 Found 90 hits of Enzyme Inhibition Constant Data
Found 90 hits of Enzyme Inhibition Constant Data Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kJ/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357914
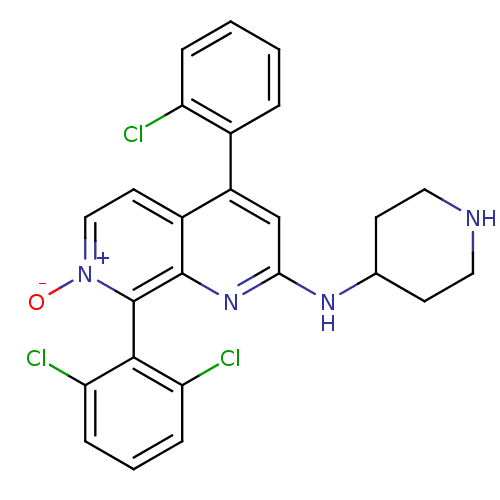
(CHEMBL1916528)Show SMILES [O-][n+]1ccc2c(cc(NC3CCNCC3)nc2c1-c1c(Cl)cccc1Cl)-c1ccccc1Cl |(20.25,-36.83,;21.59,-36.06,;21.59,-34.52,;22.92,-33.75,;24.25,-34.51,;25.59,-33.73,;26.93,-34.51,;26.93,-36.06,;28.26,-36.83,;29.59,-36.07,;30.92,-36.85,;32.25,-36.09,;32.27,-34.55,;30.93,-33.77,;29.59,-34.53,;25.58,-36.83,;24.25,-36.06,;22.92,-36.83,;22.93,-38.37,;21.59,-39.14,;20.26,-38.37,;21.59,-40.68,;22.93,-41.45,;24.27,-40.66,;24.26,-39.13,;25.59,-38.35,;25.59,-32.2,;26.93,-31.43,;26.93,-29.89,;25.59,-29.12,;24.25,-29.9,;24.26,-31.44,;22.93,-32.21,)| Show InChI InChI=1S/C25H21Cl3N4O/c26-19-5-2-1-4-16(19)18-14-22(30-15-8-11-29-12-9-15)31-24-17(18)10-13-32(33)25(24)23-20(27)6-3-7-21(23)28/h1-7,10,13-15,29H,8-9,11-12H2,(H,30,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.130 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357913
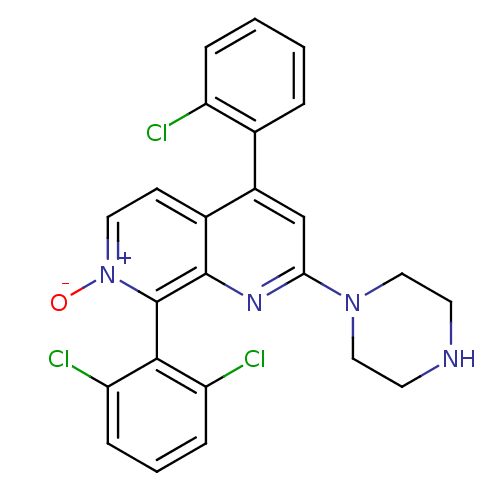
(CHEMBL1916527)Show SMILES [O-][n+]1ccc2c(cc(nc2c1-c1c(Cl)cccc1Cl)N1CCNCC1)-c1ccccc1Cl |(6.31,-36.87,;7.64,-36.1,;7.64,-34.56,;8.97,-33.79,;10.31,-34.55,;11.64,-33.77,;12.98,-34.55,;12.98,-36.1,;11.64,-36.87,;10.3,-36.1,;8.98,-36.87,;8.98,-38.4,;7.64,-39.18,;6.31,-38.41,;7.65,-40.71,;8.98,-41.48,;10.32,-40.7,;10.31,-39.17,;11.64,-38.39,;14.3,-36.87,;14.29,-38.41,;15.62,-39.18,;16.96,-38.42,;16.97,-36.88,;15.63,-36.1,;11.64,-32.24,;12.98,-31.47,;12.98,-29.93,;11.64,-29.16,;10.31,-29.94,;10.31,-31.48,;8.98,-32.25,)| Show InChI InChI=1S/C24H19Cl3N4O/c25-18-5-2-1-4-15(18)17-14-21(30-12-9-28-10-13-30)29-23-16(17)8-11-31(32)24(23)22-19(26)6-3-7-20(22)27/h1-8,11,14,28H,9-10,12-13H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357909
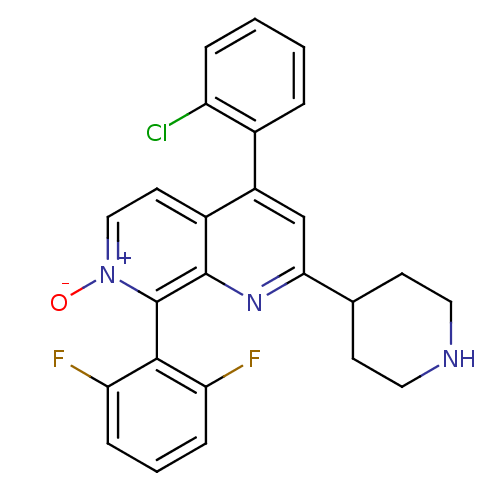
(CHEMBL1916523)Show SMILES [O-][n+]1ccc2c(cc(nc2c1-c1c(F)cccc1F)C1CCNCC1)-c1ccccc1Cl |(-8.74,-20.09,;-7.4,-19.32,;-7.4,-17.78,;-6.07,-17.01,;-4.74,-17.77,;-3.4,-16.99,;-2.06,-17.77,;-2.07,-19.32,;-3.41,-20.09,;-4.74,-19.32,;-6.07,-20.09,;-6.06,-21.63,;-7.4,-22.4,;-8.73,-21.63,;-7.4,-23.94,;-6.06,-24.71,;-4.72,-23.92,;-4.73,-22.39,;-3.4,-21.61,;-.73,-20.09,;-.74,-21.63,;.59,-22.4,;1.93,-21.64,;1.93,-20.1,;.6,-19.32,;-3.4,-15.46,;-2.07,-14.69,;-2.06,-13.15,;-3.4,-12.38,;-4.74,-13.16,;-4.73,-14.7,;-6.06,-15.47,)| Show InChI InChI=1S/C25H20ClF2N3O/c26-19-5-2-1-4-16(19)18-14-22(15-8-11-29-12-9-15)30-24-17(18)10-13-31(32)25(24)23-20(27)6-3-7-21(23)28/h1-7,10,13-15,29H,8-9,11-12H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.270 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357911
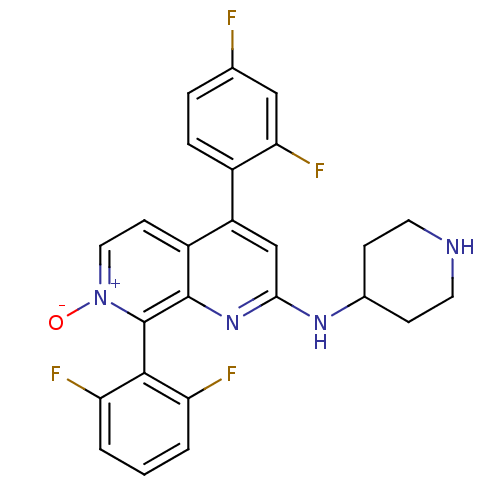
(CHEMBL1916525)Show SMILES [O-][n+]1ccc2c(cc(NC3CCNCC3)nc2c1-c1c(F)cccc1F)-c1ccc(F)cc1F |(23.08,-19.61,;24.41,-18.84,;24.41,-17.3,;25.74,-16.53,;27.08,-17.29,;28.41,-16.51,;29.75,-17.29,;29.75,-18.84,;31.08,-19.62,;32.42,-18.85,;33.75,-19.63,;35.08,-18.87,;35.09,-17.33,;33.76,-16.55,;32.41,-17.31,;28.41,-19.61,;27.08,-18.84,;25.75,-19.61,;25.75,-21.15,;27.08,-21.91,;28.41,-21.13,;27.09,-23.45,;25.76,-24.23,;24.42,-23.46,;24.42,-21.92,;23.08,-21.15,;28.41,-14.98,;29.75,-14.21,;29.75,-12.67,;28.42,-11.9,;28.41,-10.36,;27.08,-12.68,;27.08,-14.22,;25.75,-14.99,)| Show InChI InChI=1S/C25H20F4N4O/c26-14-4-5-16(21(29)12-14)18-13-22(31-15-6-9-30-10-7-15)32-24-17(18)8-11-33(34)25(24)23-19(27)2-1-3-20(23)28/h1-5,8,11-13,15,30H,6-7,9-10H2,(H,31,32) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 0.280 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357916
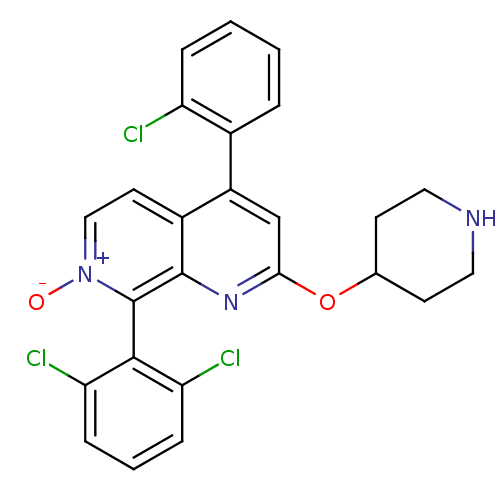
(CHEMBL1916530)Show SMILES [O-][n+]1ccc2c(cc(OC3CCNCC3)nc2c1-c1c(Cl)cccc1Cl)-c1ccccc1Cl |(8.24,-4.99,;9.58,-4.22,;9.58,-2.68,;10.91,-1.9,;12.24,-2.67,;13.58,-1.89,;14.92,-2.67,;14.91,-4.22,;16.25,-4.99,;17.58,-4.23,;18.91,-5.01,;20.25,-4.25,;20.26,-2.71,;18.93,-1.93,;17.58,-2.69,;13.57,-4.99,;12.24,-4.22,;10.91,-4.99,;10.92,-6.52,;9.58,-7.3,;8.25,-6.53,;9.58,-8.83,;10.92,-9.6,;12.26,-8.82,;12.25,-7.29,;13.58,-6.51,;13.58,-.36,;14.91,.41,;14.92,1.95,;13.58,2.72,;12.24,1.94,;12.25,.4,;10.92,-.37,)| Show InChI InChI=1S/C25H20Cl3N3O2/c26-19-5-2-1-4-16(19)18-14-22(33-15-8-11-29-12-9-15)30-24-17(18)10-13-31(32)25(24)23-20(27)6-3-7-21(23)28/h1-7,10,13-15,29H,8-9,11-12H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.460 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357903
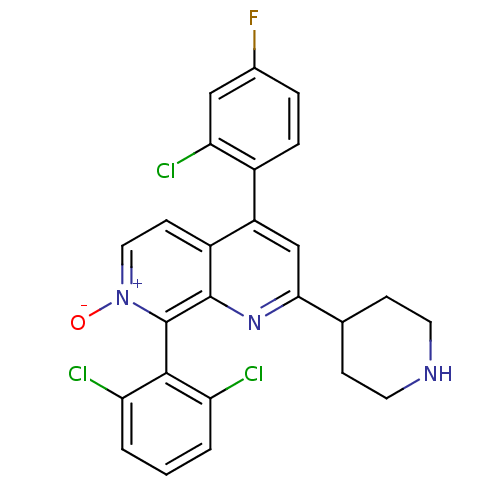
(CHEMBL1916360)Show SMILES [O-][n+]1ccc2c(cc(nc2c1-c1c(Cl)cccc1Cl)C1CCNCC1)-c1ccc(F)cc1Cl |(-9.19,-39.37,;-7.86,-38.61,;-7.86,-37.06,;-6.53,-36.29,;-5.19,-37.05,;-3.86,-36.28,;-2.52,-37.06,;-2.52,-38.6,;-3.87,-39.37,;-5.2,-38.6,;-6.53,-39.38,;-6.52,-40.91,;-7.86,-41.68,;-9.19,-40.91,;-7.85,-43.22,;-6.52,-43.99,;-5.18,-43.21,;-5.19,-41.67,;-3.86,-40.89,;-1.19,-39.37,;-1.2,-40.91,;.13,-41.69,;1.47,-40.93,;1.47,-39.38,;.14,-38.6,;-3.86,-34.75,;-2.52,-33.98,;-2.52,-32.44,;-3.86,-31.67,;-3.86,-30.13,;-5.2,-32.45,;-5.19,-33.98,;-6.52,-34.76,)| Show InChI InChI=1S/C25H19Cl3FN3O/c26-19-2-1-3-20(27)23(19)25-24-17(8-11-32(25)33)18(16-5-4-15(29)12-21(16)28)13-22(31-24)14-6-9-30-10-7-14/h1-5,8,11-14,30H,6-7,9-10H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.490 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357915
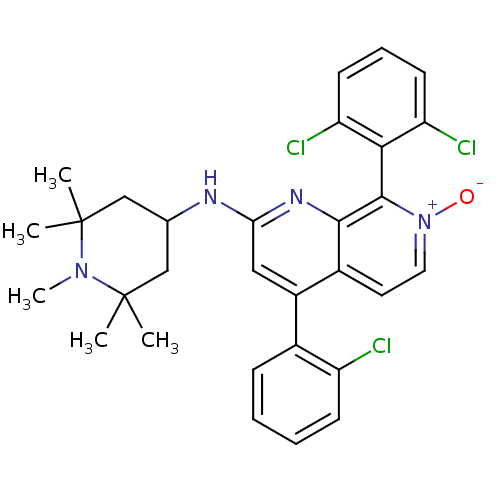
(CHEMBL1916529)Show SMILES CN1C(C)(C)CC(CC1(C)C)Nc1cc(-c2ccccc2Cl)c2cc[n+]([O-])c(-c3c(Cl)cccc3Cl)c2n1 |(4.35,-2.11,;3.01,-2.87,;2.99,-4.41,;4.53,-4.41,;3.76,-5.74,;1.66,-5.17,;.33,-4.39,;.33,-2.86,;1.67,-2.09,;.89,-.75,;2.44,-.75,;-1.01,-5.16,;-2.34,-4.38,;-2.34,-2.84,;-3.67,-2.06,;-3.67,-.52,;-2.34,.24,;-2.34,1.78,;-3.67,2.55,;-5.01,1.78,;-5,.24,;-6.33,-.53,;-5.01,-2.83,;-6.34,-2.07,;-7.67,-2.84,;-7.67,-4.38,;-9.01,-5.15,;-6.34,-5.15,;-6.33,-6.69,;-7.67,-7.46,;-9,-6.69,;-7.67,-9,;-6.33,-9.77,;-4.99,-8.98,;-5,-7.45,;-3.67,-6.67,;-5.01,-4.38,;-3.68,-5.15,)| Show InChI InChI=1S/C30H31Cl3N4O/c1-29(2)16-18(17-30(3,4)36(29)5)34-25-15-21(19-9-6-7-10-22(19)31)20-13-14-37(38)28(27(20)35-25)26-23(32)11-8-12-24(26)33/h6-15,18H,16-17H2,1-5H3,(H,34,35) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357906
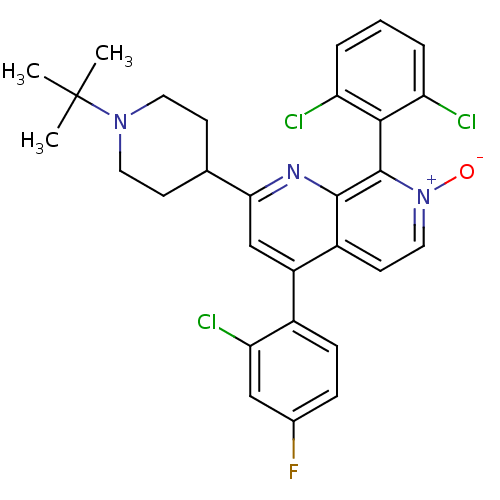
(CHEMBL1916520)Show SMILES CC(C)(C)N1CCC(CC1)c1cc(-c2ccc(F)cc2Cl)c2cc[n+]([O-])c(-c3c(Cl)cccc3Cl)c2n1 |(2.33,-7.59,;2.34,-6.04,;3.68,-5.28,;3.67,-6.81,;1.02,-5.27,;-.33,-6.03,;-1.65,-5.25,;-1.64,-3.71,;-.31,-2.94,;1.02,-3.72,;-2.98,-2.94,;-2.97,-1.4,;-4.31,-.62,;-4.31,.91,;-2.98,1.68,;-2.97,3.22,;-4.31,3.99,;-4.31,5.53,;-5.65,3.22,;-5.64,1.68,;-6.97,.91,;-5.65,-1.39,;-6.98,-.63,;-8.31,-1.4,;-8.31,-2.94,;-9.65,-3.71,;-6.98,-3.72,;-6.97,-5.25,;-8.31,-6.02,;-9.64,-5.25,;-8.31,-7.56,;-6.97,-8.33,;-5.63,-7.55,;-5.64,-6.01,;-4.31,-5.23,;-5.65,-2.94,;-4.32,-3.71,)| Show InChI InChI=1S/C29H27Cl3FN3O/c1-29(2,3)35-12-9-17(10-13-35)25-16-21(19-8-7-18(33)15-24(19)32)20-11-14-36(37)28(27(20)34-25)26-22(30)5-4-6-23(26)31/h4-8,11,14-17H,9-10,12-13H2,1-3H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.530 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357905
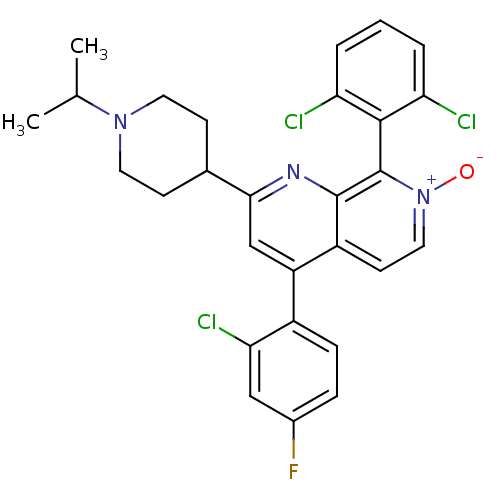
(CHEMBL1916519)Show SMILES CC(C)N1CCC(CC1)c1cc(-c2ccc(F)cc2Cl)c2cc[n+]([O-])c(-c3c(Cl)cccc3Cl)c2n1 |(32.53,-43.15,;32.54,-41.61,;33.88,-40.85,;31.21,-40.83,;29.87,-41.59,;28.54,-40.82,;28.55,-39.28,;29.88,-38.51,;31.22,-39.29,;27.23,-38.51,;27.23,-36.96,;25.89,-36.19,;25.89,-34.65,;27.23,-33.88,;27.23,-32.35,;25.89,-31.57,;25.89,-30.03,;24.55,-32.35,;24.56,-33.89,;23.23,-34.66,;24.56,-36.96,;23.22,-36.2,;21.89,-36.97,;21.89,-38.51,;20.56,-39.28,;23.22,-39.28,;23.23,-40.82,;21.89,-41.59,;20.56,-40.82,;21.9,-43.13,;23.23,-43.89,;24.57,-43.11,;24.56,-41.58,;25.89,-40.8,;24.55,-38.51,;25.88,-39.28,)| Show InChI InChI=1S/C28H25Cl3FN3O/c1-16(2)34-11-8-17(9-12-34)25-15-21(19-7-6-18(32)14-24(19)31)20-10-13-35(36)28(27(20)33-25)26-22(29)4-3-5-23(26)30/h3-7,10,13-17H,8-9,11-12H2,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.590 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357904
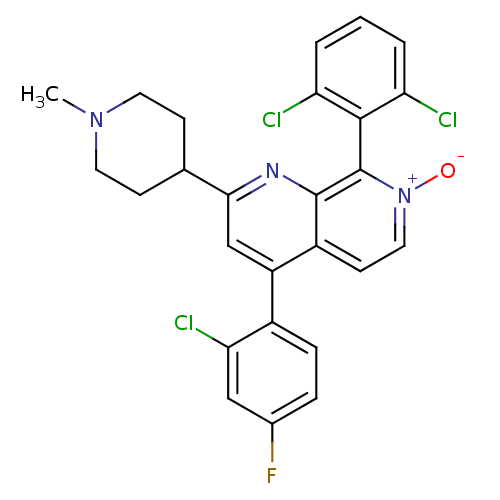
(CHEMBL1916361)Show SMILES CN1CCC(CC1)c1cc(-c2ccc(F)cc2Cl)c2cc[n+]([O-])c(-c3c(Cl)cccc3Cl)c2n1 |(17.77,-41.9,;16.45,-41.12,;15.11,-41.88,;13.78,-41.11,;13.79,-39.56,;15.12,-38.8,;16.45,-39.58,;12.46,-38.8,;12.47,-37.25,;11.13,-36.47,;11.13,-34.94,;12.46,-34.17,;12.47,-32.63,;11.13,-31.86,;11.13,-30.32,;9.79,-32.64,;9.8,-34.18,;8.47,-34.95,;9.79,-37.25,;8.46,-36.48,;7.13,-37.26,;7.13,-38.8,;5.79,-39.57,;8.46,-39.57,;8.47,-41.1,;7.13,-41.88,;5.8,-41.11,;7.13,-43.41,;8.47,-44.18,;9.81,-43.4,;9.8,-41.87,;11.13,-41.09,;9.79,-38.8,;11.12,-39.57,)| Show InChI InChI=1S/C26H21Cl3FN3O/c1-32-10-7-15(8-11-32)23-14-19(17-6-5-16(30)13-22(17)29)18-9-12-33(34)26(25(18)31-23)24-20(27)3-2-4-21(24)28/h2-6,9,12-15H,7-8,10-11H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 0.720 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM15242
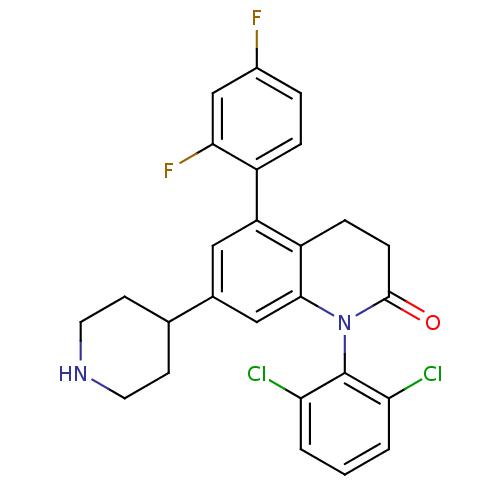
(1-(2,6-dichlorophenyl)-5-(2,4-difluorophenyl)-7-(4...)Show SMILES Fc1ccc(c(F)c1)-c1cc(cc2N(C(=O)CCc12)c1c(Cl)cccc1Cl)C1CCNCC1 Show InChI InChI=1S/C26H22Cl2F2N2O/c27-21-2-1-3-22(28)26(21)32-24-13-16(15-8-10-31-11-9-15)12-20(19(24)6-7-25(32)33)18-5-4-17(29)14-23(18)30/h1-5,12-15,31H,6-11H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| DrugBank
MMDB
PC cid
PC sid
PDB
UniChem
Patents
Similars
| DrugBank
MMDB
PDB
Article
PubMed
| n/a | n/a | 0.740 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha MAP kinase |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357902
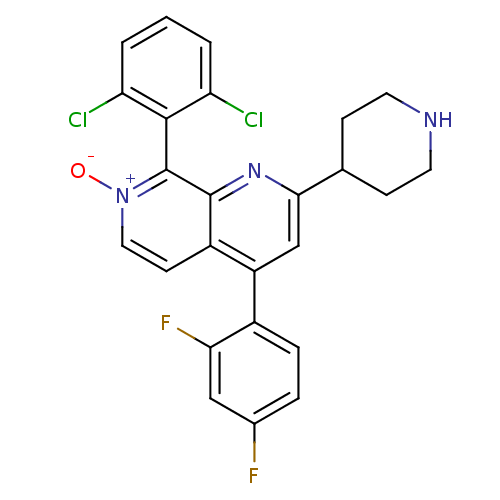
(CHEMBL1738839)Show SMILES [O-][n+]1ccc2c(cc(nc2c1-c1c(Cl)cccc1Cl)C1CCNCC1)-c1ccc(F)cc1F |(3.73,1.38,;2.66,.77,;2.66,-.77,;1.33,-1.54,;,-.77,;-1.33,-1.54,;-2.68,-.77,;-2.68,.77,;-1.33,1.54,;,.77,;1.33,1.54,;1.33,3.08,;-0,3.85,;-1.07,3.23,;-.01,5.39,;1.33,6.16,;2.66,5.39,;2.66,3.85,;3.73,3.24,;-4.01,1.54,;-5.35,.78,;-6.68,1.56,;-6.67,3.1,;-5.33,3.86,;-4,3.08,;-1.33,-3.08,;.01,-3.84,;.01,-5.38,;-1.32,-6.16,;-1.32,-7.39,;-2.65,-5.39,;-2.66,-3.85,;-3.73,-3.24,)| Show InChI InChI=1S/C25H19Cl2F2N3O/c26-19-2-1-3-20(27)23(19)25-24-17(8-11-32(25)33)18(16-5-4-15(28)12-21(16)29)13-22(31-24)14-6-9-30-10-7-14/h1-5,8,11-14,30H,6-7,9-10H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 0.820 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357917
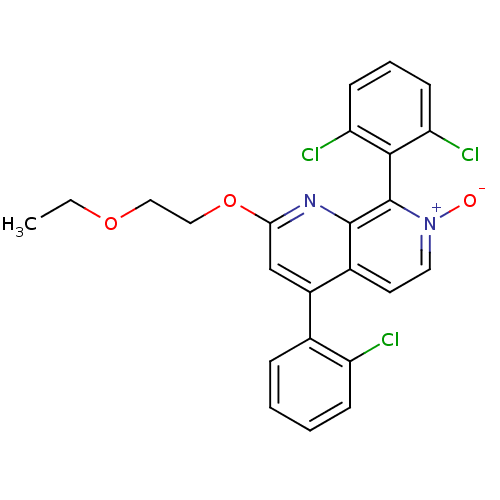
(CHEMBL1916531)Show SMILES CCOCCOc1cc(-c2ccccc2Cl)c2cc[n+]([O-])c(-c3c(Cl)cccc3Cl)c2n1 |(39.4,-4.5,;38.07,-5.26,;36.74,-4.49,;35.4,-5.26,;34.07,-4.48,;32.73,-5.25,;31.4,-4.48,;31.4,-2.93,;30.07,-2.15,;30.07,-.62,;31.4,.15,;31.4,1.69,;30.07,2.46,;28.73,1.68,;28.74,.15,;27.41,-.63,;28.73,-2.93,;27.4,-2.16,;26.07,-2.93,;26.07,-4.48,;24.73,-5.25,;27.4,-5.25,;27.41,-6.78,;26.07,-7.55,;24.74,-6.78,;26.07,-9.09,;27.41,-9.86,;28.75,-9.08,;28.74,-7.54,;30.07,-6.77,;28.73,-4.47,;30.06,-5.24,)| Show InChI InChI=1S/C24H19Cl3N2O3/c1-2-31-12-13-32-21-14-17(15-6-3-4-7-18(15)25)16-10-11-29(30)24(23(16)28-21)22-19(26)8-5-9-20(22)27/h3-11,14H,2,12-13H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 9 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357912
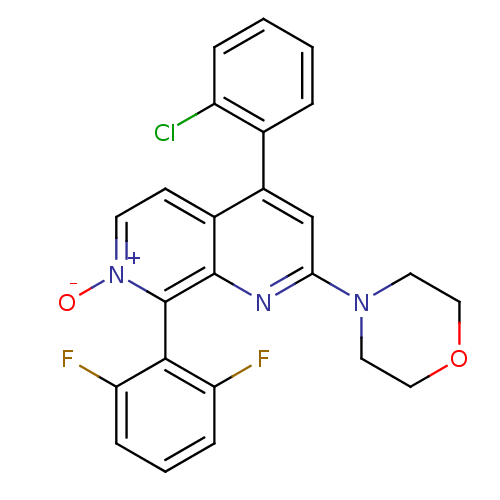
(CHEMBL1916526)Show SMILES [O-][n+]1ccc2c(cc(nc2c1-c1c(F)cccc1F)N1CCOCC1)-c1ccccc1Cl |(-7.92,-37.22,;-6.58,-36.45,;-6.58,-34.91,;-5.25,-34.14,;-3.92,-34.9,;-2.59,-34.12,;-1.25,-34.9,;-1.25,-36.45,;-2.59,-37.22,;-3.92,-36.45,;-5.25,-37.22,;-5.25,-38.75,;-6.58,-39.53,;-7.92,-38.76,;-6.58,-41.06,;-5.24,-41.83,;-3.91,-41.05,;-3.91,-39.52,;-2.58,-38.74,;.09,-37.22,;.08,-38.76,;1.4,-39.53,;2.74,-38.77,;2.75,-37.23,;1.42,-36.45,;-2.58,-32.59,;-1.25,-31.82,;-1.25,-30.28,;-2.58,-29.51,;-3.92,-30.29,;-3.91,-31.83,;-5.25,-32.6,)| Show InChI InChI=1S/C24H18ClF2N3O2/c25-18-5-2-1-4-15(18)17-14-21(29-10-12-32-13-11-29)28-23-16(17)8-9-30(31)24(23)22-19(26)6-3-7-20(22)27/h1-9,14H,10-13H2 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357908
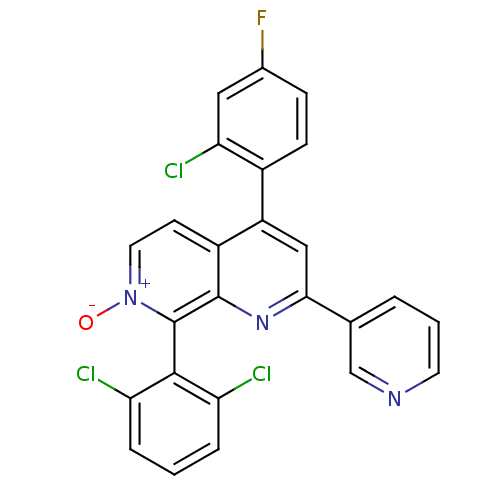
(CHEMBL1916522)Show SMILES [O-][n+]1ccc2c(cc(nc2c1-c1c(Cl)cccc1Cl)-c1cccnc1)-c1ccc(F)cc1Cl |(20.56,-4.2,;21.9,-3.43,;21.9,-1.88,;23.23,-1.11,;24.57,-1.88,;25.9,-1.1,;27.24,-1.88,;27.24,-3.43,;25.9,-4.19,;24.56,-3.42,;23.23,-4.2,;23.24,-5.73,;21.9,-6.5,;20.57,-5.74,;21.91,-8.04,;23.24,-8.81,;24.58,-8.03,;24.57,-6.49,;25.9,-5.72,;28.57,-4.19,;28.56,-5.74,;29.89,-6.51,;31.23,-5.74,;31.23,-4.2,;29.9,-3.43,;25.9,.43,;27.24,1.2,;27.24,2.74,;25.9,3.51,;25.9,5.05,;24.56,2.73,;24.57,1.2,;23.24,.42,)| Show InChI InChI=1S/C25H13Cl3FN3O/c26-19-4-1-5-20(27)23(19)25-24-17(8-10-32(25)33)18(16-7-6-15(29)11-21(16)28)12-22(31-24)14-3-2-9-30-13-14/h1-13H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357910

(CHEMBL1916524)Show SMILES CC(=O)N1CCC(CC1)c1cc(-c2ccccc2Cl)c2cc[n+]([O-])c(-c3c(F)cccc3F)c2n1 |(19.42,-20.86,;18.08,-21.62,;18.07,-23.16,;16.75,-20.84,;15.41,-21.6,;14.08,-20.83,;14.09,-19.29,;15.42,-18.52,;16.76,-19.3,;12.77,-18.52,;12.77,-16.98,;11.43,-16.2,;11.43,-14.66,;12.77,-13.9,;12.77,-12.36,;11.43,-11.59,;10.1,-12.36,;10.1,-13.9,;8.77,-14.67,;10.1,-16.97,;8.76,-16.21,;7.43,-16.98,;7.43,-18.52,;6.1,-19.29,;8.77,-19.29,;8.77,-20.83,;7.43,-21.6,;6.1,-20.83,;7.44,-23.14,;8.77,-23.91,;10.11,-23.12,;10.1,-21.59,;11.43,-20.81,;10.09,-18.52,;11.43,-19.29,)| Show InChI InChI=1S/C27H22ClF2N3O2/c1-16(34)32-12-9-17(10-13-32)24-15-20(18-5-2-3-6-21(18)28)19-11-14-33(35)27(26(19)31-24)25-22(29)7-4-8-23(25)30/h2-8,11,14-15,17H,9-10,12-13H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM31619
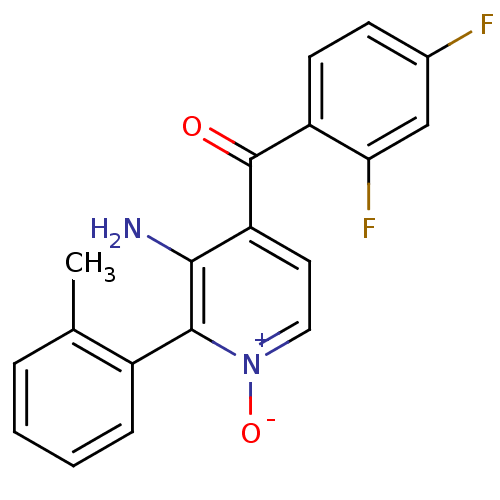
(Aminopyridine N-oxide, 45)Show SMILES Cc1ccccc1-c1c(N)c(cc[n+]1[O-])C(=O)c1ccc(F)cc1F |(1.08,6.55,;1.08,5.01,;2.42,4.23,;2.42,2.69,;1.08,1.92,;-.25,2.69,;-.25,4.23,;-1.58,5,;-1.58,6.55,;-.25,7.32,;-2.92,7.32,;-4.25,6.55,;-4.25,5,;-2.92,4.23,;-2.92,2.69,;-2.92,8.86,;-1.58,9.63,;-4.25,9.63,;-5.59,8.87,;-6.92,9.66,;-6.9,11.2,;-8.22,11.99,;-5.55,11.95,;-4.23,11.16,;-2.89,11.92,)| Show InChI InChI=1S/C19H14F2N2O2/c1-11-4-2-3-5-13(11)18-17(22)15(8-9-23(18)25)19(24)14-7-6-12(20)10-16(14)21/h2-10H,22H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
PDB
UniChem
Similars
| PDB
Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of p38alpha MAP kinase |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | 
3D Structure (crystal) |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357907
(CHEMBL1916521)Show SMILES [O-][n+]1ccc2c(cc(nc2c1-c1c(Cl)cccc1Cl)-c1ccncc1)-c1ccc(F)cc1Cl |(7.36,-4.61,;8.69,-3.84,;8.7,-2.3,;10.03,-1.52,;11.36,-2.29,;12.7,-1.51,;14.04,-2.29,;14.03,-3.84,;12.69,-4.61,;11.36,-3.84,;10.03,-4.61,;10.03,-6.14,;8.7,-6.92,;7.36,-6.15,;8.7,-8.46,;10.04,-9.23,;11.37,-8.44,;11.37,-6.91,;12.7,-6.13,;15.36,-4.61,;15.36,-6.15,;16.69,-6.92,;18.03,-6.16,;18.03,-4.61,;16.69,-3.84,;12.7,.02,;14.03,.79,;14.03,2.33,;12.7,3.1,;12.7,4.64,;11.36,2.32,;11.37,.79,;10.03,.01,)| Show InChI InChI=1S/C25H13Cl3FN3O/c26-19-2-1-3-20(27)23(19)25-24-17(8-11-32(25)33)18(16-5-4-15(29)12-21(16)28)13-22(31-24)14-6-9-30-10-7-14/h1-13H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357901
(CHEMBL1916358)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccccc1Cl |(1.89,-22.15,;3.22,-21.39,;3.23,-19.84,;4.55,-19.07,;5.89,-19.83,;7.22,-19.06,;8.56,-19.84,;8.56,-21.38,;7.22,-22.15,;5.89,-21.38,;4.56,-22.16,;4.56,-23.69,;3.23,-24.46,;1.89,-23.69,;3.23,-26,;4.57,-26.77,;5.9,-25.99,;5.89,-24.45,;7.22,-23.67,;7.22,-17.53,;8.56,-16.76,;8.56,-15.22,;7.23,-14.45,;5.89,-15.23,;5.89,-16.76,;4.56,-17.54,)| Show InChI InChI=1S/C20H11Cl3N2O/c21-15-5-2-1-4-13(15)12-8-10-24-19-14(12)9-11-25(26)20(19)18-16(22)6-3-7-17(18)23/h1-11H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357892

(CHEMBL1916349)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1F |(1.17,-37.04,;2.5,-36.27,;2.5,-34.73,;3.83,-33.96,;5.17,-34.72,;6.5,-33.95,;7.84,-34.72,;7.84,-36.27,;6.49,-37.04,;5.16,-36.27,;3.83,-37.04,;3.84,-38.58,;2.5,-39.35,;1.17,-38.58,;2.51,-40.89,;3.84,-41.65,;5.18,-40.87,;5.17,-39.34,;6.5,-38.56,;6.5,-32.41,;5.17,-31.65,;5.16,-30.11,;6.5,-29.33,;6.5,-27.79,;7.84,-30.11,;7.84,-31.64,;9.17,-32.42,)| Show InChI InChI=1S/C20H10Cl2F2N2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(23)10-17(13)24/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 41 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357893
(CHEMBL1916350)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(F)cccc1F)-c1ccc(F)cc1F |(11.83,-37.21,;13.16,-36.44,;13.17,-34.9,;14.49,-34.13,;15.83,-34.89,;17.16,-34.12,;18.5,-34.9,;18.5,-36.44,;17.16,-37.21,;15.83,-36.44,;14.5,-37.21,;14.5,-38.75,;13.17,-39.52,;11.83,-38.75,;13.17,-41.06,;14.51,-41.83,;15.84,-41.04,;15.83,-39.51,;17.16,-38.73,;17.16,-32.58,;15.83,-31.82,;15.83,-30.28,;17.17,-29.51,;17.16,-27.97,;18.5,-30.28,;18.5,-31.82,;19.83,-32.59,)| Show InChI InChI=1S/C20H10F4N2O/c21-11-4-5-13(17(24)10-11)12-6-8-25-19-14(12)7-9-26(27)20(19)18-15(22)2-1-3-16(18)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 66 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357886
(CHEMBL1916344)Show SMILES Cc1ccccc1-c1[n+]([O-])ccc2c(ccnc12)-c1ccc(F)cc1F |(26.55,-4.96,;27.88,-5.73,;27.89,-7.27,;29.22,-8.04,;30.56,-7.26,;30.55,-5.72,;29.22,-4.96,;29.21,-3.43,;27.88,-2.66,;26.54,-3.43,;27.88,-1.11,;29.21,-.34,;30.54,-1.11,;31.88,-.33,;33.22,-1.11,;33.22,-2.66,;31.87,-3.42,;30.54,-2.65,;31.88,1.2,;30.55,1.97,;30.54,3.5,;31.88,4.28,;31.88,5.82,;33.22,3.51,;33.22,1.97,;34.55,1.2,)| Show InChI InChI=1S/C21H14F2N2O/c1-13-4-2-3-5-15(13)21-20-18(9-11-25(21)26)16(8-10-24-20)17-7-6-14(22)12-19(17)23/h2-12H,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
CHEMBL
MCE
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 68 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357898
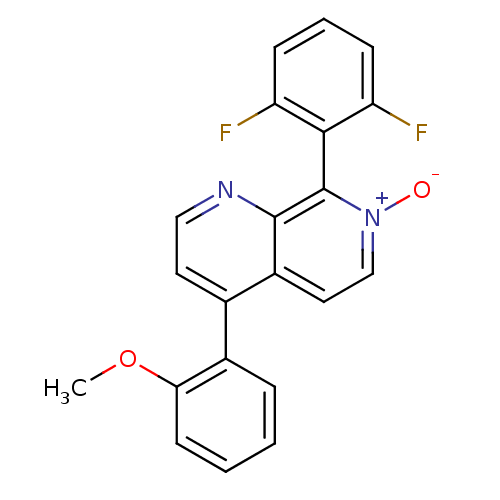
(CHEMBL1916355)Show SMILES COc1ccccc1-c1ccnc2c(-c3c(F)cccc3F)[n+]([O-])ccc12 |(15.24,2.65,;16.58,1.89,;17.91,2.66,;17.9,4.2,;19.24,4.97,;20.58,4.2,;20.58,2.66,;19.24,1.89,;19.24,.36,;20.58,-.42,;20.58,-1.96,;19.23,-2.73,;17.9,-1.96,;16.57,-2.74,;16.58,-4.27,;15.24,-5.04,;13.91,-4.27,;15.25,-6.58,;16.58,-7.35,;17.92,-6.57,;17.91,-5.03,;19.24,-4.25,;15.24,-1.96,;13.91,-2.73,;15.24,-.42,;16.57,.35,;17.91,-.41,)| Show InChI InChI=1S/C21H14F2N2O2/c1-27-18-8-3-2-5-14(18)13-9-11-24-20-15(13)10-12-25(26)21(20)19-16(22)6-4-7-17(19)23/h2-12H,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 77 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357897

(CHEMBL1916354)Show SMILES Cc1cc(NS(C)(=O)=O)ccc1-c1[n+]([O-])ccc2c(ccnc12)-c1ccc(F)cc1F |(7.61,-4.28,;6.28,-5.06,;6.29,-6.6,;4.95,-7.38,;4.96,-8.92,;6.29,-9.68,;7.63,-8.91,;7.05,-11.01,;5.52,-11.01,;3.62,-6.61,;3.62,-5.07,;4.95,-4.3,;4.95,-2.77,;3.61,-2,;2.28,-2.76,;3.61,-.45,;4.94,.32,;6.28,-.44,;7.61,.33,;8.95,-.45,;8.95,-1.99,;7.61,-2.76,;6.28,-1.99,;7.61,1.86,;6.28,2.63,;6.28,4.16,;7.61,4.94,;7.61,6.48,;8.95,4.17,;8.95,2.63,;10.28,1.86,)| Show InChI InChI=1S/C22H17F2N3O3S/c1-13-11-15(26-31(2,29)30)4-6-16(13)22-21-19(8-10-27(22)28)17(7-9-25-21)18-5-3-14(23)12-20(18)24/h3-12,26H,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 81 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357894

(CHEMBL1916351)Show SMILES Cc1cc(O)ccc1-c1[n+]([O-])ccc2c(ccnc12)-c1ccc(F)cc1F |(22.05,-38.39,;23.39,-39.16,;23.39,-40.7,;24.73,-41.47,;24.73,-43.01,;26.06,-40.69,;26.05,-39.15,;24.72,-38.39,;24.72,-36.86,;23.38,-36.09,;22.05,-36.85,;23.39,-34.54,;24.71,-33.77,;26.05,-34.53,;27.38,-33.76,;28.72,-34.54,;28.72,-36.08,;27.38,-36.85,;26.05,-36.08,;27.38,-32.23,;26.05,-31.46,;26.05,-29.93,;27.39,-29.15,;27.38,-27.61,;28.72,-29.92,;28.72,-31.46,;30.05,-32.23,)| Show InChI InChI=1S/C21H14F2N2O2/c1-12-10-14(26)3-5-15(12)21-20-18(7-9-25(21)27)16(6-8-24-20)17-4-2-13(22)11-19(17)23/h2-11,26H,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 90 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357891

(CHEMBL1916348)Show SMILES COc1cccc(OC)c1-c1[n+]([O-])ccc2c(ccnc12)-c1ccc(F)cc1F |(-10.9,-39.4,;-9.56,-38.63,;-8.23,-39.39,;-8.23,-40.93,;-6.89,-41.7,;-5.55,-40.92,;-5.56,-39.38,;-4.23,-38.61,;-2.9,-39.37,;-6.89,-38.62,;-6.9,-37.09,;-8.23,-36.32,;-9.57,-37.09,;-8.23,-34.77,;-6.9,-34,;-5.57,-34.77,;-4.23,-33.99,;-2.9,-34.77,;-2.9,-36.32,;-4.24,-37.08,;-5.57,-36.31,;-4.23,-32.46,;-5.56,-31.7,;-5.57,-30.16,;-4.23,-29.38,;-4.23,-27.84,;-2.9,-30.15,;-2.9,-31.69,;-1.56,-32.46,)| Show InChI InChI=1S/C22H16F2N2O3/c1-28-18-4-3-5-19(29-2)20(18)22-21-16(9-11-26(22)27)14(8-10-25-21)15-7-6-13(23)12-17(15)24/h3-12H,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 141 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357887
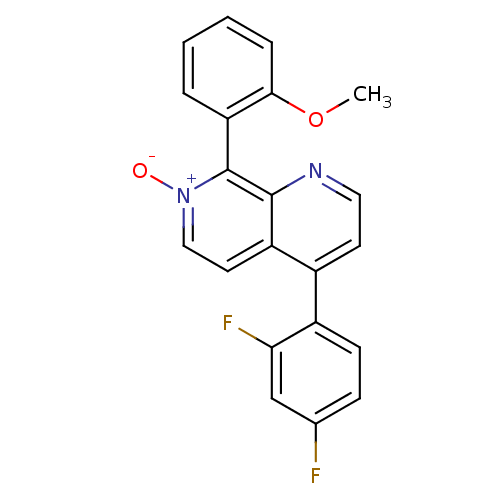
(CHEMBL1914478)Show SMILES COc1ccccc1-c1[n+]([O-])ccc2c(ccnc12)-c1ccc(F)cc1F |(-8.7,-22.73,;-7.37,-21.96,;-6.04,-22.73,;-6.03,-24.26,;-4.7,-25.03,;-3.36,-24.25,;-3.37,-22.72,;-4.7,-21.95,;-4.71,-20.42,;-6.04,-19.65,;-7.37,-20.42,;-6.04,-18.11,;-4.71,-17.34,;-3.37,-18.1,;-2.04,-17.32,;-.7,-18.1,;-.7,-19.65,;-2.05,-20.42,;-3.38,-19.65,;-2.04,-15.79,;-3.37,-15.03,;-3.38,-13.49,;-2.04,-12.71,;-2.04,-11.17,;-.7,-13.48,;-.7,-15.02,;.63,-15.79,)| Show InChI InChI=1S/C21H14F2N2O2/c1-27-19-5-3-2-4-17(19)21-20-16(9-11-25(21)26)14(8-10-24-20)15-7-6-13(22)12-18(15)23/h2-12H,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 144 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357890

(CHEMBL1916347)Show SMILES Cc1cccc(C)c1-c1[n+]([O-])ccc2c(ccnc12)-c1ccc(F)cc1F |(25.95,-22.11,;27.28,-22.87,;27.29,-24.41,;28.62,-25.18,;29.96,-24.4,;29.95,-22.86,;31.28,-22.09,;28.62,-22.1,;28.61,-20.57,;27.28,-19.8,;25.95,-20.57,;27.28,-18.25,;28.61,-17.48,;29.95,-18.25,;31.28,-17.47,;32.62,-18.25,;32.62,-19.8,;31.27,-20.56,;29.94,-19.79,;31.28,-15.94,;29.95,-15.18,;29.94,-13.64,;31.28,-12.86,;31.28,-11.32,;32.62,-13.63,;32.62,-15.17,;33.95,-15.94,)| Show InChI InChI=1S/C22H16F2N2O/c1-13-4-3-5-14(2)20(13)22-21-18(9-11-26(22)27)16(8-10-25-21)17-7-6-15(23)12-19(17)24/h3-12H,1-2H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 148 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
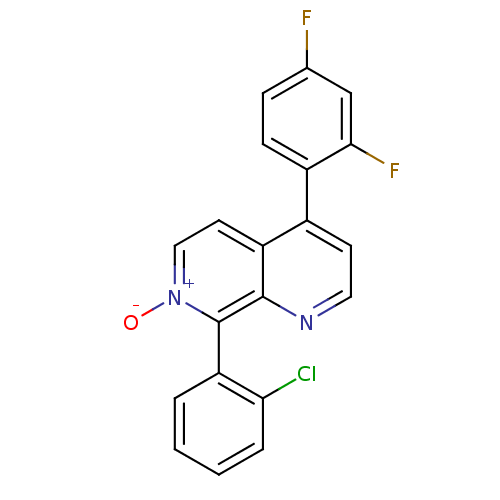
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357888
(CHEMBL1916345)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1ccccc1Cl)-c1ccc(F)cc1F |(3.33,-20.74,;4.66,-19.97,;4.66,-18.43,;5.99,-17.65,;7.33,-18.42,;8.66,-17.64,;10,-18.42,;10,-19.97,;8.66,-20.74,;7.33,-19.97,;6,-20.74,;6,-22.27,;7.33,-23.04,;7.34,-24.57,;6,-25.35,;4.67,-24.58,;4.67,-23.05,;3.33,-22.28,;8.66,-16.11,;7.33,-15.35,;7.33,-13.81,;8.66,-13.03,;8.66,-11.49,;10,-13.8,;10,-15.34,;11.33,-16.11,)| Show InChI InChI=1S/C20H11ClF2N2O/c21-17-4-2-1-3-16(17)20-19-15(8-10-25(20)26)13(7-9-24-19)14-6-5-12(22)11-18(14)23/h1-11H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 195 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357889

(CHEMBL1916346)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1ccccc1F)-c1ccc(F)cc1F |(14.31,-20.58,;15.64,-19.81,;15.65,-18.26,;16.98,-17.49,;18.31,-18.26,;19.64,-17.48,;20.98,-18.26,;20.98,-19.81,;19.64,-20.57,;18.31,-19.8,;16.98,-20.58,;16.98,-22.11,;18.31,-22.87,;18.32,-24.41,;16.99,-25.19,;15.65,-24.42,;15.65,-22.88,;14.31,-22.11,;19.64,-15.95,;18.31,-15.18,;18.31,-13.65,;19.65,-12.87,;19.65,-11.33,;20.98,-13.64,;20.98,-15.18,;22.31,-15.95,)| Show InChI InChI=1S/C20H11F3N2O/c21-12-5-6-14(18(23)11-12)13-7-9-24-19-15(13)8-10-25(26)20(19)16-3-1-2-4-17(16)22/h1-11H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 333 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357895
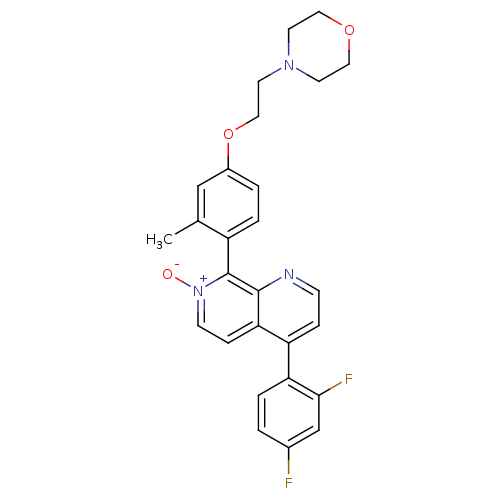
(CHEMBL1916352)Show SMILES Cc1cc(OCCN2CCOCC2)ccc1-c1[n+]([O-])ccc2c(ccnc12)-c1ccc(F)cc1F |(39.03,-36.8,;37.7,-37.57,;37.71,-39.11,;36.38,-39.89,;36.38,-41.43,;35.05,-42.2,;33.71,-41.44,;32.38,-42.21,;31.05,-41.44,;29.73,-42.21,;29.72,-43.75,;31.06,-44.52,;32.4,-43.75,;35.04,-39.12,;35.04,-37.58,;36.37,-36.81,;36.37,-35.28,;35.03,-34.51,;33.7,-35.28,;35.04,-32.96,;36.37,-32.19,;37.7,-32.96,;39.03,-32.18,;40.37,-32.96,;40.37,-34.51,;39.03,-35.27,;37.7,-34.5,;39.03,-30.65,;37.7,-29.88,;37.7,-28.35,;39.04,-27.57,;39.04,-26.03,;40.37,-28.34,;40.37,-29.88,;41.7,-30.65,)| Show InChI InChI=1S/C27H25F2N3O3/c1-18-16-20(35-15-12-31-10-13-34-14-11-31)3-5-21(18)27-26-24(7-9-32(27)33)22(6-8-30-26)23-4-2-19(28)17-25(23)29/h2-9,16-17H,10-15H2,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 422 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357885
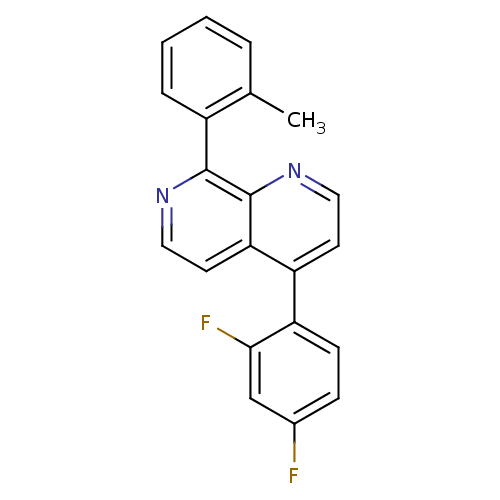
(CHEMBL1916343)Show InChI InChI=1S/C21H14F2N2/c1-13-4-2-3-5-15(13)20-21-18(9-11-24-20)16(8-10-25-21)17-7-6-14(22)12-19(17)23/h2-12H,1H3 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357896
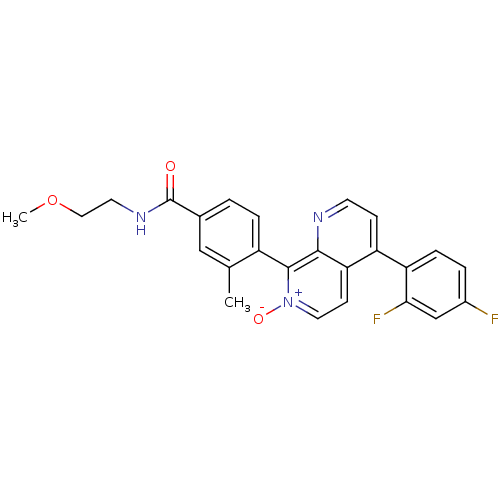
(CHEMBL1916353)Show SMILES COCCNC(=O)c1ccc(c(C)c1)-c1[n+]([O-])ccc2c(ccnc12)-c1ccc(F)cc1F |(-1.08,-9.67,;-2.41,-8.9,;-3.75,-9.67,;-5.08,-8.91,;-6.41,-9.68,;-7.75,-8.92,;-9.08,-9.69,;-7.75,-7.38,;-9.09,-6.61,;-9.09,-5.07,;-7.76,-4.3,;-6.43,-5.06,;-5.1,-4.28,;-6.42,-6.6,;-7.76,-2.77,;-9.1,-2,;-10.43,-2.76,;-9.09,-.45,;-7.77,.32,;-6.43,-.44,;-5.1,.33,;-3.76,-.45,;-3.76,-1.99,;-5.1,-2.7,;-6.43,-1.99,;-5.1,1.86,;-6.43,2.63,;-6.43,4.16,;-5.09,4.94,;-5.1,6.48,;-3.76,4.17,;-3.76,2.63,;-2.43,1.86,)| Show InChI InChI=1S/C25H21F2N3O3/c1-15-13-16(25(31)29-10-12-33-2)3-5-18(15)24-23-21(8-11-30(24)32)19(7-9-28-23)20-6-4-17(26)14-22(20)27/h3-9,11,13-14H,10,12H2,1-2H3,(H,29,31) | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 1.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human MAPK2 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
RAF proto-oncogene serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human c-RAF |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Epidermal growth factor receptor
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human EGFR |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Vascular endothelial growth factor receptor 1
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human Flt1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Homeodomain-interacting protein kinase 1
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human HIPK1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase JAK3
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human JAK3 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 1
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human MAPK1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
MAP kinase-interacting serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human Mnk2 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human Abl |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 13
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human SAPK4 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mast/stem cell growth factor receptor Kit
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human cKit |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Ephrin type-A receptor 2
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human EphA2 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase pim-1
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human Pim-1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Nuclear receptor subfamily 2 group C member 2
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human TAK1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Dual specificity tyrosine-phosphorylation-regulated kinase 2
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human DYRK2 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 8
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human JNK1alpha1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human Lck |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase 9
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human MLK1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Ribosomal protein S6 kinase alpha-5
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human MSK1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase BTK
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human BTK |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase Nek2
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human NEK2 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Protein kinase C alpha type
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human PKCalpha |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human cSRC |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Aurora kinase A
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human Aurora-A |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357899

(CHEMBL1916356)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1cccc(c1)C(F)(F)F |(25.31,-3.57,;26.64,-2.8,;26.64,-1.25,;27.97,-.48,;29.31,-1.25,;30.64,-.47,;31.98,-1.25,;31.98,-2.8,;30.64,-3.56,;29.31,-2.79,;27.98,-3.57,;27.98,-5.1,;26.65,-5.87,;25.31,-5.1,;26.65,-7.41,;27.98,-8.18,;29.32,-7.4,;29.31,-5.86,;30.64,-5.09,;30.64,1.06,;31.98,1.83,;31.98,3.37,;30.64,4.14,;29.31,3.36,;29.31,1.83,;27.97,4.13,;27.96,5.67,;26.64,3.35,;26.63,4.9,)| Show InChI InChI=1S/C21H11Cl2F3N2O/c22-16-5-2-6-17(23)18(16)20-19-15(8-10-28(20)29)14(7-9-27-19)12-3-1-4-13(11-12)21(24,25)26/h1-11H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Glycogen synthase kinase-3 beta
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human GSK3-beta |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 2
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human MAPKAP-K2 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
ALK tyrosine kinase receptor
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human ALK |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
RAC-alpha serine/threonine-protein kinase
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human PKBalpha |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Receptor-interacting serine/threonine-protein kinase 2
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human RIPK2 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
BDNF/NT-3 growth factors receptor
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human TrkB |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Rho-associated protein kinase 2
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human ROCK-2 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 10
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human JNK3 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase kinase kinase 5
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human ASK1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 11
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human SAPK2b |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
SRSF protein kinase 1
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human SRPK1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase Chk1
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human CHK1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human SAPK2a |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Fibroblast growth factor receptor 1
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human FGFR1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Interleukin-1 receptor-associated kinase 1
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human IRAK1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 9
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human JNK2alpha2 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Myosin light chain kinase, smooth muscle
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human MLCK |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Cyclin-dependent kinase 6
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human CDK6 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 14
(Homo sapiens (Human)) | BDBM50357900

(CHEMBL1916357)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(cc1)C(F)(F)F |(-9.35,-24.55,;-8.02,-23.78,;-8.01,-22.24,;-6.68,-21.47,;-5.35,-22.23,;-4.02,-21.45,;-2.68,-22.23,;-2.68,-23.78,;-4.02,-24.55,;-5.35,-23.78,;-6.68,-24.55,;-6.68,-26.08,;-8.01,-26.86,;-9.35,-26.09,;-8.01,-28.39,;-6.67,-29.16,;-5.34,-28.38,;-5.35,-26.85,;-4.02,-26.07,;-4.02,-19.92,;-2.68,-19.15,;-2.68,-17.61,;-4.01,-16.84,;-5.35,-17.62,;-5.35,-19.16,;-4.01,-15.3,;-2.68,-14.53,;-5.35,-14.53,;-4.03,-13.76,)| Show InChI InChI=1S/C21H11Cl2F3N2O/c22-16-2-1-3-17(23)18(16)20-19-15(9-11-28(20)29)14(8-10-27-19)12-4-6-13(7-5-12)21(24,25)26/h1-11H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human p38alpha MAP kinase after 1 hr by FRET analysis |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
MAP kinase-activated protein kinase 5
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human PRAK |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Ribosomal protein S6 kinase alpha-1
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human Rsk1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Mitogen-activated protein kinase 12
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human SAPK3 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase SYK
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human Syk |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase D2
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human PKD2 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Ephrin type-B receptor 1
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human EphB1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Inhibitor of nuclear factor kappa-B kinase subunit beta
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human IKK-beta |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Calcium/calmodulin-dependent protein kinase type IV
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human CaMK4 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Casein kinase I isoform gamma-1
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human CK1gamma1 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Casein kinase I isoform delta
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human CK1delta |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor beta
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human PDGFRbeta |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |
Serine/threonine-protein kinase N2
(Homo sapiens (Human)) | BDBM50357884

(CHEMBL1916359)Show SMILES [O-][n+]1ccc2c(ccnc2c1-c1c(Cl)cccc1Cl)-c1ccc(F)cc1Cl |(12.75,-22.82,;14.08,-22.05,;14.08,-20.51,;15.41,-19.74,;16.75,-20.5,;18.08,-19.73,;19.42,-20.51,;19.42,-22.05,;18.08,-22.82,;16.74,-22.05,;15.42,-22.83,;15.42,-24.36,;14.08,-25.13,;12.75,-24.36,;14.09,-26.67,;15.42,-27.44,;16.76,-26.66,;16.75,-25.12,;18.08,-24.34,;18.08,-18.2,;19.42,-17.43,;19.42,-15.89,;18.08,-15.12,;18.08,-13.58,;16.75,-15.89,;16.75,-17.43,;15.42,-18.2,)| Show InChI InChI=1S/C20H10Cl3FN2O/c21-15-2-1-3-16(22)18(15)20-19-14(7-9-26(20)27)12(6-8-25-19)13-5-4-11(24)10-17(13)23/h1-10H | PDB
NCI pathway
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Rhône-Poulenc Rorer
Curated by ChEMBL
| Assay Description
Inhibition of human PRK2 |
J Med Chem 54: 7899-910 (2011)
Article DOI: 10.1021/jm200975u
BindingDB Entry DOI: 10.7270/Q2MW2HKR |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data