)Show SMILES Cc1cc(cc2cn[nH]c12)C(=O)N1CCC2(CC1)CC(=O)c1cc(ncc1O2)-c1cnn(C)c1 Show InChI InChI=1S/C25H24N6O3/c1-15-7-16(8-17-11-27-29-23(15)17)24(33)31-5-3-25(4-6-31)10-21(32)19-9-20(26-13-22(19)34-25)18-12-28-30(2)14-18/h7-9,11-14H,3-6,10H2,1-2H3,(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACC2 expressed in CHO cells after 1 hr by fluorescence reader |
Bioorg Med Chem Lett 20: 2383-8 (2010)
Article DOI: 10.1016/j.bmcl.2009.04.091
BindingDB Entry DOI: 10.7270/Q22N52DW |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50314932
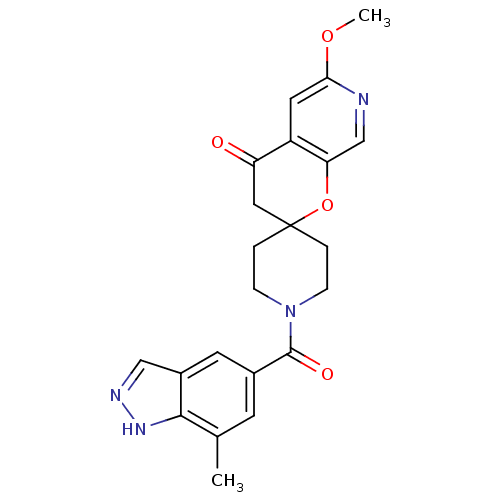
(6'-methoxy-1-(7-methyl-1H-indazole-5-carbonyl)spir...)Show SMILES COc1cc2C(=O)CC3(CCN(CC3)C(=O)c3cc(C)c4[nH]ncc4c3)Oc2cn1 Show InChI InChI=1S/C22H22N4O4/c1-13-7-14(8-15-11-24-25-20(13)15)21(28)26-5-3-22(4-6-26)10-17(27)16-9-19(29-2)23-12-18(16)30-22/h7-9,11-12H,3-6,10H2,1-2H3,(H,24,25) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.310 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACC2 expressed in CHO cells after 1 hr by fluorescence reader |
Bioorg Med Chem Lett 20: 2383-8 (2010)
Article DOI: 10.1016/j.bmcl.2009.04.091
BindingDB Entry DOI: 10.7270/Q22N52DW |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226096
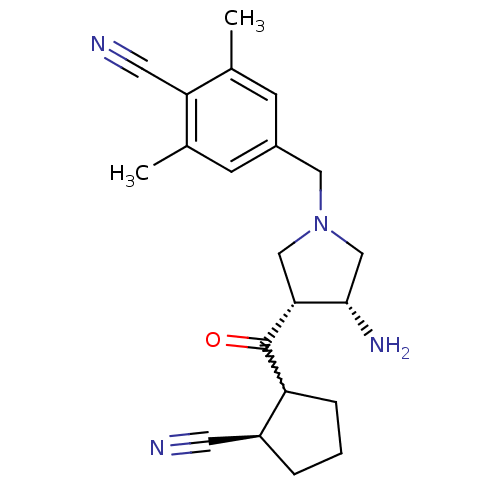
(4-(((3R,4R)-3-amino-4-((2R)-2-cyanocyclopentanecar...)Show SMILES Cc1cc(CN2C[C@H](N)[C@@H](C2)C(=O)C2CCC[C@H]2C#N)cc(C)c1C#N |w:13.13| Show InChI InChI=1S/C21H26N4O/c1-13-6-15(7-14(2)18(13)9-23)10-25-11-19(20(24)12-25)21(26)17-5-3-4-16(17)8-22/h6-7,16-17,19-20H,3-5,10-12,24H2,1-2H3/t16-,17?,19+,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226064
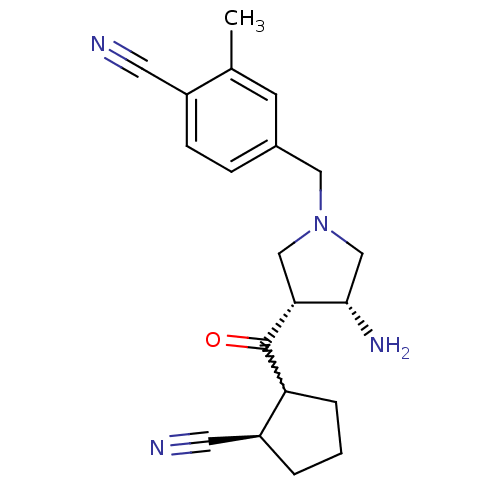
(4-(((3R,4R)-3-amino-4-((2R)-2-cyanocyclopentanecar...)Show SMILES Cc1cc(CN2C[C@H](N)[C@@H](C2)C(=O)C2CCC[C@H]2C#N)ccc1C#N |w:13.13| Show InChI InChI=1S/C20H24N4O/c1-13-7-14(5-6-15(13)8-21)10-24-11-18(19(23)12-24)20(25)17-4-2-3-16(17)9-22/h5-7,16-19H,2-4,10-12,23H2,1H3/t16-,17?,18+,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50338507
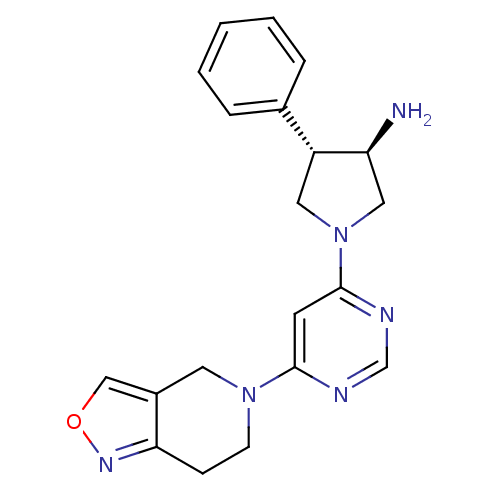
((3R,4S)-1-(6-(6,7-dihydroisoxazolo[4,3-c]pyridin-5...)Show SMILES N[C@H]1CN(C[C@@H]1c1ccccc1)c1cc(ncn1)N1CCc2nocc2C1 |r| Show InChI InChI=1S/C20H22N6O/c21-17-11-26(10-16(17)14-4-2-1-3-5-14)20-8-19(22-13-23-20)25-7-6-18-15(9-25)12-27-24-18/h1-5,8,12-13,16-17H,6-7,9-11,21H2/t16-,17+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 21: 1810-4 (2011)
Article DOI: 10.1016/j.bmcl.2011.01.055
BindingDB Entry DOI: 10.7270/Q28W3DM7 |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50439646

(CHEMBL2419600 | US8993586, 110)Show SMILES CCCNc1ccc2ccc(cc2n1)C(=O)N1CCC2(CC1)Cc1cn(nc1C(=O)N2)C(C)(C)C Show InChI InChI=1S/C27H34N6O2/c1-5-12-28-22-9-8-18-6-7-19(15-21(18)29-22)25(35)32-13-10-27(11-14-32)16-20-17-33(26(2,3)4)31-23(20)24(34)30-27/h6-9,15,17H,5,10-14,16H2,1-4H3,(H,28,29)(H,30,34) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human ACC2 using acetyl-CoA as substrate assessed as [14C]malonyl-CoA synthesis preincubated for 10 mins prior to substrate addition me... |
J Med Chem 56: 7110-9 (2013)
Article DOI: 10.1021/jm401033t
BindingDB Entry DOI: 10.7270/Q2JW8G9D |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50439646

(CHEMBL2419600 | US8993586, 110)Show SMILES CCCNc1ccc2ccc(cc2n1)C(=O)N1CCC2(CC1)Cc1cn(nc1C(=O)N2)C(C)(C)C Show InChI InChI=1S/C27H34N6O2/c1-5-12-28-22-9-8-18-6-7-19(15-21(18)29-22)25(35)32-13-10-27(11-14-32)16-20-17-33(26(2,3)4)31-23(20)24(34)30-27/h6-9,15,17H,5,10-14,16H2,1-4H3,(H,28,29)(H,30,34) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human ACC1 using acetyl-CoA as substrate assessed as [14C]malonyl-CoA synthesis preincubated for 10 mins prior to substrate addition me... |
J Med Chem 56: 7110-9 (2013)
Article DOI: 10.1021/jm401033t
BindingDB Entry DOI: 10.7270/Q2JW8G9D |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50118009

((6,7-Diethoxy-quinazolin-4-yl)-(3-thiazol-2-yl-phe...)Show InChI InChI=1S/C21H20N4O2S/c1-3-26-18-11-16-17(12-19(18)27-4-2)23-13-24-20(16)25-15-7-5-6-14(10-15)21-22-8-9-28-21/h5-13H,3-4H2,1-2H3,(H,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Central Research
Curated by ChEMBL
| Assay Description
Inhibition of (EGFR) epidermal growth factor receptor tyrosine kinase |
J Med Chem 45: 3865-77 (2002)
BindingDB Entry DOI: 10.7270/Q2PN94Z0 |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50439642
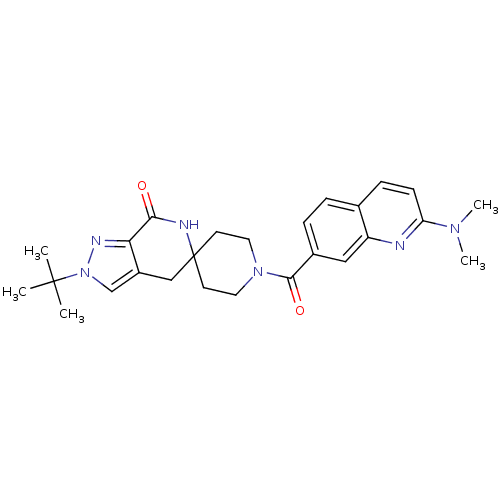
(CHEMBL2419589 | US8993586, 105)Show SMILES CN(C)c1ccc2ccc(cc2n1)C(=O)N1CCC2(CC1)Cc1cn(nc1C(=O)N2)C(C)(C)C Show InChI InChI=1S/C26H32N6O2/c1-25(2,3)32-16-19-15-26(28-23(33)22(19)29-32)10-12-31(13-11-26)24(34)18-7-6-17-8-9-21(30(4)5)27-20(17)14-18/h6-9,14,16H,10-13,15H2,1-5H3,(H,28,33) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human ACC2 using acetyl-CoA as substrate assessed as [14C]malonyl-CoA synthesis preincubated for 10 mins prior to substrate addition me... |
J Med Chem 56: 7110-9 (2013)
Article DOI: 10.1021/jm401033t
BindingDB Entry DOI: 10.7270/Q2JW8G9D |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226086

((S)-1-((3R,4R)-1-(4-cyanobenzyl)-3-aminopyrrolidin...)Show SMILES N[C@H]1CN(Cc2ccc(cc2)C#N)C[C@H]1C(=O)N1CCC[C@H]1C#N Show InChI InChI=1S/C18H21N5O/c19-8-13-3-5-14(6-4-13)10-22-11-16(17(21)12-22)18(24)23-7-1-2-15(23)9-20/h3-6,15-17H,1-2,7,10-12,21H2/t15-,16+,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50117952
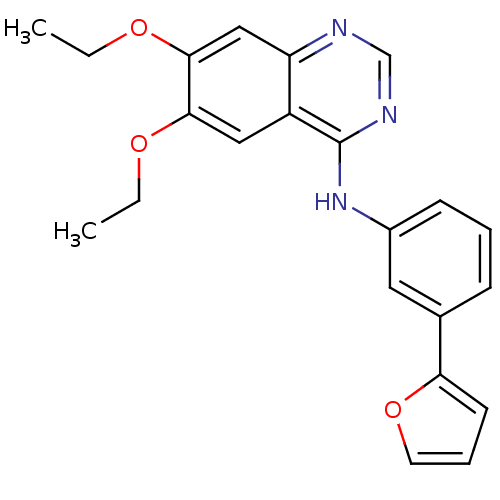
((6,7-Diethoxy-quinazolin-4-yl)-(3-furan-2-yl-pheny...)Show InChI InChI=1S/C22H21N3O3/c1-3-26-20-12-17-18(13-21(20)27-4-2)23-14-24-22(17)25-16-8-5-7-15(11-16)19-9-6-10-28-19/h5-14H,3-4H2,1-2H3,(H,23,24,25) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Central Research
Curated by ChEMBL
| Assay Description
Inhibition of (EGFR) epidermal growth factor receptor tyrosine kinase |
J Med Chem 45: 3865-77 (2002)
BindingDB Entry DOI: 10.7270/Q2PN94Z0 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226077
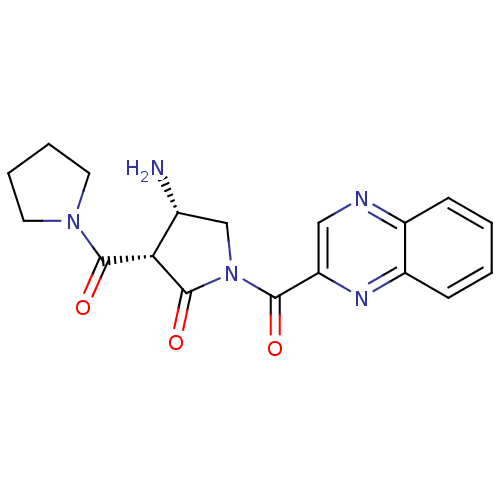
((3S,4S)-4-amino-3-(pyrrolidine-1-carbonyl)-1-(quin...)Show SMILES N[C@@H]1CN(C(=O)c2cnc3ccccc3n2)C(=O)[C@@H]1C(=O)N1CCCC1 Show InChI InChI=1S/C18H19N5O3/c19-11-10-23(18(26)15(11)17(25)22-7-3-4-8-22)16(24)14-9-20-12-5-1-2-6-13(12)21-14/h1-2,5-6,9,11,15H,3-4,7-8,10,19H2/t11-,15+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226093

((1R)-2-((3R,4R)-1-(1-(4-(1H-1,2,4-triazol-1-yl)phe...)Show SMILES CC(N1C[C@H](N)[C@@H](C1)C(=O)C1CCC[C@H]1C#N)c1ccc(cc1)-n1cncn1 |w:10.10,1.0| Show InChI InChI=1S/C21H26N6O/c1-14(15-5-7-17(8-6-15)27-13-24-12-25-27)26-10-19(20(23)11-26)21(28)18-4-2-3-16(18)9-22/h5-8,12-14,16,18-20H,2-4,10-11,23H2,1H3/t14?,16-,18?,19+,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50439644
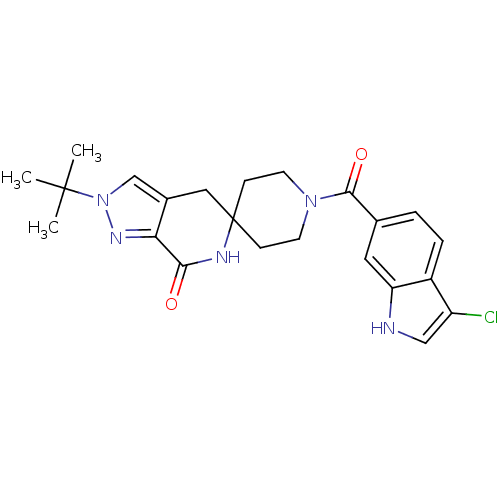
(CHEMBL2419593 | US8993586, 86)Show SMILES CC(C)(C)n1cc2CC3(CCN(CC3)C(=O)c3ccc4c(Cl)c[nH]c4c3)NC(=O)c2n1 Show InChI InChI=1S/C23H26ClN5O2/c1-22(2,3)29-13-15-11-23(26-20(30)19(15)27-29)6-8-28(9-7-23)21(31)14-4-5-16-17(24)12-25-18(16)10-14/h4-5,10,12-13,25H,6-9,11H2,1-3H3,(H,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human ACC2 using acetyl-CoA as substrate assessed as [14C]malonyl-CoA synthesis preincubated for 10 mins prior to substrate addition me... |
J Med Chem 56: 7110-9 (2013)
Article DOI: 10.1021/jm401033t
BindingDB Entry DOI: 10.7270/Q2JW8G9D |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226065
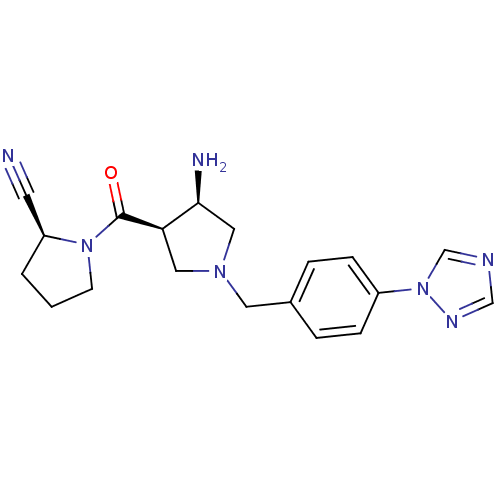
((S)-1-((3R,4R)-1-(4-(1H-1,2,4-triazol-1-yl)benzyl)...)Show SMILES N[C@H]1CN(Cc2ccc(cc2)-n2cncn2)C[C@H]1C(=O)N1CCC[C@H]1C#N Show InChI InChI=1S/C19H23N7O/c20-8-16-2-1-7-25(16)19(27)17-10-24(11-18(17)21)9-14-3-5-15(6-4-14)26-13-22-12-23-26/h3-6,12-13,16-18H,1-2,7,9-11,21H2/t16-,17+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226067
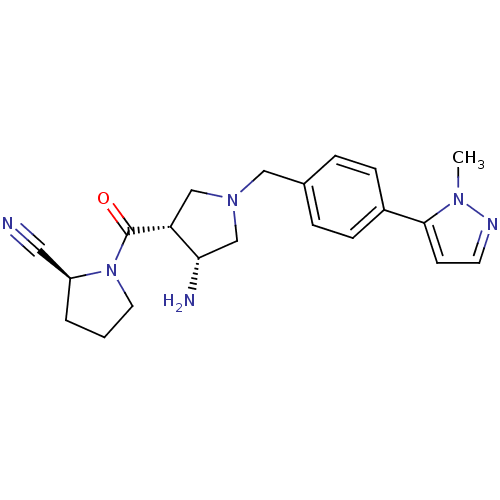
((S)-1-((3R,4R)-1-(4-(1-methyl-1H-pyrazol-5-yl)benz...)Show SMILES Cn1nccc1-c1ccc(CN2C[C@H](N)[C@@H](C2)C(=O)N2CCC[C@H]2C#N)cc1 Show InChI InChI=1S/C21H26N6O/c1-25-20(8-9-24-25)16-6-4-15(5-7-16)12-26-13-18(19(23)14-26)21(28)27-10-2-3-17(27)11-22/h4-9,17-19H,2-3,10,12-14,23H2,1H3/t17-,18+,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226078

(1-((3R,4R)-3-amino-4-((2R)-2-cyanocyclopentanecarb...)Show SMILES N[C@H]1CN(C[C@H]1C(=O)C1CCC[C@H]1C#N)C1CCc2cc(ccc12)C#N |w:8.8,15.16| Show InChI InChI=1S/C21H24N4O/c22-9-13-4-6-16-14(8-13)5-7-20(16)25-11-18(19(24)12-25)21(26)17-3-1-2-15(17)10-23/h4,6,8,15,17-20H,1-3,5,7,11-12,24H2/t15-,17?,18+,19-,20?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226074
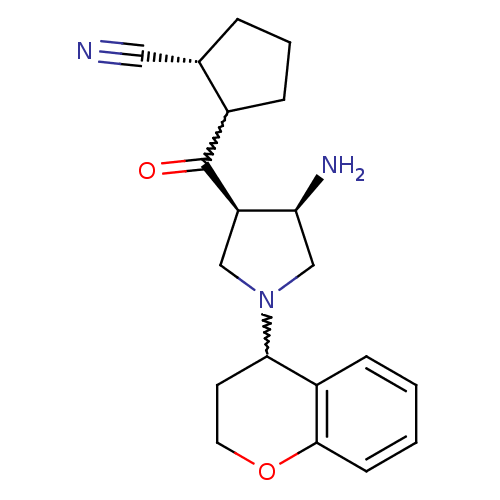
((1R)-2-((3R,4R)-3-amino-1-(chroman-4-yl)pyrrolidin...)Show SMILES N[C@H]1CN(C[C@H]1C(=O)C1CCC[C@H]1C#N)C1CCOc2ccccc12 |w:8.8,15.16| Show InChI InChI=1S/C20H25N3O2/c21-10-13-4-3-6-14(13)20(24)16-11-23(12-17(16)22)18-8-9-25-19-7-2-1-5-15(18)19/h1-2,5,7,13-14,16-18H,3-4,6,8-9,11-12,22H2/t13-,14?,16+,17-,18?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226084
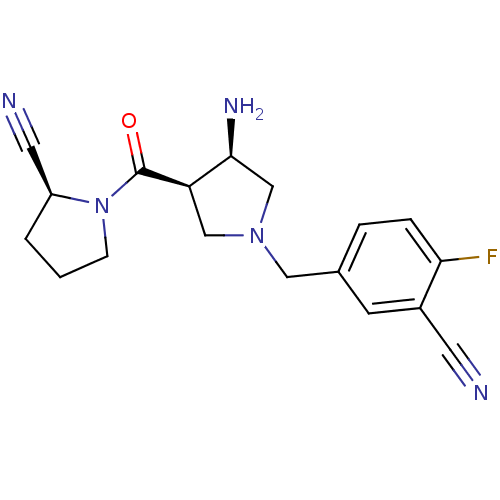
((S)-1-((3R,4R)-1-(3-cyano-4-fluorobenzyl)-3-aminop...)Show SMILES N[C@H]1CN(Cc2ccc(F)c(c2)C#N)C[C@H]1C(=O)N1CCC[C@H]1C#N Show InChI InChI=1S/C18H20FN5O/c19-16-4-3-12(6-13(16)7-20)9-23-10-15(17(22)11-23)18(25)24-5-1-2-14(24)8-21/h3-4,6,14-15,17H,1-2,5,9-11,22H2/t14-,15+,17-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226076
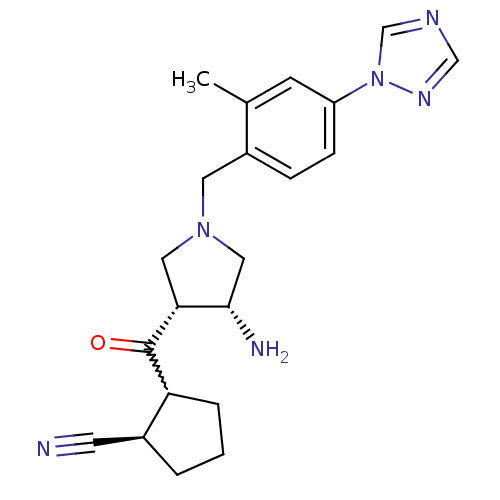
((1R)-2-((3R,4R)-1-(2-methyl-4-(1H-1,2,4-triazol-1-...)Show SMILES Cc1cc(ccc1CN1C[C@H](N)[C@@H](C1)C(=O)C1CCC[C@H]1C#N)-n1cncn1 |w:16.17| Show InChI InChI=1S/C21H26N6O/c1-14-7-17(27-13-24-12-25-27)6-5-16(14)9-26-10-19(20(23)11-26)21(28)18-4-2-3-15(18)8-22/h5-7,12-13,15,18-20H,2-4,9-11,23H2,1H3/t15-,18?,19+,20-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 3.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50095256

((6,7-Dimethoxy-quinazolin-4-yl)-(3-ethynyl-4-fluor...)Show InChI InChI=1S/C18H14FN3O2/c1-4-11-7-12(5-6-14(11)19)22-18-13-8-16(23-2)17(24-3)9-15(13)20-10-21-18/h1,5-10H,2-3H3,(H,20,21,22) | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Central Research
Curated by ChEMBL
| Assay Description
Inhibition of (EGFR) epidermal growth factor receptor tyrosine kinase |
J Med Chem 45: 3865-77 (2002)
BindingDB Entry DOI: 10.7270/Q2PN94Z0 |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50439634
(CHEMBL2419596 | US8993586, 71)Show SMILES COc1ccc2ccc(cc2n1)C(=O)N1CCC2(CC1)Cc1cn(nc1C(=O)N2)C(C)(C)C Show InChI InChI=1S/C25H29N5O3/c1-24(2,3)30-15-18-14-25(27-22(31)21(18)28-30)9-11-29(12-10-25)23(32)17-6-5-16-7-8-20(33-4)26-19(16)13-17/h5-8,13,15H,9-12,14H2,1-4H3,(H,27,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human ACC2 using acetyl-CoA as substrate assessed as [14C]malonyl-CoA synthesis preincubated for 10 mins prior to substrate addition me... |
J Med Chem 56: 7110-9 (2013)
Article DOI: 10.1021/jm401033t
BindingDB Entry DOI: 10.7270/Q2JW8G9D |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50278106
((R)-(4-(1-amino-2-(2,4,5-trifluorophenyl)ethyl)pip...)Show SMILES Cc1cc2ncc(C(=O)N3CCC(CC3)[C@H](N)Cc3cc(F)c(F)cc3F)c(C)n2n1 |r| Show InChI InChI=1S/C22H24F3N5O/c1-12-7-21-27-11-16(13(2)30(21)28-12)22(31)29-5-3-14(4-6-29)20(26)9-15-8-18(24)19(25)10-17(15)23/h7-8,10-11,14,20H,3-6,9,26H2,1-2H3/t20-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 19: 2220-3 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.099
BindingDB Entry DOI: 10.7270/Q2RN37QF |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50439641
(CHEMBL2419597 | US8993586, 55)Show SMILES COc1nccc2ccc(cc12)C(=O)N1CCC2(CC1)Cc1cn(nc1C(=O)N2)C(C)(C)C Show InChI InChI=1S/C25H29N5O3/c1-24(2,3)30-15-18-14-25(27-21(31)20(18)28-30)8-11-29(12-9-25)23(32)17-6-5-16-7-10-26-22(33-4)19(16)13-17/h5-7,10,13,15H,8-9,11-12,14H2,1-4H3,(H,27,31) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human ACC2 using acetyl-CoA as substrate assessed as [14C]malonyl-CoA synthesis preincubated for 10 mins prior to substrate addition me... |
J Med Chem 56: 7110-9 (2013)
Article DOI: 10.1021/jm401033t
BindingDB Entry DOI: 10.7270/Q2JW8G9D |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50439643
(CHEMBL2419598 | US8993586, 76)Show SMILES CNc1ccc2ccc(cc2n1)C(=O)N1CCC2(CC1)Cc1cn(nc1C(=O)N2)C(C)(C)C Show InChI InChI=1S/C25H30N6O2/c1-24(2,3)31-15-18-14-25(28-22(32)21(18)29-31)9-11-30(12-10-25)23(33)17-6-5-16-7-8-20(26-4)27-19(16)13-17/h5-8,13,15H,9-12,14H2,1-4H3,(H,26,27)(H,28,32) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human ACC2 using acetyl-CoA as substrate assessed as [14C]malonyl-CoA synthesis preincubated for 10 mins prior to substrate addition me... |
J Med Chem 56: 7110-9 (2013)
Article DOI: 10.1021/jm401033t
BindingDB Entry DOI: 10.7270/Q2JW8G9D |
More data for this
Ligand-Target Pair | |
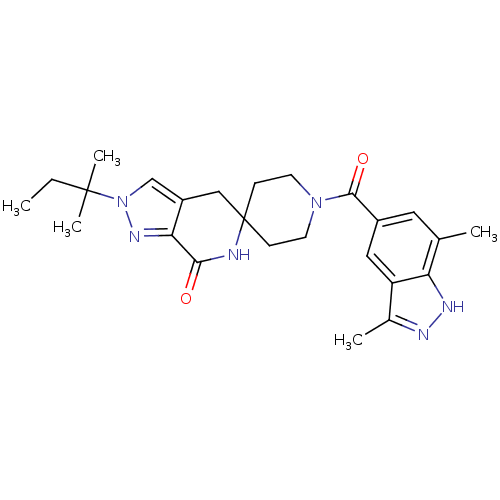
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50439645
(CHEMBL2419607)Show SMILES CCC(C)(C)n1cc2CC3(CCN(CC3)C(=O)c3cc(C)c4[nH]nc(C)c4c3)NC(=O)c2n1 Show InChI InChI=1S/C25H32N6O2/c1-6-24(4,5)31-14-18-13-25(26-22(32)21(18)29-31)7-9-30(10-8-25)23(33)17-11-15(2)20-19(12-17)16(3)27-28-20/h11-12,14H,6-10,13H2,1-5H3,(H,26,32)(H,27,28) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human ACC2 using acetyl-CoA as substrate assessed as [14C]malonyl-CoA synthesis preincubated for 10 mins prior to substrate addition me... |
J Med Chem 56: 7110-9 (2013)
Article DOI: 10.1021/jm401033t
BindingDB Entry DOI: 10.7270/Q2JW8G9D |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50314905
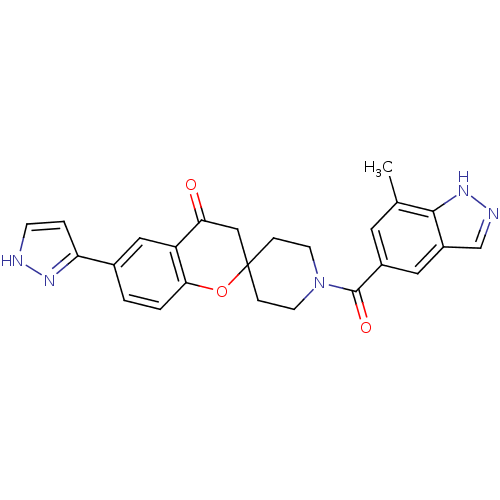
(1'-(7-methyl-1H-indazole-5-carbonyl)-6-(1H-pyrazol...)Show SMILES Cc1cc(cc2cn[nH]c12)C(=O)N1CCC2(CC1)CC(=O)c1cc(ccc1O2)-c1cc[nH]n1 Show InChI InChI=1S/C25H23N5O3/c1-15-10-17(11-18-14-27-29-23(15)18)24(32)30-8-5-25(6-9-30)13-21(31)19-12-16(2-3-22(19)33-25)20-4-7-26-28-20/h2-4,7,10-12,14H,5-6,8-9,13H2,1H3,(H,26,28)(H,27,29) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACC2 expressed in CHO cells after 1 hr by fluorescence reader |
Bioorg Med Chem Lett 20: 2383-8 (2010)
Article DOI: 10.1016/j.bmcl.2009.04.091
BindingDB Entry DOI: 10.7270/Q22N52DW |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50314938
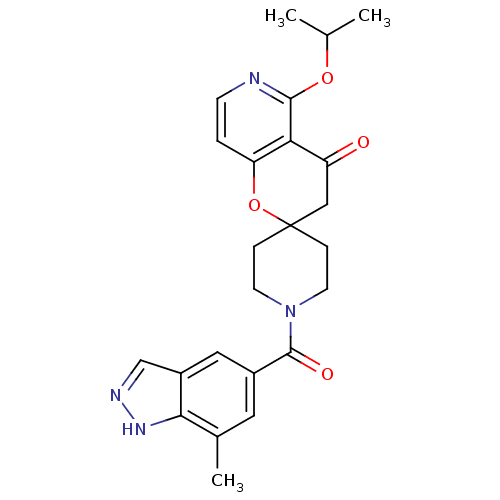
(5'-isopropoxy-1-(7-methyl-1H-indazole-5-carbonyl)s...)Show SMILES CC(C)Oc1nccc2OC3(CCN(CC3)C(=O)c3cc(C)c4[nH]ncc4c3)CC(=O)c12 Show InChI InChI=1S/C24H26N4O4/c1-14(2)31-22-20-18(29)12-24(32-19(20)4-7-25-22)5-8-28(9-6-24)23(30)16-10-15(3)21-17(11-16)13-26-27-21/h4,7,10-11,13-14H,5-6,8-9,12H2,1-3H3,(H,26,27) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant ACC2 expressed in CHO cells after 1 hr by fluorescence reader |
Bioorg Med Chem Lett 20: 2383-8 (2010)
Article DOI: 10.1016/j.bmcl.2009.04.091
BindingDB Entry DOI: 10.7270/Q22N52DW |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226102

(4-(((3R,4R)-3-amino-4-((2R)-2-cyanocyclopentanecar...)Show SMILES CCc1cc(CN2C[C@H](N)[C@@H](C2)C(=O)C2CCC[C@H]2C#N)cc(CC)c1C#N |w:14.14| Show InChI InChI=1S/C23H30N4O/c1-3-16-8-15(9-17(4-2)20(16)11-25)12-27-13-21(22(26)14-27)23(28)19-7-5-6-18(19)10-24/h8-9,18-19,21-22H,3-7,12-14,26H2,1-2H3/t18-,19?,21+,22-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50249516
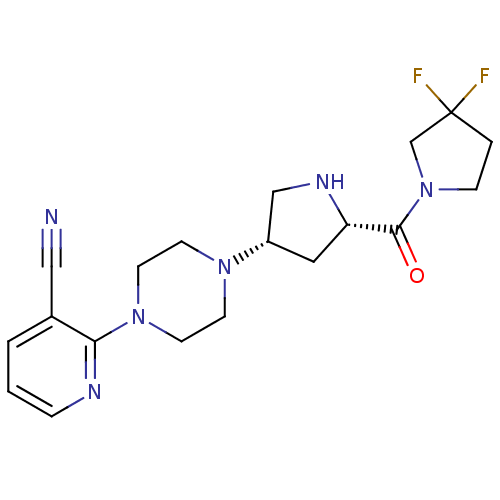
(CHEMBL514011 | cis-2-(4-((3S,5S)-5-(3,3-difluoropy...)Show SMILES FC1(F)CCN(C1)C(=O)[C@@H]1C[C@@H](CN1)N1CCN(CC1)c1ncccc1C#N |r| Show InChI InChI=1S/C19H24F2N6O/c20-19(21)3-5-27(13-19)18(28)16-10-15(12-24-16)25-6-8-26(9-7-25)17-14(11-22)2-1-4-23-17/h1-2,4,15-16,24H,3,5-10,12-13H2/t15-,16-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 19: 1991-5 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.041
BindingDB Entry DOI: 10.7270/Q2FB52S4 |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50439642

(CHEMBL2419589 | US8993586, 105)Show SMILES CN(C)c1ccc2ccc(cc2n1)C(=O)N1CCC2(CC1)Cc1cn(nc1C(=O)N2)C(C)(C)C Show InChI InChI=1S/C26H32N6O2/c1-25(2,3)32-16-19-15-26(28-23(33)22(19)29-32)10-12-31(13-11-26)24(34)18-7-6-17-8-9-21(30(4)5)27-20(17)14-18/h6-9,14,16H,10-13,15H2,1-5H3,(H,28,33) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human ACC1 using acetyl-CoA as substrate assessed as [14C]malonyl-CoA synthesis preincubated for 10 mins prior to substrate addition me... |
J Med Chem 56: 7110-9 (2013)
Article DOI: 10.1021/jm401033t
BindingDB Entry DOI: 10.7270/Q2JW8G9D |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM15494
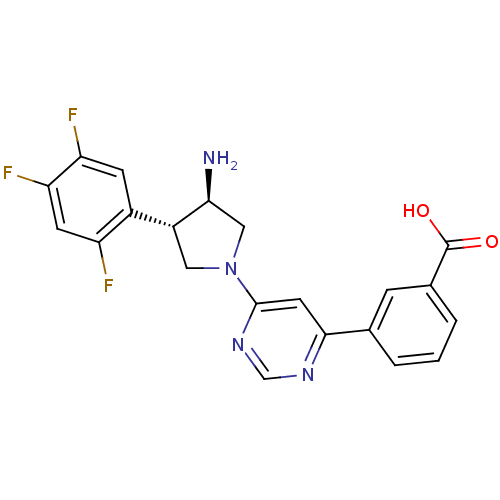
(3-{6-[(3R,4S)-3-amino-4-(2,4,5-trifluorophenyl)pyr...)Show SMILES N[C@H]1CN(C[C@@H]1c1cc(F)c(F)cc1F)c1cc(ncn1)-c1cccc(c1)C(O)=O |r| Show InChI InChI=1S/C21H17F3N4O2/c22-15-6-17(24)16(23)5-13(15)14-8-28(9-18(14)25)20-7-19(26-10-27-20)11-2-1-3-12(4-11)21(29)30/h1-7,10,14,18H,8-9,25H2,(H,29,30)/t14-,18+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human recombinant DPP4 |
Bioorg Med Chem Lett 17: 5638-42 (2007)
Article DOI: 10.1016/j.bmcl.2007.07.081
BindingDB Entry DOI: 10.7270/Q2319WRX |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 2
(Homo sapiens (Human)) | BDBM50439635
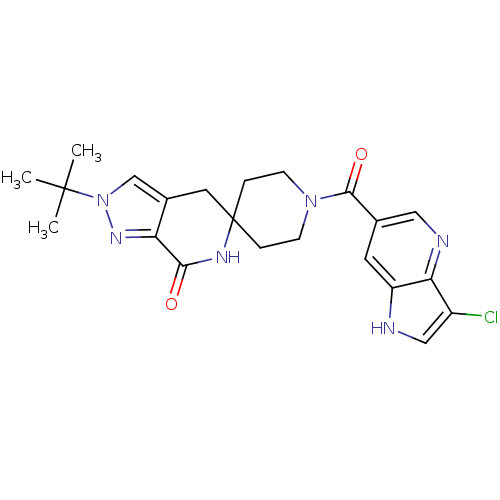
(CHEMBL2419594 | US8993586, 88)Show SMILES CC(C)(C)n1cc2CC3(CCN(CC3)C(=O)c3cnc4c(Cl)c[nH]c4c3)NC(=O)c2n1 Show InChI InChI=1S/C22H25ClN6O2/c1-21(2,3)29-12-14-9-22(26-19(30)17(14)27-29)4-6-28(7-5-22)20(31)13-8-16-18(25-10-13)15(23)11-24-16/h8,10-12,24H,4-7,9H2,1-3H3,(H,26,30) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human ACC2 using acetyl-CoA as substrate assessed as [14C]malonyl-CoA synthesis preincubated for 10 mins prior to substrate addition me... |
J Med Chem 56: 7110-9 (2013)
Article DOI: 10.1021/jm401033t
BindingDB Entry DOI: 10.7270/Q2JW8G9D |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50249515
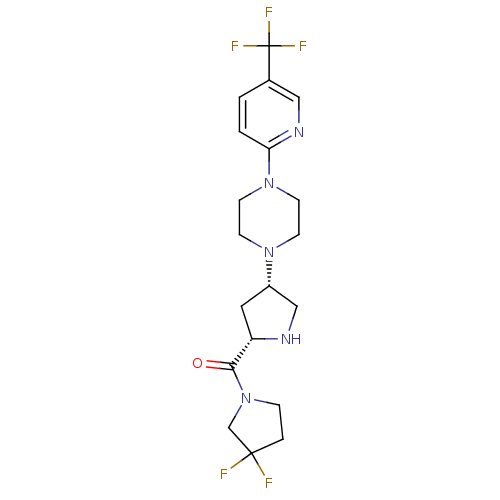
(CHEMBL474410 | cis-(3,3-difluoropyrrolidin-1-yl)((...)Show SMILES FC(F)(F)c1ccc(nc1)N1CCN(CC1)[C@@H]1CN[C@@H](C1)C(=O)N1CCC(F)(F)C1 |r| Show InChI InChI=1S/C19H24F5N5O/c20-18(21)3-4-29(12-18)17(30)15-9-14(11-25-15)27-5-7-28(8-6-27)16-2-1-13(10-26-16)19(22,23)24/h1-2,10,14-15,25H,3-9,11-12H2/t14-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 19: 1991-5 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.041
BindingDB Entry DOI: 10.7270/Q2FB52S4 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50249548
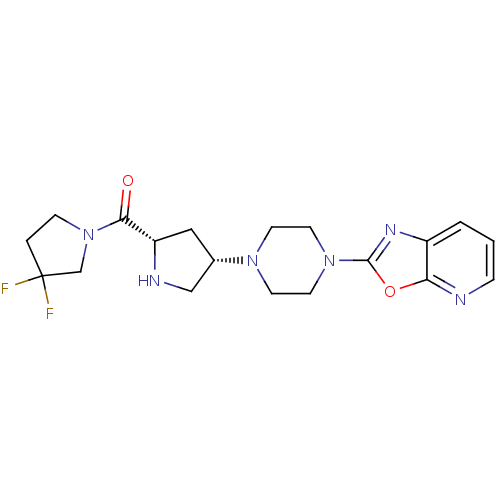
(CHEMBL475586 | cis-(3,3-difluoropyrrolidin-1-yl)((...)Show SMILES FC1(F)CCN(C1)C(=O)[C@@H]1C[C@@H](CN1)N1CCN(CC1)c1nc2cccnc2o1 |r| Show InChI InChI=1S/C19H24F2N6O2/c20-19(21)3-5-27(12-19)17(28)15-10-13(11-23-15)25-6-8-26(9-7-25)18-24-14-2-1-4-22-16(14)29-18/h1-2,4,13,15,23H,3,5-12H2/t13-,15-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 19: 1991-5 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.041
BindingDB Entry DOI: 10.7270/Q2FB52S4 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226092
((1R,3S)-2-((3R,4R)-1-(4-(5-methyl-1,3,4-oxadiazol-...)Show SMILES Cc1nnc(o1)-c1ccc(CN2C[C@H](N)[C@@H](C2)C(=O)C2[C@@H](F)CC[C@H]2C#N)cc1 |w:19.20| Show InChI InChI=1S/C21H24FN5O2/c1-12-25-26-21(29-12)14-4-2-13(3-5-14)9-27-10-16(18(24)11-27)20(28)19-15(8-23)6-7-17(19)22/h2-5,15-19H,6-7,9-11,24H2,1H3/t15-,16+,17-,18-,19?/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226066

((S)-1-((3R,4R)-1-(4-(3-methyl-1H-1,2,4-triazol-1-y...)Show SMILES Cc1ncn(n1)-c1ccc(CN2C[C@H](N)[C@@H](C2)C(=O)N2CCC[C@H]2C#N)cc1 Show InChI InChI=1S/C20H25N7O/c1-14-23-13-27(24-14)16-6-4-15(5-7-16)10-25-11-18(19(22)12-25)20(28)26-8-2-3-17(26)9-21/h4-7,13,17-19H,2-3,8,10-12,22H2,1H3/t17-,18+,19-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226075

((S)-1-((3R,4R)-1-(4-(2H-1,2,3-triazol-2-yl)benzyl)...)Show SMILES N[C@H]1CN(Cc2ccc(cc2)-n2nccn2)C[C@H]1C(=O)N1CCC[C@H]1C#N Show InChI InChI=1S/C19H23N7O/c20-10-16-2-1-9-25(16)19(27)17-12-24(13-18(17)21)11-14-3-5-15(6-4-14)26-22-7-8-23-26/h3-8,16-18H,1-2,9,11-13,21H2/t16-,17+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50226088

((S)-1-((3R,4R)-3-amino-1-(1-(4-cyanophenyl)ethyl)p...)Show SMILES CC(N1C[C@H](N)[C@@H](C1)C(=O)N1CCC[C@H]1C#N)c1ccc(cc1)C#N |w:1.0| Show InChI InChI=1S/C19H23N5O/c1-13(15-6-4-14(9-20)5-7-15)23-11-17(18(22)12-23)19(25)24-8-2-3-16(24)10-21/h4-7,13,16-18H,2-3,8,11-12,22H2,1H3/t13?,16-,17+,18-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development
Curated by ChEMBL
| Assay Description
Inhibition of human DPP4 |
Bioorg Med Chem Lett 17: 6707-13 (2007)
Article DOI: 10.1016/j.bmcl.2007.10.063
BindingDB Entry DOI: 10.7270/Q2ZS2W8B |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50249514

(CHEMBL474409 | cis-(3,3-difluoropyrrolidin-1-yl)((...)Show SMILES FC1(F)CCN(C1)C(=O)[C@@H]1C[C@@H](CN1)N1CCN(CC1)c1cnccn1 |r| Show InChI InChI=1S/C17H24F2N6O/c18-17(19)1-4-25(12-17)16(26)14-9-13(10-22-14)23-5-7-24(8-6-23)15-11-20-2-3-21-15/h2-3,11,13-14,22H,1,4-10,12H2/t13-,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 5.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 19: 1991-5 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.041
BindingDB Entry DOI: 10.7270/Q2FB52S4 |
More data for this
Ligand-Target Pair | |
Dipeptidyl peptidase 4
(Homo sapiens (Human)) | BDBM50249513
(CHEMBL475574 | cis-(3,3-difluoropyrrolidin-1-yl)((...)Show SMILES FC1(F)CCN(C1)C(=O)[C@@H]1C[C@@H](CN1)N1CCN(CC1)c1nc2cnccc2o1 |r| Show InChI InChI=1S/C19H24F2N6O2/c20-19(21)2-4-27(12-19)17(28)14-9-13(10-23-14)25-5-7-26(8-6-25)18-24-15-11-22-3-1-16(15)29-18/h1,3,11,13-14,23H,2,4-10,12H2/t13-,14-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research & Development
Curated by ChEMBL
| Assay Description
Inhibition of human plasma DPP4 |
Bioorg Med Chem Lett 19: 1991-5 (2009)
Article DOI: 10.1016/j.bmcl.2009.02.041
BindingDB Entry DOI: 10.7270/Q2FB52S4 |
More data for this
Ligand-Target Pair | |
Acetyl-CoA carboxylase 1
(Homo sapiens (Human)) | BDBM50439643

(CHEMBL2419598 | US8993586, 76)Show SMILES CNc1ccc2ccc(cc2n1)C(=O)N1CCC2(CC1)Cc1cn(nc1C(=O)N2)C(C)(C)C Show InChI InChI=1S/C25H30N6O2/c1-24(2,3)31-15-18-14-25(28-22(32)21(18)29-31)9-11-30(12-10-25)23(33)17-6-5-16-7-8-20(26-4)27-19(16)13-17/h5-8,13,15H,9-12,14H2,1-4H3,(H,26,27)(H,28,32) | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc.
Curated by ChEMBL
| Assay Description
Inhibition of human ACC1 using acetyl-CoA as substrate assessed as [14C]malonyl-CoA synthesis preincubated for 10 mins prior to substrate addition me... |
J Med Chem 56: 7110-9 (2013)
Article DOI: 10.1021/jm401033t
BindingDB Entry DOI: 10.7270/Q2JW8G9D |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data