 Found 993 hits with Last Name = 'mohammed' and Initial = 'r'
Found 993 hits with Last Name = 'mohammed' and Initial = 'r' Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50138119
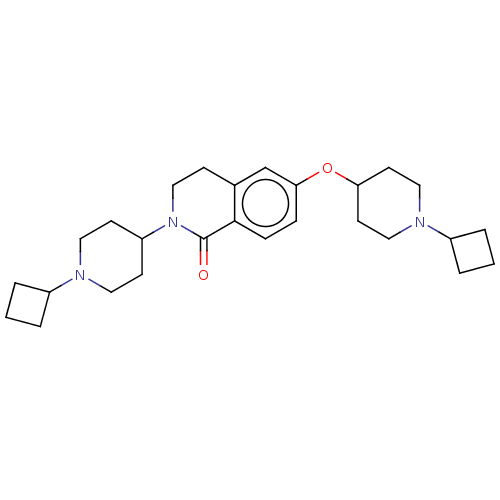
(CHEMBL3753475)Show SMILES O=C1N(CCc2cc(OC3CCN(CC3)C3CCC3)ccc12)C1CCN(CC1)C1CCC1 Show InChI InChI=1S/C27H39N3O2/c31-27-26-8-7-25(32-24-12-16-29(17-13-24)22-5-2-6-22)19-20(26)9-18-30(27)23-10-14-28(15-11-23)21-3-1-4-21/h7-8,19,21-24H,1-6,9-18H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to recombinant human H3 receptor |
Eur J Med Chem 108: 655-62 (2016)
Article DOI: 10.1016/j.ejmech.2015.12.005
BindingDB Entry DOI: 10.7270/Q2F191KF |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM221048
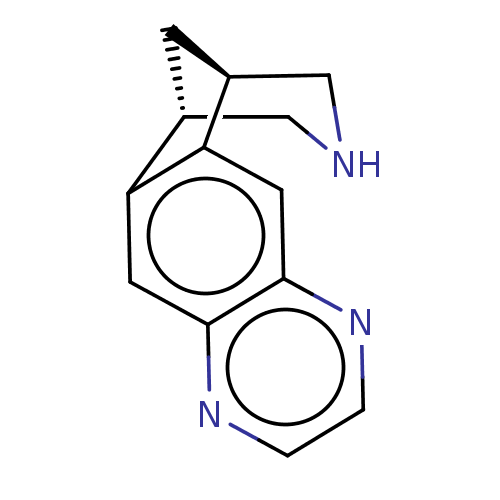
(US9284322, varenicline | US9303017, Varenicline)Show InChI InChI=1S/C13H13N3/c1-2-16-13-5-11-9-3-8(6-14-7-9)10(11)4-12(13)15-1/h1-2,4-5,8-9,14H,3,6-7H2/t8-,9+ | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
PC cid
PC sid
PDB
UniChem
| PDB
Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from human alpha4beta2 nAChR expressed in CHOK1 cell membrane by microbeta scintillation counting method |
J Med Chem 63: 2833-2853 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00790
BindingDB Entry DOI: 10.7270/Q29027BJ |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM21995
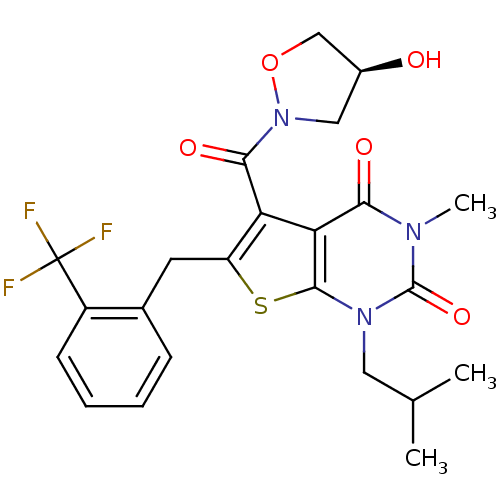
(5-{[(4S)-4-hydroxy-1,2-oxazolidin-2-yl]carbonyl}-3...)Show SMILES CC(C)Cn1c2sc(Cc3ccccc3C(F)(F)F)c(C(=O)N3C[C@H](O)CO3)c2c(=O)n(C)c1=O |r| Show InChI InChI=1S/C23H24F3N3O5S/c1-12(2)9-28-21-18(19(31)27(3)22(28)33)17(20(32)29-10-14(30)11-34-29)16(35-21)8-13-6-4-5-7-15(13)23(24,25)26/h4-7,12,14,30H,8-11H2,1-3H3/t14-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.0900 | -56.8 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
J Med Chem 50: 254-63 (2007)
Article DOI: 10.1021/jm060995h
BindingDB Entry DOI: 10.7270/Q2GT5KGF |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50138124
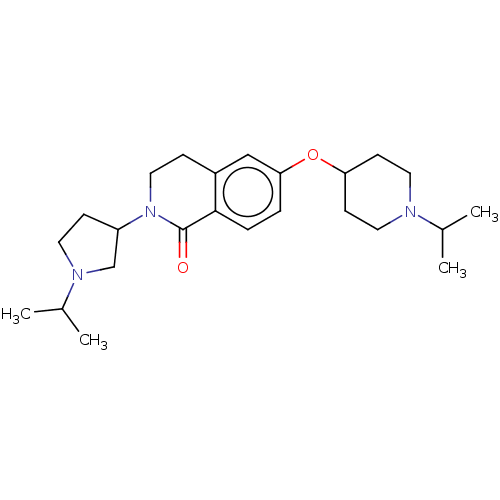
(CHEMBL3753901)Show SMILES CC(C)N1CCC(C1)N1CCc2cc(OC3CCN(CC3)C(C)C)ccc2C1=O Show InChI InChI=1S/C24H37N3O2/c1-17(2)25-12-9-21(10-13-25)29-22-5-6-23-19(15-22)7-14-27(24(23)28)20-8-11-26(16-20)18(3)4/h5-6,15,17-18,20-21H,7-14,16H2,1-4H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to recombinant human H3 receptor |
Eur J Med Chem 108: 655-62 (2016)
Article DOI: 10.1016/j.ejmech.2015.12.005
BindingDB Entry DOI: 10.7270/Q2F191KF |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM22000
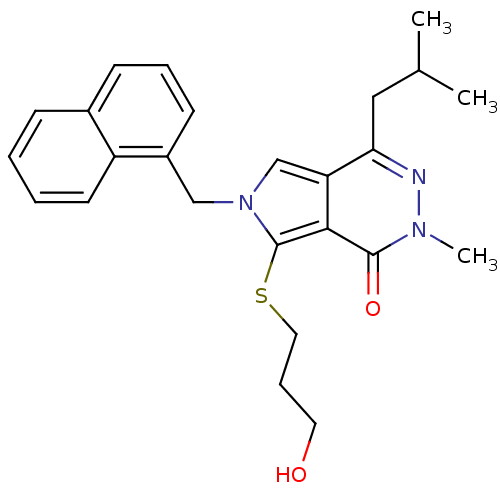
(7-[(3-hydroxypropyl)sulfanyl]-2-methyl-4-(2-methyl...)Show SMILES CC(C)Cc1nn(C)c(=O)c2c(SCCCO)n(Cc3cccc4ccccc34)cc12 Show InChI InChI=1S/C25H29N3O2S/c1-17(2)14-22-21-16-28(15-19-10-6-9-18-8-4-5-11-20(18)19)25(31-13-7-12-29)23(21)24(30)27(3)26-22/h4-6,8-11,16-17,29H,7,12-15H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.100 | -56.5 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
Bioorg Med Chem Lett 16: 2260-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.024
BindingDB Entry DOI: 10.7270/Q2C24TQK |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50538045
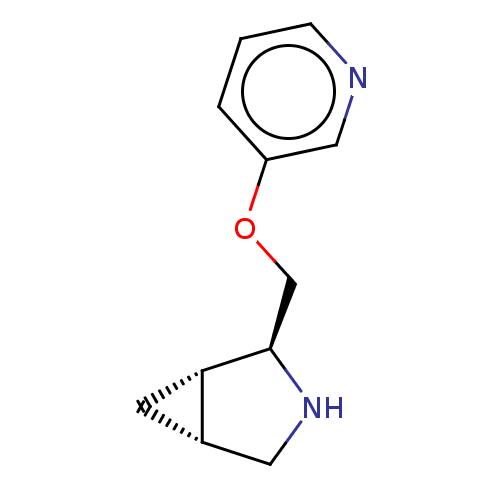
(CHEMBL4636163)Show SMILES Cl.[H][C@@]12C[C@]1([H])[C@@H](COc1cccnc1)NC2 |r| Show InChI InChI=1S/C11H14N2O.ClH/c1-2-9(6-12-3-1)14-7-11-10-4-8(10)5-13-11;/h1-3,6,8,10-11,13H,4-5,7H2;1H/t8-,10-,11+;/m0./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from human alpha4beta2 nAChR expressed in CHOK1 cell membrane by microbeta scintillation counting method |
J Med Chem 63: 2833-2853 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00790
BindingDB Entry DOI: 10.7270/Q29027BJ |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50538008
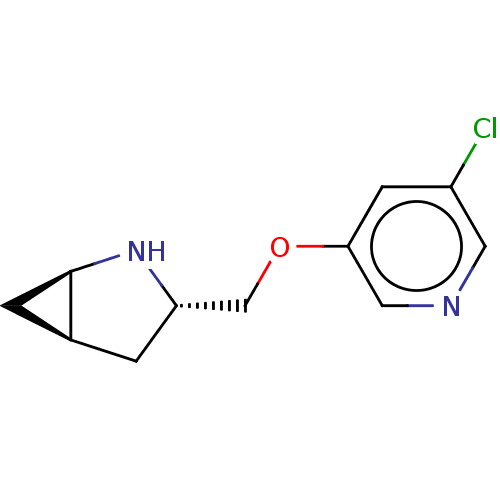
(CHEMBL4638732)Show SMILES Cl.[H][C@]12C[C@@]1([H])N[C@H](COc1cncc(Cl)c1)C2 |r| Show InChI InChI=1S/C11H13ClN2O.ClH/c12-8-3-10(5-13-4-8)15-6-9-1-7-2-11(7)14-9;/h3-5,7,9,11,14H,1-2,6H2;1H/t7-,9-,11+;/m0./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from human alpha4beta2 nAChR expressed in CHOK1 cell membrane by microbeta scintillation counting method |
J Med Chem 63: 2833-2853 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00790
BindingDB Entry DOI: 10.7270/Q29027BJ |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50538047
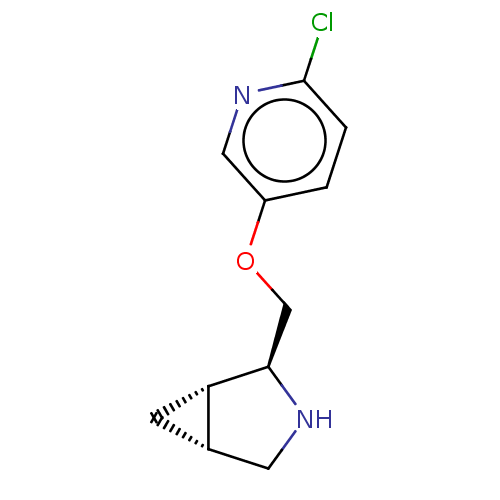
(CHEMBL4647019)Show SMILES [H][C@@]12C[C@]1([H])[C@@H](COc1ccc(Cl)nc1)NC2 |r| Show InChI InChI=1S/C11H13ClN2O/c12-11-2-1-8(5-14-11)15-6-10-9-3-7(9)4-13-10/h1-2,5,7,9-10,13H,3-4,6H2/t7-,9-,10+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from human alpha4beta2 nAChR expressed in CHOK1 cell membrane by microbeta scintillation counting method |
J Med Chem 63: 2833-2853 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00790
BindingDB Entry DOI: 10.7270/Q29027BJ |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50538046
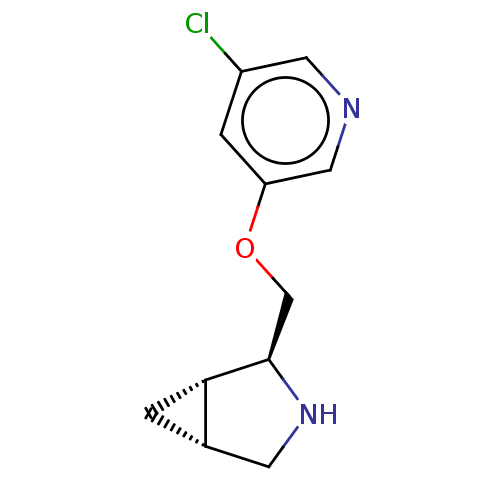
(CHEMBL4647198)Show SMILES Cl.[H][C@@]12C[C@]1([H])[C@@H](COc1cncc(Cl)c1)NC2 |r| Show InChI InChI=1S/C11H13ClN2O.ClH/c12-8-2-9(5-13-4-8)15-6-11-10-1-7(10)3-14-11;/h2,4-5,7,10-11,14H,1,3,6H2;1H/t7-,10-,11+;/m0./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from human alpha4beta2 nAChR expressed in CHOK1 cell membrane by microbeta scintillation counting method |
J Med Chem 63: 2833-2853 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00790
BindingDB Entry DOI: 10.7270/Q29027BJ |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50318633

(3-benzenesulfonyl-8-piperazin-1-ylquinoline | CHEM...)Show InChI InChI=1S/C19H19N3O2S/c23-25(24,16-6-2-1-3-7-16)17-13-15-5-4-8-18(19(15)21-14-17)22-11-9-20-10-12-22/h1-8,13-14,20H,9-12H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Herpes simplex virus type 1 thymidine kinase(HSV-1 TK) |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM22001
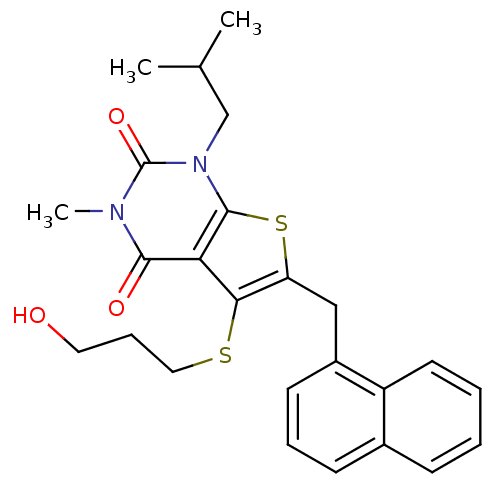
(5-[(3-hydroxypropyl)sulfanyl]-3-methyl-1-(2-methyl...)Show SMILES CC(C)Cn1c2sc(Cc3cccc4ccccc34)c(SCCCO)c2c(=O)n(C)c1=O Show InChI InChI=1S/C25H28N2O3S2/c1-16(2)15-27-24-21(23(29)26(3)25(27)30)22(31-13-7-12-28)20(32-24)14-18-10-6-9-17-8-4-5-11-19(17)18/h4-6,8-11,16,28H,7,12-15H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.280 | -54.0 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
Bioorg Med Chem Lett 16: 2260-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.024
BindingDB Entry DOI: 10.7270/Q2C24TQK |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM22009
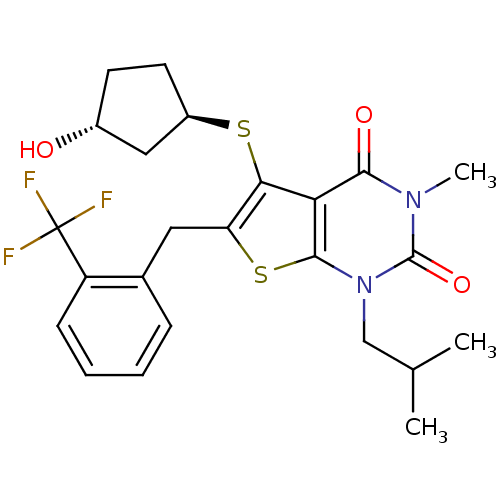
(5-{[(1R,3R)-3-hydroxycyclopentyl]sulfanyl}-3-methy...)Show SMILES CC(C)Cn1c2sc(Cc3ccccc3C(F)(F)F)c(S[C@@H]3CC[C@@H](O)C3)c2c(=O)n(C)c1=O |r| Show InChI InChI=1S/C24H27F3N2O3S2/c1-13(2)12-29-22-19(21(31)28(3)23(29)32)20(33-16-9-8-15(30)11-16)18(34-22)10-14-6-4-5-7-17(14)24(25,26)27/h4-7,13,15-16,30H,8-12H2,1-3H3/t15-,16-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.290 | -53.9 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
Bioorg Med Chem Lett 16: 2260-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.024
BindingDB Entry DOI: 10.7270/Q2C24TQK |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50483134
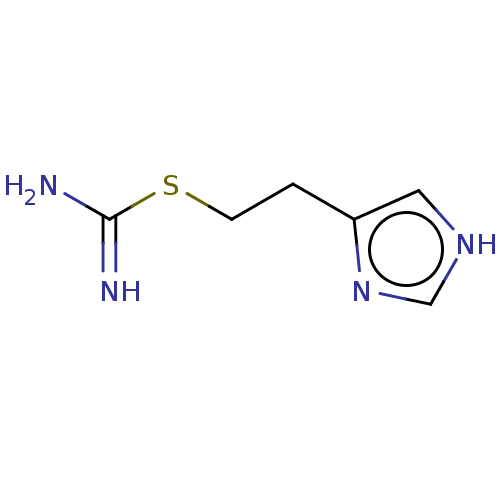
(CHEBI:64156 | Imetit)Show InChI InChI=1S/C6H10N4S/c7-6(8)11-2-1-5-3-9-4-10-5/h3-4H,1-2H2,(H3,7,8)(H,9,10) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
KEGG
PC cid
PC sid
UniChem
| Article
PubMed
| 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3]-RAMH from recombinant human histamine H3 receptor expressed in CHO-K1 cell membranes after 60 mins by scintillation counting |
J Med Chem 62: 1203-1217 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01280
BindingDB Entry DOI: 10.7270/Q2XD152G |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM21992
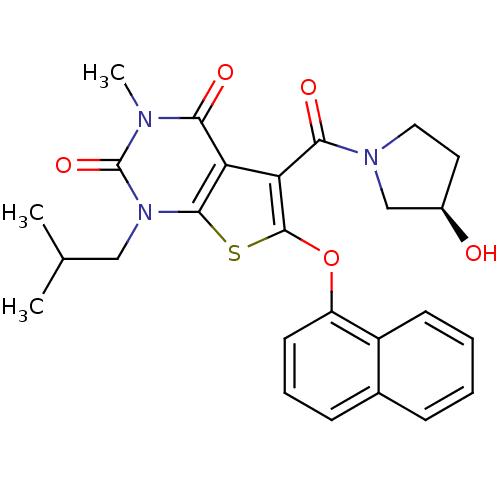
(5-{[(3R)-3-hydroxypyrrolidin-1-yl]carbonyl}-3-meth...)Show SMILES CC(C)Cn1c2sc(Oc3cccc4ccccc34)c(C(=O)N3CC[C@@H](O)C3)c2c(=O)n(C)c1=O |r| Show InChI InChI=1S/C26H27N3O5S/c1-15(2)13-29-24-20(22(31)27(3)26(29)33)21(23(32)28-12-11-17(30)14-28)25(35-24)34-19-10-6-8-16-7-4-5-9-18(16)19/h4-10,15,17,30H,11-14H2,1-3H3/t17-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.310 | -53.7 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
J Med Chem 50: 254-63 (2007)
Article DOI: 10.1021/jm060995h
BindingDB Entry DOI: 10.7270/Q2GT5KGF |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM21986
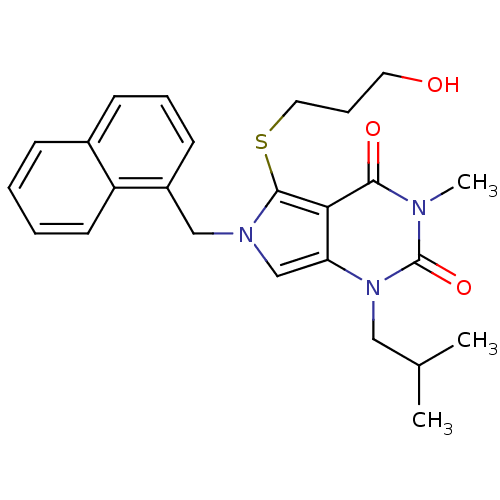
(5-[(3-hydroxypropyl)sulfanyl]-3-methyl-1-(2-methyl...)Show SMILES CC(C)Cn1c2cn(Cc3cccc4ccccc34)c(SCCCO)c2c(=O)n(C)c1=O Show InChI InChI=1S/C25H29N3O3S/c1-17(2)14-28-21-16-27(15-19-10-6-9-18-8-4-5-11-20(18)19)24(32-13-7-12-29)22(21)23(30)26(3)25(28)31/h4-6,8-11,16-17,29H,7,12-15H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.330 | -53.6 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
Bioorg Med Chem Lett 16: 2260-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.024
BindingDB Entry DOI: 10.7270/Q2C24TQK |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM22002
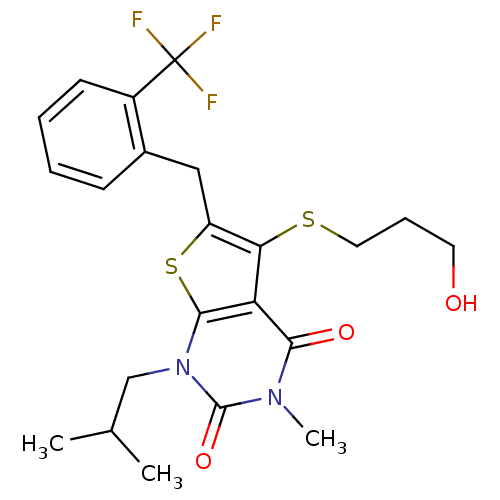
(5-[(3-hydroxypropyl)sulfanyl]-3-methyl-1-(2-methyl...)Show SMILES CC(C)Cn1c2sc(Cc3ccccc3C(F)(F)F)c(SCCCO)c2c(=O)n(C)c1=O Show InChI InChI=1S/C22H25F3N2O3S2/c1-13(2)12-27-20-17(19(29)26(3)21(27)30)18(31-10-6-9-28)16(32-20)11-14-7-4-5-8-15(14)22(23,24)25/h4-5,7-8,13,28H,6,9-12H2,1-3H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.350 | -53.4 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
Bioorg Med Chem Lett 16: 2260-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.024
BindingDB Entry DOI: 10.7270/Q2C24TQK |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50538027
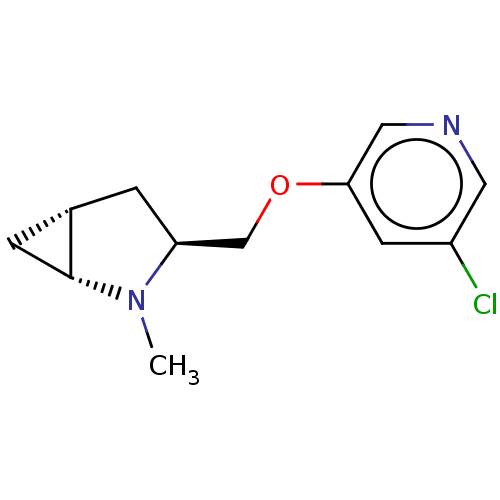
(CHEMBL4644169)Show SMILES [H][C@]12C[C@@]1([H])N(C)[C@H](COc1cncc(Cl)c1)C2 |r| Show InChI InChI=1S/C12H15ClN2O/c1-15-10(2-8-3-12(8)15)7-16-11-4-9(13)5-14-6-11/h4-6,8,10,12H,2-3,7H2,1H3/t8-,10-,12+/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from human alpha4beta2 nAChR expressed in CHOK1 cell membrane by microbeta scintillation counting method |
J Med Chem 63: 2833-2853 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00790
BindingDB Entry DOI: 10.7270/Q29027BJ |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM22010
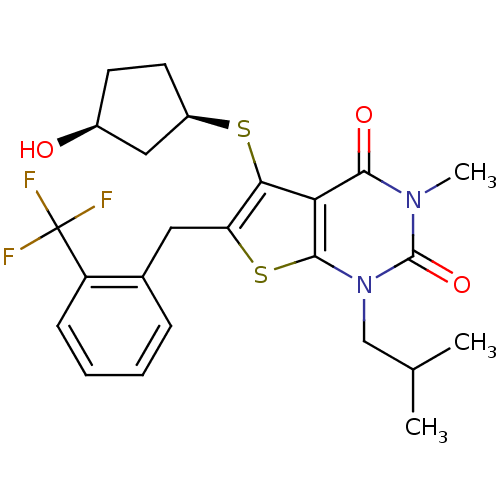
(5-{[(1R,3S)-3-hydroxycyclopentyl]sulfanyl}-3-methy...)Show SMILES CC(C)Cn1c2sc(Cc3ccccc3C(F)(F)F)c(S[C@@H]3CC[C@H](O)C3)c2c(=O)n(C)c1=O |r| Show InChI InChI=1S/C24H27F3N2O3S2/c1-13(2)12-29-22-19(21(31)28(3)23(29)32)20(33-16-9-8-15(30)11-16)18(34-22)10-14-6-4-5-7-17(14)24(25,26)27/h4-7,13,15-16,30H,8-12H2,1-3H3/t15-,16+/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.420 | -53.0 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
Bioorg Med Chem Lett 16: 2260-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.024
BindingDB Entry DOI: 10.7270/Q2C24TQK |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM21996
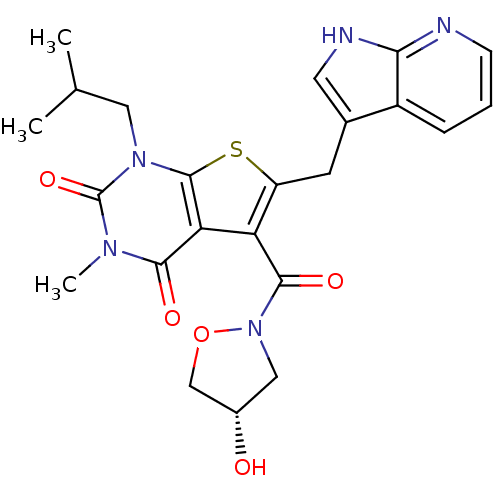
(5-{[(4S)-4-hydroxy-1,2-oxazolidin-2-yl]carbonyl}-3...)Show SMILES CC(C)Cn1c2sc(Cc3c[nH]c4ncccc34)c(C(=O)N3C[C@H](O)CO3)c2c(=O)n(C)c1=O |r| Show InChI InChI=1S/C23H25N5O5S/c1-12(2)9-27-22-18(20(30)26(3)23(27)32)17(21(31)28-10-14(29)11-33-28)16(34-22)7-13-8-25-19-15(13)5-4-6-24-19/h4-6,8,12,14,29H,7,9-11H2,1-3H3,(H,24,25)/t14-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.430 | -52.9 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
J Med Chem 50: 254-63 (2007)
Article DOI: 10.1021/jm060995h
BindingDB Entry DOI: 10.7270/Q2GT5KGF |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM21994

(5-{[(4S)-4-hydroxy-1,2-oxazolidin-2-yl]carbonyl}-3...)Show SMILES CC(C)Cn1c2sc(Cc3ccnc4ccccc34)c(C(=O)N3C[C@H](O)CO3)c2c(=O)n(C)c1=O |r| Show InChI InChI=1S/C25H26N4O5S/c1-14(2)11-28-24-21(22(31)27(3)25(28)33)20(23(32)29-12-16(30)13-34-29)19(35-24)10-15-8-9-26-18-7-5-4-6-17(15)18/h4-9,14,16,30H,10-13H2,1-3H3/t16-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 0.520 | -52.5 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
J Med Chem 50: 254-63 (2007)
Article DOI: 10.1021/jm060995h
BindingDB Entry DOI: 10.7270/Q2GT5KGF |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM22011
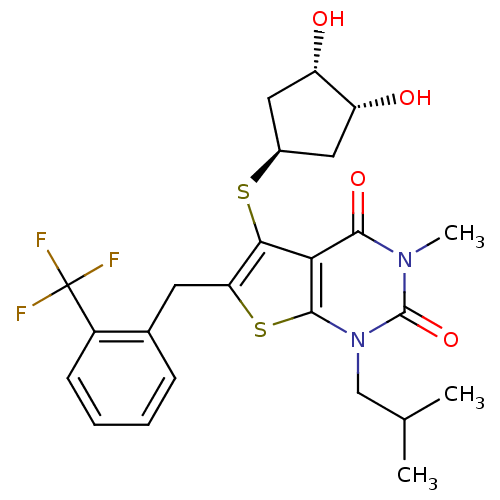
(5-{[(1S,3R,4S)-3,4-dihydroxycyclopentyl]sulfanyl}-...)Show SMILES CC(C)Cn1c2sc(Cc3ccccc3C(F)(F)F)c(S[C@H]3C[C@H](O)[C@H](O)C3)c2c(=O)n(C)c1=O |r| Show InChI InChI=1S/C24H27F3N2O4S2/c1-12(2)11-29-22-19(21(32)28(3)23(29)33)20(34-14-9-16(30)17(31)10-14)18(35-22)8-13-6-4-5-7-15(13)24(25,26)27/h4-7,12,14,16-17,30-31H,8-11H2,1-3H3/t14-,16-,17+ | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.680 | -51.8 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
Bioorg Med Chem Lett 16: 2260-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.024
BindingDB Entry DOI: 10.7270/Q2C24TQK |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM22025

(5-{[(3R)-3-hydroxypyrrolidin-1-yl]carbonyl}-6-(1H-...)Show SMILES CC(C)Cn1c2sc(Cc3c[nH]c4ccccc34)c(C(=O)N3CC[C@@H](O)C3)c2c(=O)n(C)c1=O |r| Show InChI InChI=1S/C25H28N4O4S/c1-14(2)12-29-24-21(22(31)27(3)25(29)33)20(23(32)28-9-8-16(30)13-28)19(34-24)10-15-11-26-18-7-5-4-6-17(15)18/h4-7,11,14,16,26,30H,8-10,12-13H2,1-3H3/t16-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.790 | -51.4 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
AstraZeneca
| Assay Description
A radioligand-binding assay was developed using scintillation proximity assay (SPA) technology. The wheat germ agglutinin SPA beads (Amersham) (0.2 m... |
Bioorg Med Chem Lett 16: 2260-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.024
BindingDB Entry DOI: 10.7270/Q2C24TQK |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50138119

(CHEMBL3753475)Show SMILES O=C1N(CCc2cc(OC3CCN(CC3)C3CCC3)ccc12)C1CCN(CC1)C1CCC1 Show InChI InChI=1S/C27H39N3O2/c31-27-26-8-7-25(32-24-12-16-29(17-13-24)22-5-2-6-22)19-20(26)9-18-30(27)23-10-14-28(15-11-23)21-3-1-4-21/h7-8,19,21-24H,1-6,9-18H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3H]R-alpha-methyl histamine from recombinant human H3 receptor |
Eur J Med Chem 108: 655-62 (2016)
Article DOI: 10.1016/j.ejmech.2015.12.005
BindingDB Entry DOI: 10.7270/Q2F191KF |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM21987

(6-[(6-fluoroquinolin-4-yl)methyl]-5-{[(3R)-3-hydro...)Show SMILES CC(C)Cn1c2sc(Cc3ccnc4ccc(F)cc34)c(C(=O)N3CC[C@@H](O)C3)c2c(=O)n(C)c1=O |r| Show InChI InChI=1S/C26H27FN4O4S/c1-14(2)12-31-25-22(23(33)29(3)26(31)35)21(24(34)30-9-7-17(32)13-30)20(36-25)10-15-6-8-28-19-5-4-16(27)11-18(15)19/h4-6,8,11,14,17,32H,7,9-10,12-13H2,1-3H3/t17-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.810 | -51.4 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
J Med Chem 50: 254-63 (2007)
Article DOI: 10.1021/jm060995h
BindingDB Entry DOI: 10.7270/Q2GT5KGF |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50019754
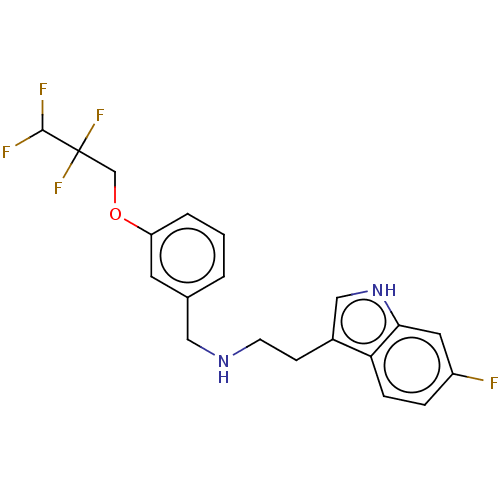
(IDALOPIRDINE | LU-AE58054)Show SMILES FC(F)C(F)(F)COc1cccc(CNCCc2c[nH]c3cc(F)ccc23)c1 Show InChI InChI=1S/C20H19F5N2O/c21-15-4-5-17-14(11-27-18(17)9-15)6-7-26-10-13-2-1-3-16(8-13)28-12-20(24,25)19(22)23/h1-5,8-9,11,19,26-27H,6-7,10,12H2 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.830 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Antagonist activity at 5-HT6 receptor (unknown origin) |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50138120
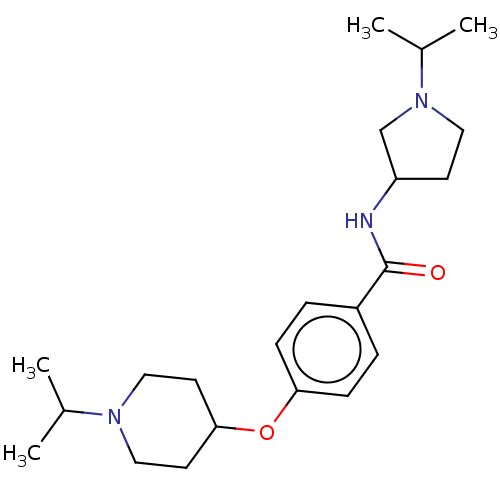
(CHEMBL3753093)Show SMILES CC(C)N1CCC(C1)NC(=O)c1ccc(OC2CCN(CC2)C(C)C)cc1 Show InChI InChI=1S/C22H35N3O2/c1-16(2)24-13-10-21(11-14-24)27-20-7-5-18(6-8-20)22(26)23-19-9-12-25(15-19)17(3)4/h5-8,16-17,19,21H,9-15H2,1-4H3,(H,23,26) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to recombinant human H3 receptor |
Eur J Med Chem 108: 655-62 (2016)
Article DOI: 10.1016/j.ejmech.2015.12.005
BindingDB Entry DOI: 10.7270/Q2F191KF |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50517508
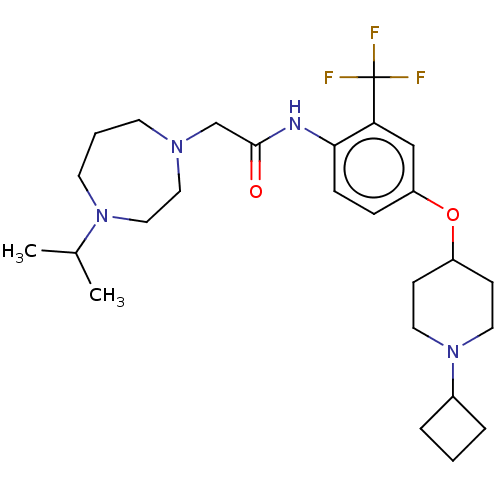
(CHEMBL4594107)Show SMILES CC(C)N1CCCN(CC(=O)Nc2ccc(OC3CCN(CC3)C3CCC3)cc2C(F)(F)F)CC1 Show InChI InChI=1S/C26H39F3N4O2/c1-19(2)32-12-4-11-31(15-16-32)18-25(34)30-24-8-7-22(17-23(24)26(27,28)29)35-21-9-13-33(14-10-21)20-5-3-6-20/h7-8,17,19-21H,3-6,9-16,18H2,1-2H3,(H,30,34) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 0.960 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3]-RAMH from recombinant human histamine H3 receptor expressed in CHO-K1 cell membranes after 60 mins by scintillation counting |
J Med Chem 62: 1203-1217 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01280
BindingDB Entry DOI: 10.7270/Q2XD152G |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50138122
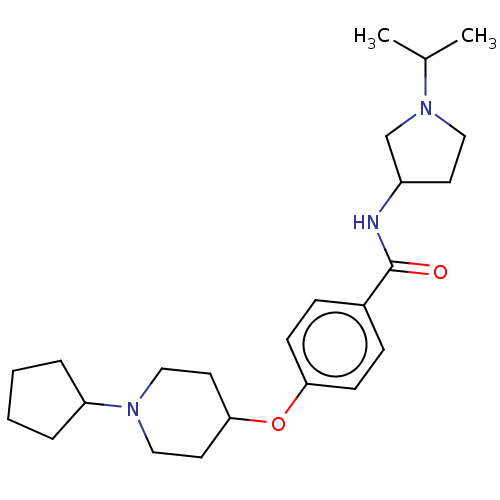
(CHEMBL3752655)Show SMILES CC(C)N1CCC(C1)NC(=O)c1ccc(OC2CCN(CC2)C2CCCC2)cc1 Show InChI InChI=1S/C24H37N3O2/c1-18(2)27-14-11-20(17-27)25-24(28)19-7-9-22(10-8-19)29-23-12-15-26(16-13-23)21-5-3-4-6-21/h7-10,18,20-21,23H,3-6,11-17H2,1-2H3,(H,25,28) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to recombinant human H3 receptor |
Eur J Med Chem 108: 655-62 (2016)
Article DOI: 10.1016/j.ejmech.2015.12.005
BindingDB Entry DOI: 10.7270/Q2F191KF |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50538033
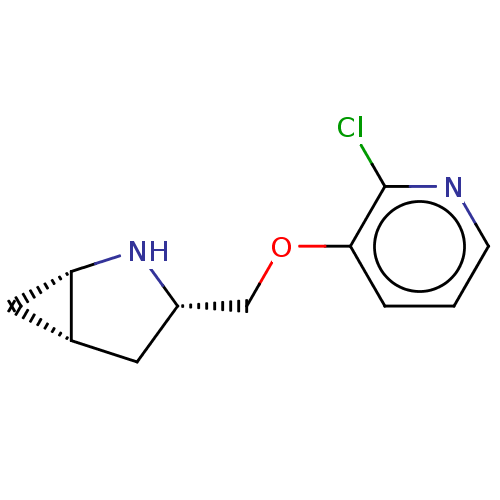
(CHEMBL4648557)Show SMILES Cl.[H][C@@]12C[C@]1([H])N[C@H](COc1cccnc1Cl)C2 |r| Show InChI InChI=1S/C11H13ClN2O.ClH/c12-11-10(2-1-3-13-11)15-6-8-4-7-5-9(7)14-8;/h1-3,7-9,14H,4-6H2;1H/t7-,8+,9+;/m1./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from human alpha4beta2 nAChR expressed in CHOK1 cell membrane by microbeta scintillation counting method |
J Med Chem 63: 2833-2853 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00790
BindingDB Entry DOI: 10.7270/Q29027BJ |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50138123

(CHEMBL3753703)Show SMILES CC(C)CN1CCC(C1)NC(=O)c1ccc(OC2CCN(CC2)C(C)C)cc1 Show InChI InChI=1S/C23H37N3O2/c1-17(2)15-25-12-9-20(16-25)24-23(27)19-5-7-21(8-6-19)28-22-10-13-26(14-11-22)18(3)4/h5-8,17-18,20,22H,9-16H2,1-4H3,(H,24,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3H]R-alpha-methyl histamine from recombinant human H3 receptor |
Eur J Med Chem 108: 655-62 (2016)
Article DOI: 10.1016/j.ejmech.2015.12.005
BindingDB Entry DOI: 10.7270/Q2F191KF |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50517531
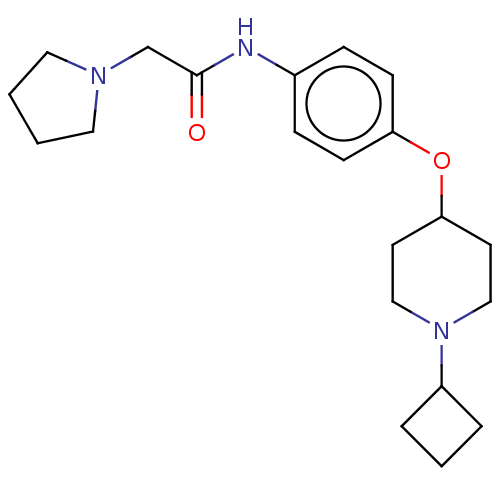
(CHEMBL4445638)Show SMILES Cl.Cl.O=C(CN1CCCC1)Nc1ccc(OC2CCN(CC2)C2CCC2)cc1 Show InChI InChI=1S/C21H31N3O2/c25-21(16-23-12-1-2-13-23)22-17-6-8-19(9-7-17)26-20-10-14-24(15-11-20)18-4-3-5-18/h6-9,18,20H,1-5,10-16H2,(H,22,25) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3]-RAMH from recombinant human histamine H3 receptor expressed in CHO-K1 cell membranes after 60 mins by scintillation counting |
J Med Chem 62: 1203-1217 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01280
BindingDB Entry DOI: 10.7270/Q2XD152G |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50517534
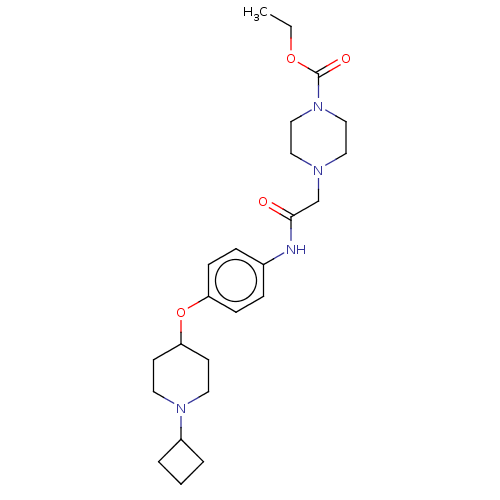
(CHEMBL4476066)Show SMILES Cl.Cl.CCOC(=O)N1CCN(CC(=O)Nc2ccc(OC3CCN(CC3)C3CCC3)cc2)CC1 Show InChI InChI=1S/C24H36N4O4/c1-2-31-24(30)28-16-14-26(15-17-28)18-23(29)25-19-6-8-21(9-7-19)32-22-10-12-27(13-11-22)20-4-3-5-20/h6-9,20,22H,2-5,10-18H2,1H3,(H,25,29) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3]-RAMH from recombinant human histamine H3 receptor expressed in CHO-K1 cell membranes after 60 mins by scintillation counting |
J Med Chem 62: 1203-1217 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01280
BindingDB Entry DOI: 10.7270/Q2XD152G |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50538030
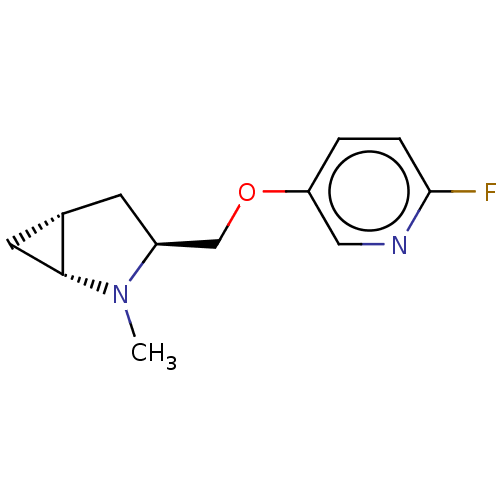
(CHEMBL4638919)Show SMILES Cl.[H][C@]12C[C@@]1([H])N(C)[C@H](COc1ccc(F)nc1)C2 |r| Show InChI InChI=1S/C12H15FN2O.ClH/c1-15-9(4-8-5-11(8)15)7-16-10-2-3-12(13)14-6-10;/h2-3,6,8-9,11H,4-5,7H2,1H3;1H/t8-,9-,11+;/m0./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from human alpha4beta2 nAChR expressed in CHOK1 cell membrane by microbeta scintillation counting method |
J Med Chem 63: 2833-2853 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00790
BindingDB Entry DOI: 10.7270/Q29027BJ |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM22014
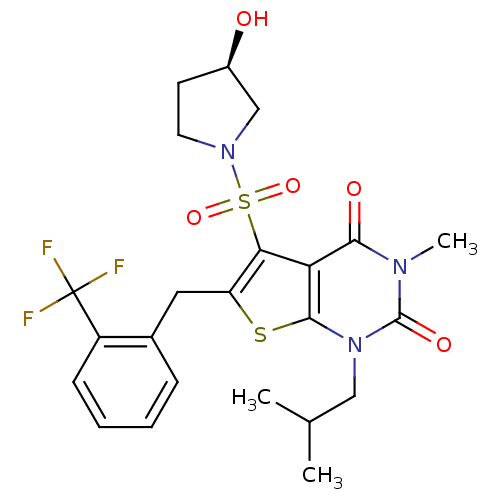
(5-{[(3R)-3-hydroxypyrrolidine-1-]sulfonyl}-3-methy...)Show SMILES CC(C)Cn1c2sc(Cc3ccccc3C(F)(F)F)c(c2c(=O)n(C)c1=O)S(=O)(=O)N1CC[C@@H](O)C1 |r| Show InChI InChI=1S/C23H26F3N3O5S2/c1-13(2)11-29-21-18(20(31)27(3)22(29)32)19(36(33,34)28-9-8-15(30)12-28)17(35-21)10-14-6-4-5-7-16(14)23(24,25)26/h4-7,13,15,30H,8-12H2,1-3H3/t15-/m1/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.10 | -50.6 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
Bioorg Med Chem Lett 16: 2260-5 (2006)
Article DOI: 10.1016/j.bmcl.2006.01.024
BindingDB Entry DOI: 10.7270/Q2C24TQK |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50138117
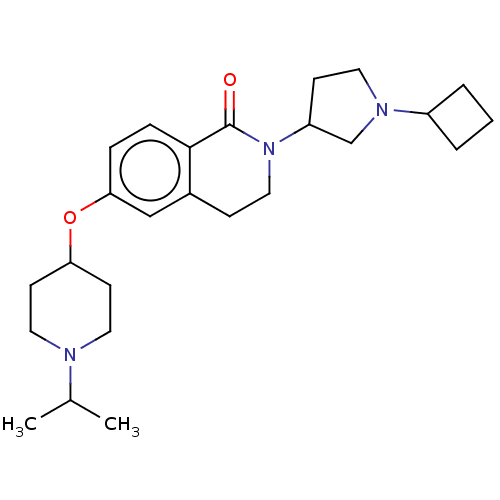
(CHEMBL3753315)Show SMILES CC(C)N1CCC(CC1)Oc1ccc2C(=O)N(CCc2c1)C1CCN(C1)C1CCC1 Show InChI InChI=1S/C25H37N3O2/c1-18(2)26-13-10-22(11-14-26)30-23-6-7-24-19(16-23)8-15-28(25(24)29)21-9-12-27(17-21)20-4-3-5-20/h6-7,16,18,20-22H,3-5,8-15,17H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3H]R-alpha-methyl histamine from recombinant human H3 receptor |
Eur J Med Chem 108: 655-62 (2016)
Article DOI: 10.1016/j.ejmech.2015.12.005
BindingDB Entry DOI: 10.7270/Q2F191KF |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50517529
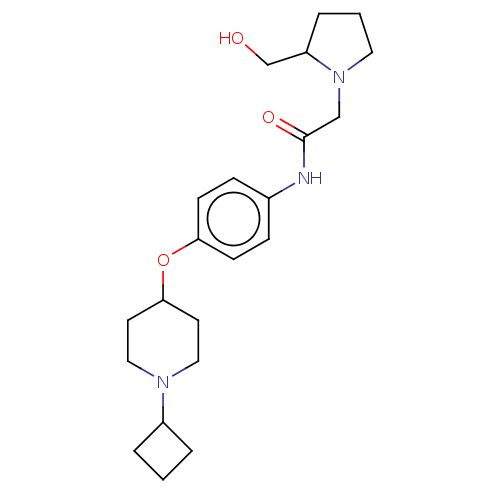
(CHEMBL4587541)Show SMILES Cl.Cl.OCC1CCCN1CC(=O)Nc1ccc(OC2CCN(CC2)C2CCC2)cc1 Show InChI InChI=1S/C22H33N3O3.2ClH/c26-16-19-5-2-12-25(19)15-22(27)23-17-6-8-20(9-7-17)28-21-10-13-24(14-11-21)18-3-1-4-18;;/h6-9,18-19,21,26H,1-5,10-16H2,(H,23,27);2*1H | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3]-RAMH from recombinant human histamine H3 receptor expressed in CHO-K1 cell membranes after 60 mins by scintillation counting |
J Med Chem 62: 1203-1217 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01280
BindingDB Entry DOI: 10.7270/Q2XD152G |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50138123

(CHEMBL3753703)Show SMILES CC(C)CN1CCC(C1)NC(=O)c1ccc(OC2CCN(CC2)C(C)C)cc1 Show InChI InChI=1S/C23H37N3O2/c1-17(2)15-25-12-9-20(16-25)24-23(27)19-5-7-21(8-6-19)28-22-10-13-26(14-11-22)18(3)4/h5-8,17-18,20,22H,9-16H2,1-4H3,(H,24,27) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Binding affinity to recombinant human H3 receptor |
Eur J Med Chem 108: 655-62 (2016)
Article DOI: 10.1016/j.ejmech.2015.12.005
BindingDB Entry DOI: 10.7270/Q2F191KF |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50538038
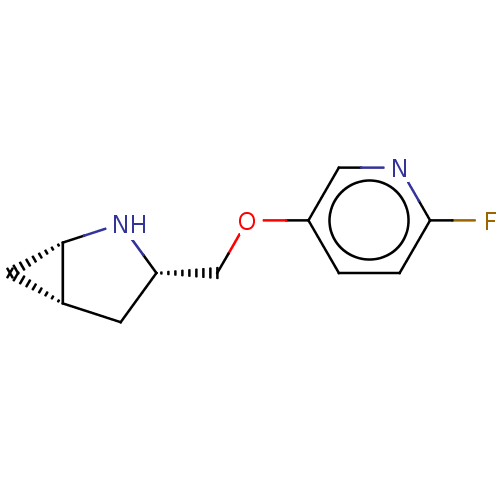
(CHEMBL4648098)Show SMILES Cl.[H][C@@]12C[C@]1([H])N[C@H](COc1ccc(F)nc1)C2 |r| Show InChI InChI=1S/C11H13FN2O.ClH/c12-11-2-1-9(5-13-11)15-6-8-3-7-4-10(7)14-8;/h1-2,5,7-8,10,14H,3-4,6H2;1H/t7-,8+,10+;/m1./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.20 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from human alpha4beta2 nAChR expressed in CHOK1 cell membrane by microbeta scintillation counting method |
J Med Chem 63: 2833-2853 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00790
BindingDB Entry DOI: 10.7270/Q29027BJ |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM21998
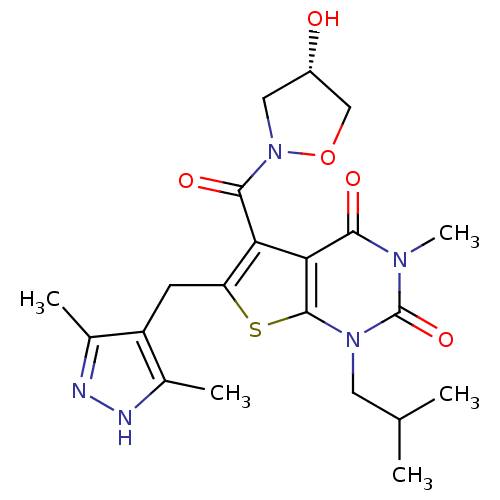
(6-[(3,5-dimethyl-1H-pyrazol-4-yl)methyl]-5-{[(4S)-...)Show SMILES CC(C)Cn1c2sc(Cc3c(C)n[nH]c3C)c(C(=O)N3C[C@H](O)CO3)c2c(=O)n(C)c1=O |r| Show InChI InChI=1S/C21H27N5O5S/c1-10(2)7-25-20-17(18(28)24(5)21(25)30)16(19(29)26-8-13(27)9-31-26)15(32-20)6-14-11(3)22-23-12(14)4/h10,13,27H,6-9H2,1-5H3,(H,22,23)/t13-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.20 | -50.4 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
J Med Chem 50: 254-63 (2007)
Article DOI: 10.1021/jm060995h
BindingDB Entry DOI: 10.7270/Q2GT5KGF |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50044616
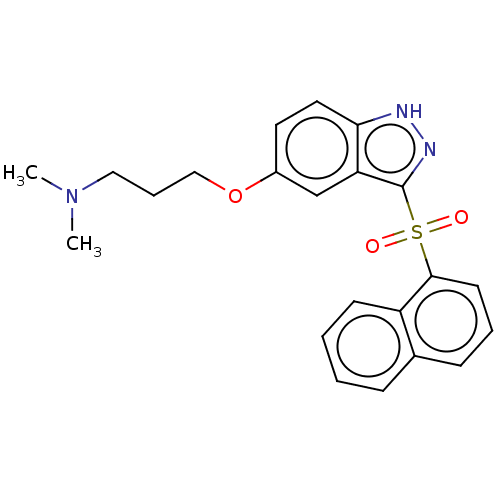
(Cerlapirdine | PF-05212365 | SAM-531 | WAY-262531)Show SMILES CN(C)CCCOc1ccc2[nH]nc(c2c1)S(=O)(=O)c1cccc2ccccc12 Show InChI InChI=1S/C22H23N3O3S/c1-25(2)13-6-14-28-17-11-12-20-19(15-17)22(24-23-20)29(26,27)21-10-5-8-16-7-3-4-9-18(16)21/h3-5,7-12,15H,6,13-14H2,1-2H3,(H,23,24) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
MCE
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Effective concentration required for agonistic activity at Metabotropic glutamate receptor 2 |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50236846
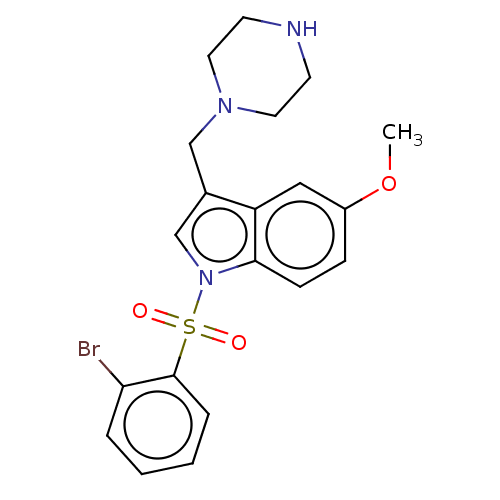
(CHEMBL4080401)Show SMILES O.O.CS(O)(=O)=O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCNCC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C20H22BrN3O3S/c1-27-16-6-7-19-17(12-16)15(13-23-10-8-22-9-11-23)14-24(19)28(25,26)20-5-3-2-4-18(20)21/h2-7,12,14,22H,8-11,13H2,1H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.30 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Inhibition of recombinant mammalian Geranylgeranyl transferase type I expressed in baculovirus |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50538036
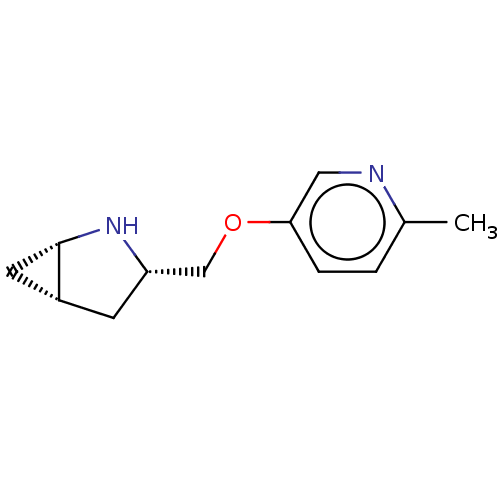
(CHEMBL4634253)Show SMILES Cl.[H][C@@]12C[C@]1([H])N[C@H](COc1ccc(C)nc1)C2 |r| Show InChI InChI=1S/C12H16N2O.ClH/c1-8-2-3-11(6-13-8)15-7-10-4-9-5-12(9)14-10;/h2-3,6,9-10,12,14H,4-5,7H2,1H3;1H/t9-,10+,12+;/m1./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from human alpha4beta2 nAChR expressed in CHOK1 cell membrane by microbeta scintillation counting method |
J Med Chem 63: 2833-2853 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00790
BindingDB Entry DOI: 10.7270/Q29027BJ |
More data for this
Ligand-Target Pair | |
Neuronal acetylcholine receptor subunit alpha-4/beta-2
(Homo sapiens (Human)) | BDBM50538063
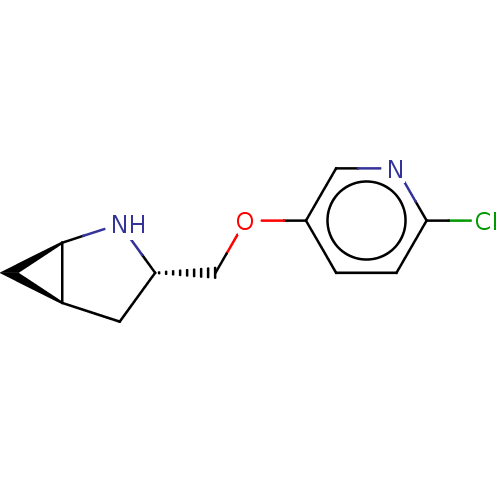
(CHEMBL4642118)Show SMILES Cl.[H][C@]12C[C@@]1([H])N[C@H](COc1ccc(Cl)nc1)C2 |r| Show InChI InChI=1S/C11H13ClN2O.ClH/c12-11-2-1-9(5-13-11)15-6-8-3-7-4-10(7)14-8;/h1-2,5,7-8,10,14H,3-4,6H2;1H/t7-,8-,10+;/m0./s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
antibodypedia
GoogleScholar
AffyNet 
| Purchase
PC cid
PC sid
UniChem
| Article
PubMed
| 1.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd.
Curated by ChEMBL
| Assay Description
Displacement of [3H]cytisine from human alpha4beta2 nAChR expressed in CHOK1 cell membrane by microbeta scintillation counting method |
J Med Chem 63: 2833-2853 (2020)
Article DOI: 10.1021/acs.jmedchem.9b00790
BindingDB Entry DOI: 10.7270/Q29027BJ |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50138106
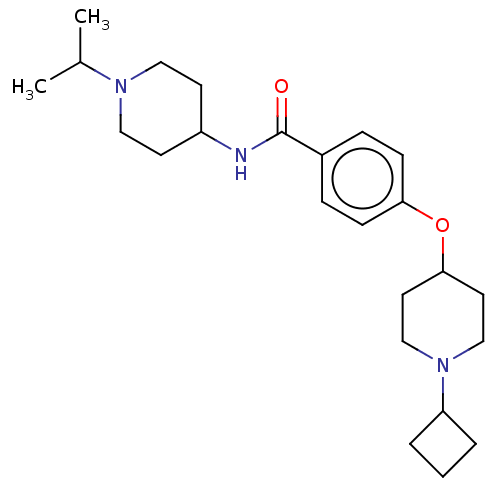
(CHEMBL3753814)Show SMILES CC(C)N1CCC(CC1)NC(=O)c1ccc(OC2CCN(CC2)C2CCC2)cc1 Show InChI InChI=1S/C24H37N3O2/c1-18(2)26-14-10-20(11-15-26)25-24(28)19-6-8-22(9-7-19)29-23-12-16-27(17-13-23)21-4-3-5-21/h6-9,18,20-21,23H,3-5,10-17H2,1-2H3,(H,25,28) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3H]R-alpha-methyl histamine from recombinant human H3 receptor |
Eur J Med Chem 108: 655-62 (2016)
Article DOI: 10.1016/j.ejmech.2015.12.005
BindingDB Entry DOI: 10.7270/Q2F191KF |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50236844
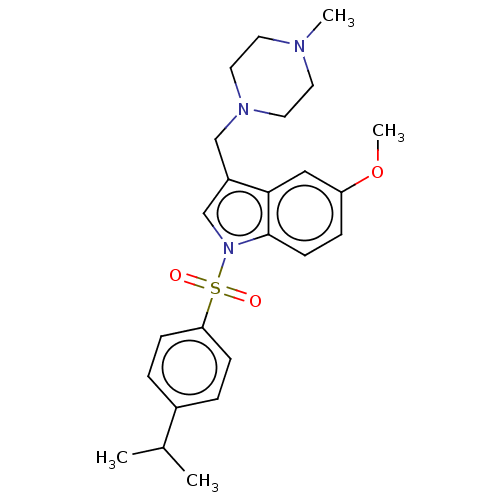
(CHEMBL4100484)Show SMILES CS(O)(=O)=O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccc(cc1)C(C)C Show InChI InChI=1S/C24H31N3O3S/c1-18(2)19-5-8-22(9-6-19)31(28,29)27-17-20(16-26-13-11-25(3)12-14-26)23-15-21(30-4)7-10-24(23)27/h5-10,15,17-18H,11-14,16H2,1-4H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3H]-LSD from recombinant human 5-HT6 receptor expressed in HEK293 cells after 60 mins |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50517507
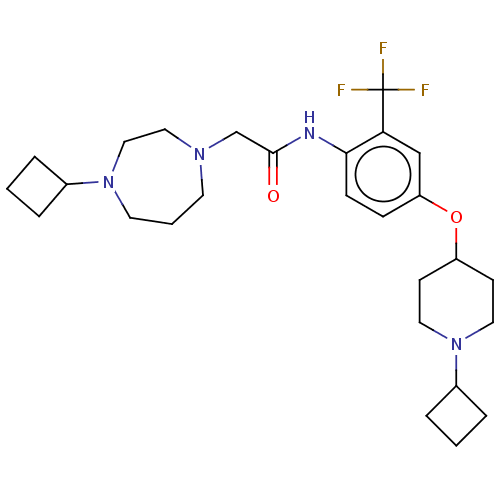
(CHEMBL4456037)Show SMILES FC(F)(F)c1cc(OC2CCN(CC2)C2CCC2)ccc1NC(=O)CN1CCCN(CC1)C1CCC1 Show InChI InChI=1S/C27H39F3N4O2/c28-27(29,30)24-18-23(36-22-10-14-34(15-11-22)21-6-2-7-21)8-9-25(24)31-26(35)19-32-12-3-13-33(17-16-32)20-4-1-5-20/h8-9,18,20-22H,1-7,10-17,19H2,(H,31,35) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3]-RAMH from recombinant human histamine H3 receptor expressed in CHO-K1 cell membranes after 60 mins by scintillation counting |
J Med Chem 62: 1203-1217 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01280
BindingDB Entry DOI: 10.7270/Q2XD152G |
More data for this
Ligand-Target Pair | |
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50236780
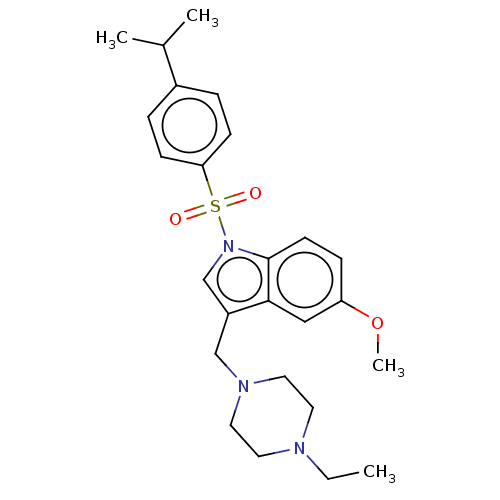
(CHEMBL4068232)Show SMILES CCN1CCN(Cc2cn(c3ccc(OC)cc23)S(=O)(=O)c2ccc(cc2)C(C)C)CC1 Show InChI InChI=1S/C25H33N3O3S/c1-5-26-12-14-27(15-13-26)17-21-18-28(25-11-8-22(31-4)16-24(21)25)32(29,30)23-9-6-20(7-10-23)19(2)3/h6-11,16,18-19H,5,12-15,17H2,1-4H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3H]-LSD from recombinant human 5-HT6 receptor expressed in HEK293 cells after 60 mins |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Monocarboxylate transporter 1
(Homo sapiens (Human)) | BDBM21997
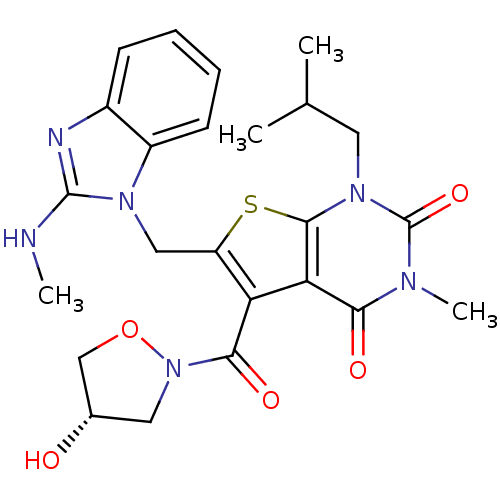
(5-{[(4S)-4-hydroxy-1,2-oxazolidin-2-yl]carbonyl}-3...)Show SMILES CNc1nc2ccccc2n1Cc1sc2n(CC(C)C)c(=O)n(C)c(=O)c2c1C(=O)N1C[C@H](O)CO1 |r| Show InChI InChI=1S/C24H28N6O5S/c1-13(2)9-29-22-19(20(32)27(4)24(29)34)18(21(33)30-10-14(31)12-35-30)17(36-22)11-28-16-8-6-5-7-15(16)26-23(28)25-3/h5-8,13-14,31H,9-12H2,1-4H3,(H,25,26)/t14-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| 1.70 | -49.6 | n/a | n/a | n/a | n/a | n/a | 7.8 | 22 |
AstraZeneca
| Assay Description
Jurkat cell membranes were incubated with [3H]-labeled ligand in the absence or presence of increasing concentrations of test compound. The reagents ... |
J Med Chem 50: 254-63 (2007)
Article DOI: 10.1021/jm060995h
BindingDB Entry DOI: 10.7270/Q2GT5KGF |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50138118
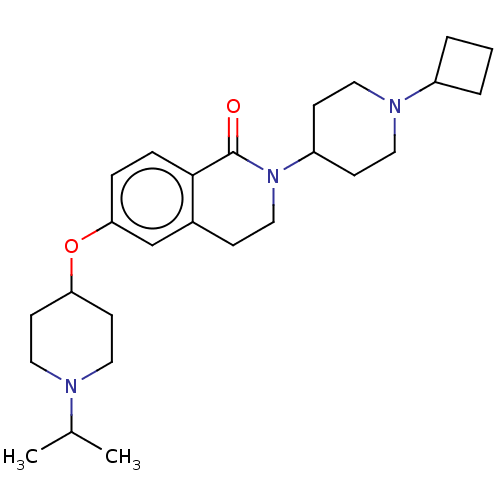
(CHEMBL3754200)Show SMILES CC(C)N1CCC(CC1)Oc1ccc2C(=O)N(CCc2c1)C1CCN(CC1)C1CCC1 Show InChI InChI=1S/C26H39N3O2/c1-19(2)27-15-11-23(12-16-27)31-24-6-7-25-20(18-24)8-17-29(26(25)30)22-9-13-28(14-10-22)21-4-3-5-21/h6-7,18-19,21-23H,3-5,8-17H2,1-2H3 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3H]R-alpha-methyl histamine from recombinant human H3 receptor |
Eur J Med Chem 108: 655-62 (2016)
Article DOI: 10.1016/j.ejmech.2015.12.005
BindingDB Entry DOI: 10.7270/Q2F191KF |
More data for this
Ligand-Target Pair | |
Histamine H3 receptor
(Homo sapiens (Human)) | BDBM50517523
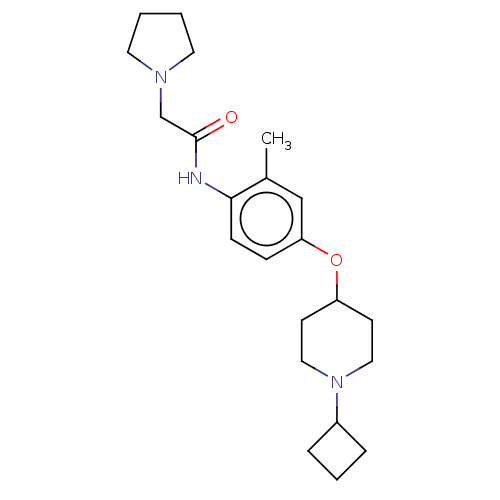
(CHEMBL4518550)Show SMILES Cl.Cl.Cc1cc(OC2CCN(CC2)C2CCC2)ccc1NC(=O)CN1CCCC1 Show InChI InChI=1S/C22H33N3O2/c1-17-15-20(27-19-9-13-25(14-10-19)18-5-4-6-18)7-8-21(17)23-22(26)16-24-11-2-3-12-24/h7-8,15,18-19H,2-6,9-14,16H2,1H3,(H,23,26) | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 1.90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3]-RAMH from recombinant human histamine H3 receptor expressed in CHO-K1 cell membranes after 60 mins by scintillation counting |
J Med Chem 62: 1203-1217 (2019)
Article DOI: 10.1021/acs.jmedchem.8b01280
BindingDB Entry DOI: 10.7270/Q2XD152G |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data