

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kJ/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104706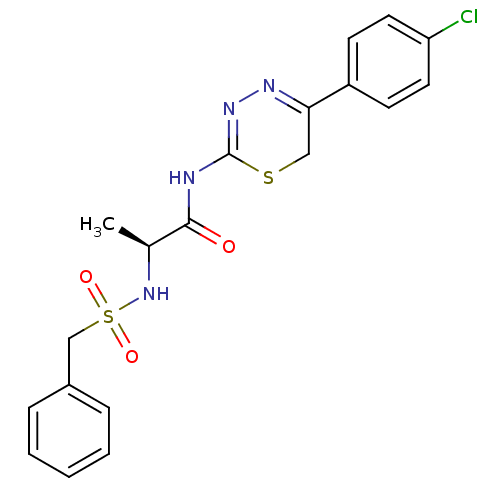 (CHEMBL111179 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104718 ((R)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 40 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104710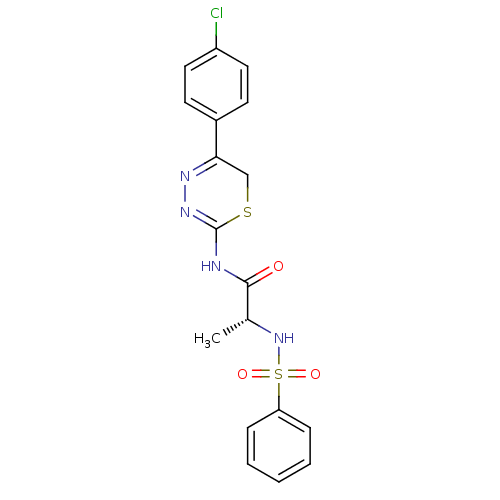 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibition of matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104717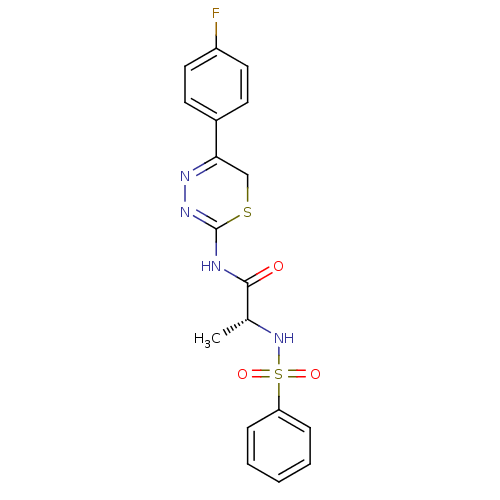 ((R)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 50 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104721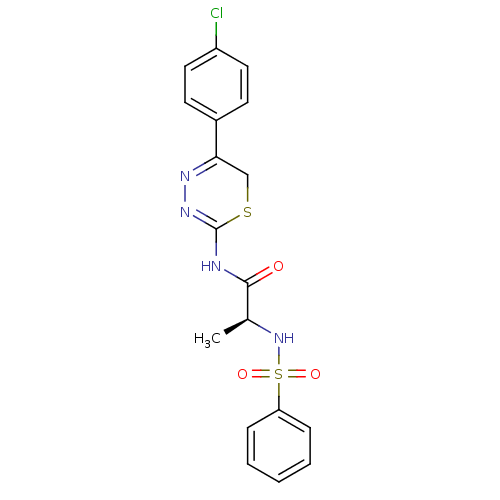 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104714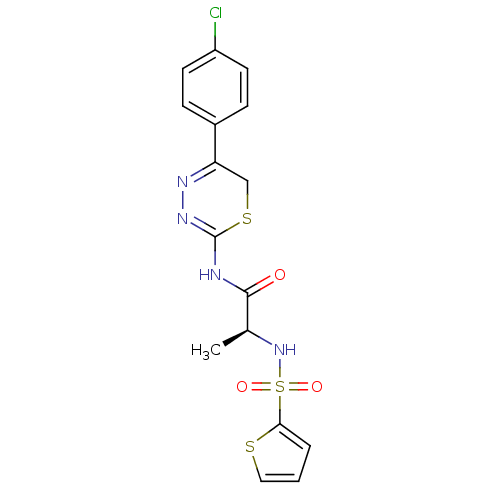 (CHEMBL109861 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 60 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104707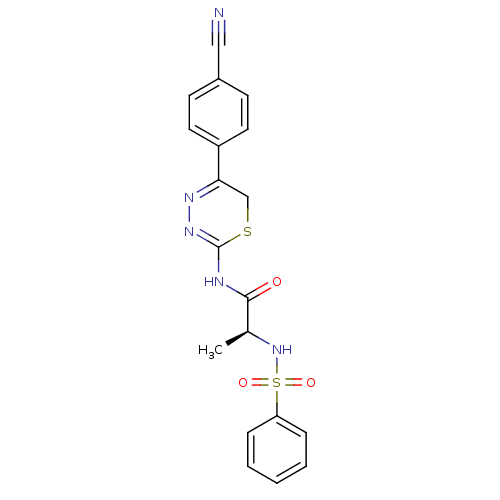 ((S)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104710 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104709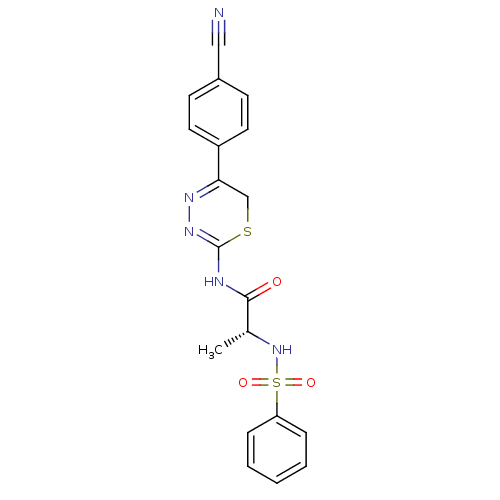 ((R)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104723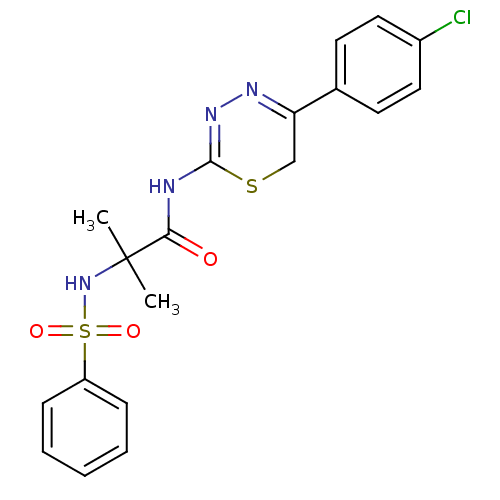 (2-Benzenesulfonylamino-N-[5-(4-chloro-phenyl)-6H-[...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 80 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104716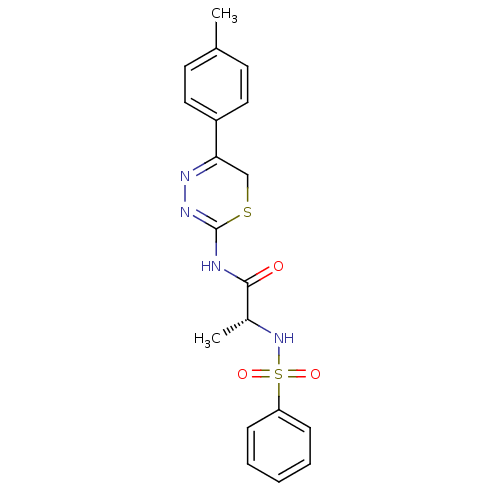 ((R)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104723 (2-Benzenesulfonylamino-N-[5-(4-chloro-phenyl)-6H-[...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104706 (CHEMBL111179 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 90 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104721 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 100 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104723 (2-Benzenesulfonylamino-N-[5-(4-chloro-phenyl)-6H-[...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the ectodomain of the human matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104712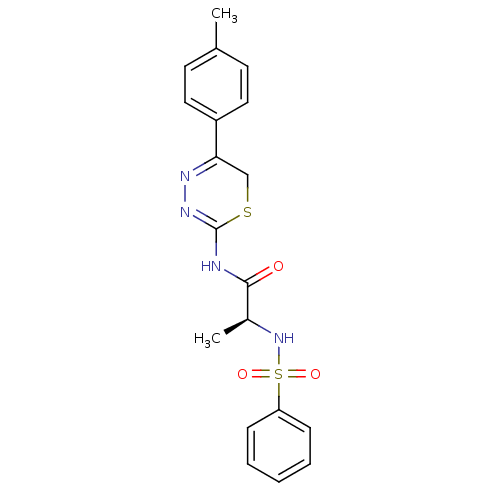 ((S)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104722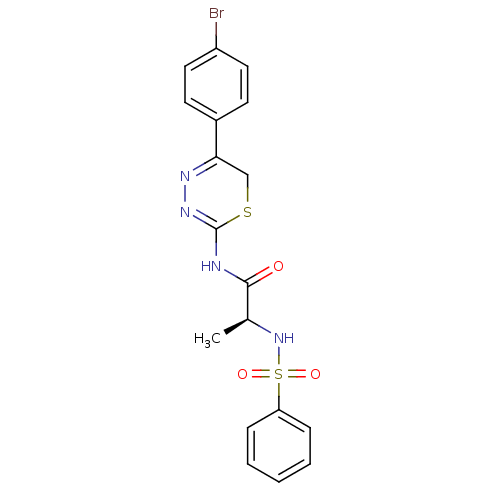 ((S)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 110 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104706 (CHEMBL111179 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 120 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104718 ((R)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104713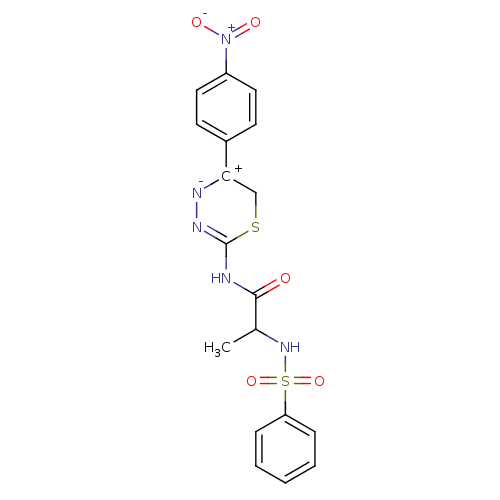 ((S)-N-(5-(4-nitrophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 130 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104710 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104721 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 140 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104710 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104718 ((R)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104723 (2-Benzenesulfonylamino-N-[5-(4-chloro-phenyl)-6H-[...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104716 ((R)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the ectodomain of the human matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104704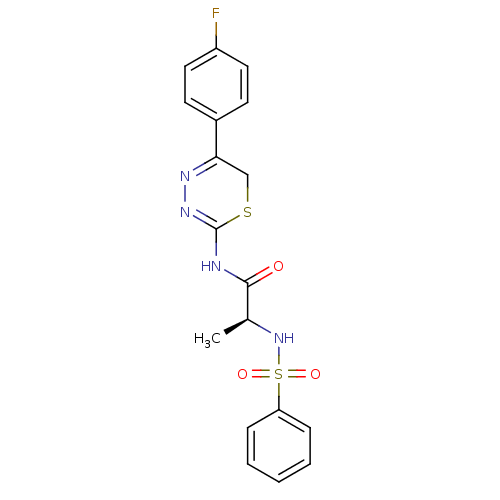 ((S)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104722 ((S)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104712 ((S)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 160 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104711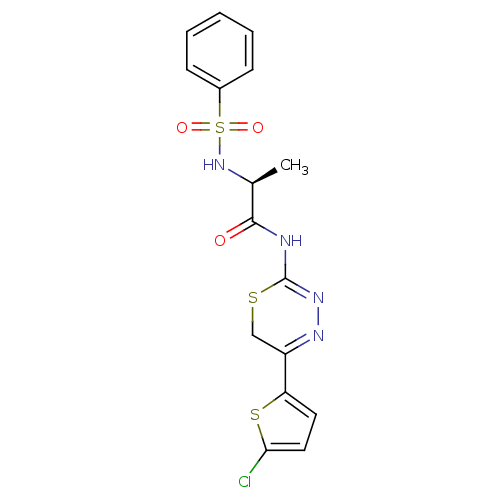 (2-Benzenesulfonylamino-N-[5-(5-chloro-thiophen-2-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104713 ((S)-N-(5-(4-nitrophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 170 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104708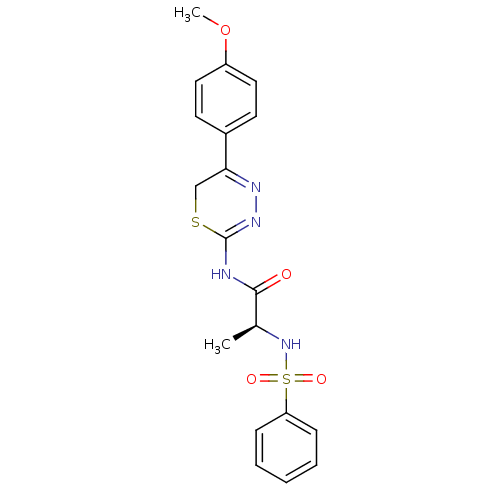 ((S)-N-(5-(4-methoxyphenyl)-6H-1,3,4-thiadiazin-2-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104718 ((R)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the ectodomain of the human matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104711 (2-Benzenesulfonylamino-N-[5-(5-chloro-thiophen-2-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104723 (2-Benzenesulfonylamino-N-[5-(4-chloro-phenyl)-6H-[...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104706 (CHEMBL111179 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the ectodomain of the human matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104721 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 180 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104706 (CHEMBL111179 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104704 ((S)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 190 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104711 (2-Benzenesulfonylamino-N-[5-(5-chloro-thiophen-2-y...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the ectodomain of the human matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104723 (2-Benzenesulfonylamino-N-[5-(4-chloro-phenyl)-6H-[...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104706 (CHEMBL111179 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104719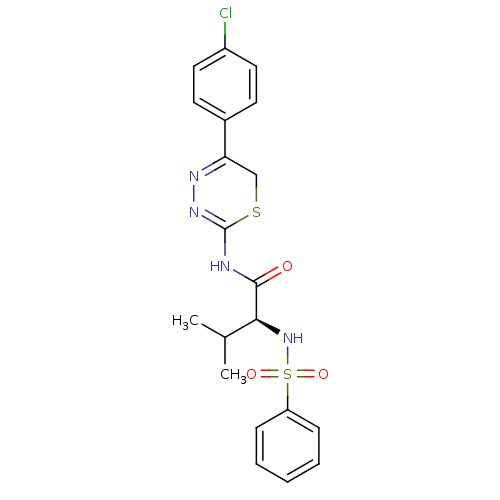 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104718 ((R)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 200 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibition of matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104716 ((R)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104709 ((R)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104709 ((R)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104721 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the ectodomain of the human matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104718 ((R)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104710 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the ectodomain of the human matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104710 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 210 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104716 ((R)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104707 ((S)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 220 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104709 ((R)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 230 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104707 ((S)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104711 (2-Benzenesulfonylamino-N-[5-(5-chloro-thiophen-2-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104709 ((R)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibition of matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104715 ((S)-2-(phenylsulfonamido)-N-(5-(4-(trifluoromethyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104707 ((S)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 240 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibition of matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104707 ((S)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104714 (CHEMBL109861 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 250 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104715 ((S)-2-(phenylsulfonamido)-N-(5-(4-(trifluoromethyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104713 ((S)-N-(5-(4-nitrophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104713 ((S)-N-(5-(4-nitrophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 260 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104722 ((S)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 270 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104704 ((S)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104713 ((S)-N-(5-(4-nitrophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 280 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104716 ((R)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104713 ((S)-N-(5-(4-nitrophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 290 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104717 ((R)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104713 ((S)-N-(5-(4-nitrophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104723 (2-Benzenesulfonylamino-N-[5-(4-chloro-phenyl)-6H-[...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104715 ((S)-2-(phenylsulfonamido)-N-(5-(4-(trifluoromethyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104712 ((S)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104716 ((R)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104708 ((S)-N-(5-(4-methoxyphenyl)-6H-1,3,4-thiadiazin-2-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104710 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104711 (2-Benzenesulfonylamino-N-[5-(5-chloro-thiophen-2-y...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 320 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104720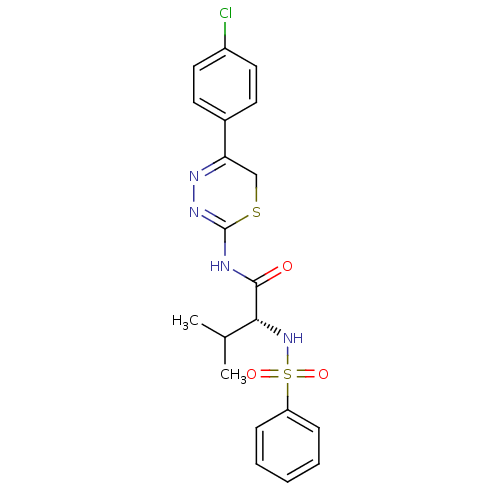 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the ectodomain of the human matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104717 ((R)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the ectodomain of the human matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104717 ((R)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104708 ((S)-N-(5-(4-methoxyphenyl)-6H-1,3,4-thiadiazin-2-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104718 ((R)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104722 ((S)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 340 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibition of matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104720 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104717 ((R)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104717 ((R)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 350 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104709 ((R)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104706 (CHEMBL111179 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104714 (CHEMBL109861 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 360 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104707 ((S)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104722 ((S)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 370 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104719 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104712 ((S)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 380 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104707 ((S)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104717 ((R)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104715 ((S)-2-(phenylsulfonamido)-N-(5-(4-(trifluoromethyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 390 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the ectodomain of the human matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104709 ((R)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104709 ((R)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the ectodomain of the human matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104707 ((S)-N-(5-(4-cyanophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the ectodomain of the human matrix metalloproteinase-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104705 ((S)-N-(5-Adamantan-1-yl-6H-[1,3,4]thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104712 ((S)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 410 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104718 ((R)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 420 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104715 ((S)-2-(phenylsulfonamido)-N-(5-(4-(trifluoromethyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 430 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104722 ((S)-N-(5-(4-bromophenyl)-6H-1,3,4-thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104704 ((S)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104704 ((S)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104716 ((R)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 440 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104711 (2-Benzenesulfonylamino-N-[5-(5-chloro-thiophen-2-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104704 ((S)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 450 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104716 ((R)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 460 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104711 (2-Benzenesulfonylamino-N-[5-(5-chloro-thiophen-2-y...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 470 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104723 (2-Benzenesulfonylamino-N-[5-(4-chloro-phenyl)-6H-[...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104706 (CHEMBL111179 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 480 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104710 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 500 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104708 ((S)-N-(5-(4-methoxyphenyl)-6H-1,3,4-thiadiazin-2-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Macrophage metalloelastase (Homo sapiens (Human)) | BDBM50104721 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 520 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-12 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104714 (CHEMBL109861 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104720 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 550 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104715 ((S)-2-(phenylsulfonamido)-N-(5-(4-(trifluoromethyl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 570 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104715 ((S)-2-(phenylsulfonamido)-N-(5-(4-(trifluoromethyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104708 ((S)-N-(5-(4-methoxyphenyl)-6H-1,3,4-thiadiazin-2-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 580 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104720 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104708 ((S)-N-(5-(4-methoxyphenyl)-6H-1,3,4-thiadiazin-2-y...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 590 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104712 ((S)-2-(phenylsulfonamido)-N-(5-p-tolyl-6H-1,3,4-th...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 620 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104717 ((R)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 630 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104721 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104704 ((S)-N-(5-(4-fluorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 650 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104721 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 730 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104719 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 740 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104719 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 780 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104714 (CHEMBL109861 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 800 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104720 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 850 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104719 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 880 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Interstitial collagenase (Homo sapiens (Human)) | BDBM50104714 (CHEMBL109861 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.04E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-1 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104708 ((S)-N-(5-(4-methoxyphenyl)-6H-1,3,4-thiadiazin-2-y...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 1.33E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104711 (2-Benzenesulfonylamino-N-[5-(5-chloro-thiophen-2-y...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 1.87E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-14 (Homo sapiens (Human)) | BDBM50104705 ((S)-N-(5-Adamantan-1-yl-6H-[1,3,4]thiadiazin-2-yl)...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | 3.07E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of human matrix metalloprotease-14 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104714 (CHEMBL109861 | N-[5-(4-Chloro-phenyl)-6H-[1,3,4]th...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.81E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104720 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 4.37E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104720 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104705 ((S)-N-(5-Adamantan-1-yl-6H-[1,3,4]thiadiazin-2-yl)...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 72 kDa type IV collagenase (Homo sapiens (Human)) | BDBM50104705 ((S)-N-(5-Adamantan-1-yl-6H-[1,3,4]thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibitory activity against the catalytic domain of the human matrix metalloprotease-2 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neutrophil collagenase (Homo sapiens (Human)) | BDBM50104705 ((S)-N-(5-Adamantan-1-yl-6H-[1,3,4]thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description Inhibitory activity against matrix metalloprotease-8 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104720 ((R)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Collagenase 3 (Homo sapiens (Human)) | BDBM50104719 ((S)-N-(5-(4-chlorophenyl)-6H-1,3,4-thiadiazin-2-yl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In Vitro inhibitory activity against the catalytic domain of the matrix metalloprotease-13 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Matrix metalloproteinase-9 (Homo sapiens (Human)) | BDBM50104705 ((S)-N-(5-Adamantan-1-yl-6H-[1,3,4]thiadiazin-2-yl)...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | PubMed | >1.50E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universität Bielefeld Curated by ChEMBL | Assay Description In vitro inhibition of human matrix metalloprotease-9 | J Med Chem 44: 3231-43 (2001) BindingDB Entry DOI: 10.7270/Q2RN3758 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||