

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM31826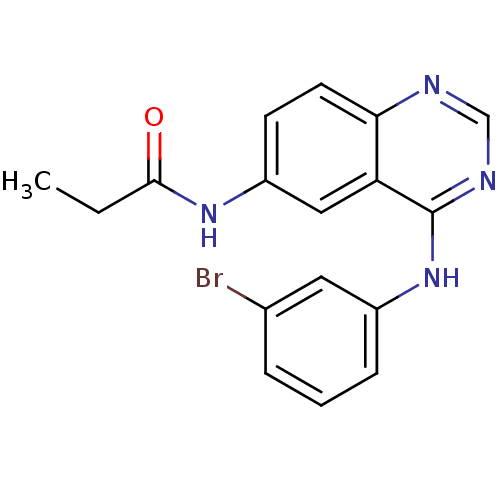 (4-aminoquinazoline, 2a | BMC163482 Compound 3 | CH...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of wild type EGFR by HTRF assay | J Med Chem 53: 2892-901 (2010) Article DOI: 10.1021/jm901877j BindingDB Entry DOI: 10.7270/Q2T43T7N | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM5446 (CHEMBL553 | ERLOTINIB HYDROCHLORIDE | Erlotinib | ...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | n/a | n/a | 0.200 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of wild type EGFR by HTRF assay | J Med Chem 53: 2892-901 (2010) Article DOI: 10.1021/jm901877j BindingDB Entry DOI: 10.7270/Q2T43T7N | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50314991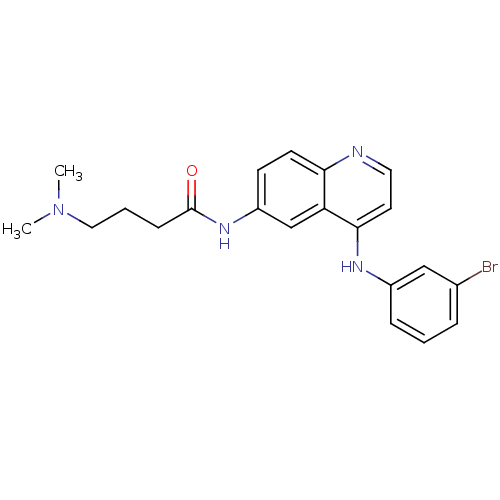 (CHEMBL1089524 | N-(4-(3-Bromophenylamino)quinolin-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of wild type EGFR by HTRF assay | J Med Chem 53: 2892-901 (2010) Article DOI: 10.1021/jm901877j BindingDB Entry DOI: 10.7270/Q2T43T7N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50314992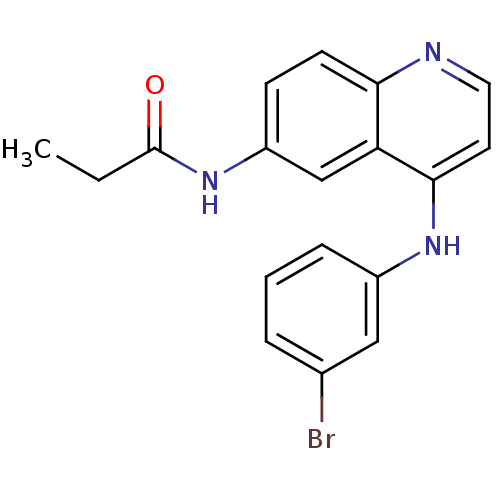 (CHEMBL1092382 | N-(4-(3-Bromophenylamino)quinolin-...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of wild type EGFR by HTRF assay | J Med Chem 53: 2892-901 (2010) Article DOI: 10.1021/jm901877j BindingDB Entry DOI: 10.7270/Q2T43T7N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50314989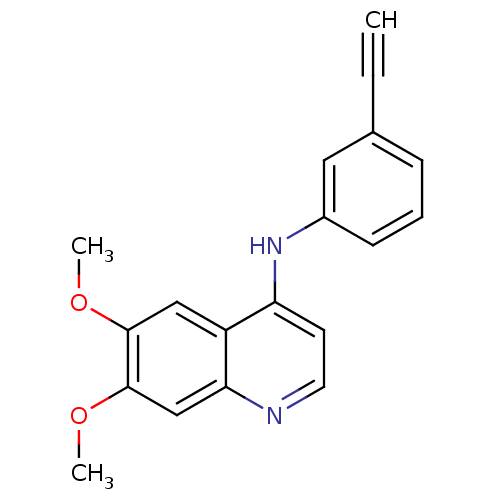 (CHEMBL1092250 | N-(3-Ethynylphenyl)-6,7-dimethoxyq...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of wild type EGFR by HTRF assay | J Med Chem 53: 2892-901 (2010) Article DOI: 10.1021/jm901877j BindingDB Entry DOI: 10.7270/Q2T43T7N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 9 (Homo sapiens (Human)) | BDBM13533 (1-[2-(4-methylphenyl)-5-tert-butyl-pyrazol-3-yl]-3...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 27 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Epidermal growth factor receptor (Homo sapiens (Human)) | BDBM50314990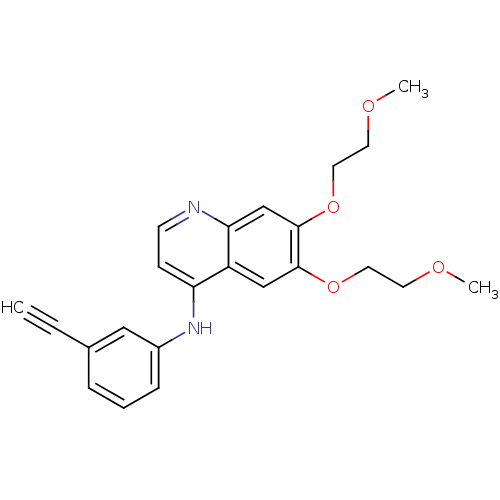 (CHEMBL1089203 | N-(3-Ethynylphenyl)-6,7-bis(2-meth...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of wild type EGFR by HTRF assay | J Med Chem 53: 2892-901 (2010) Article DOI: 10.1021/jm901877j BindingDB Entry DOI: 10.7270/Q2T43T7N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303597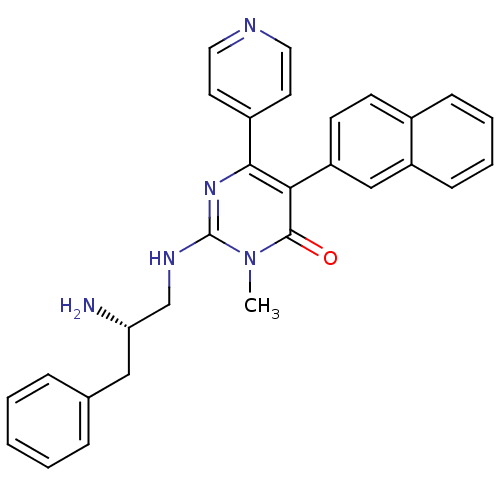 ((S)-2-(2-amino-3-phenylpropylamino)-3-methyl-5-(na...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | 35 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50266947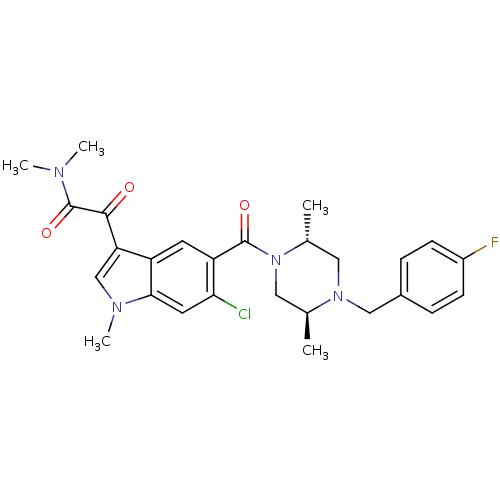 (2-(6-chloro-5-((2R,5S)-4-(4-fluorobenzyl)-2,5-dime...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303586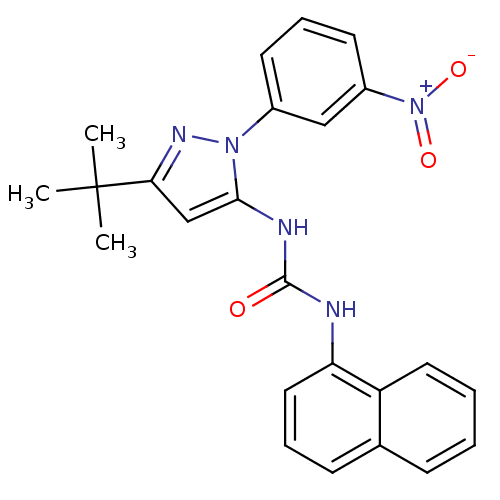 (1-(3-tert-Butyl-1-(3-nitrophenyl)-1H-pyrazol-5-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 45 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 9 (Homo sapiens (Human)) | BDBM31825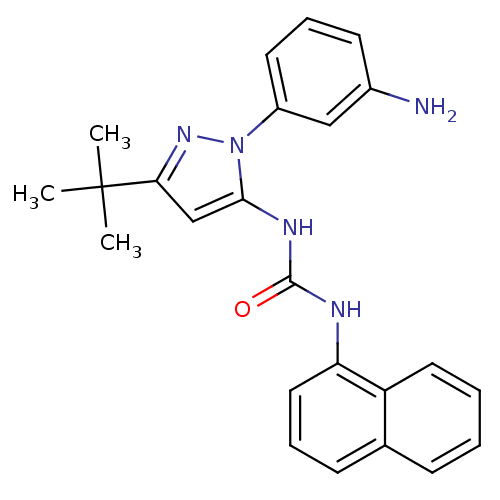 (1-(1-(3-Aminophenyl)-3-tert-butyl-1H-pyrazol-5-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303597 ((S)-2-(2-amino-3-phenylpropylamino)-3-methyl-5-(na...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | 75 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 9 (Homo sapiens (Human)) | BDBM50303597 ((S)-2-(2-amino-3-phenylpropylamino)-3-methyl-5-(na...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | 76 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of JNK2 active form by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM31825 (1-(1-(3-Aminophenyl)-3-tert-butyl-1H-pyrazol-5-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 9 (Homo sapiens (Human)) | BDBM31825 (1-(1-(3-Aminophenyl)-3-tert-butyl-1H-pyrazol-5-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of JNK2 active form by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303594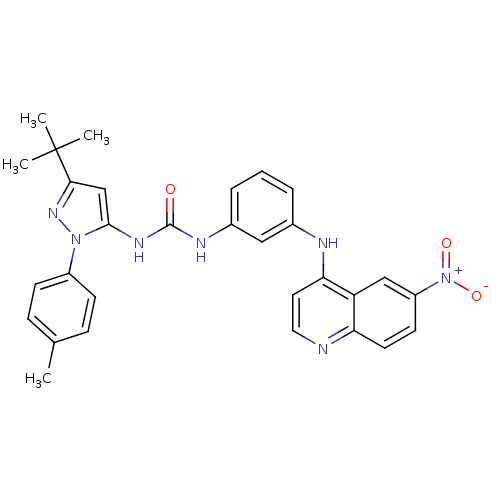 (1-(3-tert-butyl-1-p-tolyl-1H-pyrazol-5-yl)-3-(3-(6...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 95 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50266947 (2-(6-chloro-5-((2R,5S)-4-(4-fluorobenzyl)-2,5-dime...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 96 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 9 (Homo sapiens (Human)) | BDBM50303591 (1-(4-(6-aminoquinolin-4-ylamino)phenyl)-3-(3-tert-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303584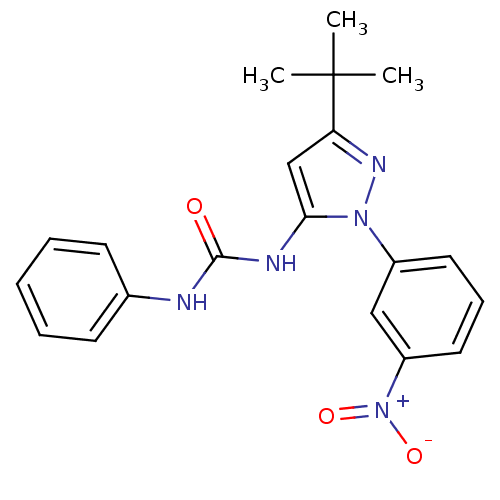 (1-(3-tert-butyl-1-(3-nitrophenyl)-1H-pyrazol-5-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 100 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM14832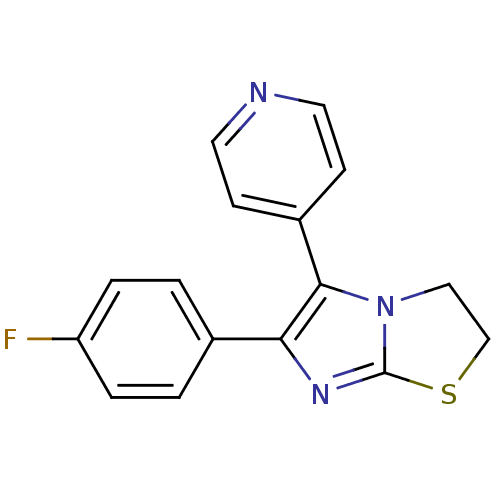 (3-(4-fluorophenyl)-2-pyridin-4-yl-6-thia-1,4-diaza...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 110 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303585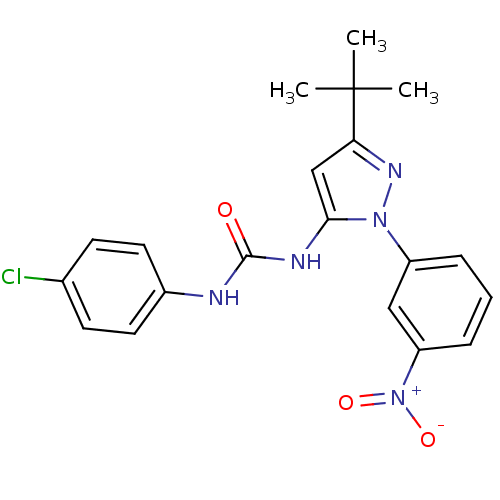 (1-(3-tert-butyl-1-(3-nitrophenyl)-1H-pyrazol-5-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 120 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303596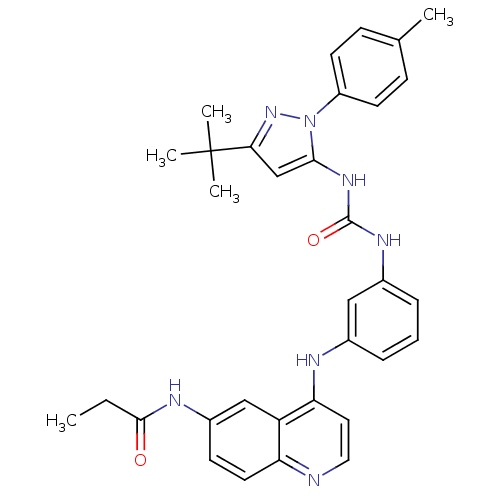 (CHEMBL566100 | N-(4-(3-(3-(3-tert-butyl-1-p-tolyl-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303586 (1-(3-tert-Butyl-1-(3-nitrophenyl)-1H-pyrazol-5-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303594 (1-(3-tert-butyl-1-p-tolyl-1H-pyrazol-5-yl)-3-(3-(6...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 9 (Homo sapiens (Human)) | BDBM13533 (1-[2-(4-methylphenyl)-5-tert-butyl-pyrazol-3-yl]-3...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | PDB Article PubMed | n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of JNK2 active form by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 9 (Homo sapiens (Human)) | BDBM31831 (quinazoline-pyrazolourea hybrid compound, 3e) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 150 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of JNK2 active form by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM31825 (1-(1-(3-Aminophenyl)-3-tert-butyl-1H-pyrazol-5-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank PDB Article PubMed | n/a | n/a | 170 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM13533 (1-[2-(4-methylphenyl)-5-tert-butyl-pyrazol-3-yl]-3...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | n/a | n/a | 220 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303585 (1-(3-tert-butyl-1-(3-nitrophenyl)-1H-pyrazol-5-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 220 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303584 (1-(3-tert-butyl-1-(3-nitrophenyl)-1H-pyrazol-5-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 220 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 9 (Homo sapiens (Human)) | BDBM50303597 ((S)-2-(2-amino-3-phenylpropylamino)-3-methyl-5-(na...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem | Article PubMed | n/a | n/a | 220 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303595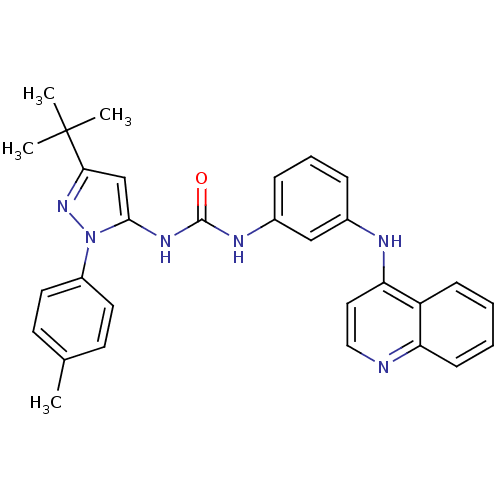 (1-(3-tert-butyl-1-p-tolyl-1H-pyrazol-5-yl)-3-(3-(q...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 230 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM13533 (1-[2-(4-methylphenyl)-5-tert-butyl-pyrazol-3-yl]-3...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | DrugBank MMDB PDB Article PubMed | n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303596 (CHEMBL566100 | N-(4-(3-(3-(3-tert-butyl-1-p-tolyl-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 250 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303593 (1-(3-(6-aminoquinolin-4-ylamino)phenyl)-3-(3-tert-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303595 (1-(3-tert-butyl-1-p-tolyl-1H-pyrazol-5-yl)-3-(3-(q...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 290 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303593 (1-(3-(6-aminoquinolin-4-ylamino)phenyl)-3-(3-tert-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 310 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM31831 (quinazoline-pyrazolourea hybrid compound, 3e) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 320 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303590 (1-(3-(6-aminoquinazolin-4-ylamino)phenyl)-3-(3-ter...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 370 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 9 (Homo sapiens (Human)) | BDBM50303591 (1-(4-(6-aminoquinolin-4-ylamino)phenyl)-3-(3-tert-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 380 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of JNK2 active form by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM16673 (4-[4-({[4-chloro-3-(trifluoromethyl)phenyl]carbamo...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL DrugBank MCE MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PDB Article PubMed | n/a | n/a | 400 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM31831 (quinazoline-pyrazolourea hybrid compound, 3e) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 420 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303592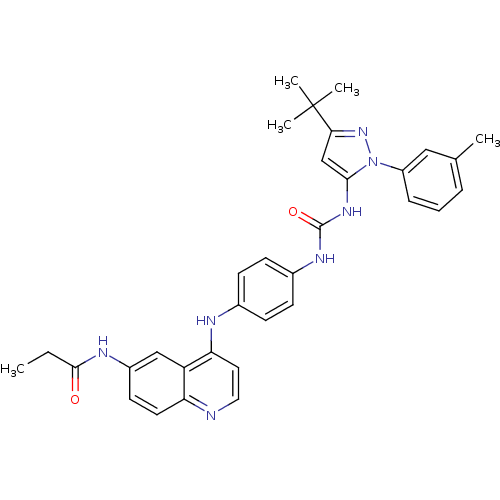 (CHEMBL568756 | N-(4-(4-(3-(3-tert-butyl-1-m-tolyl-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 420 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50115216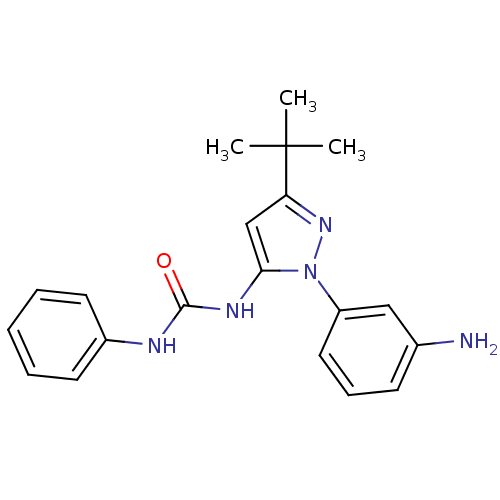 (1-(1-(3-Aminophenyl)-3-tert-butyl-1H-pyrazol-5-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Patents Similars | Article PubMed | n/a | n/a | 440 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 9 (Homo sapiens (Human)) | BDBM50303587 (1-(3-tert-butyl-1-m-tolyl-1H-pyrazol-5-yl)-3-(4-ch...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 440 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303590 (1-(3-(6-aminoquinazolin-4-ylamino)phenyl)-3-(3-ter...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 460 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of p38alpha active form expressed in Escherichia coli BL21(DE3) cells by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303591 (1-(4-(6-aminoquinolin-4-ylamino)phenyl)-3-(3-tert-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | MMDB PDB Article PubMed | n/a | n/a | 470 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Mitogen-activated protein kinase 9 (Homo sapiens (Human)) | BDBM31828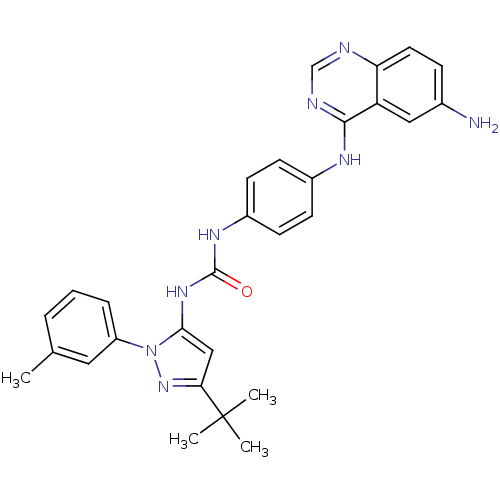 (quinazoline-pyrazolourea hybrid compound, 3b) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | n/a | n/a | 570 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Inhibition of JNK2 active form by HTRF assay | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM50303588 (1-(3-tert-butyl-1-p-tolyl-1H-pyrazol-5-yl)-3-p-tol...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 680 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mitogen-activated protein kinase 14 (Homo sapiens (Human)) | BDBM31827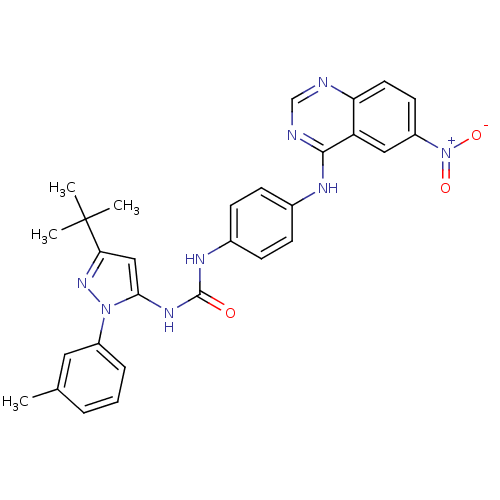 (quinazoline-pyrazolourea hybrid compound, 3a) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 760 | n/a | n/a | n/a | n/a | n/a | n/a |
Chemical Genomics Centre of the Max Planck Society Curated by ChEMBL | Assay Description Displacemnt of N,N'-(2,2'-(3,3'-disulfanediylbis(2,5-dioxopyrrolidine-3,1-diyl))bis(ethane-2,1-diyl))bis(2-(3-(3-tert-butyl-5-(3-naphthalen-1-ylureid... | J Med Chem 53: 357-67 (2010) Article DOI: 10.1021/jm901297e BindingDB Entry DOI: 10.7270/Q29W0FKC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Displayed 1 to 50 (of 83 total ) | Next | Last >> |