 Found 1013 hits with Last Name = 'timmerman' and Initial = 'h'
Found 1013 hits with Last Name = 'timmerman' and Initial = 'h' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM50079527
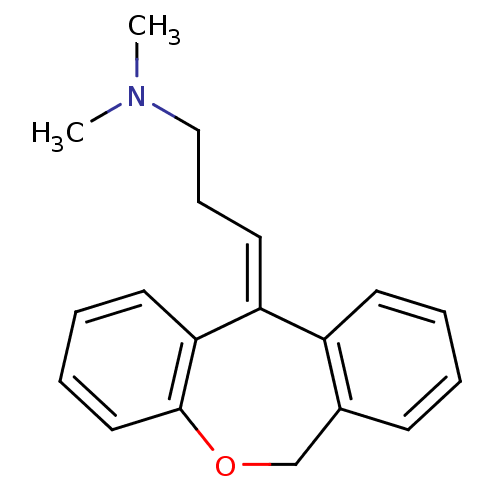
((3E)-3-dibenzo[b,e]oxepin-11(6H)-ylidene-N,N-dimet...)Show InChI InChI=1S/C19H21NO/c1-20(2)13-7-11-17-16-9-4-3-8-15(16)14-21-19-12-6-5-10-18(17)19/h3-6,8-12H,7,13-14H2,1-2H3/b17-11- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.0900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50079527

((3E)-3-dibenzo[b,e]oxepin-11(6H)-ylidene-N,N-dimet...)Show InChI InChI=1S/C19H21NO/c1-20(2)13-7-11-17-16-9-4-3-8-15(16)14-21-19-12-6-5-10-18(17)19/h3-6,8-12H,7,13-14H2,1-2H3/b17-11- | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Rattus norvegicus (rat)) | BDBM50099623
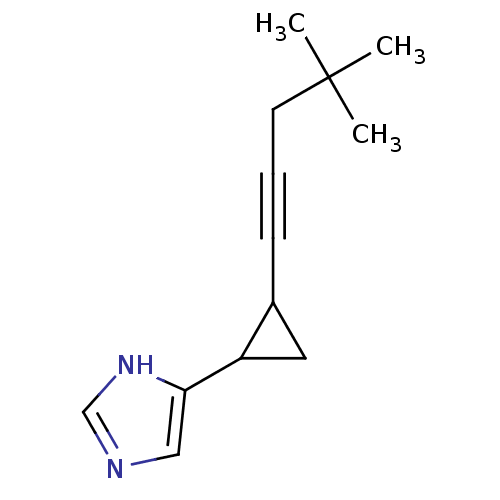
(4-[2-(4,4-Dimethyl-pent-1-ynyl)-cyclopropyl]-1H-im...)Show InChI InChI=1S/C13H18N2/c1-13(2,3)6-4-5-10-7-11(10)12-8-14-9-15-12/h8-11H,6-7H2,1-3H3,(H,14,15) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 0.180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
De Novo Pharmaceuticals
Curated by ChEMBL
| Assay Description
Ability to displace [3H]Nalpha-methylhistamine from histamine H3 receptors in homogenates of rat cerebral cortex |
J Med Chem 44: 1666-74 (2001)
BindingDB Entry DOI: 10.7270/Q2WH2QP8 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50079527

((3E)-3-dibenzo[b,e]oxepin-11(6H)-ylidene-N,N-dimet...)Show InChI InChI=1S/C19H21NO/c1-20(2)13-7-11-17-16-9-4-3-8-15(16)14-21-19-12-6-5-10-18(17)19/h3-6,8-12H,7,13-14H2,1-2H3/b17-11- | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50009073
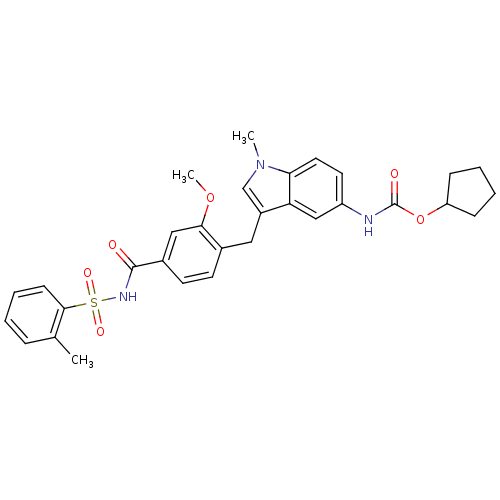
(4-(5-cyclopentyloxycarbonylamino-1-methyl-1H-indol...)Show SMILES COc1cc(ccc1Cc1cn(C)c2ccc(NC(=O)OC3CCCC3)cc12)C(=O)NS(=O)(=O)c1ccccc1C Show InChI InChI=1S/C31H33N3O6S/c1-20-8-4-7-11-29(20)41(37,38)33-30(35)22-13-12-21(28(17-22)39-3)16-23-19-34(2)27-15-14-24(18-26(23)27)32-31(36)40-25-9-5-6-10-25/h4,7-8,11-15,17-19,25H,5-6,9-10,16H2,1-3H3,(H,32,36)(H,33,35) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PDB
Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by ChEMBL
| Assay Description
Binding affinity of the compound towards Cysteinyl leukotriene D4 receptor (cysLT1) was measured by the displacement of [3H]-LTD4 radioligand |
J Med Chem 41: 1439-45 (1998)
Article DOI: 10.1021/jm970180w
BindingDB Entry DOI: 10.7270/Q2WH2P4P |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM50079527

((3E)-3-dibenzo[b,e]oxepin-11(6H)-ylidene-N,N-dimet...)Show InChI InChI=1S/C19H21NO/c1-20(2)13-7-11-17-16-9-4-3-8-15(16)14-21-19-12-6-5-10-18(17)19/h3-6,8-12H,7,13-14H2,1-2H3/b17-11- | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
KEGG
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM22541
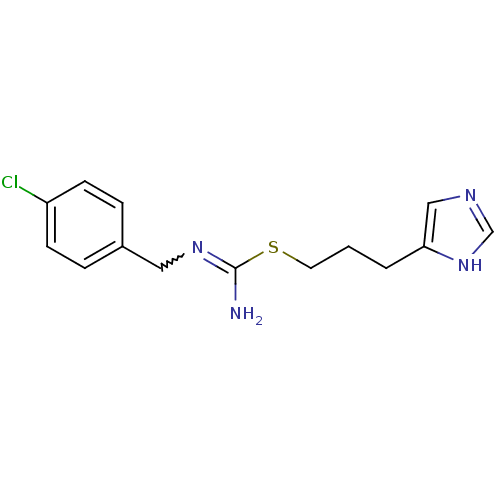
(Clobenpropit | N''-[(4-chlorophenyl)methyl]{[3-(1H...)Show SMILES NC(SCCCc1cnc[nH]1)=NCc1ccc(Cl)cc1 |w:11.12| Show InChI InChI=1S/C14H17ClN4S/c15-12-5-3-11(4-6-12)8-18-14(16)20-7-1-2-13-9-17-10-19-13/h3-6,9-10H,1-2,7-8H2,(H2,16,18)(H,17,19) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.339 | -54.1 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
VU University Amsterdam
| Assay Description
Ligand displacement assays were performed on CHO cells membranes expressing hH3R. Retained radioactivity was determined by liquid scintillation count... |
J Med Chem 51: 2944-53 (2008)
Article DOI: 10.1021/jm7014149
BindingDB Entry DOI: 10.7270/Q24F1P2W |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Rattus norvegicus (rat)) | BDBM50099622
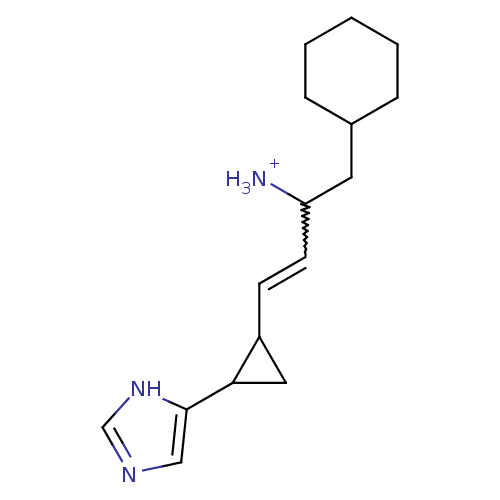
(1-Cyclohexylmethyl-3-[2-(1H-imidazol-4-yl)-cyclopr...)Show SMILES [NH3+]C(CC1CCCCC1)C=CC1CC1c1cnc[nH]1 |w:9.9| Show InChI InChI=1S/C16H25N3/c17-14(8-12-4-2-1-3-5-12)7-6-13-9-15(13)16-10-18-11-19-16/h6-7,10-15H,1-5,8-9,17H2,(H,18,19)/p+1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| PubMed
| 0.400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
De Novo Pharmaceuticals
Curated by ChEMBL
| Assay Description
Ability to displace [3H]Nalpha-methylhistamine from histamine H3 receptors in homogenates of rat cerebral cortex |
J Med Chem 44: 1666-74 (2001)
BindingDB Entry DOI: 10.7270/Q2WH2QP8 |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM22542

(4-(1H-imidazol-4-ylmethyl)piperidine | 4-(1H-imida...)Show InChI InChI=1S/C9H15N3/c1-3-10-4-2-8(1)5-9-6-11-7-12-9/h6-8,10H,1-5H2,(H,11,12) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.407 | -53.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
VU University Amsterdam
| Assay Description
Ligand displacement assays were performed on CHO cells membranes expressing hH3R. Retained radioactivity was determined by liquid scintillation count... |
J Med Chem 51: 2944-53 (2008)
Article DOI: 10.1021/jm7014149
BindingDB Entry DOI: 10.7270/Q24F1P2W |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50150945
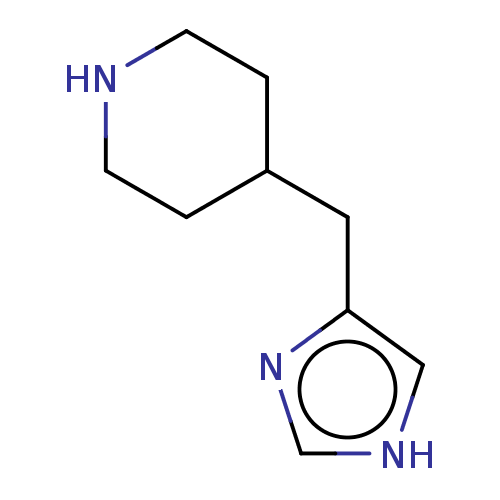
(CHEBI:81390 | Immepip)Show InChI InChI=1S/C9H15N3/c1-3-10-4-2-8(1)5-9-6-11-7-12-9/h6-8,10H,1-5H2,(H,11,12) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.479 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by ChEMBL
| Assay Description
Inhibitory activity measured by [3H]- N alpha- methyl-histamine binding to membranes of SK-N-MC cells expressing the human Histamine H3 receptor |
J Med Chem 46: 5445-57 (2003)
Article DOI: 10.1021/jm030905y
BindingDB Entry DOI: 10.7270/Q2FN18X0 |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50052024

(CHEMBL787 | montelukast)Show SMILES CC(C)(O)c1ccccc1CC[C@@H](SCC1(CC(O)=O)CC1)c1cccc(\C=C\c2ccc3ccc(Cl)cc3n2)c1 |r| Show InChI InChI=1S/C35H36ClNO3S/c1-34(2,40)30-9-4-3-7-25(30)13-17-32(41-23-35(18-19-35)22-33(38)39)27-8-5-6-24(20-27)10-15-29-16-12-26-11-14-28(36)21-31(26)37-29/h3-12,14-16,20-21,32,40H,13,17-19,22-23H2,1-2H3,(H,38,39)/b15-10+/t32-/m1/s1 | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by ChEMBL
| Assay Description
Binding affinity of the compound towards Cysteinyl leukotriene D4 receptor (cysLT1) was measured by the displacement of [3H]-LTD4 radioligand |
J Med Chem 41: 1439-45 (1998)
Article DOI: 10.1021/jm970180w
BindingDB Entry DOI: 10.7270/Q2WH2P4P |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50150945

(CHEBI:81390 | Immepip)Show InChI InChI=1S/C9H15N3/c1-3-10-4-2-8(1)5-9-6-11-7-12-9/h6-8,10H,1-5H2,(H,11,12) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
PDB
UniChem
Similars
| Article
PubMed
| 0.501 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Amsterdam
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-R-methylhistamine binding to SK-N-MC cell membranes expressing human H3 receptor |
J Med Chem 48: 2100-7 (2005)
Article DOI: 10.1021/jm049475h
BindingDB Entry DOI: 10.7270/Q28K7CVC |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM22567
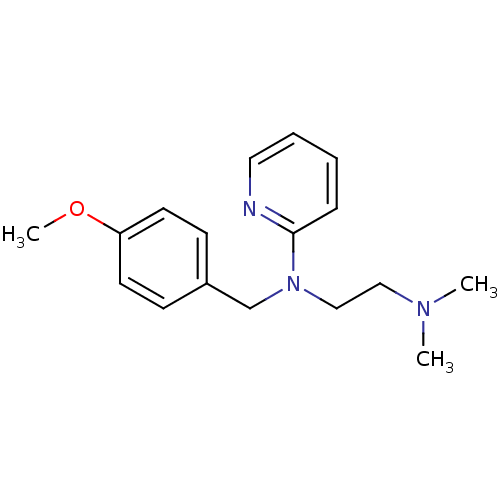
(3H]pyrilamine | CHEMBL511 | Dorantamin | Mepyramin...)Show InChI InChI=1S/C17H23N3O/c1-19(2)12-13-20(17-6-4-5-11-18-17)14-15-7-9-16(21-3)10-8-15/h4-11H,12-14H2,1-3H3 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.570 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM86032

(trans-H2-PAT(-) | trans-PAT)Show InChI InChI=1S/C18H21N/c1-19(2)16-12-15-10-6-7-11-17(15)18(13-16)14-8-4-3-5-9-14/h3-11,16,18H,12-13H2,1-2H3/t16-,18-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50292411
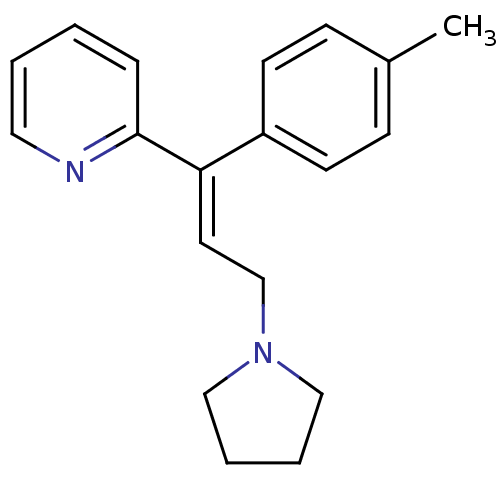
((E)-2-(3-(pyrrolidin-1-yl)-1-p-tolylprop-1-enyl)py...)Show InChI InChI=1S/C19H22N2/c1-16-7-9-17(10-8-16)18(19-6-2-3-12-20-19)11-15-21-13-4-5-14-21/h2-3,6-12H,4-5,13-15H2,1H3/b18-11+ | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.690 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM35938
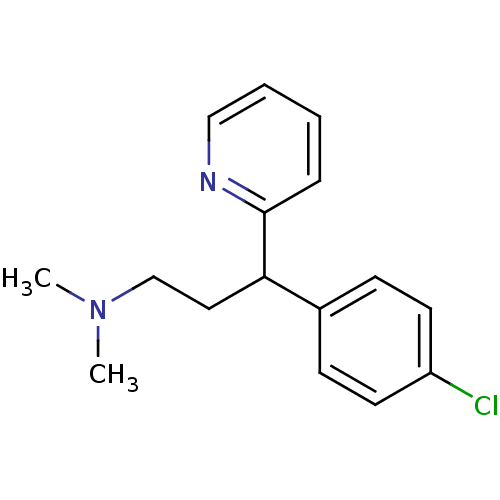
(1-(p-chlorophenyl)-1-(2-pyridyl)-3-N,N-dimethylpro...)Show InChI InChI=1S/C16H19ClN2/c1-19(2)12-10-15(16-5-3-4-11-18-16)13-6-8-14(17)9-7-13/h3-9,11,15H,10,12H2,1-2H3 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.690 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM22567

(3H]pyrilamine | CHEMBL511 | Dorantamin | Mepyramin...)Show InChI InChI=1S/C17H23N3O/c1-19(2)12-13-20(17-6-4-5-11-18-17)14-15-7-9-16(21-3)10-8-15/h4-11H,12-14H2,1-3H3 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| Article
PubMed
| 0.740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Hrh3 protein
(RAT) | BDBM22541

(Clobenpropit | N''-[(4-chlorophenyl)methyl]{[3-(1H...)Show SMILES NC(SCCCc1cnc[nH]1)=NCc1ccc(Cl)cc1 |w:11.12| Show InChI InChI=1S/C14H17ClN4S/c15-12-5-3-11(4-6-12)8-18-14(16)20-7-1-2-13-9-17-10-19-13/h3-6,9-10H,1-2,7-8H2,(H2,16,18)(H,17,19) | KEGG
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 0.870 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 299: 908-14 (2001)
BindingDB Entry DOI: 10.7270/Q2JS9P1N |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM50292411

((E)-2-(3-(pyrrolidin-1-yl)-1-p-tolylprop-1-enyl)py...)Show InChI InChI=1S/C19H22N2/c1-16-7-9-17(10-8-16)18(19-6-2-3-12-20-19)11-15-21-13-4-5-14-21/h2-3,6-12H,4-5,13-15H2,1H3/b18-11+ | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.930 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Hrh3 protein
(RAT) | BDBM22913
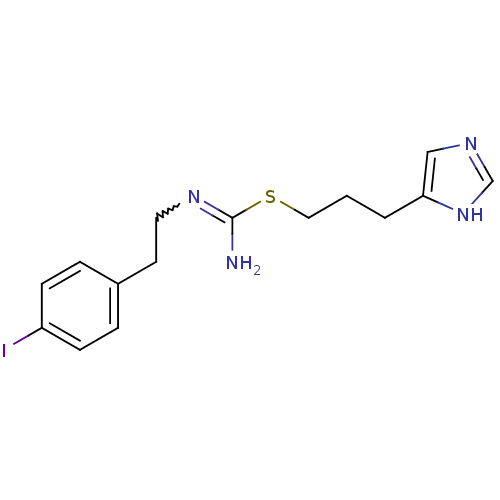
(CHEMBL1237146 | CHEMBL498770 | Iodophenpropit | {[...)Show SMILES NC(SCCCc1cnc[nH]1)=NCCc1ccc(I)cc1 |w:11.12| Show InChI InChI=1S/C15H19IN4S/c16-13-5-3-12(4-6-13)7-8-19-15(17)21-9-1-2-14-10-18-11-20-14/h3-6,10-11H,1-2,7-9H2,(H2,17,19)(H,18,20) | KEGG
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.970 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by PDSP Ki Database
| |
Br J Pharmacol 116: 2315-21 (1995)
Article DOI: 10.1111/j.1476-5381.1995.tb15071.x
BindingDB Entry DOI: 10.7270/Q2ZK5F64 |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM22548
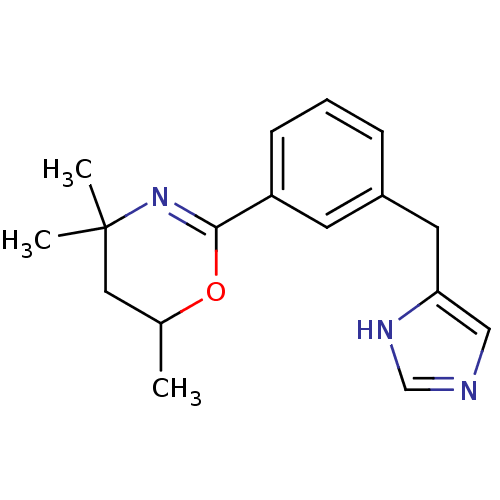
(2-[3-(1H-imidazol-4-ylmethyl)phenyl]-4,4,6-trimeth...)Show SMILES CC1CC(C)(C)N=C(O1)c1cccc(Cc2cnc[nH]2)c1 |c:6| Show InChI InChI=1S/C17H21N3O/c1-12-9-17(2,3)20-16(21-12)14-6-4-5-13(7-14)8-15-10-18-11-19-15/h4-7,10-12H,8-9H2,1-3H3,(H,18,19) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | -51.4 | n/a | n/a | 2 | n/a | n/a | 7.4 | 25 |
VU University Amsterdam
| Assay Description
Ligand displacement assays were performed on CHO cells membranes expressing hH3R. Retained radioactivity was determined by liquid scintillation count... |
J Med Chem 51: 2944-53 (2008)
Article DOI: 10.1021/jm7014149
BindingDB Entry DOI: 10.7270/Q24F1P2W |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50475340
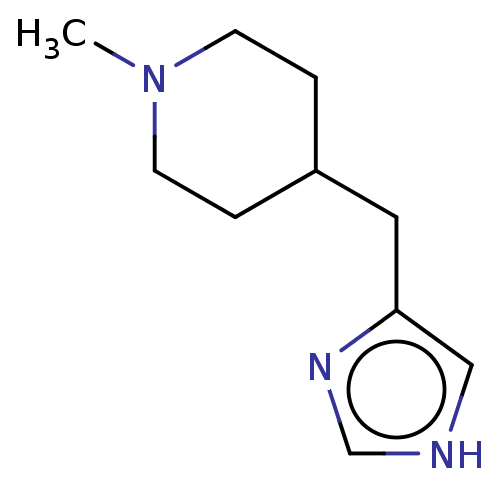
(Methimepip)Show InChI InChI=1S/C10H17N3/c1-13-4-2-9(3-5-13)6-10-7-11-8-12-10/h7-9H,2-6H2,1H3,(H,11,12) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit Amsterdam
Curated by ChEMBL
| Assay Description
Displacement of [3H]N-R-methylhistamine binding to SK-N-MC cell membranes expressing human H3 receptor |
J Med Chem 48: 2100-7 (2005)
Article DOI: 10.1021/jm049475h
BindingDB Entry DOI: 10.7270/Q28K7CVC |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM22567

(3H]pyrilamine | CHEMBL511 | Dorantamin | Mepyramin...)Show InChI InChI=1S/C17H23N3O/c1-19(2)12-13-20(17-6-4-5-11-18-17)14-15-7-9-16(21-3)10-8-15/h4-11H,12-14H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 1.01 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50064085
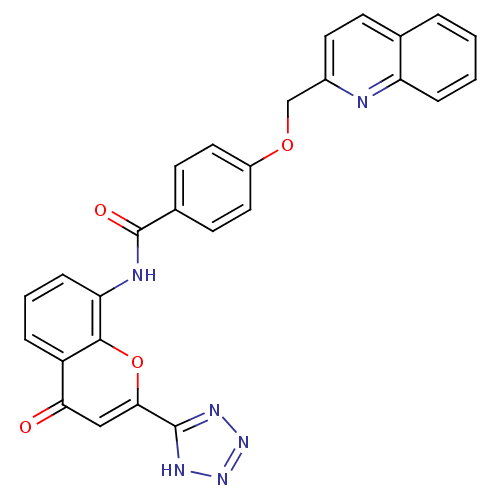
(CHEMBL285070 | N-[4-Oxo-2-(1H-tetrazol-5-yl)-4H-ch...)Show SMILES O=C(Nc1cccc2c1oc(cc2=O)-c1nnn[nH]1)c1ccc(OCc2ccc3ccccc3n2)cc1 Show InChI InChI=1S/C27H18N6O4/c34-23-14-24(26-30-32-33-31-26)37-25-20(23)5-3-7-22(25)29-27(35)17-9-12-19(13-10-17)36-15-18-11-8-16-4-1-2-6-21(16)28-18/h1-14H,15H2,(H,29,35)(H,30,31,32,33) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by ChEMBL
| Assay Description
Binding affinity of the compound towards Cysteinyl leukotriene D4 receptor (cysLT1) was measured by the displacement of [3H]-LTD4 radioligand |
J Med Chem 41: 1439-45 (1998)
Article DOI: 10.1021/jm970180w
BindingDB Entry DOI: 10.7270/Q2WH2P4P |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM50292411

((E)-2-(3-(pyrrolidin-1-yl)-1-p-tolylprop-1-enyl)py...)Show InChI InChI=1S/C19H22N2/c1-16-7-9-17(10-8-16)18(19-6-2-3-12-20-19)11-15-21-13-4-5-14-21/h2-3,6-12H,4-5,13-15H2,1H3/b18-11+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 1.11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM86032

(trans-H2-PAT(-) | trans-PAT)Show InChI InChI=1S/C18H21N/c1-19(2)16-12-15-10-6-7-11-17(15)18(13-16)14-8-4-3-5-9-14/h3-11,16,18H,12-13H2,1-2H3/t16-,18-/m1/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.15 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM22567

(3H]pyrilamine | CHEMBL511 | Dorantamin | Mepyramin...)Show InChI InChI=1S/C17H23N3O/c1-19(2)12-13-20(17-6-4-5-11-18-17)14-15-7-9-16(21-3)10-8-15/h4-11H,12-14H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 1.18 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Adenosine receptor A1
(Rattus norvegicus (rat)) | BDBM50207816
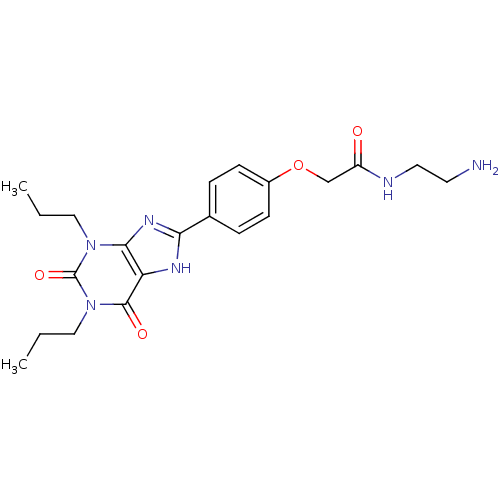
(CHEMBL273094 | N-(2-Amino-ethyl)-2-[4-(2,6-dioxo-1...)Show SMILES CCCn1c2nc([nH]c2c(=O)n(CCC)c1=O)-c1ccc(OCC(=O)NCCN)cc1 Show InChI InChI=1S/C21H28N6O4/c1-3-11-26-19-17(20(29)27(12-4-2)21(26)30)24-18(25-19)14-5-7-15(8-6-14)31-13-16(28)23-10-9-22/h5-8H,3-4,9-13,22H2,1-2H3,(H,23,28)(H,24,25) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by ChEMBL
| Assay Description
Displacement of [3H]DPCPX binding to adenosine A1 receptor of rat brain cortical membrane |
J Med Chem 44: 749-62 (2001)
BindingDB Entry DOI: 10.7270/Q2H132Q0 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM22869

(6-chloro-10-(4-methylpiperazin-1-yl)-2,9-diazatric...)Show SMILES CN1CCN(CC1)C1=c2ccccc2=Nc2ccc(Cl)cc2N1 |c:8,15| Show InChI InChI=1S/C18H19ClN4/c1-22-8-10-23(11-9-22)18-14-4-2-3-5-15(14)20-16-7-6-13(19)12-17(16)21-18/h2-7,12,21H,8-11H2,1H3 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.23 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM86033

(cis-H2-PAT(+/-) | trans-H2-PAT(+/-))Show InChI InChI=1S/C18H21N/c1-19(2)16-12-15-10-6-7-11-17(15)18(13-16)14-8-4-3-5-9-14/h3-11,16,18H,12-13H2,1-2H3 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.37 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50474424
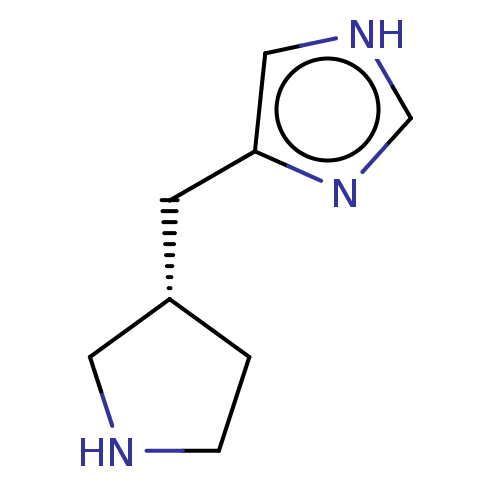
(CHEMBL153051)Show InChI InChI=1S/C8H13N3/c1-2-9-4-7(1)3-8-5-10-6-11-8/h5-7,9H,1-4H2,(H,10,11)/t7-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by ChEMBL
| Assay Description
Inhibitory activity measured by [3H]- N alpha- methyl-histamine binding to membranes of SK-N-MC cells expressing the human Histamine H3 receptor |
J Med Chem 46: 5445-57 (2003)
Article DOI: 10.1021/jm030905y
BindingDB Entry DOI: 10.7270/Q2FN18X0 |
More data for this
Ligand-Target Pair | |
Hrh3 protein
(RAT) | BDBM22913

(CHEMBL1237146 | CHEMBL498770 | Iodophenpropit | {[...)Show SMILES NC(SCCCc1cnc[nH]1)=NCCc1ccc(I)cc1 |w:11.12| Show InChI InChI=1S/C15H19IN4S/c16-13-5-3-12(4-6-13)7-8-19-15(17)21-9-1-2-14-10-18-11-20-14/h3-6,10-11H,1-2,7-9H2,(H2,17,19)(H,18,20) | KEGG
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| 1.48 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 299: 908-14 (2001)
BindingDB Entry DOI: 10.7270/Q2JS9P1N |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM86033

(cis-H2-PAT(+/-) | trans-H2-PAT(+/-))Show InChI InChI=1S/C18H21N/c1-19(2)16-12-15-10-6-7-11-17(15)18(13-16)14-8-4-3-5-9-14/h3-11,16,18H,12-13H2,1-2H3 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.53 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM50292411

((E)-2-(3-(pyrrolidin-1-yl)-1-p-tolylprop-1-enyl)py...)Show InChI InChI=1S/C19H22N2/c1-16-7-9-17(10-8-16)18(19-6-2-3-12-20-19)11-15-21-13-4-5-14-21/h2-3,6-12H,4-5,13-15H2,1H3/b18-11+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 1.53 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM22869

(6-chloro-10-(4-methylpiperazin-1-yl)-2,9-diazatric...)Show SMILES CN1CCN(CC1)C1=c2ccccc2=Nc2ccc(Cl)cc2N1 |c:8,15| Show InChI InChI=1S/C18H19ClN4/c1-22-8-10-23(11-9-22)18-14-4-2-3-5-15(14)20-16-7-6-13(19)12-17(16)21-18/h2-7,12,21H,8-11H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.54 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM22911
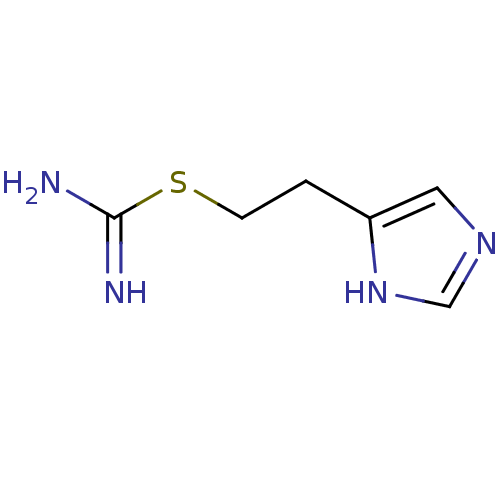
(2-(3H-imidazol-4-yl)ethylsulfanylmethanimidamide |...)Show InChI InChI=1S/C6H10N4S/c7-6(8)11-2-1-5-3-9-4-10-5/h3-4H,1-2H2,(H3,7,8)(H,9,10) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
PDB
UniChem
Patents
Similars
| PubMed
| 1.62 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 299: 908-14 (2001)
BindingDB Entry DOI: 10.7270/Q2JS9P1N |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM22869

(6-chloro-10-(4-methylpiperazin-1-yl)-2,9-diazatric...)Show SMILES CN1CCN(CC1)C1=c2ccccc2=Nc2ccc(Cl)cc2N1 |c:8,15| Show InChI InChI=1S/C18H19ClN4/c1-22-8-10-23(11-9-22)18-14-4-2-3-5-15(14)20-16-7-6-13(19)12-17(16)21-18/h2-7,12,21H,8-11H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.63 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM86032

(trans-H2-PAT(-) | trans-PAT)Show InChI InChI=1S/C18H21N/c1-19(2)16-12-15-10-6-7-11-17(15)18(13-16)14-8-4-3-5-9-14/h3-11,16,18H,12-13H2,1-2H3/t16-,18-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.65 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Rattus norvegicus (rat)) | BDBM50127605
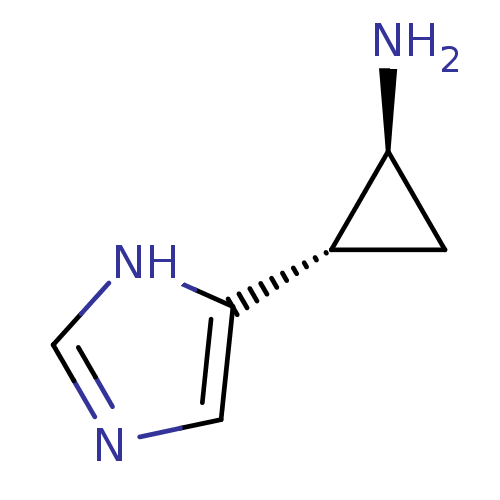
((1S,2S)-2-(1H-Imidazol-4-yl)-cyclopropylamine | (1...)Show InChI InChI=1S/C6H9N3/c7-5-1-4(5)6-2-8-3-9-6/h2-5H,1,7H2,(H,8,9)/t4-,5-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by ChEMBL
| Assay Description
In vitro binding affinity was measured against Histamine H3 receptor on rat cerebral cortex. |
J Med Chem 42: 1115-22 (1999)
Article DOI: 10.1021/jm9810912
BindingDB Entry DOI: 10.7270/Q24Q7XPW |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM35938

(1-(p-chlorophenyl)-1-(2-pyridyl)-3-N,N-dimethylpro...)Show InChI InChI=1S/C16H19ClN2/c1-19(2)12-10-15(16-5-3-4-11-18-16)13-6-8-14(17)9-7-13/h3-9,11,15H,10,12H2,1-2H3 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.98 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM22538
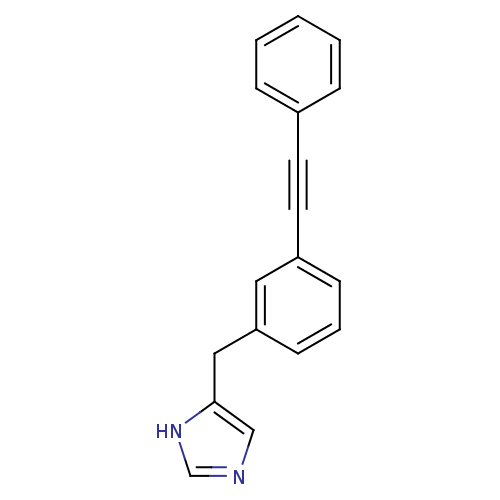
(4-Benzyl-1H-imidazole derivative, 19 | 4-{[3-(2-ph...)Show InChI InChI=1S/C18H14N2/c1-2-5-15(6-3-1)9-10-16-7-4-8-17(11-16)12-18-13-19-14-20-18/h1-8,11,13-14H,12H2,(H,19,20) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | -49.7 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
VU University Amsterdam
| Assay Description
Ligand displacement assays were performed on CHO cells membranes expressing hH3R. Retained radioactivity was determined by liquid scintillation count... |
J Med Chem 51: 2944-53 (2008)
Article DOI: 10.1021/jm7014149
BindingDB Entry DOI: 10.7270/Q24F1P2W |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Cavia porcellus (domestic guinea pig)) | BDBM22869

(6-chloro-10-(4-methylpiperazin-1-yl)-2,9-diazatric...)Show SMILES CN1CCN(CC1)C1=c2ccccc2=Nc2ccc(Cl)cc2N1 |c:8,15| Show InChI InChI=1S/C18H19ClN4/c1-22-8-10-23(11-9-22)18-14-4-2-3-5-15(14)20-16-7-6-13(19)12-17(16)21-18/h2-7,12,21H,8-11H2,1H3 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.07 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM85830

(VUF5202)Show InChI InChI=1S/C15H20ClN3/c16-14-7-5-13(6-8-14)10-17-9-3-1-2-4-15-11-18-12-19-15/h5-8,11-12,17H,1-4,9-10H2,(H,18,19) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| PubMed
| 2.34 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 299: 908-14 (2001)
BindingDB Entry DOI: 10.7270/Q2JS9P1N |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM86033

(cis-H2-PAT(+/-) | trans-H2-PAT(+/-))Show InChI InChI=1S/C18H21N/c1-19(2)16-12-15-10-6-7-11-17(15)18(13-16)14-8-4-3-5-9-14/h3-11,16,18H,12-13H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.49 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM86032

(trans-H2-PAT(-) | trans-PAT)Show InChI InChI=1S/C18H21N/c1-19(2)16-12-15-10-6-7-11-17(15)18(13-16)14-8-4-3-5-9-14/h3-11,16,18H,12-13H2,1-2H3/t16-,18-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50064083
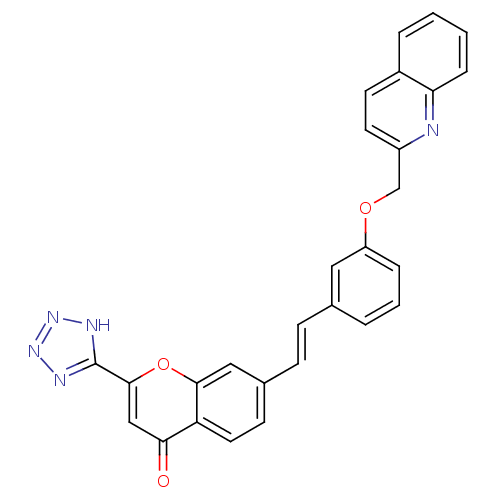
(7-{(E)-2-[3-(Quinolin-2-ylmethoxy)-phenyl]-vinyl}-...)Show SMILES O=c1cc(oc2cc(\C=C\c3cccc(OCc4ccc5ccccc5n4)c3)ccc12)-c1nnn[nH]1 Show InChI InChI=1S/C28H19N5O3/c34-25-16-27(28-30-32-33-31-28)36-26-15-19(10-13-23(25)26)9-8-18-4-3-6-22(14-18)35-17-21-12-11-20-5-1-2-7-24(20)29-21/h1-16H,17H2,(H,30,31,32,33)/b9-8+ | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| 2.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by ChEMBL
| Assay Description
Binding affinity of the compound towards Cysteinyl leukotriene D4 receptor (cysLT1) was measured by the displacement of [3H]-LTD4 radioligand |
J Med Chem 41: 1439-45 (1998)
Article DOI: 10.1021/jm970180w
BindingDB Entry DOI: 10.7270/Q2WH2P4P |
More data for this
Ligand-Target Pair | |
Histamine H1 receptor
(Homo sapiens (Human)) | BDBM35938

(1-(p-chlorophenyl)-1-(2-pyridyl)-3-N,N-dimethylpro...)Show InChI InChI=1S/C16H19ClN2/c1-19(2)12-10-15(16-5-3-4-11-18-16)13-6-8-14(17)9-7-13/h3-9,11,15H,10,12H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| 2.67 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of North Carolina at Chapel Hill
Curated by PDSP Ki Database
| |
J Pharmacol Exp Ther 302: 328-36 (2002)
Article DOI: 10.1124/jpet.302.1.328
BindingDB Entry DOI: 10.7270/Q2JQ0ZK1 |
More data for this
Ligand-Target Pair | |
Cysteinyl leukotriene receptor 1
(GUINEA PIG) | BDBM50064084
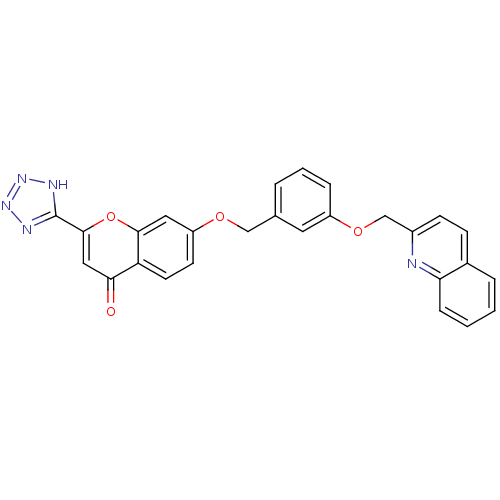
(7-[3-(Quinolin-2-ylmethoxy)-benzyloxy]-2-(1H-tetra...)Show SMILES O=c1cc(oc2cc(OCc3cccc(OCc4ccc5ccccc5n4)c3)ccc12)-c1nnn[nH]1 Show InChI InChI=1S/C27H19N5O4/c33-24-14-26(27-29-31-32-30-27)36-25-13-21(10-11-22(24)25)34-15-17-4-3-6-20(12-17)35-16-19-9-8-18-5-1-2-7-23(18)28-19/h1-14H,15-16H2,(H,29,30,31,32) | PDB
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
| Article
PubMed
| 3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by ChEMBL
| Assay Description
Binding affinity of the compound towards Cysteinyl leukotriene D4 receptor (cysLT1) was measured by the displacement of [3H]-LTD4 radioligand |
J Med Chem 41: 1439-45 (1998)
Article DOI: 10.1021/jm970180w
BindingDB Entry DOI: 10.7270/Q2WH2P4P |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM22540
(4-Benzyl-1H-imidazole derivative, 21 | 4-{[3-(pent...)Show InChI InChI=1S/C15H16N2/c1-2-3-4-6-13-7-5-8-14(9-13)10-15-11-16-12-17-15/h5,7-9,11-12H,2-3,10H2,1H3,(H,16,17) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3 | -48.6 | n/a | n/a | n/a | n/a | n/a | 7.4 | 25 |
VU University Amsterdam
| Assay Description
Ligand displacement assays were performed on CHO cells membranes expressing hH3R. Retained radioactivity was determined by liquid scintillation count... |
J Med Chem 51: 2944-53 (2008)
Article DOI: 10.1021/jm7014149
BindingDB Entry DOI: 10.7270/Q24F1P2W |
More data for this
Ligand-Target Pair | |
Adenosine receptor A2a
(Rattus norvegicus (rat)) | BDBM50067081

(9-Fluoro-2-furan-2-yl-5,6-dihydro-[1,2,4]triazolo[...)Show InChI InChI=1S/C13H8FN5O/c14-7-3-4-9-8(6-7)12-17-11(10-2-1-5-20-10)18-19(12)13(15)16-9/h1-6H,(H2,15,16) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| 3.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vrije Universiteit
Curated by ChEMBL
| Assay Description
Displacement of [3H]ZM-241385 binding in adenosine A2A receptor of rat striatal membrane |
J Med Chem 44: 749-62 (2001)
BindingDB Entry DOI: 10.7270/Q2H132Q0 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data